Abstract
The aim of this study was to fabricate cotton and wool fabrics with both of thermoregulation and thermochromic properties. For this aim, three-component thermochromic systems (TCTS) were microencapsulated and applied to the pure cotton and wool textile fabrics. In the study, TCTSs consisting of crystal violet lactone (CVL), bisphenol A (BPA), and myristyl alcohol were microencapsulated in shell of poly(methyl methacrylate-co-methacrylic acid) (P(MMA-co-MAA)) by emulsion polymerization method. Microcapsule wall structures were designed to exhibit affinity for cotton and wool fibres under appropriate reaction conditions. Microcapsules having spherical shape, compact morphology and average size of 23.24 µm were prepared successfully. These microparticles exhibited latent heat energy storage capacity of 197.7 J/g and considerable thermal stability required for textile application process conditions. P(MMA-co-MAA)/TCTS microcapsules were applied to the cotton and wool textiles by exhaustion method. In the method, a substrate takes the chemicals in the bath and the affinity between the chemicals and substrate molecules is determinant on exhaustion process. The structural, colorimetric and thermal characteristics of the microcapsule applied fabrics were analyzed. The fabrics had reversible color change with a function of temperature. Their colors were blue at room temperature and original color of the fabric at temperatures above activation point of the thermochromic system. Additionally, the cotton fabric stored the latent heat of 23.4 J/g while the wool fabric stored the latent heat of 62.1 J/g. They exhibited thermoregulation property resulting from the absorbed latent heat.
Keywords
Introduction
Smart materials are materials that react to stimulants in their environment. They can detect and respond to environmental warnings. They have been gaining a lot of interest because of they are the basis of many applications, including sensors and actuators [1]. There are a number of types of smart material, of which are already common. Smart materials can be exemplified as phase change materials, shape memory polymers and chromic materials. Recently, textile scientists have been focused on developing smart textiles which not only exhibited general needs such as basic protection and covering, but also could act as a sensor or signal detector. Smart clothing based on fabrics or fibres having dynamic thermoregulation function have being used to keep body temperature, especially in situations of sudden temperature changes. Chromic materials respond to the stimuli by showing reversible changes in their colors. They are classified as thermochromic, photochromic, piezochromic and electrochromic according to temperature, light, pressure, electric current etc. stimulus [2].
Among the chromic dyes, photo and thermo stimulated chromic dyes changing their color rapidly and reversibly according to changes in light and temperature changes are the most common ones used in textile field [3,4]. Thermochromic colorants have been also used in chameleon-type building coatings, thermographic recording materials, strip thermometers, aerospace, medical or food technology products, military, engineering science, thermal mapping and cosmetic industry (nail polish and hair dyes) [5,6]. Thermochromic dyes offer important potential to textile producers for manufacturing of nice aesthetic and functional textiles. Thermochromic textiles can change their color reversibly from colorless to colored state when they activate by change in temperature. Thermochromic textiles can be designed to be used as a high added value products in various applications including medical textiles, flexible communicative display for clothing, sportswear [6].
Many research centers and commercial organizations have focused on formulation and textile applications of the thermochromic colorants. Scientists and technology experts in the smart apparel industry make researches for making thermochromic technologies popular for end users and helping to reduce costs [7]. In the last few decades, many research reports and patents have been published on the chemistry, mechanisms and applications of thermochromic materials. Today, thermochromic materials are eventually attracting considerable interest in textile industry [8–10].
Temrochromic materials that can be used in the textile field are leuco dye based and liquid crystal type materials [8,9,11,12]. Commercially available thermochromic dyes are leuco dye based three-component thermochromic systems which generally consist of a colorant (leuco dye in general), a color-developer and a non-volatile solvent. The system, which is colored in solid form, loses its color as the temperature increases and returns to its colored state when the temperature decreases again. Color former which is a component of the system, is a pH-sensitive compound that receives proton to create the colored merocyanine form via openning of the ring structure of leuco dye. Leuco dyes switch between a colourless spiropyran (SP) form and a deeply coloured merocyanine (MC) form. The ring-opened structure of crystal violet lactone dye is called the merocyanine (MC) form and is intensely blue/violet in colour. The open-ring coloured merocyanine form transform to closed ring which indicates spiropyran (SP) form when the temperature rises. The colorants are generally crystal violet lactone, fluoran dyes, diphenylmethane components or spiropyran components [8]. The most commonly used colorants were fluorescent dyes and crystal violet lactone analogues which were type of diarylphthalide components [8,13]. The color developer, another component of the system, gives the proton necessary for the leuco dye to be colored. The function of the solvent in the system is to present the appropriate medium for creating interaction between the system components namely colorant and the color developer. The non-volatile solvent controls activation temperature at which the system changes its color. The melting point of the thermochromic system corresponds to temperature which initiates discoloration of the system. The solvent is also a phase change material which has latent heat storing/releasing property. It can absorbs and stores the latent heat during its melting and releases the latent heat during its crystallization. The long aliphatic chain fatty acids, amides or alcohols are used as solvent. When the temperature is below the solidification point of the solvent, a strong interaction between the components of the system namely colorant and the color developer occurs, which makes the system colorful. On the contrary, while the system is heated to temperature above the melting temperature of the solvent, the interaction between the colorant and the color developer ends and it causes to lose color of the system [8,10,14]. Thermochromic materials have to encapsulate in a transparent shell before application to textiles. In this way, the components are protected against undesirable environmental impacts and the application areas of these materials are expanded [9].
In the literature, there are studies on microencapsulation of thermochromic materials with different methods. However, the investigation reported on application to textiles of microencapsulated thermochromic materials are limited. Ma et al. produced the thermochromic microcapsules with urea formaldehyde shell by emulsion polymerization method for usage wall coverings of buildings [15]. Li et al. encapsulated the thermochromic system consisting of CVL, BPA and myristyl alcohol in poly(methyl methacrylate) (PMMA) nanofibers [16]. Wei et al. encapsulated the thermochromic materials into gelatin-arabic gum and urea-formaldehyde shells by complex coacervation method [17]. Wang et al. sythesized the microcapsules which having both of thermochromic and thermoregulation properties using suspension polymerization method. The thermochromic pigments were dispersed in n-octadecane phase change material then microencapsulated into PMMA shell [18]. Geng et al. produced microcapsules exhibiting color change as a function of temperature besides storing thermal energy. The shell material of microcapsules which was sythesized via in-situ polymerization method were melamine formaldehyde and methylated melamine formaldehyde [19,20].
