Abstract
Electrospinning is an inexpensive and versatile technique for fabricating micro- and nano- scaled fibers. There have been limited attempts to employ it for the fabrication of thermochromic (TC) fibers, and the fabrication of a three-component (dye, developer, and solvent) TC material has required the use of a more complicated coaxial electrospinning technique. Herein, a simple and novel method for creating thermochromic fibers by electrospinning single strands of poly (methyl methacrylate) (PMMA) with embedded thermochromic powder of a polymer encapsulated three-component system was employed. Unlike past leuco dye-based thermochromic fibers, an unmodified syringe tip can be used for the spinning process and only one flow rate needs to be determined. A solution of solvent (either N-dimethylformamide or chloroform), PMMA, and a commercially available black thermochromic powder was prepared and spun using a custom-made electrospinning apparatus. The spun fibers exhibited a clear color transition from grey to white and had average diameters of 2.53 µm and 1.96 µm for chloroform and N-dimethylformamide based fibers, respectively. The fibers were characterized by scanning electron and optical microscopy to determine their morphology, Fourier transform infrared spectroscopy to determine their chemical composition, and differential scanning calorimetry and thermogravimetric analysis to characterize their thermal properties.
Introduction
Functional materials, such as thermochromic (TC), electrochromic, and piezo electric materials have proven to be an interesting area of research for their versatility and inherent utility [1–3]. Thermochromic materials in particular are quite useful for their applications in energy saving coatings, sensors, and smart textiles [4–6]. Because of this, there is great interest in understanding more about their fundamental operating principles as well as their applications. Materials with an optical response to temperature change are quite varied as this property can be found in inorganic metal oxides such as VO2 as well as conjugated polymers, liquid crystals, and quantum dots [5]. However, many thermochromic materials are quite impractical due to their high cost, operating temperature, or difficulty in manufacturing. Leuco dye-based materials do not suffer these drawbacks as they are inexpensive and easy to produce [5]. Because of this, they are often used in commercial products such as thermochromic inks and novelty items [7].
Leuco dye based thermochromic materials normally consist of a pH sensitive organic dye, a weakly acidic color developer, and an organic solvent. When the solvent changes phase, it allows for electrons to transfer between the dye and developer, leading to a change in the chemical structure of the dye and thus, a change in the observed color [8]. However, because this mechanism is dependent upon the phase change of the solvent, the thermochromic material must be encapsulated in order to be used for applications like thermochromic inks or textiles, as the solvent most often will be changing between solid and liquid phase for this reaction to occur [9]. Most commercially available thermochromic dyes are sold as powders whose individual particles are polymer encapsulated through complex coacervation (using gelatin and gum arabic) or interfacial polymerization (with melamine or urea formaldehyde) [10]. If they are not encapsulated already, their incorporation into their intended application must be done in a manner to prevent the loss of solvent to melting or evaporation.
In order to be applied to fabrics, encapsulated leuco dyes are traditionally applied by mixing the dye with a binder, often with a standard acrylic printing paste. While this does lead to a visual color change, it also leads to dilution of the final color as well as longevity issues, since the dye itself is subject to degradation through normal usage and washing out due to being on the exterior of the fibers [10]. A method of alleviating this is by incorporating the dye into the fiber, which has been done through both melt spinning and electrospinning using various types of thermochromic materials [11–16]. The melt spinning process, while studied more, involves more expensive, difficult to use equipment operating at high temperatures. For lab settings, it is easier and less costly to custom build an electrospinning apparatus to spin fibers. In our previous research efforts, the authors have successfully electrospun polyaniline fibers for storing hydrogen reversibly and released the stored hydrogen via the proton exchange membranes in hydrogen-fuel cells [17].
Electrospinning is a process that uses a high voltage between a metal syringe containing a polymer solution and a conductive substrate to slowly draw-out micro- or nano-scale fibers. In addition to being able to control the fiber morphology by varying the solvent and changing other parameters such as voltage, flow rate, and separation between the syringe tip and substrate, the method is also relatively inexpensive and can be used on an industrial scale [18]. Electrospun fibers have been used extensively for tissue scaffolding, drug delivery, battery electrodes, and to create UV protective composites [18–20]. The ease of use and cost effectiveness makes it a viable approach for this intended application.
