Abstract
This paper presents a textile-based pH sensor with high flexibility fabricated by printing a polymer composite as a working electrode and Ag/AgCl/solid electrolyte as a reference electrode on a textile substrate. The textile working electrode is composed of polyaniline, carbon nanotubes, and agarose printed on the textile. A thermoplastic polyurethane overlayer hot-pressed on the textile substrates provides a smooth hydrophobic surface, enabling a more stable formation of the composite films with a reliable output signal. The textile reference electrode is fabricated by printing Ag/AgCl paste and solid electrolyte. The fully textile-based pH sensor, by integrating the textile working and reference electrodes, exhibits a good sensitivity of 45.9 mV/pH with high linearity. The textile pH sensor maintains excellent performance and repeatability with 93% retention even in a bent state and after 1000 bending cycles. Finally, it is demonstrated that the textile pH sensor can detect the pH change on a piece of porcine skin.

Introduction
Monitoring the pH of human skin has generated great interest because it provides direct and indirect information related to human health. For example, skin diseases such as dermatitis and fungal infections change the pH of human skin. 1 The change in the pH of skin may also indicate a state of dehydration due to strenuous exercise, heatstroke, etc. The pH of healthy skin is in the range of 4.2–5.6 while the pH of dehydrated skin shifts to weakly basic. 2 In particular, the pH of skin is useful for determining the phases of wound healing. Under normal conditions, the surface of undamaged healthy skin is slightly acidic depending on the physiological and anatomical conditions of the human body and age. Once the skin is damaged by a wound, the biochemical reactions associated with blood clotting and inflammation increase the pH above 7. As the wound heals, the skin becomes acidic again. 3 For more careful treatment of skin diseases, wounds, and dehydration state, pH sensors that can be conformally attached to the skin could be a promising way to monitor the phases of the symptoms continuously and to prevent their deterioration.
Flexibility is one of the essential requirements of skin pH sensors to ensure conformal contact with the skin for reliable and accurate monitoring.4,5 The flexible skin pH sensors reported so far primarily use polymeric films such as polyimide (PI), polyethylene terephthalate (PET), and parylene as substrates to provide flexible performance. For example, Li et al. reported a pH sensor fabricated by spin-coating polyaniline (PANI) doped with dodecylbenzenesulfonic acid on a PI substrate. 6 Yoon et al. also produced a flexible pH sensor on a PET film consisting of a working electrode with a PANI nanopillar array and a Ag/AgCl reference electrode. 7 In addition to flexibility, the polymer films have the advantage of offering physical and structural stability sufficient to enable various fabrication processes including soft lithography, printing, casting, and deposition.7–9 However, their flexibility may not be sufficient to form a conformal contact with the skin of curved body parts such as fingers, shoulders, ankles, and heels.
Utilizing a textile substrate instead of a film substrate could be a promising approach to improve the flexibility and breathability of wearable sensors. The textile substrates for monitoring skin pH are generally nontoxic and are highly deformable without changing the intrinsic physical or chemical properties.10,11 Recently, there have been a couple of studies on textile-based pH sensors. For instance, Zamora et al. presented a textile-based sensor that measures the pH of sweat. 12 Different types of conductive fabric such as a stainless steel mesh with an electrodeposited iridium oxide film showed good sensitivity to the pH change from 4 to 8. Despite its high sensitivity, the sensor based on rigid inorganic materials may have some limitations on its manufacturing process, flexibility, and durability against repeated deformation. Also, only the sensing electrode was fabricated on the steel mesh and a commercial Ag/AgCl electrode was used as a reference electrode. Maniakkal et al. proposed a pH sensor with a sensing electrode made of graphite and polyurethane composite. With a Ag/AgCl reference electrode, the electrodes were printed on a cellulose-polyester blend cloth. The pH sensor was highly flexible and showed a fast response time of ≈5 s, while the sensitivity was relatively low, less than 4 mV/pH in the range of pH 6–9. 13 Mariani et al. exhibited a textile-based pH sensor based on a bio-ceramic fabric. Poly(3,4-ethylenedioxythiophene):poly(styrene sulfonate) (PEDOT:PSS) was printed on the fabric, and then it was functionalized with PEDOT doped with bromothymol blue (PEDOT:BTB) by electrodeposition as a pH-sensitive layer. However, the voltage should be provided FOR current measurement due to its chemo-resistive like mechanism, and the sensitivity was relatively low. 14 Thus, although a textile could be one of the most suitable substrates for a skin-conformal pH sensor, several challenging issues remain.