In this study, it was aimed to fabricate double function cotton and wool fabrics using microcapsules having both of thermochromic and thermal energy storage system (TES) properties. The aim was to develop textile fabrics to be used as flexible TES materials and visual temperature indicators. For this, reversibly color changed three-component thermochromic system (TCTS) was prepared. The system was microencapsulated in poly(methyl methacrylate-co-methacrylic acid) (P(MMA-co-MAA)) copolymer. MMA monomer was copolymerized with MAA monomer in order to create functional groups on outer surfaces of the microcapsules offering opportunity to form interaction between their shell polymer molecules and fibres molecules. P(MMA-co-MAA) microcapsules were applied to pure wool and cotton textile fabrics by exhaustion process. In an exhaustion principle, a substrate takes the chemicals in the bath at a certain temperature during a certain time period till the equilibrium with a solution containing the chemicals. In the application method of the microcapsules by exhaustion principle to the fabrics, the affinity between the shell polymer molecules of the microcapsules and fibre molecules is determinant. According to the literature survey, microcapsules have been usually applied to the fabrics by pad-dry-cure and coating methods because of the lack of the afinity between the fibre molecules and shell material of the microcapsules. In this study, P(MMA-co-MAA)/TCTS microcapsules were able to applied the textile fabrics by exhaustion method because of their reactive shell exhibiting affinity to the cotton cellulose and wool ceratin molecules. Besides, in the study, cotton fabric was cationized before microcapsule application to increase its electrostatic interaction with anionic microcapsule shell resulting from existence of anionic methacrylic acid monomer. In the study, the morphologies, chemical structures, thermal properties, as well as thermochromic performances of the produced microcapsules were characterized. Furthermore, thermochromic, TES and thermoregulating properties of the P(MMA-co-MAA) microcapsule applied pure cotton and wool textile fabrics including the latent heat storage/releasing, thermo-regulating and color changing as a function of temperature were investigated.
Experimental
Materials
Scoured and bleached pure cotton and pure wool fabrics were used for microcapsule application and their specifications are summarized in Tables 1 and 2, respectively. Setamordant T was supplied from Setaş Colorcenter Company. Fixapret Resin F-Eco and magnesium chloride was supplied from BASF Corporation. Sodium hydroxide (NaOH) and Triton X-100 were used together with the cationizing agent during the process and purchased from Sigma Aldrich Company.
Specifications of the cotton fabric used for microcapsule application.

Specifications of the wool fabric used for microcapsule application.

Setamordant T was used to insert cationic groups on the surface of cellulosic fibres. To bind P(MMA-co-MAA)/TCTS microcapsules to pure cotton and wool textile fabrics, Fixapret Resin F-Eco was used as received. Magnesium chloride was employed as catalyst. In the application of P(MMA-co-MAA)/TCTS microcapsules to pure cotton textile, sodium chloride was used to be able to increase the affinity of microcapsules to the fabrics.
Method
Preparation of the microcapsules
P(MMA-co-MAA)/TCTS microcapsules were produced by emulsion polymerization method as in our previous study. In our previous study, we produced P(MMA-co-MAA)/TCTS microcapsules with shell/core ratios of 1/1, 2/1 and 4/1. The characterization results of the produced microcapsules showed that microcapsules with thinner shell structure made the colour change more visible. Additionally, microcapsules which produced at low shell/core ratio had higher heat storage capacity [21]. According to this finding, in this study, shell/core ratio of developed thermochromic microcapsules was chosen as 0.5/1 and investigated convenience of their properties for fabric applications. The aim was to develop thermochromic effect offering high color contrast and maximum energy storage capacity in the fabrics. The thermochromic system consisting of CVL, BPA and myristyl alcohol was used as core material. The weight ratio of the CVL, BPA, and myristyl alcohol was selected as 1/4/70, respectively [21].
Cationization of pure cotton textile
The cationization of the cotton fabric was performed in order to increase the electrostatic interaction of capsules to the fabric as a result of the formation of cationic groups on the fabric surface. The bath consisting of a solution of NaOH (50 g/l), Setamordant T (150 g/l) and Triton X100 (3 g/l) was prepared. The fabrics were immersed in the bath and the bath was exhausted at 80 °C for 40 minutes. After acidification with aqueous solution containing 2 g/l of acetic acid, it was washed with a bath containing 3 g/l of phosphate free detergent. Then, pre-treated fabric was rinsed with sufficient amount of cold water and dried till constant weight at room temperature [22].
Microcapsule application to the fabrics by bath exhaustion method
The thermochromic microcapsules were applied to the fabrics using bath exhaustion method. Firstly, P(MMA-co-MAA)/TCTS microcapsules were emulsified in distilled water containing cross-linker and catalyst using a mechanical homogenizer (IKA Ultra Turrax T25) at a speed of 10,000 rpm for 1 hour. Then, prepared microcapsule solution was homogenized for 1 hour by ultrasonic homogenizer (Bandaline Sonuplus UV 2200, 40 W) (14% amplitude, 1 s pulse on, 1 s pulse off). Microcapsule application conditions were given in Table 3. The fabrics were immersed in the prepared microcapsule bath and the bath was exhausted for 1 hour at 60 °C. Microcapsule imparted fabrics were dried at around 80 °C for 5 minutes. The cotton fabric was cured at 120 °C for 1 minute. The wool was cured at 100 °C for 1 minute [22].
P(MMA-co-MAA)/TCTS microcapsule application conditions.

Characterization
Characterization of microcapsules
To determine the morphology of the produced microcapsules and the distribution and settlement of microcapsules on fabrics structure, scanning electron microscope (SEM, LEO 440 Computer Controlled Digital) analysis was carried out. The particle size of P(MMA-co-MAA)/TCTS microcapsules was measured using a particle size distribution analyzer instrument (Malvern MS2000E). To characterize the chemical structure of microcapsules, the spectroscopic analyses were performed by Fourier Transform Infrared (FT-IR, Perkin Elmer Spectrum BX) spectroscopy. Thermochromic properties of the produced microcapsules were examined by an Ultraviolet-Visible-Near-infrared spectrophotometer (Shimadzu UV-3800). Moreover, color changing behavior of the microcapsules was investigated using photographs taken by a digital camera at 25 °C and 50 °C. Thermal properties of the microcapsules and fabrics were determined by differential scanning calorimetry (DSC, Netsch Polyma) under nitrogen atmosphere [21]. Thermal stability of the microcapsules was investigated by thermal gravimetric (TG) analysis using a SETARAM instrument. Analyses were conducted under nitrogen atmosphere with a 10 °C/min scanning rate, in the range of 0–500°C.
