Abstract
Poloxamer188 composite poly L-lactic acid (PLLA) electrospun fibrous nonwovens (EFNW) were prepared by simple letrozole-poloxamer188-PLLA co-solution electrospinning and used as subcutaneous implants to sustained release letrozole. Contents of poloxamer188 and letrozole were varied to investigate the effect of the two compounds’ amounts on drug release behavior. Materials’ combination and drug incorporation in fibers were investigated by X-ray diffraction and differential thermal calorimetry. Letrozole contents in vitro and in vivo were all determined by high-performance liquid chromatography-ultraviolet spectrometry. Meanwhile, in order to analyze the mechanism of drug release behavior and show modulated drug release by electrospun fibers, letrozole-poloxamer188 solid dispersions (SDs) and letrozole-poloxamer188poly L-lactic acid-dichloromethane solution cast films (CFs) were also studied. Plasma letrozole concentration curves of subcutaneous implanting EFNW and daily oral administration of poloxamer188-letrozole SD on female rabbits were investigated and compared. Fibers of desirable morphology were obtained. During the whole release process of all EFNWs, water-insoluble letrozole could be released in the form of being dissolved. Letrozole release rates increased with increasing poloxamer188 content and decreased with increasing letrozole content. When letrozole and poloxamer188 content in fibers was 30% and 150% (EFNW-30-150%), respectively (with respect to PLLA in mass ratio), the release curve presented a desirable profile. After subcutaneous implant of EFNW-30-150%, the plasma concentration curve presented a later peak time and lower maximum value, and a comparable bioavailability with daily oral administration of poloxamer188-letrozole SD solution within 15 days. The research provided primary and encouraging information to use EFNW as a novel subcutaneous implant for sustained delivery of water-insoluble drugs.
Introduction
In a clinic, there are a considerable amount of drugs which need to be administrated daily for a considerably long time (at least more than 1 month), while the daily dosages needed by the body are very small (normally less than 10 mg). This kind of drugs includes endogenous hormones, synthesized hormone receptor agonists/antagonists, and hormone-transforming enzyme inhibitors. One shortcoming for oral administration dosage forms, such as tablets, pills, and millimeter particles, for these drugs is that they need to be taken every day. This would cause many inconveniences, bad compliance, and missing doses during the course of therapy.1–3 To solve these problems, scientists have always been trying to utilize subcutaneously implantable matrices as substitutes for sustained release of these drugs for considerably long time periods by small molecule diffusion and macromolecule matrices blocking-erosion mechanisms. To accomplish these purposes, macromolecules with properties of water-insolubility and degradability have been selected and used. Such polymers include silicones, polyurethanes, and polyesters, etc., which were manufactured into microspheres, hydrogels, little sticks, membranes, and so on.4,5 However, there have still been many shortcomings in these drug delivery systems. For example, microspheres were usually prepared by emulsion solvent evaporation method in systems of oil phase in aqueous phase. Thus, in the preparation process, drug molecules would easily leak from the oil phase into the aqueous phase and tend to accumulate on the surface of oil phase-aqueous phase, resulting in low encapsulation efficiency and considerable burst release. Hydrogels generally presented low loading efficiency to water-insoluble drugs and considerable burst release due to aqueous-aqueous phase contact and pervasion. Porous matrices and membranes often presented undesirable release profiles and considerable amounts of remaining unreleased drug molecules during the release process. Therefore, developing novel subcutaneous drug delivery systems which present desirable properties remains a significant and interesting task.6–8
In the last 15 years, micro/nanofibers prepared by the electrospinning technique have emerged as desirable candidates for efficient drug incorporation and controlled release. Because electrospinning was generally performed in a dry gas atmosphere, there would not be liquidl-iquid surface effects causing extraction of drug molecules from solidifying polymer organic solvent solutions and consequently resulting in undesirable low drug incorporation efficiency. So, electrospun fibers usually presented high drug incorporation efficiency and could actualize many more kinds of composites of hydrophobic polymers with hydrophilic polymers.9,10 In the field of pharmaceutics, many electrospun micro/nanofibers have been developed and used to incorporate and deliver antibiotics, cytotoxic anticancer drugs, and anti-allergic drugs in the form of patches or implants, due to their shape and physico-chemical properties.11,12
Although there have been a lot of polymers desirable for being electrospun into fibers to prepare various drug delivery systems, aliphatic polyesters (APEs) have been still the most frequently used ones among them.13–15 APEs can be dissolved in many organic solvents (dichloromethane, chloroform, acetone, methanol, hexafluoroisopropanol, etc.), so any drug that can be dissolved in these organic solvents is ready to be incorporated successfully in APE fibers after electrospinning drugs/APE co-solutions prepared from these solvents. And, among these organic-solvent-dissolvable drugs, the ones with water-soluble properties could present sustained release behavior based on the diffusion mechanism of hydrophilic small molecules passing through hydrophobic polymeric matrices. Meanwhile, there was still a problem that remained: water-insoluble drugs could not be released from APE electrospun fibers in dissolved form in an aqueous medium as water-soluble drugs did. For example, paclitaxel or carmustine, both water-insoluble drugs, could hardly be released from poly L-lactic acid electrospun fibers. Only water-insoluble tiny solid aggregates containing combined paclitaxel or carmustine molecules could peel off from poly L-lactic acid fibrous matrices under the degrading effect by proteinase K. Without really dissolving in an aqueous medium in the molecular state but drug content continuously accumulating in the release medium in the form of precipitates, this was seemingly a release phenomenon, in fact. After all, the tiny solid aggregates could neither interact well with biomembranes nor develop the best pharmacological effects on cells.16,17
To solve this problem, someone had incorporated a few kinds of surfactants with poly L-lactic acid (PLLA) by co-solution electrospinning and expected the selected surfactants to solubilize water-insoluble drugs from water-insoluble fibrous matrices to realize water-insoluble drugs’ release in the form of being solubilized. Among the candidates, it was discovered that poloxamer188, one of polyoxyethylene-polyoxypropylene block copolymers could form composites well with poly L-lactic acid easily obtaining microscale and diameter-homogeneous fibers by a simple co-solution electrospinning process. 18 What is more, it was discovered that water-insoluble drugs such as docetaxel could be easily incorporated into poloxamer188/PLLA composite electrospun fibers with a high loading amount and could be released from the fibers in the form of being solubilized under the condition that a considerable amount of Poloxamer188 had been incorporated in fibers. 19
The previous research inspired us to try incorporating some other water-insoluble drugs into poloxamer188/PLLA composite electrospun fibers. After preliminary studies in our laboratory, it was discovered that several water-insoluble drugs with diazole or triazole groups and less than 500-Dalton molecular weight could be released from poloxamer188/PLLA composite electrospun fibers in the form of being solubilized. Moreover, these drugs’ release profile could be improved compared with cast films of the same materials’ composite rates and could be modulated by poloxamer188/drug incorporation content. Aromatase inhibitor letrozole was among the drugs.