Examples exist of both coaxially spun thermochromic fibers as well as fibers electrospun with embedded thermochromic materials [13–16]. In co-axially spun fibers, a co-axial needle is employed with a polymer solution flowing through the outer layer and a liquid or dissolved solution of thermochromic material in the inner layer of the needle. This is, however, a difficult process to control as co-axial spinning requires the fine tuning of two different flowrates and solutions, making it susceptible to damage of the TC core material if the fiber breaks, as well as potential chemical reactions between the shell layer and thermochromic core layer [14]. Instead, it is possible to embed thermochromic particles directly inside the fiber. This allows for a much simpler spinning procedure as well as potentially improved longevity as there is more protection for the particles themselves. Embedded thermochromic fibers have been produced with liquid crystal and inorganic metal oxide based thermochromic materials before but have yet to be produced with an embedded leuco dye-based material [15,16]. Besides being incorporated into clothing for their visual change, these leuco-dye based thermochromic fibers can be used as phase change materials for energy storage and thermal insulation, with potential applications including heat resistant clothing for fire fighters and temperature sensitive sensors [15,21].
In this work, thermochromic fibers embedded with a leuco dye-based thermochromic powder were successfully synthesized through electrospinning and the effects that different solvents have on the fabrication of these fibers was investigated. Based on previous studies, N,N-Dimethylformamide (DMF) and chloroform were chosen as solvents and Poly-(methyl methacrylate) (PMMA) was chosen as the polymer to be spun because of its low cost and transparency in the visible spectrum [22–24]. The spun fibers produced were characterized for their surface, interface, chemical, optical, thermal properties, and the correlated color change behavior.
Experimental details
Materials used
Poly(methyl methacrylate) (PMMA, Mw = 120,000 g/mol), N,N-Dimethylformamide (DMF), and chloroform were purchased from Sigma-Aldrich. Black thermochromic pigment with a color change temperature of 31°C was purchased from Uniglow. The above materials were used without further purification.
Fabrication of thermochromic fibers
The thermochromic dye was mixed with chloroform or DMF for one hour using a magnetic stirrer at ambient temperature. PMMA was then added to the solution and allowed to dissolve for an additional 24 hours. The concentrations of thermochromic dye and PMMA were kept at 2 wt.% and 8 wt.%, respectively. These solutions were then loaded into a 3 mL syringe and electrospun with a custom-made electrospinning apparatus (see Figure 1) with flow rate and voltage set to 0.5 mL/hr and 13 kV, respectively. These values were based on previous work with PMMA fibers and were optimized to prevent unspun droplets and obtain a stable polymer jet [16,25]. An aluminum foil substrate was used to collect the fiber 15 cm away from the syringe tip.