Conductive polymers other than inorganic or carbon materials can also exhibit pH-sensing properties. Conductive polymers have advantages when applied to wearable sensors due to their flexibility, ease of processing, and excellent electrical properties. Among a variety of conductive polymers, PANI is one of the most promising substances to be the sensing electrode of a pH sensor due to its reversible doping–dedoping.15–18 In addition to the high pH sensitivity, PANI can be easily synthesized at a low cost and can be processed into thin films using simple casting or printing techniques such as inkjet printing, screen printing, and blade coating.19–21
This paper introduces a highly flexible and sensitive potentiometric pH sensor based on a woven textile substrate. A composite paste composed of carbon nanotubes (CNTs), agarose, and PANI (CAP) forms the pH-sensing film of the textile working electrode by doctor-blading. Cotton, normal polyester, and waterproof polyester are compared as the textile substrate. A thermoplastic polyurethane (TPU) overlayer is adopted to improve further the CAP film stability. The pH-dependent potential of the textile working electrode is investigated. The textile reference electrode is prepared by printing Ag/AgCl paste and solid electrolyte on the textile substrate. To maintain the flexibility of the reference electrode, a silicone-based solid electrolyte is coated on the surface of the textile reference electrode. The fully textile-based pH sensor is fabricated by integrating the textile working and reference electrodes. The pH-sensing performance of the textile sensor is discussed in terms of sensitivity and linearity. The physical durability and stability of the sensor are also analyzed by comparing the pH-sensing performance before and after repeated bending cycles and observing the surface morphology of the electrodes. Finally, the pH sensing of the textile sensor on a piece of porcine skin is demonstrated.
Experimental
Materials
Ammonium persulfate (ACS reagent, ≥98.0%), aniline (ACS reagent, ≥99.5%), phytic acid (50% (w/w) in H2O), agarose, potassium ferricyanide (K3Fe(CN)6) (powder, < 10 µm, 99%), and phosphate-buffered saline (pH 7.4) were purchased from Sigma-Aldrich, Republic of Korea. Carbon nanotubes (3 wt% multiwalled CNTs, a diameter of 20–30 nm and a length of 10–30 µm) were purchased from Nanostructured & Amorphous Materials Inc., USA. The fabric was purchased from Derhappy Ltd. Republic of Korea. Thermoplastic polyurethane (TPU, 3080w) film was purchased from Sealon, Republic of Korea. DM-ENC-2500 from Dycotec Materials Ltd., UK was used as the encapsulant paste. The silver paste (Loctite® EDAG 479SS E&C) used in working electrode and the Ag/AgCl paste (011464 Ag/AgCl ink) were purchased from Henkel, Germany and BAS Inc., Japan, respectively. Potassium chloride (KCl, 99.0%) and buffer solution (pH 5–9 ± 0.02, 25 °C) were purchased from Samchun, Republic of Korea. Ecoflex (00-30) from Smooth-on, USA, was used as the matrix of solid electrolyte.