In clinical practice, novel delivery systems for letrozole still need to be developed which would present sustained release (1 month or longer) after single-time administration to overcome incompliance and inconvenience of its common dosage forms such as oral tablets. Moreover, after providing the information of small-molecule drugs’ incorporation and release behaviors in poloxamer188/PLLA composite electrospun fibers, this study would inspire many research areas in the application of pharmaceutical composite fibers and novel implant development for sustained drug release.
Therefore, in this paper, three highlighted elements, including novel drug delivery system, desirable preparation technique, and selection of suitable drugs and material for the system, are combined together to generate a novel letrozole delivery system called electrospun fibrous nonwoven (EFNW) for subcutaneous implants. Since the previous studies have revealed that incorporating contents of both poloxamer188 and drugs could affect drugs’ release profiles, a series of contents of letrozole and poloxamer188 were specially investigated in this study. At last, sustained release of letrozole for more than 40 days was realized in vitro, and the EFNW with the most letrozole released was subcutaneously implanted in female rabbits, and the curves of letrozole plasma concentration (ng·mL−1) versus time (h) were investigated in order to further characterize and present the improvement of the novel delivery system compared with daily oral administration.
Materials and methods
Materials
Poly L-lactic acid (Mw = 150 kDa) was synthesized in our laboratory by ring-opening polymerization from L-lactide (98.0%, Energy Chemical Company, Shanghai, China) with 1-Dodecanol (Macklin Reagent Company, Shanghai, China) as the initiator, obtaining PLLA with the structure such that one terminal group was the hydroxyl group and another terminal group was the dodecyl group. Letrozole (99.6%) and poloxamer188 were purchased from Meilunbio Company, Dalian, China. Dichloromethane (analytical pure) was purchased from Damao Chemical Reagent Company, Tianjin, China. Acetonitrile (chromatographic pure) was purchased from Guangfu Chemical Reagent Company, Tianjin, China.
Preparation of films and fibers
First, a series of homogeneous solutions with materials dissolved were prepared to be cast and electrospun. First, all solutions were prepared by magnetic stirring solid poly L-lactic acid raw powder, poloxamer188 raw powder, and letrozole raw powder of designed amounts into 5 mL dichloromethane for 6 h to obtain thick homogeneous solutions in which all materials had been thoroughly dissolved. And then, extra amount of dichloromethane was precisely added to thick solutions to make the final volume of all solutions 6 mL. After another 6 h of mixing by magnetic stirring, the final solutions ready for casting and electrospinning were obtained. 12
Letrozole and poloxamer188 incorporating contents (mass ratio to poly L-lactic acid) and average diameters of CFs and EFNWs.

All preparations were carried out in 1 atm air. The circumstance temperature was 20 ± 2°C and the relative humidity was 60 ± 5%. Films were prepared by casting the solutions on dry smooth glass plates. Then, chloromethane was evaporated until dry layers formed. Meanwhile, for fibers preparation, all solutions were loaded into 30 mL syringes with a stainless steel needle of 0.41-mm internal diameter. Solutions’ fluid was supplied at a constant flow rate of 2.5 ± 0.2 mL/h. Voltage on the needle was set at 15 kV. After electric field was applied, Taylor cones were soon formed and ultra-fine fibers were successfully spun. Poly L-lactic acid-dichloromethane solution has high surface tension and cannot be formed into Taylor cones and be electrospun successfully. However, in this study, non-ionic surfactant was added in the solutions, causing significant surface tension decrease which would facilitate electrospinning. Produced nonwoven fibers were collected on sheets of static tin foil. The distance between the needle tip and tin foil collector was 30 cm.
After completion of casting and electrospinning, films and fibers were removed from the carriers and placed under vacuum for 24 h at 40 ± 2°C to remove any remaining solvent. All samples were then stored in sealed nitrogen-aerated polyethylene bags at 4°C for further study, and all samples were used within 10 days.
Solubilization of letrozole by poloxamer188 solid dispersion (SD)
Letrozole and poloxamer188 composite contents.

Morphology and materials composite
Morphology
Scanning electron microscope (SEM) was used to study the surface morphology of electrospun fibers. Samples were mounted on a steel stub with double-sided electroconductive adhesive tapes and were coated by sputtering under vacuum with an aurum layer (HITACHI E−1010). Fiber diameters in images obtained from SEM were measured 100 times randomly in a single sample and frequency histograms of fiber diameters were plotted after measuring by using Nano Measurer 1.2.0. Software.
SEM characterization could present fibers’ morphology, especially their average diameter and homogeneity which are very important factors for drug release behavior. Moreover, SEM presents the state of the fiber surface. If the drug has not been well incorporated, it would separate from the fiber’s surface forming tiny raised aggregates or even crystals composed by the drug itself or drug-polymer composites. And, if the materials used for forming fibers have not combined well, there would be phenomena of materials’ separation, with surface morphology just like bulges, ripples, cracks, etc. SEM could only provide primary information about drug incorporation and materials’ combination. Further characterizations are necessary.
