Abstract
In order to facilitate the combination of composite phase change materials (CPCMs) and fabrics, granular CPCMs were prepared, a simple vacuum impregnation method was used to adsorb n-octadecane with disc-shaped diatomite as a carrier. To improve the adsorption capacity of n-octadecane on diatomite, the average pore size of its single point was increased from 13.35 nm to 25.54 nm through acidification. The microemulsion of n-octadecane liquid with particle size of less than 200 nm was immersed into the diatomite under vacuum conditions, and after demulsification, the particle type CPCMs with a particle size of about 30 μm was prepared. The stability, thermal properties of particle type CPCM, and its thermoregulation on textile were systematically studied. The particle type CPCM with the latent heat energy of 34.69 J/g revealed no phase leakage when heated for 30 min at 50°C. The impregnation rate of acidified diatomite was increased by 51.7% compared with original diatomite. The CPCMs mixed with polyurethane was coated on the surface of cotton fabric and evaluated the thermal properties of the fabric. The fabric was allowed to absorb heat at 40°C to reach saturation. Analysis showed that the heat storage performance of the fabric was increased by 7.9%, and the temperature difference was adjusted to 2.9–3.1°C, which effectively slowed down thermoregulatory in a relatively low temperature environment. Moreover, the coated particle type CPCM did not alter the coldness of the cotton fabric.
Keywords
Introduction
The human skin plays a key role in keeping the body warm; however, it is very sensitive to cold and must be covered by an additional layer through clothing. 1 In addition to protecting and decorating the human body, the primary function of clothing is to insulate against cold and keep the body warm. Traditional clothing achieves thermal insulation by protecting the human body from cold and reducing convection. 2 However, traditional clothing cannot be adjusted automatically to variations in external temperatures. Smart textiles have increasingly emerged with the development of science and technology. The textiles can respond to environmental stimulation and achieve dynamic temperature control in the form of energy storage and release. 3
PCMs are recognized as one of the most effective materials for energy storage and release. 4 The energy is stored in the form of latent heat and released based on temperature variations. Temperature regulation is achieved through the storage and release of thermal energy. 5 The energy storage characteristics of PCMs can be used to improve the thermal properties of fabrics and subsequently prepare thermoregulatory fabrics. 6 Organic PCMs are more suitable for the preparation of thermoregulatory fabrics when compared with inorganic PCMs due to their properties, such as corrosion-resistance, reusability, relatively low cost, limited or zero subcooling, and high latent heat. 7
Alkane-based organic PCMs are essential organic PCMs with high latent heat, a wide melting point range and thermal stability, are non-corrosive, non-toxic, and have stable chemical properties. 8 Straight-chain alkanes composed of normal alkanes (CH3-(CH2)n-CH3) are widely used as PCMs. The melting temperatures of such alkanes fall within the temperature range that is comfortable for the human body, which makes them suitable for the maintenance of the thermal comfort of textiles.9,10 However, the preparation process of CPCM based on PCMs generally involves solid-liquid phase changes, which lead to poor stability and easy leakage, in turn making them unsuitable for direct application in textiles. 11
The most widespread method for preparing CPCM is impregnation. 12 Large gaps exist between the multi-layer structure of expanded graphite, which provides adsorption space for PCMs. The porous structure cannot completely fix PCMs through capillary action and surface tension, especially at high loading rates. 13 Moreover, the three-dimensional reticular-structured material with a rich porous structure and large specific surface area allows adsorption onto pores without leakage during the conversion of PCMs from solid to liquid state. 14