Synthesis of polyaniline
For the synthesis of PANI, we refer to the procedure reported by Pan et al. 22 The solution of the initiator was prepared by mixing 0.286 g of ammonium persulfate and 1 mL of deionized (DI) water. The solution of monomer and crosslinker was prepared by mixing 0.46 mL of aniline, 0.92 mL of phytic acid solution, and 2 mL of DI water. The two solutions were mixed thoroughly to form a hydrogel. The PANI hydrogel was washed with DI water to remove unreacted reactants and impurities and then fully dried in an oven at 60 °C overnight. The dried PANI was ground with a mortar to form a powder.
Preparation of CAP composite paste
0.1 g of the synthesized PANI powder was mixed with 6.67 g of an aqueous CNT suspension and 3.03 g of DI water. After the suspension of CNTs and PANI was sonicated for 10 min in an ultrasonic water bath (Power sonic 505, 40 kHz, Hwashin Tech Co., Ltd., Republic of Korea), it was heated on a hot plate set at 110 °C for ≈10 min under continuous stirring. Subsequently, 0.2 g of agarose powder was added to the suspension while being stirred. After the agarose was completely dissolved, the temperature of the hot plate was decreased to 80 °C, so that the viscous CAP paste was formed. The relative amounts of each component in the prepared CAP composite paste are 2:2:1 for CNT, agarose, and PANI, respectively.
Manufacturing of the textile working electrode
The fabrication process of the textile working electrode is illustrated in Figure 1(a) and Figure S1(a). The different types of fabric, cotton, normal polyester, and polyurethane-treated waterproof polyester were compared as a textile substrate. First, if applicable, the 80 µm-thick TPU film was hot-pressed to a rectangular piece of fabric at 170–180 °C. After aligning a mask with a thickness of 100 µm on the textile substrate, silver paste was printed using a blade coater and cured at 60 °C for 1 h. In the same way, the CAP paste was printed on the silver area by using the mask with a thickness of 200 µm and then dried at room temperature for ≈2 h. The area of working electrode is 304 mm2 (8 mm × 38 mm). To expose only the sensing area and to protect the rest of the electrode, a urethane-based resin encapsulant paste was applied on the part of the CAP film and then cured at 120 °C for ≈10 min. Fabrication process of the textile pH sensor. (a) The textile working electrode and (b) the textile reference electrode.
Manufacturing of the textile reference electrode
The fabrication process of the reference electrode is illustrated in Figure 1(b) and Figure S1(b). First, the TPU film was hot-pressed on the waterproof polyester at 170–180 °C. After applying a 300 µm thick mask on the textile substrate, a Ag/AgCl paste was printed using a blade coater and then cured at 60 °C for 1 h. To form a solid electrolyte layer on the Ag/AgCl electrode, a paste was prepared by mixing KCl powder and Ecoflex in a 1:1 mass ratio. Subsequently, the solid electrolyte paste was applied by doctor-blading, and then dried at room temperature for 2 h. As for the working electrode, a part of the reference electrode was coated with the encapsulant paste and cured at 120 °C for ≈10 min. The encapsulant paste helps to protect the solid electrolyte and to prevent KCl in the solid electrolyte from quickly leaking out.
Characterization and methods
Surface morphology observation
The structural morphologies of the CAP working electrode and the solid electrolyte of the reference electrode were investigated using a scanning electron microscope (SEM, SU8010, Hitachi High Technologies Corporation, Japan).
Electrochemical measurement
For the potentiometric measurement in response to pH, that is, the potential difference between the working electrode and the reference electrode, a potentiostat (SP-200, Biologic, France) and software (EC-Lab, V11.20) were used. To maintain the electrical connection securely between the electrodes and the potentiostat, we attached copper tape at the ends of the electrodes and then connected the tape to the measuring device.
When the pH-sensing ability of the textile working electrode was individually analyzed, the potentiometric measurement was carried out using a commercial Ag/AgCl reference electrode (CHI111, Qrins Inc., Republic of Korea). To confirm that the textile reference electrode operated properly, cyclic voltammetry (CV) measurements were conducted using a three-electrode system. A gold-deposited wafer and a platinum wire were used as the working electrode and the counter electrode, respectively. 1 mM potassium ferricyanide (K3Fe(CN)6) in 1 M potassium chloride (KCl) aqueous solution was used as the electrolyte. The scan range was from −0.1 to 0.6 V with scan rates of 5, 25, 50, 100, 150, and 200 mV/s.