X-Ray diffraction and differential scanning calorimetry
States of letrozole and poloxamer188 existing on fibers’ surfaces and being incorporated in fibers’ matrices were investigated by wide-angle X-ray diffraction (XRD, Rigaku UltimaIV, Japan), in which all samples were scanned from 5o to 80o at a scanning rate of 2o/min at 25°C, and differential thermal analysis (DTA, Netzsch, 200F3, Germany) in which all samples were scanned from 25o to 400o at a scanning rate of 5o/min under the N2 atmosphere.
XRD could provide further information about drug incorporation and materials’ combination. If the drug was well incorporated and the materials’ combination was desirable, characteristic peaks of every raw material would not exist in XRD, otherwise, characteristic peaks of raw materials would exist. As to DTA, favorable drug incorporation and materials’ combination would cause shifting of raw materials’ characteristic peaks.
In vitro letrozole release
Letrozole released amounts from CFs and EFNWs in vitro, and concentrations in female rabbit plasma were all determined by using a combination of a column (Elite Hypersil C18, ϕ4.5 mm×200 mm, 5μ), a HITACHI ELITE Lachrom L-2400 ultraviolet-detector, and an L-2130 pump. The mobile phase was a 47/53(V/V) mixture of acetonitrile and pure water. The flow rate was 1.0 mL/min, and the detection wavelength was 230 nm. 21
To study the drug release kinetics in the release medium in vitro, CFs and EFNWs were cut into square shapes of about 5 cm × 5 cm with an average weight of about 50 mg for each one. All samples were placed into 10 mL pure water in glass vials in a thermostatic oscillator of 100 r·min−1 at 37°C. At predesigned time intervals, a total of 10 mL release medium in every vial was withdrawn and filtrated through 0.8 μm polycarbonate membranes for letrozole quantification by the high-performance liquid chromatography-ultraviolet spectrometry (HPLC-UV) method as mentioned above. After this, 10 mL fresh release medium was re-added, and the sampling procedures were re-done for the next time intervals.10,16
At the same time, curves of letrozole solubilizing amount-time were also plotted by the same method mentioned above to understand the solubilizing ability of Poloxamer188 to letrozole.
The release percentage of letrozole for each sampling time point was calculated by the formula as below:
r% = Cn/M×p%;
where r% is the percentage of released letrozole to the total incorporated letrozole in the sample, Cn is the content of letrozole in the release medium for every sampling time point, M is the total mass of the sample to be investigated, and p% is the letrozole composite percentage in the sample.
After obtaining the release mass amounts and percentages of every sampling time point, the accumulated amounts and percentages were also calculated. At last, letrozole release curves of absolute released content and released percentage to total incorporated content were plotted to time.
Drug administration and plasma concentration
Female albino rabbits (10 weeks old, 250 ± 5g) were supplied by the Laboratory Animal Center of Jinzhou Medical University and fed standard rabbit food in individually ventilated cages with a constant temperature (20 ± 5°C) and humidity (45 ± 5%) on a controlled 12-h light/dark cycle. They were fasted for 12 h before drug administration, during which only water was freely available. The experimental protocol was approved by the University Ethics Committee for the use of experimental animals and conformed with the Guide for Care and Use of Laboratory Animals.
EFNW-30%-150% was administered by subcutaneous implanting into the back skin of five female albino rabbits, respectively. Meanwhile, SD-10%-100% was administered orally to another five female albino rabbits to simulate the current practical clinic administration of letrozole for the control study. The administration was carried out daily for 1.5 months during which every day’s dosage of letrozole was the same, and the summation amount of letrozole was equal to the content of letrozole in EFNW-30%-150%.
Since the dosage of letrozole oral administration for adult women patients have been 50 μg·kg−1 per day in current clinical practice, the total letrozole dosage for 1.5 months should be 2.25 mg·kg−1, and by mathematical conversion after considering rabbit mass and letrozole content in EFNW, the dosage for every rabbit should be around 11.25 mg and the nonwoven (EFNW-30%-150%) mass for only-one-time subcutaneous administration should be 105 mg which was about 2.5 cm × 2.5 cm. So, the mass and volume of implanted EFNW-30%-150% was not so big and could be used conveniently in the subcutaneous implanting process.
After administrations, blood samples were collected from rabbits’ auricular veins and centrifuged at 2000 r·min−1 to get plasma at every predesigned time interval. Then 1 mL plasma of every sample was transferred to 10 mL heparinized glass tube and added with 200 μL internal standard diethylstilbestrol (4 μg/mL). After being vortex-mixed for 3 min, 3 mL diethyl ether was added and vortex-mixed for another 3 min to extract letrozole and internal standard into the organic layer. Then, the system was centrifuged for 5 min at 2000 r·min−1 and 2 mL of the obtained clear supernatant layer was transferred into a 5-mL tube and evaporated by nitrogen gas flow in a 40°C water bath. Finally, the residue was dissolved by 200 μL HPLC mobile phase and 20 μL of clear reconstitution was injected for HPLC-UV analysis as described in the section. Pharmacokinetics (PK) data were generated using non-compartmental analysis. 22
Inflammation reaction
To further evaluate cutaneous and subcutaneous response, EFNW-30%-150% was subcutaneously implanted into a female rabbit. Meanwhile, pure poly L-lactic acid cast film, Poloxamer188 composite poly L-lactic acid cast film and Poloxamer188 composite poly L-lactic acid electrospun fibers were also prepared to investigate the effects of shape, Poloxamer188 composition, and drug incorporation to inflammation reaction. The rabbit was sacrificed at predetermined intervals of 3, 7, and 28 days when the skin of implanted position was taken from the rabbit body and immersed into 4% (w/v) paraformaldehyde water solution for 3 days. And then, the fixed specimens were serially dehydrated in a graded series of ethanol washes and embedded in paraffin. Further, the dealt skin tissue was prepared into tissue slices by solid paraffin sectioning-hematoxylin-eosin (H&E) staining process for microscope observation on a Nikon Digital Camera (Eclipse Ci-L apparatus with Nikon DS-F12 soft system, Nikon Co., Tokyo, Japan). The host response was determined by measuring inflammatory cell infiltration.