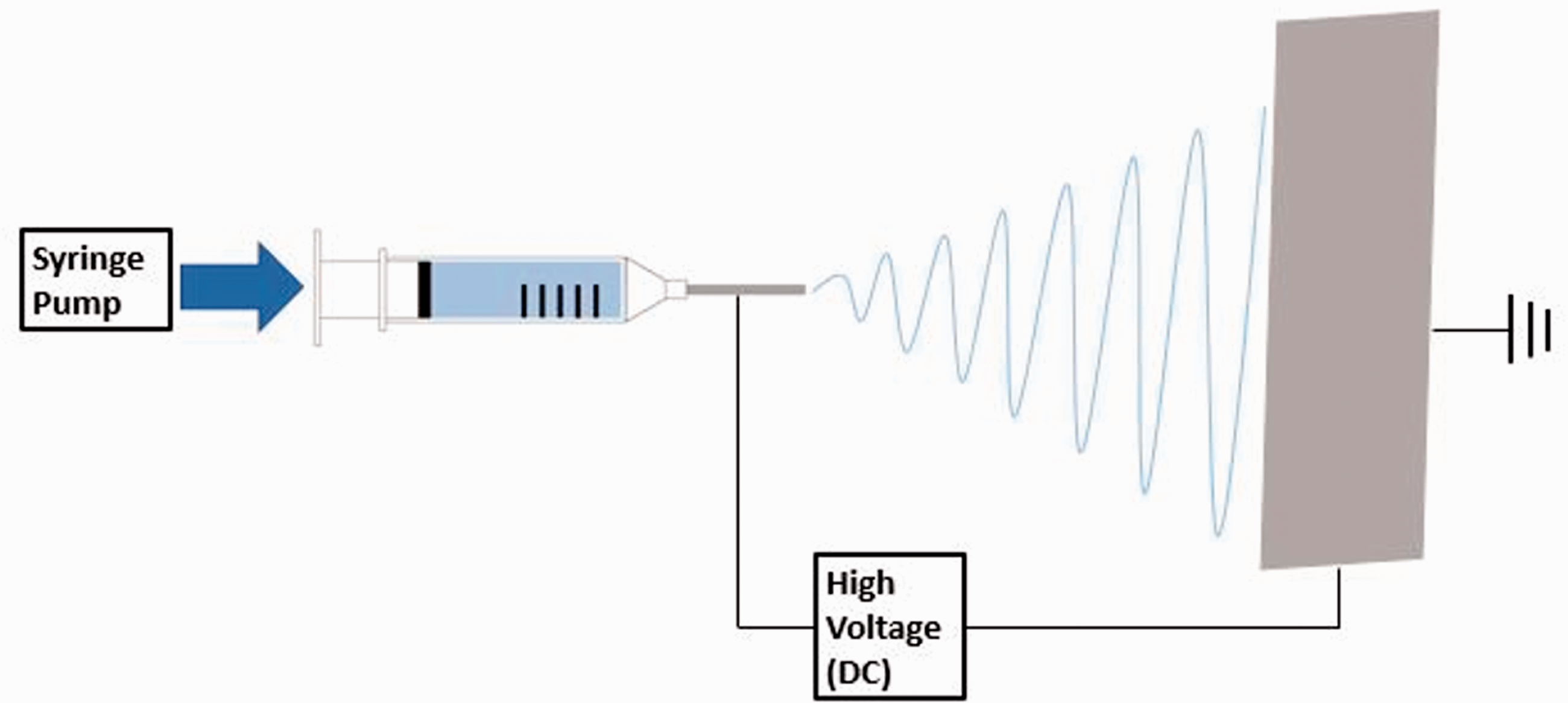
Schematic arrangement for electrospinning of fibers.
Characterization
The surface morphology was analyzed by scanning electron microscopy (Hitachi SEM SU-3500) and optical microscopy (Ziess Axio Vert.A1). For the SEM studies, a voltage of 25 kV, current of 80–90 µA, a spot size of 30–40 µ, objective aperture of 2 and a working distance of 5 mm were selected for optimum imagery. To determine average diameters of fibers and dye, 100 measurements were made and averaged using imageJ software. Fourier transform infrared spectroscopy (Nicolet iS50 FTIR) was performed to determine the bonding environment. For the FTIR experiments, an ATR cell was used for the fibers placed on the diamond window and applied a pressure using a screw gauge. The transmission mode was chosen to collect IR signals between 400 to 4000 cm−1 wavenumbers. The thermal properties of these fibers were analyzed by differential scanning calorimetry (TA Instruments DSC 2500) and thermo-gravimetric analysis (Mettler Toledo, TGA/DSC 1 STARe system). For the TGA studies, the fiber sample size of <10 mg was treated from room temperature to 600°C with a ramping rate of 10°C/min under nitrogen flow of 50 mL/min. To determine the visual color change, fibers were placed on white filter paper on top of a hot plate. The hot plate's temperature was adjusted in increments of 5°C while the actual temperature of the fibers was determined with an infrared thermometer. Every 15 minutes, a photo was taken and cropped to obtain an image of the color. No filtering or other tools were deployed on these photographs, to capture the actual color change due to thermochromism.