To assess the pH-sensing performance of the textile-based pH sensor, buffer solutions with different pH were used. Before all experiments, the pH of the buffer solutions was verified with a commercial pH meter (PHS-3D-02, Sanxin, China), and the sensors were rinsed with DI water and blow-dried with nitrogen. Even during the rinsing and drying process, the films of the sensors were stable without peeling or detachment.
Cyclic bending test of the textile pH sensor
To investigate the flexibility and durability of the textile pH sensor, we conducted a cyclic bending test using a custom-made apparatus that had two clamps to hold the sample with motion-control software (Bending & Stretchable Machine System, SnM, Republic of Korea). One clamp is stationary and the other one was moved back and forth by a motor to generate a radius of curvature of 4 mm. The speed of motion was 22 mm/s and the bending process was repeated 1000 times.
pH sensing on real animal skin
To demonstrate the pH-sensing ability of the textile pH sensor on real animal skin, square pieces of porcine skin was purchased from Apures Co., Ltd. Republic of Korea. The size of the skin was 3 cm × 3 cm, and the thickness was 1200 μm. The porcine skin was rinsed with phosphate-buffered saline to remove impurities on its surface. Two pieces of skin were immersed in pH 5 and 7 buffer solutions and then stored in a refrigerator at 4 °C for 24 h to let the skin absorb the buffer solution. After the preconditioning process, the textile pH sensor was placed on the skin and the potential between the two electrodes of the sensor was measured using a digital multimeter (Fluke Corporation, USA). For the consecutive measurement for the skin of pH 5 and 7, the pH sensor was not rinsed with DI water or the saline solution to mimic the case of real wounded skin.
Results and discussion
Textile working electrode
For fabrication of the textile-based pH sensor with better processability and reliable performance, it is essential to choose the proper textile substrate. We fabricated and tested the textile working electrodes with three different types of woven textile: cotton, polyester, and waterproof polyester. The waterproof polyester is polyester coated with polyurethane resin and PET through thermoplastic processing coating technology.
23
When the CAP composite paste is printed on a cotton substrate, the water-based paste partly infuses into the porous structure of the hygroscopic cotton (Figure S2). After drying, many millimeter-to micrometer-sized cracks were formed on the CAP film (Figure 2(a)) and thus the potential of the resulting working electrode was not measurable. On the waterproof polyester substrate, the CAP film is most reliably formed without significant crack formation (Figure 2(b)). Thus, for the CAP paste printing for fabrication of the textile working electrode, a more hydrophobic textile or introduction of hydrophobic overlayer is preferred. Top-viewed images of CAP film on (a) cotton and (b) waterproof polyester. (c) Section-and (d) top-viewed images of waterproof polyester with TPU overlayer.
TPU film hot-pressed on textile substrates, as shown in Figure 2(c), can provide a smooth hydrophobic surface for the CAP printing, regardless of the textile types. Indeed, with the TPU overlayer, the CAP film is well printed without cracks for all textile substrates tested (Figure 2(d) and Figure S3). During the potential measurement in samples of different pH, however, the cotton substrate is likely to swell by absorbing a large amount of water, causing deformation of the CAP film, and even delamination of the TPU overlayer from cotton. As a result, the potential measured by the CAP on cotton with TPU is not measurable or not stabilized for a long time (Figure S4). In contrast, the CAP paste forms and maintains a stable film on both the polyester and the waterproof polyester substrates with a TPU overlayer, even in the buffer solutions. During repetitive measurements, no delamination or cracks were observed. The resulting output potential is stable and reliable (Figure S4).