Characterization of fabrics
Thermochromic properties of the P(MMA-co-MAA)/TCTS microcapsule applied fabrics were determined by examination of photographs taken at different temperatures, and color measurements based on CIELab system. A Minolta CR400 spectrophotometer was used to measure colorimetric parameters. Measurements were performed under D65-illumination and 10° standard observer. The calculated colorimetric parameters using CIELAB colour space were L* (lightness index), a* (red-green index), b* (yellow-blue index), C* (chroma index), h* (hue index). Measurements were performed at 25 °C and 50 °C and the color parameters measured on the fabric at 25 °C were regarded as references. Besides, total color difference (ΔE) calculated using equation (1) was used. A negative value of ΔL indicates that the color of the compared sample is darker than the reference, and a positive value indicates that it is lighter. While positive a and b values were red and yellow, negative a and b values were green and blue.
ΔE = √(ΔL*) 2 + (Δa*) 2 + (Δb*) 2 = Sum of color difference(1)
ΔL* = (L*sample – L*reference) = (difference in lightness and darkness)
Δa* = (a*sample – a*reference) = (difference in red and green)
Δb* = (b*sample – b*reference) = (difference in yellow and blue)
T-History measurements of microcapsules and fabrics
T-history measurements were made to investigate the thermo-regulation properties of the P(MMA-co-MAA)/TCTS microcapsules and microcapsule applied cotton and wool fabrics. In T-History test, a system consisting of a big insulated and two small polyurethane boxes were designed. Additionally, K-type thermocouples, a temperature and humidity sensor and a data-logger (Ahlborn, MA25903S model) were included in the system [23]. Microcapsules were settled in locked bags. Microcapsule containing TCTS was defined as sample, microcapsule without TS was defined as reference. Two thermocouples measured the temperature of the reference and sample microcapsules simultaneously. The reference fabrics without microcapsules and sample fabrics with P(MMA-co-MAA)/TCTS microcapsules, which were wrapped around the thermocouples in contact, were tested simultaneously. To heat the big polyurethane box to a constant temperature a 40-watt lamp which connecting to thermostat was utilized. Before temperature measurement, all materials were conditioned between ice moulds. Same amounts of microcapsules and the same size of fabrics were tested. The results were evaluated comparing the temperatures measured for reference and sample.
Results and discussions
Results of the analyses of the P(MMA-co-MAA)/TCTS microcapsules
To investigate morphology of the P(MMA-co-MAA)/TCTS microcapsules, SEM analysis was performed. According to the SEM images given Figure 1, the microcapsules had almost spherical shape and very smooth surface. Besides, microcapsules were nano-sized and their particle size distribution was relatively uniform.

SEM image of the P(MMA-co-MAA)/TCTS microcapsules.
Particle size distribution histograms of the P(MMA-co-MAA)/TCTS microcapsules were presented in Figure 2. According to Figure 2, microcapsules exhibited narrow and unimodal size distributions. Their average particle size was 23.24 µm and the sizes varied between 4.69-50.91 µm. Particle size measurement results showed that microcapsules had single uniform peak distribution and suitable particle sizes for textile applications [23,24]. However, it is well known that the particle sizes of microcapsules were found bigger than the particle sizes seen on the SEM images due to agglomerated nano-sized capsules in the water during particle size measurement [25,26]. Besides, according to the results given in the literature, the average particle sizes of the P(MMA-co-MAA)/TCTS usually varied between the 1–7 µm [19,20,27,28].

Particle size distribution histogram of the microcapsules.
FT-IR spectroscopy analysis was performed to show microencapsulation of TCTS in P(MMA-co-MAA) shell. In Figure 3, IR spectra of MMA, MAA, TCTS system, and P(MMA-co-MAA)/TCTS microcapsules were presented. In the IR spectrum of microencapsulated TCTS, the peaks at 2959 cm−1, 2920 cm−1 and 2850 cm−1 corresponded to C–H stretching peaks and the peaks at 1473–1464 cm−1 corresponded to characteristic C–H bending vibration peaks. Besides, a C-O stretching peak at 1064 cm−1 and binary peaks at 730 cm−1 and 720 cm−1 were evidences of the presence of P(MMA-co-MAA)/TCTS. The broad band at 3200-3600 cm−1 in spectrum of the microcapsules originated from O-H stretching bands of myristic alcohol and Bisphenol A, and carboxylic acid groups in the MAA monomer structure. Additionally, the peaks at 1375-1360 cm−1 attributed to symmetrical carboxylate stretching peaks, and the peaks at 1582 cm−1 and 1544 cm−1 attributed to asymmetric carboxylate stretching peak of coloured dye. These were revealed opening of the lactone ring of the colorant and coloration of leuco dye at room temperature. The peak at 1637 cm−1 in the spectrum of the MMA monomer and at 1631 cm−1 in the spectrum of the MAA monomer belonged to the C = C double bond peak and these peaks disappeared in the spectra of the microcapsules. The disappearance of these peaks proved the opening of C = C double bonds in the structure of MA and MMA monomers during addition polymerization.

FT-IR spectra of MMA (a), MAA (b), TC (c), P(MMA-co-MAA)/TCTS microcapsules (d).
The UV-Vis spectrophotometer analysis was made to determine thermochromic behaviour of microcapsules. UV-Vis spectra obtained from measurements at 25 °C and 50 °C was given in Figure 4. The absorbance recorded in wavelength of 520–630 nm corresponded to the wavelength where the blue color was visible. According to Figure 4, it was seen that absorbance value measured on sample at 25 °C at this wavelength was higher than that of the sample heated to 50 °C. The maximum absorbance value was 2.08 for the sample at 25 °C, while it decreased to 0.89 for the sample heated to 50 °C. The decreasing absorbance value demonstrated that color fading of the microcapsules depending on increase in temperature.

The UV-Vis spectra of P(MMA-co-MAA)/TCTS microcapsules at 25 °C and 50 °C.
In the study, to investigate reversible color change of the P(MMA-co-MAA)/TCTS microcapsules depending on change in temperature, their photographs taken at 50 °C and 25 °C were studied. As seen from photographs given in Figure 5, microcapsules were blue color at 25 °C and lost their color when they heated to 50 °C. When they were cooled again to 25 °C they were coloured to blue and exhibited reversible color change.

The photographs illustrating the reversible color change of the P(MMA-co-MAA)/TCTS microcapsules.
DSC analysis was realized to investigate phase transition temperatures and latent heat capacities of the microencapsulated system. According to DSC curve in Figure 6, microcapsules stored 197.7 J/g of latent heat at 33 °C. While their liquid-solid crystallization latent heat was -90.8 J/g at 35 °C, the solid-solid phase change latent heat was -82.5 J/g at 29 °C. The reason of the two crystallization peaks appeared during cooling process was two-step crystallization of fatty alcohol (myristyl alcohol) [29,30].