Results and discussion
Fiber morphology
The thermochromic dye encapsulated polymer fibers with a visible color change were successfully spun by the electrospinning method. The mats formed contained webs of fibers as seen in SEM images, with diameters ranging from 1.44 µm to 4.47 µm and an average diameter of 2.53 µm for fibers spun with chloroform as a solvent (Figure 2(a) and (b)), as well as fibers with diameters ranging from 0.95 µm to 4.40 µm and an average diameter of 1.96 µm for fibers spun with DMF as a solvent (Figure 2(c) and (d)).

SEM images of (a, b) chloroform fibers, (c, d) DMF fibers, and (e,f) unembedded TC dye.
Regardless of the solvent used, a beaded morphology could be observed for the fibers spun with this method as demonstrated in the SEM images of Figure 2(a) to (d). The beads possessed an average diameter of 4.25 µm in the fibers spun in chloroform and 3.34 µm in the fibers spun with DMF. These beads are likely entrapped thermochromic dye particles, due to their shape and the larger average diameter of the unembedded dye particles of 3.86 µm (Figure 2(e) and (f)). A similar morphology has been observed for electrospun fibers with embedded, encapsulated liquid crystal thermochromic materials, with a similar mismatch in fiber and dye diameters [15]. While it is possible these beads are occurring as a result of the electrospinning process itself (as beads tend to form for lower wt% polymer solutions), the optical microscope images tend to support the argument that these beads were a result of the embedded dye [18]. The optical microscope image (Figure 3(a)) of the fibers spun with chloroform as a solvent show the dye particles are entrapped throughout the fibers, which are optically transparent due to being constructed from PMMA. They are relatively evenly distributed, while the fibers themselves are randomly oriented throughout the matt. For fibers spun with DMF as a solvent (Figure 3(b)), some dye particles can still be seen in the optical microscope image, but they are far less concentrated when compared to the fibers spun with chloroform.

Optical Microscope images of (a) chloroform fibers and (b) DMF fibers at 10× magnification.
IR spectra
FTIR on the fibers (Figure 4) showed several peaks between 2000 cm−1 and 1000 cm−1 that can be attributed to PMMA, particularly the 1446 cm−1 peak for the carbonyl group. Peaks between 2916 cm−1 and 2922 cm−1 present on both types of fibers and the unembedded commercial thermochromic can be attributed to the dye, as well as the 967 cm−1 peak present in all samples. Various functional groups of bending and stretching modes that are assigned for the FTIR spectra of the as-spun fibers are shown in Table 1 [26].

FTIR spectra of unembedded dye as well as fibers spun with chloroform and DMF.
FTIR spectral peak’s wavenumbers and assigned functional groups.

Thermal properties
The thermochromic dye’s color change occurs due to the three-component system’s solvent phase change from solid to liquid, allowing the dye and developer to interact and change color. Differential scanning calorimetry (DSC) was employed to characterize the candidate materials such as unembedded TC dye, and the TC fibers spun using DMF and chloroform. At the outset, all the samples exhibited reversible thermal behavior in both heating and subsequent cooling cycles. The parameter related to temperature of endothermic melting transitions during heating run, exothermic crystallization temperature in cooling run and their corresponding enthalpy values are given in Table 2. The following interpretations of DSC thermal characteristics (Figure 5(a)) are now discussed in detail and correlated with the visual color change properties as discussed below.
Thermal values of plain TCM and DMF/chloroform encapsulated TCM fibers.


DSC profiles of (a) unembedded TC dye, (b) fibers spun with chloroform, and (c) fibers spun with DMF (DSC peak down is endothermic phase transition). (d) TGA profiles of all three samples.
The unembedded TC dye undergoes solid to solid phase transition and solid to liquid phase transitions (or melting) at around 29.2°C and 32°C with enthalpy values of 15.1 J/g and 81.8 J/g, respectively. In the cooling phase, the plain dye demonstrates the high degree of reversibility with an enthalpy value of 88.9 J/g for its liquid to solid phase transition (or crystallization) at around 26°C.