The results indicate that the two parameters, the surface hydrophobicity and hygroscopicity of the textile substrate, are important to choose the textile substrate for the CAP-based pH sensor. The more hydrophobic and less hygroscopic textile is more suitable as the substrate for the pH sensor. The TPU film helps to smoothen the surface of textile substrates typically having high surface roughness due to the woven structure. In this study, the waterproof polyester with the TPU film was used as the substrate of the pH sensor for reliable film formation and potential measurement.
Figure 3(a) shows the surface morphology of the CAP composite of the textile working electrode. The hairy threads are CNTs dispersed well in the agarose polymer matrix. To test the performance of the textile working electrode, we measured the potential using a two-electrode system composed of the textile working electrode and a Ag/AgCl commercial reference electrode. When the two electrodes were immersed in the five buffer solutions with different pH values, the potential between them over time was as shown in Figure 3(b). The potential decreases for the first few minutes, and then reaches equilibrium without significant change even after 1000 s. The CAP composite of the textile working electrode maintains the stable film after the potentiometric measurements in the various pH buffer solutions (Figure S5). To evaluate the potential response of the textile working electrode in a pH-varying environment, the sensor was immersed consecutively in buffer solutions with increasing pH values. Figure 3(c) shows the resulting potential changes of the textile working electrode. The potential shows step changes in response to change in pH values with a short response time. The potential response was also reversible when the pH of buffer solution decreased from 9 to 5. The reason for the faster equilibrium in Figure 3(c) compared with the result in Figure 3(b) is that the sensor in Figure 3(b) is initially in a dry state before being immersed in the buffer solutions and takes time to be hydrated, whereas the sensor in Figure 3(c) is exposed to a gradual pH change in the hydrated state. (a) SEM image of the surface of the CAP composite for the textile working electrode. (b, c) Potential of the textile working electrode with respect to a commercial Ag/AgCl reference electrode for different pH buffer solutions. (b) Time-dependent potential of the textile working electrodes in different pH buffer solutions. (c) Change of potential of the textile working electrode upon increasing pH conditions. (d) Potential responses at various pH levels between 5 and 9. The potential values are the average of the equilibrium potentials from three times of measurement. The red line represents the trendline of the data points.
Figure 3(d) shows the equilibrium potentials of the textile electrodes relative to the reference electrode as a function of pH. The average potentials show negligible standard deviations and the red trendline in the plot shows a linear response (r2 = 0.99055) with a sensitivity of 57.1 mV/pH. The linear potential change can be explained by the modified Nernst equation, which is written as
Each component of the CAP composite is essential for the pH-sensing ability of the textile working electrode. PANI and CNTs improve the sensitivity of the sensor due to amino functional groups and high electrical conductivity, respectively. As the pH increases, the electrical conductivity of PANI decreases due to the deprotonation of the nitrogen atoms in the polymer chain of PANI, resulting in the high sensitivity of the polymer as a pH sensor. 26 The agarose possibly contributes to an increase in the shape stability. To investigate systematically the effect of each component on the performance of the textile working electrode, composites without each component were prepared and their pH-dependent potentials were measured (Figure S6). First, without PANI, the potential response to the pH change tends to be more slowly stabilized. The dependence of the potential on pH change is also nonlinear. 27 For the composite without CNTs, little dependence of potential on pH is observed. Finally, without agarose, the sensitivity of the textile working electrode shows a stable sensing ability with a sensitivity of 51.4 mV/pH. However, the response time (i.e., the time for the sensor to equilibrate at each pH) is longer than for the sensor based on the CAP composite, and the sensitivity degrades by ≈18% after repetitive bending cycles of 1000 times (Figure S7). Moreover, agarose increases the viscosity of the CAP composite and allows the formation of the CAP paste, improving the processability of the composite.