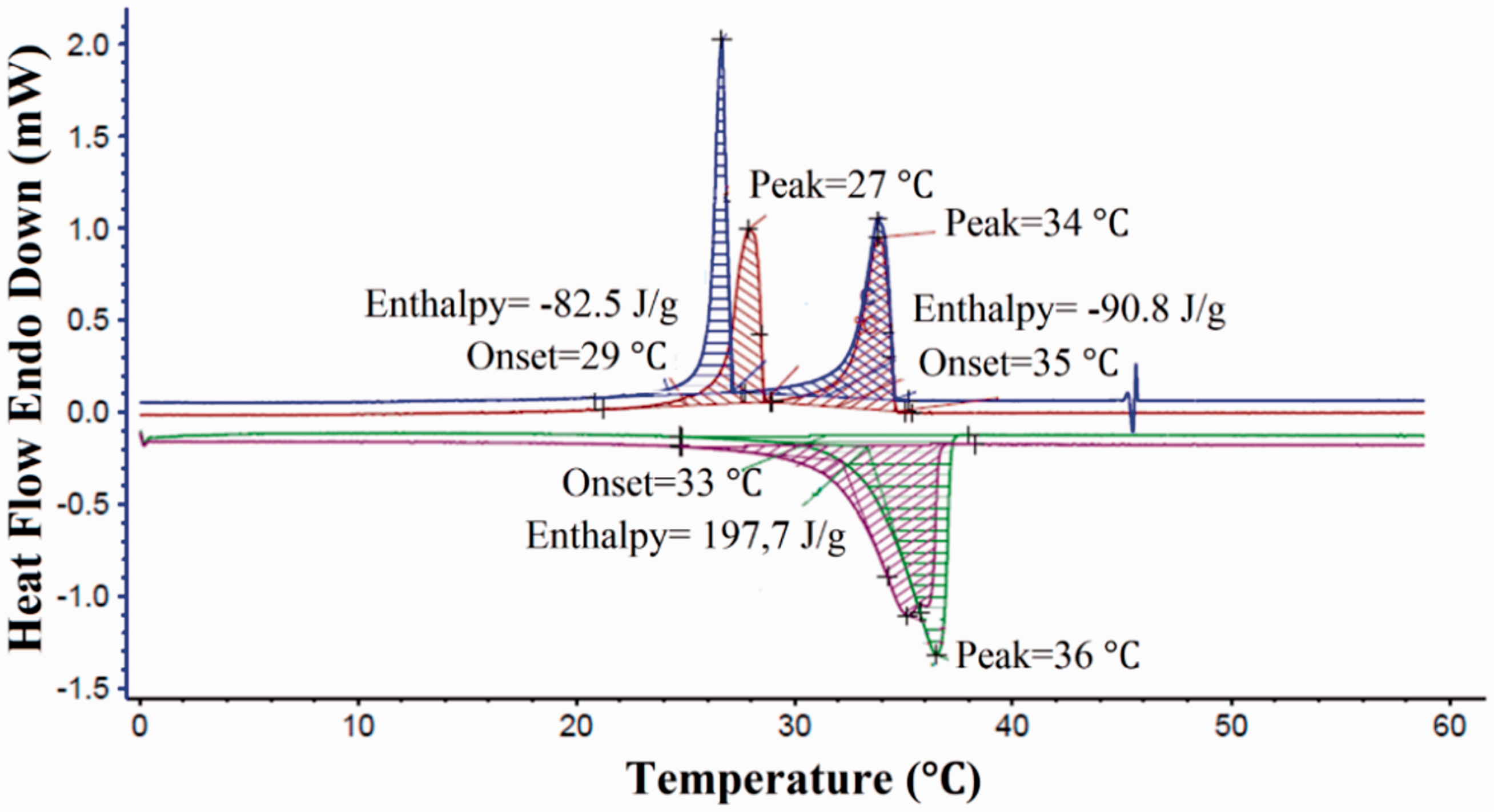
DSC curve of P(MMA-co-MAA)/TCTS microcapsules.
TG analysis was employed for determination of thermal degradation behaviour of the microcapsules. The TG analysis thermogram of the P(MMA-co-MAA)/TCTS microcapsules was presented in Figure 7. According to thermogram, microcapsules exhibited two steps thermal degradation. The initial weight loss for microcapsules started at around 125 °C. This first sharp weight loss of the microcapsules was in the range of 125–265°C and this degradation related with vapour pressure of the core materials. In this step, the first weight loss which lost by microcapsules was 84.4%. The second step degradation which was degradation of the capsule shell material occurred in the range of 265–460°C and P(MMA-co-MAA)/TCTS microcapsules lost their almost all weights. According to TG analysis results, microcapsules had thermal stability up to 125 °C to maintain their structural integrity and it was concluded that they could be resistant the maximum thermal curing temperatures up to 125 °C.