Results and Discussion
Morphology and drug incorporation
All obtained letrozole-incorporated EFNWs were of white color and non-transparent. All fibers exhibited wettability soon after the water droplets were contacted to them. Their microscopic morphologies are presented in Figure 1(a) and their diameters are also presented in Table 1. Generally, all samples presented continuous microscale fibers less than 2.5 μm without any bead structure. As the incorporated amount of poloxamer188 in fibers had increased and the incorporated amount of letrozole had been fixed, the fibers’ diameter increased. (A) Morphology of poloxamer188 composite and letrozole-incorporated poly L-lactic acid electrospun fibers (bar represents 30 μm); (B) X-ray diffraction patterns; and (C) differential thermal analysis curves of raw materials and drug-incorporated EFNWs.
There are many factors affecting the diameter of electrospun fibers, such as solution viscosity/thickness, surface tension, electric charging of solution, applied electrospinning voltage, distance from needle tip to collection plate, solution flow rate, and so on. Three factors, including electrospinning voltage, distance from needle tip to collection plate, and solution flow rate, were selected following the suitable commonly used conditions reported elsewhere and fixed, respectively, in order to minimize the number of variants as much as possible. And substances in solution could not be ionized. So, the factors that affected the diameters of electrospun fibers could be solution viscosity/thickness and surface tension. As it is known that surfactants could decrease the surface tension of poly L-lactic acid/dichloromethane solution, the fibers’ diameter should increase as poloxamer188 amounts in solutions had increased. However, the increasing amount of Poloxamer188 could increase the viscosity of the solutions which would cause fiber diameter to increase. The morphological result suggests that the factor of solution viscosity increasing caused by the surfactant had counteracted the effect of the solution’s surface tension decrease, also caused by the surfactant, and further presented a dominant effect on fiber diameter increase.8–11
When poly L-lactic acid-dichloromethane solution was electrospinning without poloxamer188 being dissolved together, it could not form fibers of homogeneous diameters and non-bead structure.12,14,16 And it was discovered in previous studies that by dissolving certain kinds of surfactants within aliphatic polyesters dichloromethane solutions, the electrospinning process became more smooth and the fibers’ morphology could be significantly improved.23,24 Moreover, composite surfactants could also provide added functions, such as solubilizing water-insoluble drugs from water-insoluble fiber matrices, modulating drug release behavior, modulating fibers hydrophilicity, etc., which were all newly discovered in very recent years.
The obtained CFs were semi-transparent and white color, according to the ratios of letrozole and poloxamer188 being incorporated in them. As the content of letrozole increased, CFs became more and more opaque.
From the SEM, there was no crystal structure and materials’ separation structures on all EFNWs. However, for more accurate characterization about the composite state of materials on the surface and in internal matrices, XRD (X-ray diffraction) and DTA (differential thermal analysis) tests are necessary. From XRD patterns shown in Figure 1(b), two conclusions can be generated. First, according to the occurring of characteristic peaks when poloxamer188-incorporated contents were above 50% and letrozole-incorporated contents were fixed, it can be seen that as the ratio of Poloxamer188 had increased to a certain extent in fibers, it would be separated on the fibers surface. This phenomenon had also been discovered in previous studies both in drug-incorporated and non-drug-incorporated poloxamer188 composite poly L-lactic acid electrospun fibers.18,19,25 Due to high hydrophilic-lipophilic balance value of poloxamer188, the surfactant tends to accumulate on the surface position of gas-organic solution interface when being prepared into the micro-dispersion system. This could facilitate dissolving and releasing of letrozole. In addition, when the letrozole-incorporated ratio was 30% and poloxamer188-incorporated ratio were 50%, 75%, and 100%, letrozole had also separated from the fibers presenting its characteristic peaks in XRD patterns although no crystal-like structure existed on the fibers’ surface. In the case of being electrospun in hydrophobic poly L-lactic acid-dichloromethane solution, considerable ratio of incorporated poloxamer188 and letrozole would probably exist on the fibers’ surface and at the same, present no structure separating-out. By comprehensively analyzing the results of detectable characteristic peaks of poloxamer188 and letrozole and no crystal-like structure on the fibers’ surface, it suggests that these two substances did separate from the fiber matrices, it suggests that these two substance separated from fiber matrices in a state of order assembling around fiber surfaces. Separation-out of letrozole on the fibers caused by its amount increasing on the fiber surface may result in decreasing solubilization of the drug itself and consequently decrease the drug release rate to be corroborated in the following drug release study.
From DTA patterns shown in Figure 1(c), it can be discovered that characteristic endothermal peaks at 182.9°C of letrozole were all covered up because of its relatively small incorporation ratio in the system, suggesting desirable composite of the drug within fibers matrices; meanwhile, characteristic endothermal peaks at 57.8°C of poloxamer188 emerged significantly when its incorporated ratios were 50%, 75%, and 100%, which suggested that poloxamer188 could separate from the matrices of fibers when its ratio had been considerably high. Both of XRD and DTA characterizations had verified that poloxamer188 tended more to be separated from the matrices and dependent on its increased incorporated ratio. Although agglomeration of poloxamer188 may also happen, in our study, there was no structure of materials’ separation and crystals observed through the scanning electron microscope. While, by X-ray diffraction and differential thermal analysis, it is suggested that a single component of poloxamer188 had existed on and in some fibers. Indeed, there is no suggestion of stronger bonding or interactions between poloxamer188 and the fiber surface than those among poloxamer188 itself; that is the very reason that poloxamer188 could be hydrated and released successfully from water-insoluble poly L-lactic acid fibers. Poloxamer188 may exist on or near the fibers’ surface in an orderly assembled separated state. As to how poloxamer188 existed within fibers’ cross-section, there is still no desirable equipment characterization method. The situation may be inferred from drug release behavior.16–19
All fibers could be infiltrated immediately after a pure water droplet was placed on their surface to make perfect soakage. This would facilitate their release in aqueous release medium and application in vivo.