The textile reference electrode
For the fabrication of a potentiometric pH sensor fully integrated on textile, the reference electrode, as well as the working electrode, should also be realized on the textile substrate. The textile reference electrode is fabricated by coating the Ag/AgCl paste and the solid electrolyte as shown in Figure 1. Ecoflex, a silicone elastomer, is utilized as the flexible matrix of the solid electrolyte where KCl salt is dispersed.
28
The SEM image in Figure 4(a) shows the surface morphology of the solid electrolyte of the textile reference electrode. The KCl salt forms approximately 15–20 μm granules and they are well dispersed in the Ecoflex matrix. The surface of the electrolyte does not show any noticeable cracks although it has many wrinkles, possibly due to the localized stress around the rigid salt granules. (a) SEM images of the solid electrolyte of the textile reference electrode. (b) Cyclic voltammetry (CV) analysis for the textile reference electrode with increasing scan rate from 5 to 200 mV/s. (c) Cathodic and anodic peak currents as a function of the square root of the scan rate in the CV measurements in (b). The squares (■) and circles (•) stand for the cathodic peak current and anodic peak current, respectively.
To characterize the properties and performance of the textile reference electrode, we conducted CV analysis for a model redox couple consisting of 1 mM potassium ferricyanide (K3Fe(CN)6) in 1 M potassium chloride (KCl) while varying the scan rate (Figure 4(b)). As the scan rate increases from 5 to 200 mV/s, the absolute values of cathodic and anodic peak currents increase. The peak currents of the cathodic and anodic reactions are the same and the potentials for the peak currents are constant regardless of the scan rate, which are the typical characteristics for a reversible electrochemical reaction. The other important characteristic for the reversible redox reaction is that the peak current values are proportional to (scan rate)1/2. Figure 4(c) shows the cathodic and anodic peak currents as a function of (scan rate)1/2. The linear dependence indicates the reversible electrochemical reactions, thereby confirming that the fabricated textile reference electrode works properly. 29
Integrated textile pH sensor
After the individual analysis of the textile working electrode and reference electrode, the fully textile-based pH sensor was fabricated by integrating the two textile electrodes on a piece of textile (Figure 5(a)). The textile pH sensor is highly flexible as shown in the inset of Figure 5(b). Figure 5(b) shows the dynamic potential response while switching the buffer solutions with increasing pH. The potential is stabilized upon pH changes within 1–2 min and shows a stable response. Figure 5(c) shows the static potential response according to the pH change in the range of pH 5–9. The trendline shows the linear relationship (r2 = 0.99641) between the potential and pH with a sensitivity of 45.9 mV/pH. Thus, the textile-based pH sensor shows stable and reliable performance for both static and dynamic pH conditions. (a) A schematic view of the textile-based pH sensor (b, c) Potential with respect to the textile reference electrode for different pH buffer solutions. (b) Sensor response at the pH of buffer solution increase from 5 to 9. (c) The potential response of the textile pH sensor to various pH levels between 5 and 9. The trendline in red shows a linear response (r2=0.99641) with an average sensitivity of 45.9 mV/pH.
The flexibility of the textile-based pH sensor was tested by measuring the pH-dependent potential of the sensor in the bent state and after repetitive bending. Figure 6(a) compares the sensitivity of the textile pH sensors in the normal and bent states. Compared with that in the normal state, the sensitivity of the sensor in the bent state slightly decreases by ≈7%. For application on a human body, given that the radius of curvature of body parts and joints would be generally larger than 2.5 mm, the sensor could maintain its performance well even under physical deformations that may occur. (a) pH-dependent potential response of the textile pH sensor under normal and bent state. In bent state, the sensor is wrapped around a glass rod with radius of curvature 2.5 mm as shown in the inset. (b) pH-dependent potential response of the textile pH sensor before and after 1000 cycles of bending. The radius of curvature of the bending is 4 mm. (c–d) SEM images of the textile pH sensor after 1000 bending cycles: (c) the CAP textile working electrode, (d) the textile-reference electrode. The arrows indicate the deep wrinkles in the Ecoflex matrix.