As a type of natural amorphous mineral, diatomite with a porous structure and low thermal conductivity is predominantly composed of fossilized skeletons called diatoms. 15 Diatomite, which is widely used as a filler and adsorbent, is abundant and available at low prices in China. 16 Diatomite exhibits unique properties such as high porosity, high permeability, large specific surface area, and chemical inertness. 17 The surfaces and pores of diatomite are covered with high amounts of magnesium oxide (MgO), calcium oxide (CaO), and other minerals. Therefore, certain pretreatments are often used to enhance porosity. A previous study revealed that heating diatomite in a microwave made the particles to expand and increased pore sizes, in addition to increasing its loading rate of lauric acid by 83.22%. 18 Acidification can remove impurities from diatomite but requires a longer processing time. The microwave heating method can shorten the pretreatment time to a few minutes, and effectively remove impurities such as MgO, CaO, and phosphorus oxide (P2O3). The loading rate of lauric acid-stearic acid was increased by 36.68% when compared with that of untreated diatomite. 19
PCMs used to produce thermoregulatory fabrics should comprise linear alkanes with melting temperatures that are close to the human body temperature (approximately 30°C) to enhance the thermal comfort of textiles. N-octadecane, which has a melting point of 28°C and latent heat of 242 J/g, is often used in the preparation of CPCM. Li et al. prepared titanium dioxide (TiO2) powder with high specific surface area and mesoporous structure, which was more conducive to preventing leakage of n-octadecane. However, to successfully impregnate n-octadecane into the micropores of TiO2, n-octadecane is initially dissolved in petroleum ether and then impregnated into TiO2 micropores through a vacuum due to the small pore sizes of n-octadecane. 20 Zhang et al. used polystyrene instead of porous materials to adsorb n-octadecane, which was also initially mixed with toluene as a solvent. Finally, toluene was removed by drying and n-octadecane was coated with cured polystyrene to form solid CPCM. The full covered structure resolves the challenge of phase leakage that is associated with n-octadecane. 21
In this project, we used a brand-new method to prepare the particle type CPCM. Compared with the traditional microcapsule preparation process, the preparation method of the particle type CPCM is simple, can be produced on a large scale, and is well compatible with waterborne polyurethane. We used the vacuum impregnation method to impregnate n-octadecane into diatomite and prepare the particle type CPCM, which were further applied to thermoregulatory fabrics. Impregnating n-octadecane into diatomite pores is challenging due to its high molecular weight and low affinity for diatomite. A new simple and easy-to-operate combination method has been used, specifically, vacuum impregnation of microemulsions in which the emulsion is broken down to prepare CPCM. To begin with, sorbitan oleate and polyoxyethylene sorbitan fatty acid ester are used as emulsifiers to convert n-octadecane into an oil-in-water microemulsion, followed by impregnation of the microemulsion into diatomite pores under a negative pressure condition of −0.1 kPa. Finally, ethanol is added to break down the microemulsion and remove the emulsifier. Afterward, n-octadecane is firmly adsorbed onto diatomite pores to produce the particle type CPCM. A waterborne polyurethane (WPU) characterized by low temperature flexibility, pH stability, water resistance, excellent solvent resistance, and excellent weather resistance properties is used as an adhesive. 22 The WPU is mixed with powdered CPCM and subsequently used to coat cotton, which is in turn, used to manufacture functional fabrics with thermoregulatory effect.
Materials and methods
Materials and reagents
Cotton fabrics (yarn count (75D), warp yarn per inch and weft yarn per inch: 130×70, GSM: 293.77 g/m2) were obtained from Hebei Ningfang Group Co., Ltd, (Xingtai, Hebei, China). WPUs (analytical reagent grade, solid content:60%, [AR]) were purchased from Macklin (Shanghai, China). Diatomite (CP) was purchased from Dahua Diatomite Co., Ltd, (Jilin, China). Sorbitan oleate (Span 80), polyoxyethylene sorbitan fatty acid ester (Tween 80), concentrated hydrochloric acid (HCl, 38%), and absolute ethanol (EtOH, 98%) were all AR reagents.