Letrozole in vitro release
Although the release of letrozole incorporated within EFNWs is the main issue for this paper, letrozole release profiles of CFs were still investigated and compared with EFNWs in order to further understand the effect of poloxamer188 incorporation into letrozole release. The results are presented in Figure 2(a). Obviously, when PLLA cast films were prepared with incorporated poloxamer188, more letrozole was released and faster than those films cast by only PLLA when letrozole incorporation content was the same. This suggests that poloxmer188 facilitated letrozole release due to the polymeric surfactant providing hydrophilicity on the material surface and in material matrix, and poloxamer188 could combine letrozole and dissolve the drug out of material when in aqueous release mediums. Furthermore, it is discovered that poloxamer188 composite EFNWs facilitated more letrozole release than poloxamer188 composite CFs did by EFNWs providing more surface area than CFs. (A) Release profiles for presenting the effect of material morphology and poloxamer188 composite content to letrozole release from CFs and EFNWs; (B) release profiles for presenting the effect of poloxamer188 composite content to letrozole release rate from EFNWs when letrozole incorporation content was set as an invariant; (C) release profiles for presenting the effect of letrozole incorporation content to letrozole release from SDs when poloxamer188 composite content was an invariant; (D) release profiles for presenting the effect of letrozole composite content to letrozole release rate from EFNWs when letrozole incorporation content was set as an invariant (error bars correspond to one standard deviation from three individual measurements).
To further investigate how poloxamer188 incorporation content had affected letrozole release, its values from 12.5% to 100% were investigated. The results are presented in Figure 2(b). It is discovered that the release rate of letrozole increased as the content of poloxamer188 increased when the incorporation content of letrozole was the same. This phenomenon could be explained as more poloxamer188 providing more hydrophilicity on the surface and in the internal matrix of fibers facilitating penetration and hydration of water molecules through the fibers, and more poloxamer188 would also combine and solubilize more letrozole to let drug molecules release from fibers.
The solubilizing ability of poloxamer188 in letrozole was proved by the poloxamer188-letrozole solid dispersion dissolving test which is presented in Figure 2(c). Letrozole was nearly 100% solubilized after 30 min from poloxamer188 SD when the drug incorporation content to poloxamer188 of mass ratio was 10%. Meanwhile, letrozole of more than 20% mass ratio to poloxamer188 in SDs could not be solubilized within 3 h. The result suggests that solubilizing ability of poloxamer188 in letrozole is below 10:100 of letrozole to poloxamer188. However, no matter how much of letrozole and poloxamer188 were incorporated (letrozole from 0% to 30% and poloxamer188 from 12.5% to 100%, respectively, in this study), letrozole was released from EFNWs in the form of being solubilized all the time. This specifically suggested that the letrozole-poloxamer188 ratio was not the only decisive factor for the letrozole solubilizing-releasing process and electrospun fibers had also improved the dissolving ability in letrozole probably by the mechanism of high-dispersion effect.
Yet, Figure 2(b) is re-analyzed from the aspect of how letrozole-incorporating contents in fibers affected the release profiles of the drug itself. It is obvious that increasing letrozole incorporation content decreased the letrozole release rate for both EFNWs and SDs. Specifically, as shown in Figure 2(d), when letrozole incorporation content in EFNWs had increased under the condition of same incorporation content of poloxamer188, letrozole burst release amount and release rate would also decrease. This suggests that the higher the letrozole incorporation content was, the lesser the drug release rate would be. Thus, longer time would be needed to accomplish nearly 100% release ratio. The results could be caused by two mechanisms. First, more incorporation content of letrozole could increase hydrophobicity on the material surface and in the material matrix by impeding water molecules to hydrate and penetrate into material matrices of EFNWs which would result in slower letrozole dissolving. Second, more letrozole would consume more poloxamer188 for combining and dissolving the drug to weaken the effects of hydrophilic route-making based on poloxamer188 dissolving out from matrices of EFNWs. However, more letrozole-incorporating contents had not affected the thorough release of letrozole that release amount of letrozole from EFNW-30%-100% increased all the time from the moment it was immersed in the release medium to the end of the experiment, and it had released nearly 80% incorporated letrozole which was very close to that EFNW-10%-100% and EFNW-20%-100% had done. Therefore, the final comprehensive result was that EFNW-30%-100% presented the most desirable release profile bending toward the time-axis with smaller burst release amounts and longer sustained release phase. In the profile, the release amount increased more evenly within unit time compared with other EFNWs. Finally, a very long sustained release period of about 6 weeks became a reality.
Electrospun fibers can be considered as an unordinary solid dispersion with specific assembly structure. To further investigate the release mechanism of letrozole from EFNWs, all release curves were fitted to Korsmeyer-Peppas equation presented as
Qt/Q∞= ktn
In which, Qt is the accumulated release amount in t time, Q ∞ is the absolute drug amount in the system, k is the release index, t is the release time, and n is the geometric characteristic constant. While
k = D s C a P(2C 0 -PC a )/λ
D s is the diffusion coefficient, C a is the drug solubility in release medium, P is the degree of porosity of solid matrix, C 0 is the total drug content in matrix of unit volume, and λ is the bending factor of the micro route in solid matrix; among these parameters, D s , C a , and C 0 are invariable.