Comparison of textile-based pH sensors.

aNo result for systematic flexibility test was reported.
bTwo-terminal pH sensors with no reference electrode.
cNormalized current response value.
Textile-based pH sensor on porcine skin
One of the important objectives of the textile pH sensor is to detect the pH change of human skin. Skin is a wet solid surface and has biomaterials (i.e., sebum), which is a very different environment from a buffer solution. To simulate the pH-varying skin condition, we tested the textile pH sensor on porcine skin. Two pieces of porcine skin were immersed in two buffer solutions of pH 5 and 7 for 24 h. Excess pH buffer solution on the skin is removed before each measurement. The potential response of the textile pH sensor depending on the pH change of the porcine skin is shown in Figure 7. First, the skin of pH 5 was placed on the pH sensor to measure the potential. The potential of the porcine skin of pH 5 was 137 mV. When the porcine skin of pH 7 was placed on the pH sensor, the potential decreased by 73.9 mV. After the porcine skin of pH 5 was placed on the pH sensor again, the potential increased to 138 mV with a negligible hysteresis of 1 mV. For the porcine skin experiment, the sensitivity of the sensor on the skin is 37 mV/pH which is approximately 19% lower than the sensor in the buffer solutions. This result might be due to the presence of biomaterials on the skin, such as sebum and dermal interstitial fluid, which can affect the potential of the sensor during the measurements. Thus, the textile pH sensor could also effectively detect the pH changes on the skin. The potential response of the textile-based pH sensor on a piece of porcine skin with different pH. The surface pH of the skin switched between 5 and 7. Inset is the photo image of the porcine skin on the pH sensor.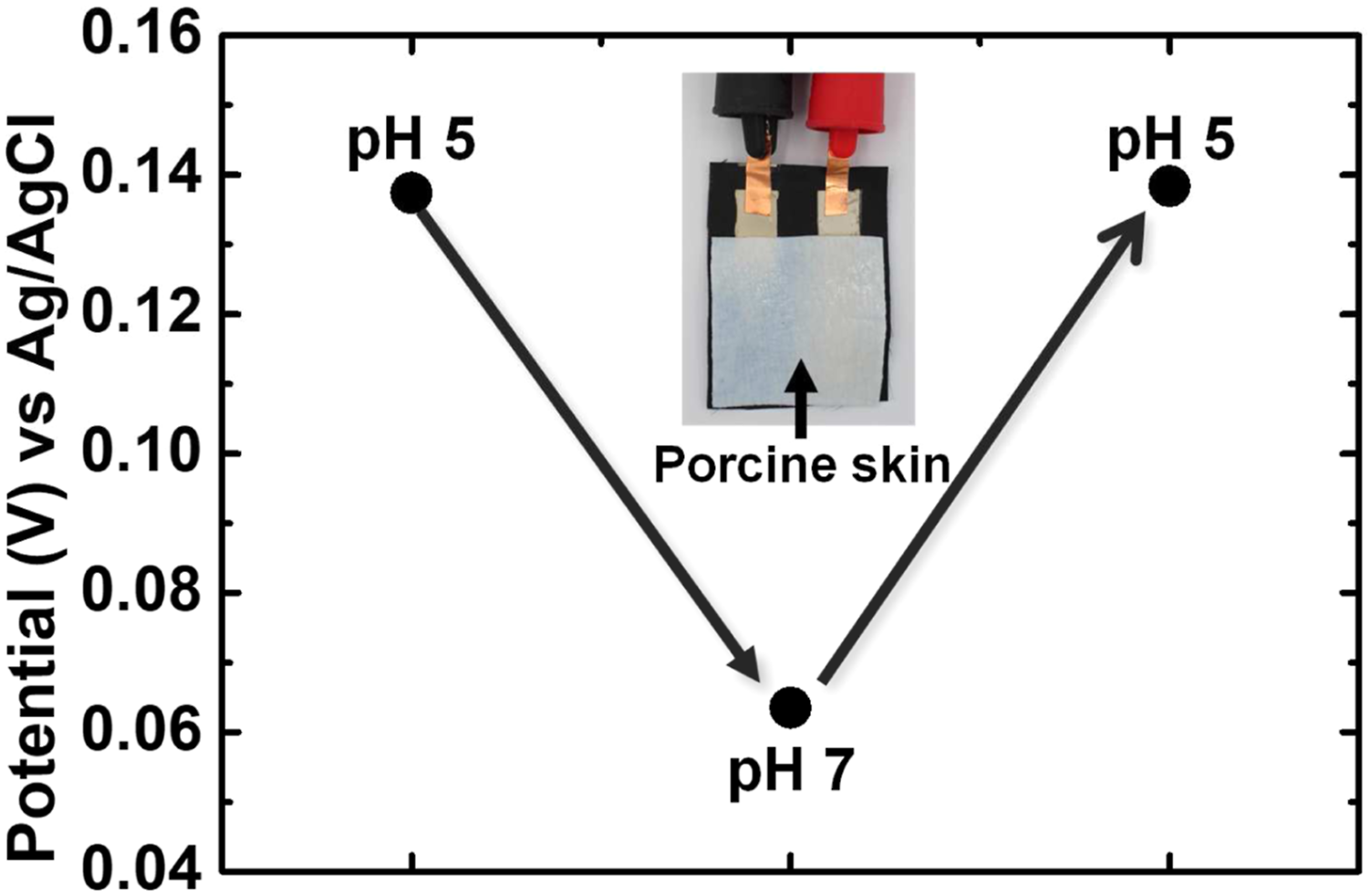
Conclusions