The phase transition temperatures during heating cycle of the TC fibers made from chloroform and DMF were 30.3°C and 30.8°C respectively, which are slightly higher than that of the unembedded TC dye. Contrarily, the crystallization temperatures of the TC fibers (which were between 18.3°C and 23.5°C for fibers spun with chloroform, and around 25.9°C for those spun with DMF) are lower than the unembedded TC dye (26°C). Therefore, the difference in the phase transition temperatures thus ensures embedding of the TC dye particles within the fibers.
The enthalpy values of melting drastically decreases for the fibers spun with chloroform and DMF (17.0 J/g and 2.1 J/g respectively) than the unembedded TC dye (81.8 J/g), due to the lower amounts of dye encapsulated by the fibers (roughly, chloroform ∼ 1/10th and DMF ∼1/100th concentration of the plain dye). These lower concentrations of dye encapsulation are due to the virtue of adhesion properties of chloroform and DMF solvents during the electrospinning process.
However, the reversibility in terms of crystallization while cooling was observed with an enthalpy value restored, for both chloroform (between 18.3 J/g and 23.5 J/g) and DMF (2.3 J/g) enabled TCM fibers. This reversibility of endothermic melting and exothermic crystallization behavior in fact reveals the thermal storage of the electrospun fibers in addition to their textile fabrics applications.
Yet another correlation is unambiguously visible that the lower concentration dye particles embedded by the DMF fibers (∼1/100th of plain dye) with the lower enthalpy of 2.1 J/g when compared to the chloroform encapsulated fibers (1/10th of plain dye) with an order of magnitude increase in the enthalpy of melting (between 18.3 J/g and 23.5 J/g) thus directly impact the visual color change behavior which is discussed in the next section.
TGA was performed on the fibers and unembedded commercial dye as shown in Figure 5(d). Again, as the fibers are primarily PMMA, there was a noticeable change in the characteristics of the mass losses likely due to the insulation of the polymer as well as the much smaller amount of thermochromic material present for the same mass of material. For both the unembedded dye as well as the fibers spun with chloroform, the onset of mass loss occurred at a similar temperature of 209°C, however the second dip in the TGA curve at 230°C is no longer visible for the fibers spun with chloroform. For the fibers spun with DMF, an initial mass loss can be observed at 210°C and a much larger loss at >250°C. This first dip can likely be attributed to residual DMF, as the solvent has a lower vapor pressure than chloroforms (3.8 mmHg vs 169 mmHg at 21°C) and evaporates at 153°C, roughly the same temperature as the initial mass loss.
Visual color change
In addition to thermal studies discussed above, color change transitions during the thermal treatment were observed and photographed for both types of fibers as well as the unembedded dye (Figure 6). The temperatures of the thermochromic materials ranged from 25°C to 40°C in increments of 5°C and provide qualitative insight as to the differences in color. It is apparent that the DMF fibers are lighter in color than either the chloroform or unembedded dye, while the chloroform fibers manage to maintain clear color contrast throughout this temperature range. This difference in color contrast can likely be attributed to the difference in amount of embedded TC dye particles as noted from the DSC results. A greater concentration of dye will lead to a color closer to that of the unembedded TC dye.

Color change behavior of plain dye and fibers made from chloroform and DMF.
Conclusions
In this study, thermochromic fibers were successfully prepared via electrospinning a solution of PMMA, commercial thermochromic dye, and either chloroform or DMF as a solvent. Fibers spun with chloroform as a solvent showed a larger average diameter, clearer color contrast, and more pronounced phase change properties when compared to fibers spun with DMF. DSC analysis showed that the fibers began to both change color around 30°C and the chloroform fibers showed higher enthalpies of phase change when compared to fibers spun with DMF. Both fibers displayed a smooth but beaded morphology that can likely be partly attributed to the incorporation of dye particles with larger diameters than the fibers themselves.
Footnotes
Acknowledgements
The authors gratefully acknowledge the DSC support provided by Yassin Kocak at the University of South Florida and the optical microscope support by Professor Richard Matyi at Florida Polytechnic University.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