Result of regression by Korsmeyer–Peppas equation of every EFNW’s release curves.

There have been some reports about the effect of drug incorporation content increase on the drug release profile in electrospun fibers. These reports pointed out that the water-soluble drug release rate would increase after drug incorporation content had increased in the electrospun fiber. The mechanism is that higher incorporation content of water-soluble molecules can generate more routes after their release from fiber matrices. This facilitated continuous movement in and their thorough release out of fiber matrices. Moreover, in the situation of hydrophilic drug incorporation in electrospun fibers, composite poloxamer188 does not take part in binding and solubilizing drugs as in the situation of hydrophobic drugs; its main function is providing a route in fiber-matrix after its release. In this experiment, one of the functions of poloxamer188 was combining and solubilizing letrozole. And, when the incorporation content of letrozole increased, drug molecules would be combined by a certain ratio of poloxamer188, resulting in depletion of certain ratios of all incorporated Poloxamer188. Thus, another rest ratio of poloxamer188 for providing a route for small molecules passing through would be lesser. This is the cause of slower letrozole release rate under the condition.
To conclude, both incorporation content of poloxamer188 and letrozole are key factors to modulate the release profile of letrozole in poly L-lactic acid electrospun fibers.
Since the obtained results suggest that more poloxamer188 incorporation content could cause bigger release rates and amounts of letrozole, EFNWs of 125%, 150%, 175%, and 200% incorporation content of poloxamer188 were prepared and studied. The result indeed presented an increased letrozole release rate. However, when composite contents of poloxamer188 were 175% and 200%, EFNWs disintegrated into tiny pieces of fibrous slices, suspended in the release medium within 3 days only during the in vitro release experiment. Meanwhile, when composite contents of Poloxamer188 were 125% and 150%, EFNWs presented total integrity during the in vitro release experiment process. So, EFNWs with 125% and 150% poloxamer188 composite and 30% letrozole-incorporated (EFNW-30%-125% and EFNW-30%-150%) were studied for the in vitro sustained release experiment, and the result is presented in Figure 3. Release profiles of the two EFNWs were very desirable in that they were similar to EFNW-30%-125%, presenting low burst release contents and more than 1000 h sustained-release phases. EFNW-30%-125% and EFNW-30%-150% released letrozole more thoroughly than EFNW-30%-100% did. Morphology of EFNWs with more than 100% poloxamer188 incorporated (A, bar represents 30 μm) and their release profiles of EFNWs(B, absolute release content of mass; C, release percentage to total incorporated content)when letrozole incorporation content was 30% and poloxamer188 composite content was 100%, 125%, and 150%, respectively (error bars correspond to one standard deviation from three individual measurements).
In order to load more letrozole in electrospun fibers, we also tried to increase the drug incorporation content to above 40%, and it was discovered that the sustained release periods became longer and longer as letrozole incorporation content in fibers increased. However, the fibers’ morphology became hard to control by presenting beads and fibers of un-homogeneous diameters; moreover, the EFNWs’ mechanical strength became considerably weak, readily disintegrating when they were operated with hands. The phenomenon may be caused by higher droplet surface tension due to letrozole lipophilicity and small molecule-mediated polymer disentangling.
Based on the results presented above, we decided to choose EFNW-30%-150% as the objective to be studied by being implanted subcutaneously onto rabbits and detecting letrozole blood concentration during 2 months to investigate whether or not in vitro sustained release would bring about long-lasting existence of in vivo blood concentration.
Letrozole in vivo pharmacokinetics after subcutaneous implanting