Diatomite purification
Diluted HCl with a concentration of 16% w/v was prepared. Diatomite and diluted HCl were mixed uniformly at a solid-to-liquid ratio of 1.0 g: 6.0 mL and then allowed to stand for 1 h. The mixture was heated to 100°C at 360 r/min with occasional agitation and heated continuously for 30 min. Subsequently, the mixture was cooled to room temperature (25°C), filtered with suction, washed using deionized water to neutrality, oven-dried at 80°C for 6 h to obtain diatomite after removing the acid, which was named
Preparation of n-octadecane microemulsion
Span 80 and Tween 80 were mixed to a certain proportion and stirred for 20 min at a speed of 200 r/min. After heating to 35°C, the molten state of n-octadecane was added to the mixture and stirred for 10 min to form a uniform oil phase. Deionized water with a temperature of 35°C was added gradually into the oil phase. The stirring speed was increased to 350 r/min after dripping was complete and stirring continued for 30 min to form the n-octadecane microemulsion.
Preparation of composite phase change materials
As shown in Figure 1, the n-octadecane microemulsion with the smallest particle size and superior stability was proportionally mixed with Dh. The mixture was added to the suction flask, sealed, evacuated, and stirred at a speed of 120 r/min for 40 min under a negative pressure of −0.1 kPa. The vacuum was released and anhydrous ethanol twice the volume of the microemulsion was added. Stirring continued for another 5 min to demulsify the emulsion. The mixture was then drained and rinsed three times in water (35°C) to remove the emulsifier and excess n-octadecane. The CPCM was kept at room temperature (25°C) for 8 h to dry. A flow chart illustrating the preparation process of composite phase change material using microemulsion vacuum impregnation method.
Preparation of thermoregulatory fabric
WPU (2.0 g) and CPCM (0.4 g, 0.6 g) were mixed. The mixture was stirred at a speed of 100 r/min for 20 min and then left to stand for 2 min to eliminate bubbles in the liquid used for coating. The front surface of a 5×5 cm cotton fabric was evenly coated with the mixed liquid and heated at 110°C for 6 min to make a thermoregulatory fabric.
Characterization
Testing of basic characterization
The X-ray powder diffraction (XRD) pattern of samples was collected by X-ray diffractometer (D/Max-2500v/pc; Rigaku, Tokyo, Japan); The specific surface area of diatomite was tested by a specific surface area analyzer (Kubo1200, Bjbiaode, China) with nitrogen (N2) adsorption-desorption isotherm. The chemical compositions of samples were analyzed using a Fourier-transform infrared spectrometer (FTIR; Thermo Scientific Nicolet 6700, Madison, WI, USA). Latent heat and melting points of samples were obtained using a differential thermal analyzer (DSC-214, NETZSCH, Germany); The thermal conductivity was obtained by thermal constants analyzer (Hot Disk TPS 2500S, Gothenburg, Sweden). The electronic fabric strength machine (YG026SJ, DaRong, China) was used to test the breaking strength and breaking elongation of the fabric.
Performance testing of composite phase change materials
The phase leakage of the CPCM was tested by heating at 50°C for 30 min; The impregnation rate of n-octadecane in CPCM was calculated using equation (1) as follows
Thermal performance testing of coated fabric
Step cooling curve test, photothermal conversion performance test and assessment of thermal stability test of coated fabric was tested using an infrared thermal imager (TiS10, Fluke Corporation, Everett, WA, USA); Heat retention capacity analysis was collected by the thermal insulation instrument (YG606 L, Yuan More, China); Contact warm-cool feeling analysis was collected by the warm-cool feeling tester (KES-F7 II, Kador International Industrial Limited, Japan); Solar thermal energy storage efficiency of the thermoregulatory fabric under simulated solar radiation conditions using an iodine tungsten lamp was calculated using equation (2) as follows
23
Results and discussion
The morphology of diatomite
Pristine diatomite was disk-shaped with numerous cavities on its surface (Figure 2(a)). Moreover, most of the pores on the diatomite surface were blocked or small. The coverings on the pores of diatomite surface fell off and the blocked pores were opened following treatment with 16% diluted HCl (Figure 2(b)). The pores had similar diameters and were evenly distributed. The impurities at the edge of diatomite surface were removed (Figure 2(c) and (d)). Although there was no complete pore structure, numerous grooves were observed, which could further increase diatomite porosity. (a) SEM images of diatomite at a magnification of ×2000; (b) SEM images of 
The nitrogen (N2) adsorption–desorption isotherm curves of diatomite and (a) Nitrogen adsorption curves of specific surface areas of diatomite and 
X-Ray diffraction (XRD) analyses were performed on diatomite and
Performance of n-octadecane microemulsion
The mass ratios of components in the microemulsion.