We developed a textile-based pH sensor with high flexibility and mechanical stability. The textile working electrode was prepared by printing the composite paste composed of PANI, CNTs, and agarose. To improve the film stability and obtain a reliable output signal, different types of fabric were compared as the textile substrates and a TPU overlayer was adopted. The textile reference electrode was prepared by printing Ag/AgCl paste and solid electrolyte. By integrating the textile working and reference electrodes, the fully textile-based potentiometric pH sensor was fabricated. The resulting sensor shows a linear dependence of potential on pH ranging from 5 to 9 with a sensitivity of 45.9 mV/pH. Because of its high flexibility, the textile sensor maintains its pH-sensing ability after repeated bending cycles. The pH sensor also shows reliable pH-dependent potential change on porcine skin with a sensitivity higher than 30 mV/pH. The textile-based pH sensor with good sensitivity, flexibility, and stability will be an important milestone in the continuous monitoring of skin conditions and diseases. Due to the advantages, the proposed pH sensor could also be used for remote care of patients when combined with a wireless communication system. Further study is currently underway to evaluate the biocompatibility and the adhesion of the textile sensor on the human skin and investigate the possibility of interference of the pH-dependent potential by sweat or other biomolecules.
Supplemental Material
sj-pdf-1-jit-10.1177_15280837211073361 – Supplemental Material for A fully textile-based skin pH sensor
Supplemental Material, sj-pdf-1-jit-10.1177_15280837211073361 for A fully textile-based skin pH sensor by Choi Minyoung, Minji Lee, Ji-Hye Kim, Sooyoung Kim, Jonghoon Choi, Ju-Hee So and Hyung-Jun Koo in Journal of Industrial Textiles
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
We gratefully acknowledge the support of this work provided by Basic Science Research Program (NRF-2021R1H1A1014396) and Nano-Material Technology Development Program (No. 2017M3A7B8061942) through the National Research Foundation of Korea funded by the Ministry of Science and ICT. This work has also been partially supported by the Korea Institute of Industrial Technology (Development of microfactory-based technology for future smartwear manufacturing, kitech EH220003).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