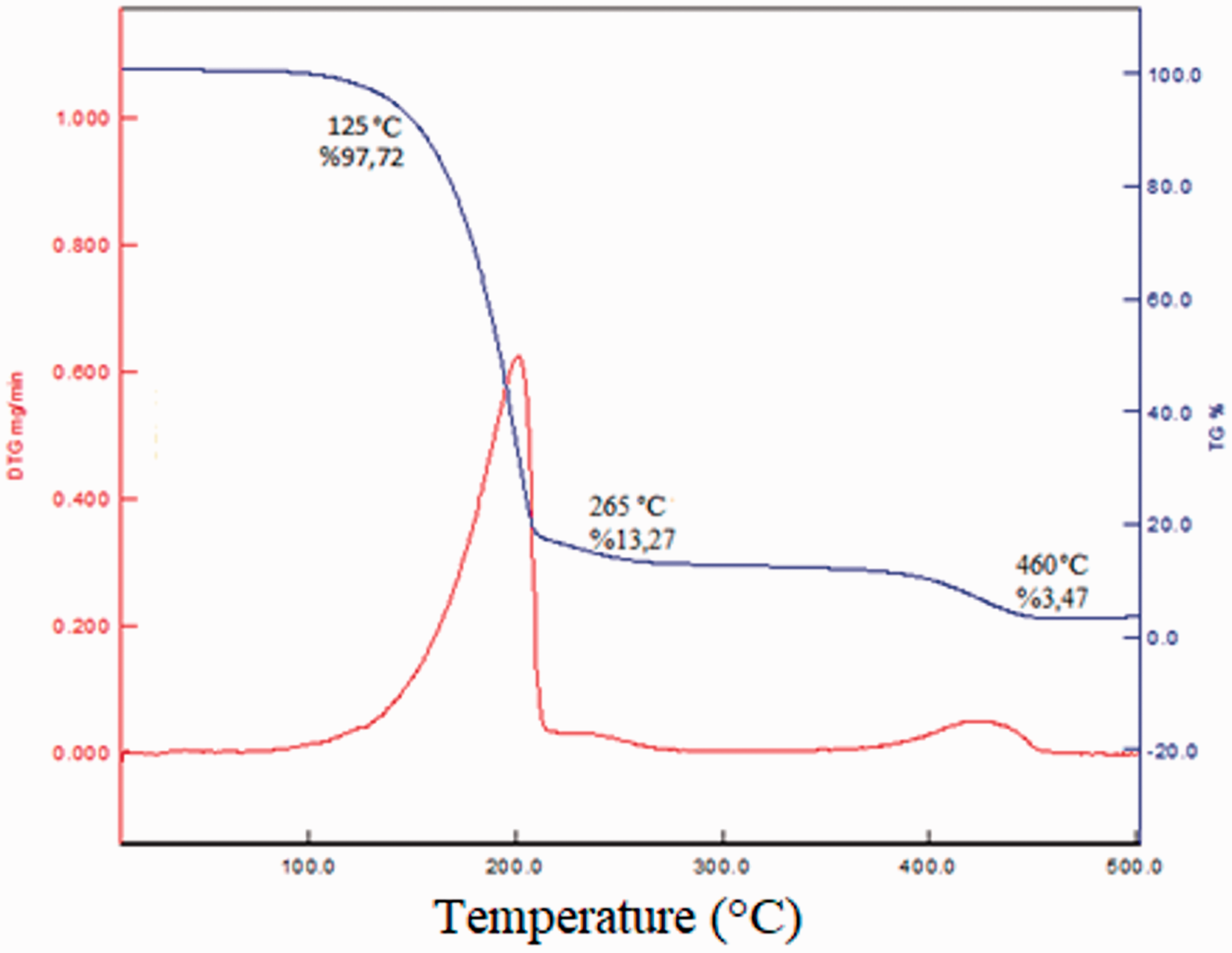
TG analysis thermogram of the P(MMA-co-MAA)/TCTS microcapsules.
Results of the fabric analyses and tests
SEM analysis of the P(MMA-co-MAA)/TCTS microcapsules applied fabrics
Morphology of pthe cotton and wool fabrics treated with P(MMA-co-MAA)/TCTS microcapsules was examined by SEM. Figure 8 showed SEM images of both cotton and wool fabrics containing P(MMA-co-MAA)/TCTS microcapsules. From SEM images, it was clearly seen the presence of the microcapsules settled between the fibres and on the fibre surfaces in the structure of the fabrics. However, microcapsules exhibited agglomeration tendency because of the affinity of particle surfaces each other. Besides, the number of microcapsules observing on the wool fabric was higher than that of the cotton fabric. Microcapsules showed more strongly affinity to the wool fibres compared to cotton fibres. The wool fibre is composed of a particular protein known as keratin consisting of long polypeptide chains having numerous different amino acids. These amino acids have reactive groups such as hydroxyl, amine and carboxylic acid. Molecular attractions occurred between the reactive carboxylic acid group in the shell molecules of the microcapsules and reactive groups on peptide chains in the structure of wool fibres. Consequently, numerous reactive groups in the keratin protein molecules caused more interactions of wool fibres with microcapsule wall at molecular level. Besides, in the microcapsule application processes to the fabrics, the pH of bath was adjusted to 1 to create an acidic medium. Wool molecules were cationic character at acidic pH values. However, shell of the microcapsules was anionic character at acidic pH. Electrostatic interaction generated between microcapsules and fibres which was other reason the affinity of the microcapsules to the wool fibres. This high affinity has enabled more microcapsules to be drawn into the fabric structure in the application by the exhaustion method. However, there are three free OH groups in each glucose unit of cellulose molecules which can form H-bonds by methacrylic acid molecules. Interaction of these groups with methacrylic acid molecules in the microcapsule wall structure provide affinity of the fibres to the microcapsules. Another source of affinity between the microcapsule and the cotton fibres was electrostatic interactions formed by cationic groups on the cationized cotton fabric with anionic microcapsules.

SEM images of the cotton (above) and wool (below) fabrics.
FT-IR analysis of the cotton and wool fabrics
The structure of the cotton and wool fabrics treated with P(MMA-co-MAA)/TCTS microcapsules was analysed by FT-IR spectroscopy to study the presence of the microcapsule in the fabric structure. Figure 9 showed the FT-IR spectra of P(MMA-co-MAA)/TCTS microcapsules, untreated cotton fabric, cationized cotton fabric and microcapsule applied cotton fabric. FT-IR spectrum of the untreated cotton fabric showed characteristic cellulose peaks around 1000–1200 cm−1. Besides, the hydrogen-bonded OH stretching at 3550–3100 cm−1, the C-H stretching at 2920 cm−1, and the C-H wagging at 1318 cm−1 were other characteristic peaks of the cellulose molecules [31]. In IR spectrum of the cationized cotton fabric, the narrowing of the H-bonded hydroxyl (O-H) stretching peak of pure cellulose was related to the reaction of O-H groups with cationizing agent. In spectrum of the cotton fabric treated with microcapsules, there were three peaks at about 2958 cm−1, 2920 cm−1 and 2850 cm−1 which were characteristic C-H stretching peaks of myristyl alcohol. The carbonyl peak at 1735 cm−1 in spectrum of the microcapsules which was carbonyl peak of the copolymer shell structure appeared at 1718 cm−1 in spectrum of the P(MMA-co-MAA)/TCTS microcapsule applied fabric. The peaks at 1578 cm−1 and 1542 cm−1 in spectrum of the microcapsule applied fabric represented the asymmetric COŌ stretching peaks of the dye molecule in the microcapsule structure. The carboxylate group occurs when the closed ring opened in the dye molecule with the interactions between the dye and the color developer (while myristyl alcohol was in solid state).

FT-IR spectra of P(MMA-co-MAA)/TCTS microcapsules (a); untreated cotton textile fabric (b); the cationized cotton textile fabric (c); cotton textile fabric treated with P(MMA-co-MAA)/TCTS microcapsules (d).