As shown in Figure 4, transparency of the microemulsions gradually increases from left to right. The stability of samples 1 and 2 is the worst after standing and stratified when standing for 2 h, and the degree of stratification increased with the extension of time. After 14 h, samples 3 and 4 did not show obvious stratification, but a layer of milky white oil precipitated on the surface of the microemulsion. In addition, the other samples showed good stability, the appearance of the microemulsion did not change during the standing process. It can be concluded that the stability and transparency of the emulsification gradually enhanced as the amount of Tween-80 increased. Comparison of stability of n-octadecane microemulsion.
The particle size analysis of samples (NO. 5–12) are shown in Figure 5. As shown in Figure 5(a), the particle size of samples (NO. 5–9) gradually decreased, but still in the range of the 10–200 nm, which meets the definition of microemulsion.
29
However, as the amount of Tween-80 increased, the particle size of samples (NO. 10–12) improved, which was caused by the hydrophilicity of Tween-80. When the amount was excessive, a lot of water molecules will be gathered, causing the increase of particle size. Although there was little difference between the particle size of samples 5 to 7. It was found that the particle size distribution of sample 7 is the most uniform and was used for preparation of CPCM. N-octadecane microemulsion: (a) median particle size analysis diagram; (b), (c), (d) particle size distribution diagram.
Shape stability of composite phase change material
The phase leakage needs to be solved when CPCM is applied in textile. The samples in Figure 6 from left to right are (i) CPCM prepared by 0.8 g Dh and 2.0 g microemulsion (Dh/ME 1); (ii) CPCM prepared by 0.7 g Dh and 3.0 g microemulsion (Dh/ME 2); (iii) CPCM prepared by 0.6 g Dh and 4.0 g microemulsion (Dh/ME 3); (iv) CPCM prepared with 0.5 g Dh and 5.0 g microemulsion (Dh/ME 4). After heated at 50°C for 30 min, the Dh/ME 4 phase leaked. Currently, the ratio of Dh to microemulsion was 0.5:5.0, and it was determined that the maximum ratio of composite phase change material thermal stability after heated at 50°C for 30 min.
With the optimal ratio of diatomite to microemulsion of 0.6:4.0, other CPCM were prepared according to the 2.4 experimental method. As shown in Figure 6, the samples from left to right are (i) CPCM prepared by 0.6 g diatomite and 4.0 g microemulsion (diatomite/ME); (ii) CPCM prepared by 0.6 g Dh and 4.0 g microemulsion without demulsification (Dh/ME -no break); (iii) CPCM prepared from 0.6 g
It can be seen from Figure 6 that the diatomite/ME and Dh/ME-no break also maintained excellent thermal stability without phase leakage, while Dh/octadecane had a serious leakage, which indicated the impregnation effect was indeed improved by the combination of microemulsion vacuum impregnation and demulsification, and further solved the phase leakage.
The samples with excellent dimensional stability were selected for DSC test. As show in Figure 7, as the proportion of microemulsion increased, the phase transition interval in CPCM gradually widen and there is no peak in the Dh/ME-no break DSC curve. The microemulsion was removed completely after washing because the CPCM does not show latent heat properties. Latent heat energy analysis diagram: (a) CPCM heating DSC curve; (b) CPCM cooling DSC curve.