Letrozole plasma concentration-time profiles of daily oral administration of SD-10%-100% and single-time subcutaneous implanted EFNW-30%-150% were drawn and compared along with pharmacokinetic parameters presented in Figure 4(a). The result suggests certain advantages of EFNW-30%-150% single-time subcutaneous implantation compared with SD-10%-100% daily oral administration. To be specific, the plasma concentration peak time of subcutaneous implanted EFNW-30%-150% was 36 h later (1.6 times later) than that of daily oral administration of SD-10%-100%. The maximum plasma concentration of subcutaneous implanted EFNW-30%-150% was 90.46% of that of daily oral administration of SD-10%-100%. All these suggest that EFNW-30%-150% have presented in vivo sustained release behavior to some extent. Area under curve (AUC) data shows that AUC0–360h of EFNW-30%-150% implantation was 99.29% of SD-10%-100% daily oral administration, suggesting that the former is comparable with the latter during 360 h (15 days, 0.5 months). There are still other advantages of EFNW-30%-150% compared to SD-10%-100% such as a faster plasma drug concentration escalating from 0 to 12 h, which may be due to the faster drug penetrating from subcutaneous soft tissues to capillary vessels than that from intestine epithelium in the gastrointestinal tract. Appearance of external skin surface after an EFNW-30%-150% subcutaneous implant on a female rabbit after 3 days (A-a), cut internal skin surface (A-b), and exposed EFNW30%–150% taken from dermis after 42 days’ release (A-c); Plasma concentration curves of letrozole and pharmacokinetic parameters of EFNW incorporated letrozole in female rabbits after daily oral administration and single-time subcutaneous implant of equivalent total content (B, error bars correspond to one standard deviation from five individual measurements).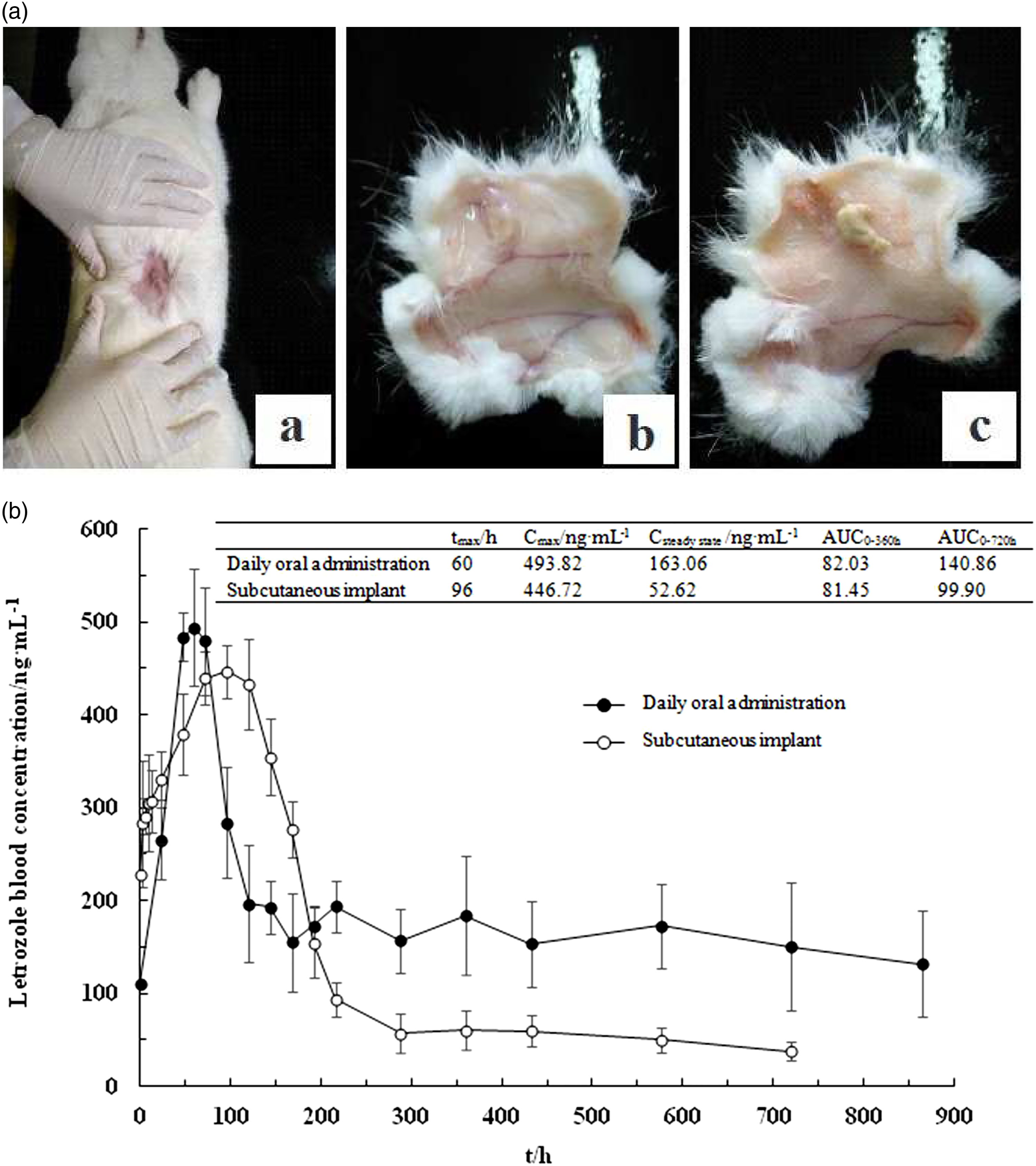
However, the disadvantages of EFNW-30%-150% subcutaneous implantation are also obvious. AUC0–720h of EFNW-30%-150% was only 70.92% to that of SD-10%-100%, and steady-state plasma concentration of EFNW-30%-150% was only 32.27% to that of SD-10%-100%. From in vitro release profiles, it is clear that EFNW-30%-150% had released 50% incorporated letrozole which would consequently penetrate from the implantation site into plasma and be distributed to organs immediately. Although letrozole has a considerable slow plasma clearance (distribution half-life > 2.5 days), the released letrozole from EFNW-30%-150% in the first 24 h would still be continually depleted. And the sustained release of another 50% letrozole during the next 1 month did not maintain plasma letrozole levels comparable with daily oral administration. Whereas, daily oral administration of SD-10%-100% would continually re-add letrozole amount day after day in plasma, and the plasma concentration exceeded EFNW-30%-150% implantation after 8 days and maintained a plasma concentration level higher than that of EFNW-30%-150% implantation by accumulation effect.
The next research work about delivering letrozole from EFNW in a desirable way should focus on reducing the release rate and amount within 24 h while keeping a considerable sustained release phase and complete release. This means that the release curve should bend more toward the time-axis by decreasing fast release amount in the former part of the release curve and increasing the lasting period and release amount of the latter part of the release curve. Consequently, continuous and more supplements would be provided to plasma day by day until reaching the level of SD-10%-100% daily oral administration.
Since the release profiles of EFNW were fitting well with Peppas’ equation, that the release mechanism followed a composite mode of diffusion and corrosion has been suggested. Both of the two modes are mostly due to poloxamer188’s effect. The drug passing through before releasing from PLLA matrix in form of being solubilized by the surfactant is the reason for the diffusion mechanism. Poloxamer188 itself was also one of the components in which the surfactant itself occupied considerable mass ratio in fiber matrix; thus, its release from PLLA matrix had resulted in considerable depletion in matrix mass, volume, and structure presenting the corrosion effect of the whole matrix. According to Peppas’ equation, increasing the value of bending factor λ could be the only certain method to decrease the value of pre-exponential factor constant k, and consequently, release curves would bend more toward the time-axis. To achieve this purpose, racemic poly lactic acids, such as copolymer of L-lactic acid and D-lactic acid, or even copolymers of racemic poly lactic acid and glycolic acid should be used to form fiber matrix of lower crystallinity degree for increasing λ value.