The FT-IR spectra of P(MMA-co-MAA)/TCTS microcapsules, untreated wool fabric and wool fabric treated with P(MMA-co-MAA)/TCTS microcapsules were given in Figure 10. A broad band at 3298 cm−1 and a weak band at 3066 cm−1 in the spectrum of untreated wool textile fabric were peaks of the N–H stretching of the peptide group (–CONH–). The peak at 1648 cm−1 was characteristic band of amide I (asymmetrical CO and C–N vibrations) and the peak at 1514 cm−1 indicated symmetrical coupling of CO and C–N vibrations and N–H deformation of amide II. The peak at 1236 cm−1 belonged to symmetrical coupling of CO and C–N vibrations and N–H deformation of amide III. Besides, C–H stretching peaks in the wavelengths of 2980–2850 cm−1 and CH2 scissoring and CH3 symmetric bending peaks at 1448 cm−1 and 1394 cm−1 arose in IR spectrum of the raw wool fabric [32]. The peaks at 2958 cm−1, 2918 cm−1 and 2850 cm−1 in IR spectrum of the wool fabric containing P(MMA-co-MAA)/TCTS microcapsules were characteristic C-H stretching peaks of microencapsulated the fatty alcohol. The peak at 1735 cm−1 in the spectrum of microcapsules which belonged to carbonyl peak (C = O) of P(MMA-co-MAA) copolymer arose at near 1726 cm−1 in the spectrum of the wool fabric. The peak at 1542 cm−1 in the spectrum of the wool fabric containing P(MMA-co-MAA)/TCTS microcapsules was the asymmetric COŌ stretching peak of the leuco dye. This peak proved that the ring in the dye molecule was opened and the system colored.

FT-IR spectra of P(MMA-co-MA)/TCTS microcapsules (a); untreated wool textile fabric (b); wool textile fabric applied with P(MMA-co-MA)/TCTS microcapsules (c).
Detection of the color change in the fabrics
The photographs of the cotton and wool fabrics heated and cooled were taken by a digital camera to investigate their thermochromic behaviours. Figure 11 showed that the fabrics were completely colorless at 50 °C. They started to colorization as the temperature decreased and turned into dark blue color when the temperature was 25 °C. The fabrics continued to change color as the temperature increased. However, the temperatures at which the fabrics began to discolor significantly (35 °C–37°C) were close to the melting temperature of myristyl alcohol contained in the microcapsules. Consequently, cotton and wool fabrics exhibiting reversible color change were obtained by application of the P(MMA-co-MAA)/TCTS microcapsules. However, the wool fabric presented darker color compared to the cotton fabric. This meant that the wool fabric took more amounts of the microcapsules from the bath during microcapsule application. SEM images confirmed this finding also.

The photos the cotton and wool fabrics taken at different temperatures.
Color analysis of the P(MMA-co-MAA)/TCTS microcapsules imparted fabrics
To determine thermochromic property of the cotton and wool fabrics, color measurements were carried out at different temperatures by spectrophotometer and colorimetric parameters were calculated using CIELAB. The L*, a*, b*, C* and h parameters of the fabrics were given in Table 4. In the evaluation, the values determined at 25 °C were used as references and the total color difference (ΔE) was calculated using the color parameters measured at 25 °C and 50 °C. According to Table 4, when the surface temperature of the fabrics increased from 25 °C to 50 °C, the L* values of the fabrics increased indicating the color of the fabrics opened. The decreasing chroma (C*) value demonstrated that brightness of the fabrics decreased with increasing temperature. The calculated ΔE values using color parameters obtained from measurements at 25 °C and 50 °C indicated that a significant color difference occurred when the fabric was heated. The negative b* values of the fabrics measured at 25 °C indicated that fabrics were blue colored. The b* values increased when the fabric was heated to 50 °C which meant that the blue color of the fabrics disappeared and turned into pale blue and yellowish white. According to the color angle (h) values calculated from a* and b* values, the color angle corresponding to the blue hue at 25 °C changed into a color angle indicating yellow or pale blue hue at 50 °C.
The color values of the textile fabrics measured at 25 °C and 50 °C.
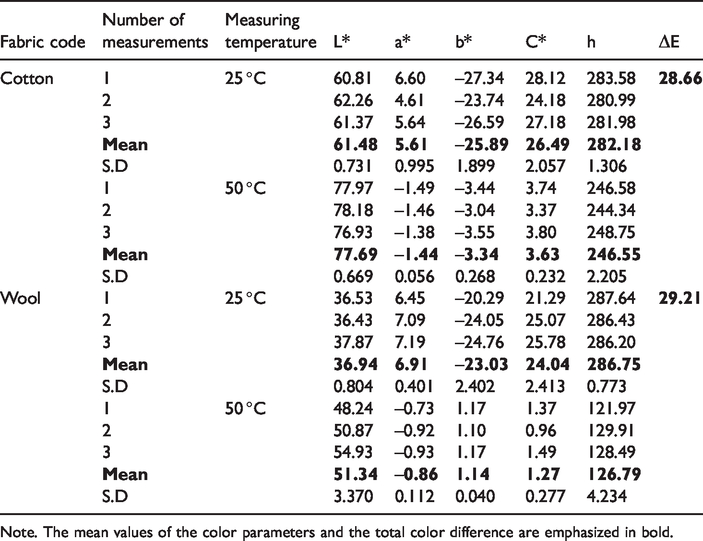
Note. The mean values of the color parameters and the total color difference are emphasized in bold.
DSC analysis of the P(MMA-co-MAA)/TCTS microcapsules imparted fabrics
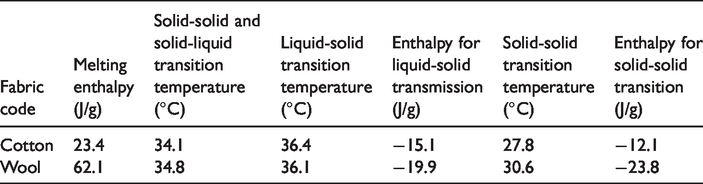
The DSC results of the fabrics were presented in Table 5. According to Table 5, cotton fabric had heat storage capacity of 23.4 J/g at 34.1 °C. The solidification and solid-solid transition temperatures during cooling of the fabric were determined as 36.4 °C and 27.8 °C, respectively. The enthalpies of crystallization for solidification and solid-solid phase transitions were measured as −15.1 J/g and −12.1 J/g, respectively. The wool fabric stored latent heat of 62.1 J/g at 34.8 °C and released latent heat of −19.9 J/g at 36.1 °C and −23.8 J/g at 30.6 °C for liquid-solid and solid-solid phase transitions, respectively. DSC analysis results indicated that heat storage capacities of the both the cotton and wool fabrics were quite high compared to the finding given in the literature [33–36]. Besides, it was concluded that the wool fabric had more enthalpy values compared to the cotton fabric. This finding was consistent with the photographs of the wool fabric as well as SEM images showing that more amount of the microcapsules settled on the wool fabric surface.
DSC results of the pure cotton and wool textile fabrics.