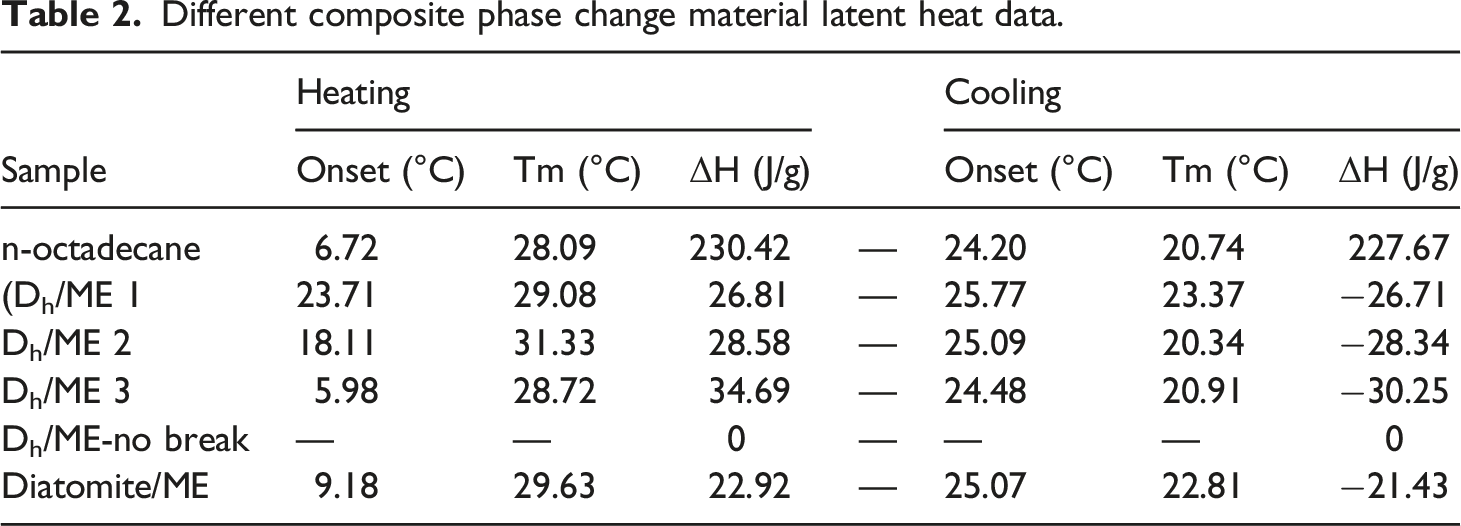
Different composite phase change material latent heat data.
Heat transfer data of Dh and composite phase change material.
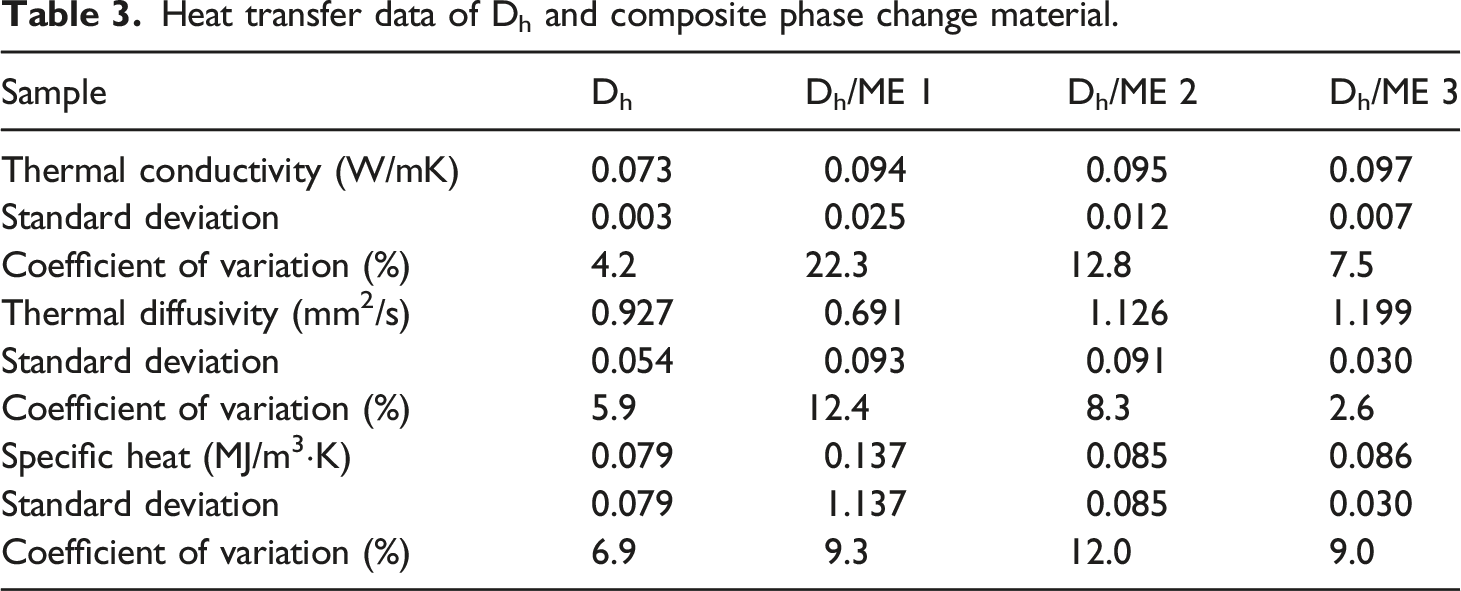
In summary, Dh/ME 3 exhibited superior overall performance among the CPCM analyzed. equation (1) was used to calculate the impregnation rate of n-octadecane, and the results revealed that the impregnation rate of Dh/ME 3 was 22.0%, while that of diatomite/ME was 14.5%. The impregnation rate of Dh after acid treatment increased by 51.7% relative to diatomite.
The XRD and FTIR spectra of n-octadecane and CPCM are illustrated in Figure 8. The diffraction peak of (a) XRD spectra of CPCM and its components; (b) FTIR spectra of CPCM and its components.
The characteristic peaks of
Mechanical and thermal properties of thermoregulatory fabric
The thermoregulatory performance of CPCM was analyzed using three functional fabrics prepared as follows: (i) b was cotton fabric coated with WPU (c+WPU); (ii) c was cotton fabric coated with 20% CPCM and WPU (c+CPCM 20%); (iii) d was cotton fabric coated with 30% CPCM and WPU (c+CPCM 30%) (Figure 9). Photos of cotton fabrics and coated fabrics.