The appearance after EFNW-30%-150% subcutaneous implanting on female rabbits after 3 days is presented in Figure 4(A-a). No severe inflammation occurred and the only side effect rubefaction was caused by wound healing. After 42 days’ release, there was also no severe inflammation found by the naked eye which are presented in Figure 4(A-b) and (A-c). However, histopathology of various kinds of the skin tissue sections adjacent to the implantation site should be further studied.
Inflammatory reaction
In Figure 5, after 3 days of implantation, the inflammatory reaction caused by pure poly L-lactic acid cast film and the letrozole-incorporated fibers EFNW-30%-150% was stronger than that of poloxamer188 composite poly L-lactic acid cast film and electrospun fibers without incorporated letrozole indicated by more detected inflammatory cells accumulating at the contact position of the skin’s inner layer to the implants. The phenomenon may be caused by lower surface hydrophilicity of pure poly L-lactic acid cast film and EFNW-30%-150%. Meanwhile, poloxamer188 composite poly L-lactic films and electrospun fibers caused a smaller number of inflammatory cells, probably due to their better hydrophilicity and their flexibility and smaller microstructural scale. A) Pure poly L-lactic acid cast film, (B) poloxamer188-incorporated poly L-lactic acid cast film, (C) poloxamer188-incorporated poly L-lactic acid electrospun fibers, (D) poloxamer188 composite poly L-lactic acid electrospun fibers with letrozole-incorporated, EFNW−30%–150% (bars represent 200 μm).
After 7 days of implantation, the inflammatory cells still gathered in the contacting position of the inner skin and the pure poly L-lactic acid implant. And, poloxamer188 composite poly L-lactic acid film and electrospun fibers without incorporated letrozole showed increment of inflammatory cells. Especially, the number increment of inflammatory cells caused by poloxamer188 composite poly L-lactic acid film was significant. The main reason for this phenomenon may be that the hydrophilicity of the implant decreased after considerable percentage of poloxamer188 had released. Meanwhile, inflammatory cell reaction caused by EFNW-30%-150% was reduced to a considerable extent probably due to the improvement of its hydrophilicity caused by large amount of letrozole that had been released (52.5%) during this period.
After 28 days of implantation, the inflammatory cells caused by pure poly L-lactic acid film had penetrated into the internal place of skin, while the inflammatory cells caused by poloxamer188 composite film still had significant concentration in the contact position between inner surface of the skin and the implant. Inflammatory cells caused by poloxamer188 composite fibers, both with or without incorporated letrozole, decreased compared with the earlier stage.
The in vivo biocompatibility study revealed that poloxamer188 composite poly L-lactic acid electrospun fibers with incorporated letrozole (EFNW-30%-150% implants) induced considerable inflammatory reaction in the earlier stage and resulted in mild inflammatory reactions, by comparing the inflammatory reaction caused by films with or without incorporating Poloxamer188 and Poloxamer188 composite fibers with or without incorporating letrozole. Although hydrophobic letrozole incorporation could result in an inflammatory reaction, letrozole releasing-out from poloxamer188 composite electrospun fibers could increase hydrophilicity to ameliorate inflammatory reaction caused by the implant. Moreover, poloxamer188 composite and fibrous structure of EFNW-30%-150% also help to improve the effect of decreasing inflammatory reaction.
Conclusions
Microscale fibrous nonwovens with desirable morphology and poloxamer188-solubilized and sustained-release letrozole were released by electrospinning letrozole/poloxamer188/poly L-lactic acid homogeneous solutions of dichloromethane. All fibrous nonwovens released letrozole faster and more than cast films did when material composite ratios were the same. The effect of poloxamer188 and letrozole contents on release behavior has been revealed. Poloxamer188 could facilitate letrozole release, meanwhile letrozole content increase slowed down the release rate of itself. Fibrous nonwoven with 150% poloxamer188 and 30% letrozole loaded (EFNW-30%-150%) could release letrozole to 50% of its total loading amount after 1 d and nearly 100% of its total loading amount after 42 days. The sustained period could last nearly 1.5 months. After subcutaneous implanted EFNW-30%-150%, the female rabbit plasma concentration curve of letrozole concentration-time presented a later tmax, a lower Cmax, and a comparable AUC with equivalent amount of SD-10%-100% solution daily oral administration within the first 15 days. However, for the continued time period of more than 15 days, AUC of the subcutaneous EFNW implant could not be compared with that of daily oral administration. And the average steady-state plasma concentration of the EFNW subcutaneous implant could not be compared with that of daily oral administration. All these suggested advantages of EFNW as a subcutaneous implant compared with oral dosage form. Although EFNW-30%-150% caused inflammation, the inflammatory reaction would be alleviated within a week and remain for nearly a month. In summary, we think EFNW-30%-150% may serve as a potential candidate of a novel delivery system for 1-month sustained release of letrozole after single-subcutaneous implanting.
Further study and effort should focus on how to elevate the average steady-state plasma concentration. To achieve this object, efforts to increase letrozole content while decreasing Poloxamer188 content should be taken to attain less hydrophilic EFNWs. Thus, decreased burst release amount and release rate may be realized. Thus, release curves with smaller k values may be attained to realize much later tmax and much lower Cmax. And, increased letrozole content in EFNWs may also obviously cause a higher average steady-state plasma concentration.
This study has not only provided some useful information and encouraging results on how to modulate the release profile of water-insoluble drugs and successfully develop electrospun fibrous nonwovens as novel subcutaneous implants for sustained drug delivery, but also provided novel ideas for launching further research on how to select surfactants as functional additives for poly L-lactic acid electrospun fibers in pharmaceutical application.4,6,11,15,20,26
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is financially supported by director fund of Liaoning Provincial Key Laboratory of Marine Bioactive Substances and Technological Innovation Center of Liaoning Pharmaceutical Action and Quality Evaluation (2020-06, 2021-X) and Open project of scientific research platform in Shandong Academy of Pharmaceutical Sciences (SPKLBP201801).