Thermal performance of the microcapsules and fabrics through T-history test
The thermo-regulation property of the microcapsules was determined by T-history test. In the test, thermo-regulation effect of the microcapsules resulting from the absorbed latent heat by myristyl alcohol during its melting was measured. Myristyl alcohol is a fatty alcohol which is a solid-liquid PCM and absorbs and releasing latent heat during its phase change processes. The heat uptake of myristyl alcohol in the P(MMA-co-MAA)/TCTS microcapsules leads to decrease in temperature of the microcapsule surfaces, which is defined as cooling effect. In this study, the cooling effect of the microcapsules and fabrics was determined by T-history measurements. Figure 12 showed surface temperatures measured on reference microcapsules without TCTS and sample microcapsules containing TCTS. As seen in Figure 12, surface temperature of the sample microcapsules was always lower than surface temperature of the reference microcapsules because of the latent heat absorbed by TCTS in sample microcapsules. The difference between reference and sample microcapsules started at about 30 °C continued for 30 minutes. The maximum surface temperature difference was recorded as 16.6 °C. Consequently, T-history test confirmed that P(MMA-co-MAA)/TCTS showed cooling effect which was defined as thermo-regulation property.
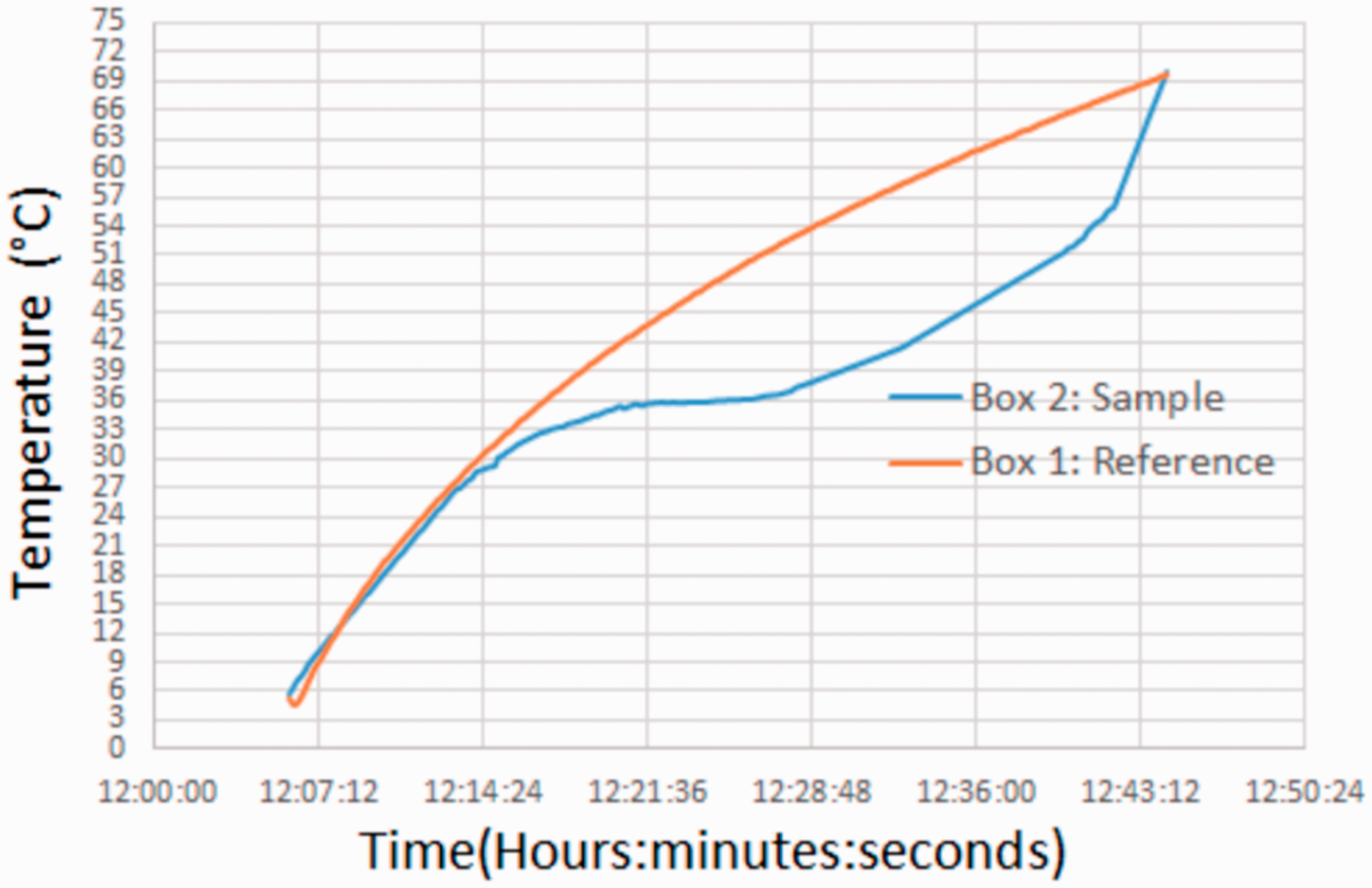
T-History results of the P(MMA-co-MAA)/TCTS microcapsules.
The cooling effect of the cotton and wool fabrics treated with thermochromic microcapsule resulted from the latent heat absorption of myristyl alcohol were determined by T-history test. In the test, changes in surface temperatures of the fabrics placed in an insulated polyurethane box during heating were recorded during a certain time and time-dependent temperature change graphs were created. According to Figure 13(a), the temperature difference between reference cotton fabric and cotton fabric treated with thermochromic microcapsules began at 15:47 and the surface temperature of the cotton fabric containing microcapsules was lower during 7 minutes. The maximum temperature difference was calculated as 3.7 °C. Figure 13(b) demonstrated that the surface temperature of the wool fabric containing thermochromic microcapsule increased slowly since 10:54 minutes and was lower than untreated wool fabrics’ for 14 minutes. The calculated maximum temperature difference was 3.9 °C between them. The surface temperature difference of the untreated and microcapsule incorporated fabrics was resulted from the latent heat absorbed by myristyl alcohol encapsulated in P(MMA-co-MAA), and defined as cooling effect. Consequently, T-history test results proved that the cotton and wool fabrics containing thermochromic microcapsules exhibited thermo-regulating property.