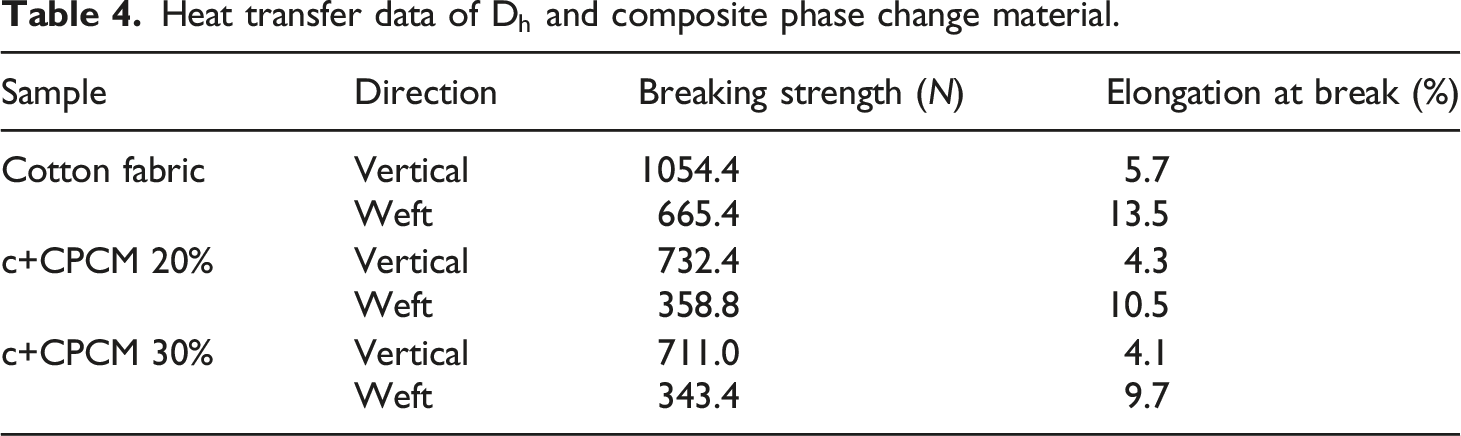
Heat transfer data of
The cooling curve of each sample was tested, and the curves are illustrated in Figure 10. After heating the fabrics at a constant temperature of 40°C, the cotton fabric exhibited almost no heat storage effect. Covering the surface of cotton fabrics with WPU coating reduced the cooling trend of the fabrics. The temperature gradually dropped from an initial 35.8°C to 21°C (Figure 10(b)). A comparison of the infrared thermal imaging results of the four samples revealed that the heat storage capacities of pure cotton fabric and c+WPU were not uniform. Furthermore, the temperature of fabric coated reached 39.6°C after constant heating at 40°C when 30% CPCM was added, which was 2.9°C higher than that of pure cotton fabric. During the cooling process, initial cooling was rapid. However, when the temperature dropped to approximately 28°C, CPCM started to undergo a phase transition process, followed by a decrease in its cooling rate. After cooling for 420 s, the temperature remained constant at 23°C, which was still higher than the 21°C. After the energy storage was saturated, the temperature of the coated cotton fabric with CPCM added was 2.1–6.2°C higher than the cotton fabric during the 450s cooling process. A comparison of step cooling curves (room temperature: 21°C) of various fabrics.
The photothermal conversion effect of samples was analyzed by simulating photothermal environment using an iodine tungsten lamp. A comparison of the heating curves of each sample (Figure 11) revealed that the heating rates of the cotton fabric and c+WPU were basically the same. The temperature increased rapidly to approximately 30°C and then gradually increased to approximately 36°C under irradiation conditions using iodine tungsten lamp, which suggests that the WPU coating did not influence photothermal conversion. Contrary to the heating curves illustrated in Figure 10 (c) and (d), n-octadecane in CPCM began changing from solid to liquid state after the temperature increased rapidly to 28°C, which reduced the heating rate. Moreover, the heating rate of c+30% CPCM was lower than that of c+20% CPCM when the amount of CPCM added to the coated fabrics was increased. The final temperature increased from 38.5°C to 39.8°C, which suggests that the photothermal conversion and temperature adjustment effects of the coated fabrics can gradually increase with an increase in the amount of CPCM. Comparison of photothermal energy storage (room temperature: 21.9°C).
Finally, c+30% CPCM was subjected to continuous temperature increase tests of 360 s and temperature drop tests of 360 s up to four cycles. The results revealed that the heating and cooling curves of the samples were basically the same. However, after each thermal cycle, the initial and final temperatures of the samples increased gradually (Figure 11(e)). The observation could be because the iodine tungsten lamp emitted bright light and more heat. As the sample was irradiated by the iodine tungsten lamp for a long time, the ambient temperature increased gradually. The phenomenon did not affect temperature increase in c+30% CPCM, which suggests that the sample exhibited excellent stability.
The photothermal conversion efficiency of c+30% CPCM was calculated using equation (2) in Thermal performance testing of coated fabric. The mass of Dh/ME 3 in the coating was 0.6 g, latent heat was 34.69 J/g, the power density of the iodine tungsten lamp was 384.62 W/m2, and the coating area of the thermoregulatory fabric was 0.0025 m2. When the thermoregulatory fabric was exposed to light for 195 s, the heating rate decreased significantly, and heat storage was saturated (Figure 11(d)). The phase change of c+30% CPCM occurred after exposure to light for 35 s, and the duration of the entire energy storage process was 160 s. The data were subsequently substituted into equation (2). The calculated photothermal conversion efficiency of c+30% CPCM was 13.5%.
The thermal insulation capacities of the fabrics were comprehensively evaluated, and the results were satisfactory. The thermal insulation rate of each sample (Figure 12(a)) was inversely proportional to the heat transfer coefficient (Figure 12(a)). The heat transfer coefficient of pure cotton fabric was 153.4 and the thermal insulation rate was 5.746%. Air permeability decreased after the cotton fabric surfaces were coated with WPU, the heat transfer coefficient was 120.1 and the thermal insulation rate increased 34.7% was 7.741%. The thermal insulation rate of c+30% CPCM increased considerably, and the heat transfer coefficient reduced to 50.74 since the c+30% CPCM surface was coated with CPCM. The thermal insulation rate of c+30% CPCM increased by 171.1% when compared with that of the cotton fabric, which increased by 15.580%. (a) Heat retention capacity analyses results; (b) A comparison of cold-hot sensation in various fabrics and their Crow values.
Cold-hot sensation of fabric and the Crow value are important indicators for evaluating the heat preservation property of a fabric. As shown in Figure 12(b), the Crow value of cotton fabric was 0.042, which increased to 0.058 after the surface was coated with water-based polyurethane. Because of the thermal insulation effect of CPCM, the Crow value of c+CPCM 30% was increased by 235.7% to 0.141. Cold-hot sensation tests of the three samples were compared. Results showed that the Q-max of cotton fabric was 0.172 W/cm2, and that of c+WPU was 0.165 W/cm2. The coolness of the fabric was not improved by the water-based polyurethane. Although the heat storage effect of the CPCM coating caused a surface of c+CPCM 30%, the Q-max was only increased by 6.4% to 0.183 W/cm2. Although thermoregulatory fabric has excellent thermal insulation properties, it has no significant effect on the coldness of the fabric.
Conclusion
The microemulsion vacuum impregnation method was used to successfully prepare the particle type CPCM with a particle size of about 30 μm and excellent stability. The research results show that the encapsulation efficiency and thermal performance of the particle type CPCM are influenced by diatomite pretreatment and the amount of impregnation of the microemulsion. After acid purification, the porosity of diatomite increased significantly. In addition, impregnation rate of the n-octadecane in diatomite increased from 14.5% to 22% after acid purification, and there was no phase leakage problem. Finally, a thermoregulatory fabric was successfully prepared by coating the mixture of particle type CPCM and water-based polyurethane on the surface of the cotton fabric. Compared with cotton fabrics, the photothermal performance of fabrics coated with 30% CPCM improved significantly, but the breaking strength of the fabric is reduced by 32.6%–48.4%. The range of the thermal regulation temperature was 2.9–3.1°C, and its photothermal conversion efficiency of 13.5% effectively slowed down changes in temperature. The coated fabric showed excellent thermal stability during test cycles of heating and cooling. It also exhibited excellent thermal insulation, with a thermal insulation rate of 15.58%. In summary, the particle type CPCM prepared in this article can be applied to the field of thermal regulation management.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Funding of Hebei Education Department (Grant No. ZD2020112), Key Research and Development Program of Hebei Province (Grant No. 20374004D) and Hebei Province Graduate Student Innovation Funding Project (Grant No. CXZZSS2021097).