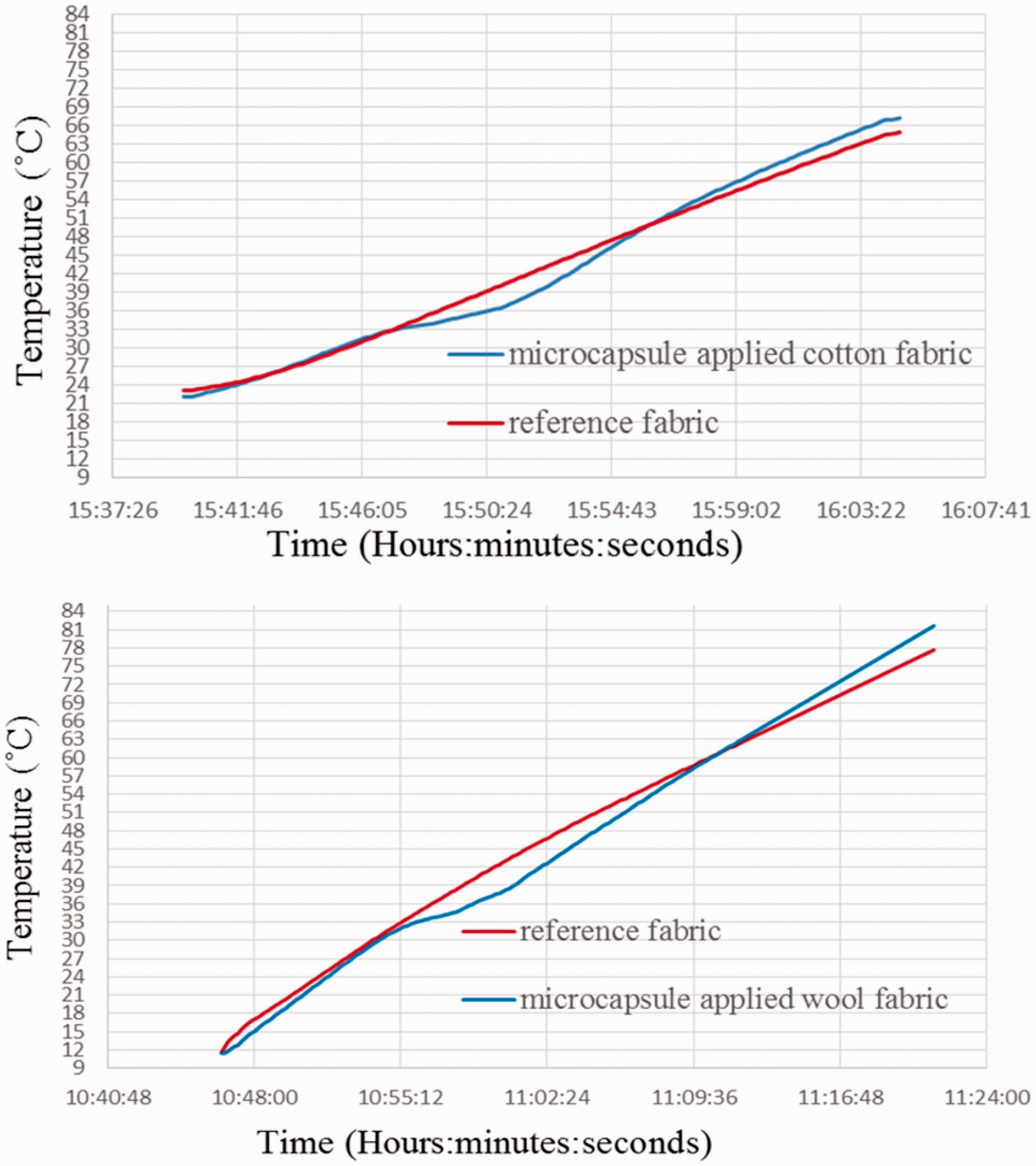
TES performance (T-History test) results of the pure cotton (a) and wool (b) fabrics.
Conclusion
In this study, the textile fabrics were designed to be used as flexible thermal energy storage materials and visual temperature indicators. These dual function cotton and wool fabrics were fabricated applying microcapsules having both thermochromic and TES properties. For this, reversibly color changed TCTSs were microencapsulated in the shell of poly(MMA-co-MAA) copolymer. In the study, the shell of the microcapsules was designed in ways to interac to the cotton and wool textile surfaces. The microcapsules with shell/core ratios of 0.5/1 containing TCTS were synthesized by emulsion polymerization method. They were spherical in shape and had smooth surfaces. They had mean particle size of 23.24 µm and exhibited a narrow particle size distribution. According to thermal analyses, they absorbed 197.7 J/g of latent heat at 33 °C and their phase change temperatures and thermal stability value were suitable for thermal comfort applications. UV-Visible spectroscopy analysis and the photographs taken at different temperatures showed that the microcapsules which were blue at room temperatures converted to light blue color over the threshold (phase change) temperature of the TCTS. They exhibited reversible color change and latent heat storage/releasing properties, depending on the change in temperature. Consequently, the results obtained by different analyses showed that microcapsules could store considerable latent heat and they can be used as thermochromic energy-storage materials in various end uses.
In the study, P(MMA-co-MAA)/TCTS microcapsules were applied to pure cotton and wool textile fabrics using bath exhaustion method. The exhaustion technique was selected as microcapsule application technique because of the affinity between the microcapsule shell structure and cotton cellulose and wool protein molecules was possible. Functionality of the shell surface enabled successful application of the microcapsules to the fabrics by exhaustion process.The carboxylic acid groups of the MAA monomer in P(MMA-co-MAA) shell interacted to the OH groups of cellulose molecule, and amid groups (I and II) and acid groups of the protein molecules. This interaction ensures the affinity between the molecules of the fabrics and microcapsules. From color analysis results and photos of the fabrics containing microcapsules, it was seen that they were blue color at room temperatures and turned into the original fabric color when they were heated above activation temperature of the sytems and this change was reversible. Besides, the calculated ΔE values using color parameters obtained from measurements at 25 °C and 50 °C were 28.66 and 29.21 for cotton and wool fabrics, respectively. It meant that the fabrics changed their color significantly depending on change in temperature. Consequently, their reversible color change property as a function of the temperature was proved. However, color depth of the wool textile fabric was more than cotton textile fabric. According to the thermal analysis results, the cotton fabric stored 23.4 J/g of latent heat while the wool textile fabric stored 62.1 J/g of latent heat. The energy stored by wool textile fabric was higher than cotton textile fabric. This meant that the more amount of microcapsules were exhausted from the bath by the wool textile fabric compared to the cotton textile fabric because of the more molecular interactions between the shell of the microcapsules and protein molecules of the wool fibres. These findings were consistent with the photographs of the wool textile fabric as well as SEM images showing that more amount of the microcapsules settled on the wool textile fabric surface. Besides, both of the microcapsule applied cotton and wool textile fabrics exhibited cooling effect. This effect was defined as thermoregulation property and resulted from absorbing of latent heat by microencapsulated P(MMA-co-MAA)/TCTS in the fabric structure. As expected the thermo-regulating property of the wool textile fabric was more noticeable.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the support by Scientific Research Projects Funding Organization of Suleyman Demirel University (Project no:4602-D2-16).
