Abstract
Flexible sensors and wearable electronics have become important in recent years. A good conductive and flexible textile is needed to develop a commercial wearable device. Conductive polymers have generally been used with limitation in reducing the surface resistance to a certain amount. In this research, a method for fabricating a stretchable highly conductive cotton/lycra knitted fabric is introduced by treating the fabric with polypyrrole (PPy), silver nanoparticles (SNPs) composites, and post-treating with poly (3,4-ethylenedioxythiophene) poly (styrenesulfonate) (PEDOT:PSS). Polypyrrole and SNPs were in situ fabricated on the cotton/lycra fabric by consecutive redox reaction of silver nitrate and pyrrole and finally covered by PEDOT:PSS solution through dip-coating. The coated textile was characterized by Fourier transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), field emission scanning electron microscopy (FESEM), X-ray mapping, and energy dispersive X-ray spectroscopy (EDX). Fourier transform infrared spectroscopy confirmed PPy-SNPs (P-S) composites on the fabric surface. Fourier transform infrared spectroscopy results, X-ray mapping, EDAX, and XRD analysis also confirmed the P-S composites and PEDOT:PSS polymeric layer on the fabric. Morphological observation showed a layer of PEDOT:PSS on the P-S caused the higher connection of coating on textiles which resulted in the higher electrical conductivity (43 s/m). Also morphological observations showed penetration of the silver particles inside fibers which represented improving in attachment and stability of the coating on the fibers. Further, the electrical conductivity of PPy-SNPs-PEDOT:PSS coated textile increased under the tension. Hence, the stretchable and highly conductive knitted cotton/lycra fabric has potentiality to be used for fabricating the flexible sensors or wearable electronics.
Introduction
Electronic textiles are known as the substrates with electronic characteristics that may be attached to the textile as various accessories. These textiles with dimensional flexibility cannot be produced with other existing electronic substrates. In other words, fabricating the electronic textiles is converging textiles and electronics to develop smart materials with a wide range of functions that usually found in non-flexible and rigid electronic devices. 1 Since most of textiles are inherently insulating materials, the electrical conductivity of a fabric is the most crucial factor to be used in the flexible electronic device. Hence, much attention has been paid to fabricate the durable and flexible conductive textiles.
The use of nanomaterials to fabricate the conductive textiles in the electronics industry, flexible monitors, healthcare displaying systems, temperature and humidity sensors, flexible energy storage devices, and power generation devices is rapidly developing. In order to impart the conductivity on textiles, the use of materials such as metal nanoparticles, nanowires, conductive polymers and carbon nanomaterials have been considered by researchers. In many published reports, flexible wearable device made of conductive metallic nanomaterials and conductive polymers have demonstrated many advantages such as high electrical conductivity, low cost, good comfort, appropriate flexibility, stretch ability, capability to sense, display, and energy storage. Among conductive polymers, polypyrrole (PPy) is one of the most widely used with optimal electrochemical properties, excellent conductivity, simple synthesis process, environmental friendliness, and good chemical and thermal resistance.2–8
In situ polymerization is used for developing polymer nanocomposites from nanoparticles. It usually occurs in polymerization mixture and comprises an initiation step, followed by a series of polymerization process, in which a hybrid is formed between polymer molecules and nanoparticles. For accomplishing the polymerization, an adequate initiator is added and in most cases it is exposed to a source of heat or radiation. 9 Polymer-metal nanoparticles composites have demonstrated improved physicochemical and biological properties compared to their individual components. Nanocomposites composed of silver nanoparticles (SNPs) and conducting polymers, especially PPy, have shown unique properties such as high conductivity and electro catalytic activity which makes them very ideal for application in sensors and electronic devices. 10 There are few reports on coating textiles with PPy-SNPs nanocomposites. Babu et al. (2012) fabricated multifunctional cotton fabric by coating with PPy-SNPs nanocomposite. The electrical conductivity of the treated cotton was reported to be about 4.83*10−3 (s/cm) meanwhile the fabric had strong antibacterial activity against microorganisms. 10 Parvinzadeh Gashti et al. (2014) reported high electromagnetic shielding of the wool fibers coated with PPy-SNPs nanocomposites. They were also claimed that nanocomposites improve the thermal resistance of the textile. 11 Electromagnetic shielding of polyethylene terephthalate (PET) nonwoven textiles coated with PPy-SNPs was reported by Erdoĝan et al. (2017). In this research, surface resistivity and electromagnetic shielding effectiveness value (EMSE) of the treated nonwovens were 15 Ω/cm2and 13–15.5 dB (frequency range of 15–3000 MHz), respectively. 12 Surface resistivity of 35 Ω-cm was also reported for the cellulosic textiles coated with PPy-SNPs through one pot photopolymerization. 13
Due to some limitations such as surface roughness on the textiles, it is impossible to form a uniform coating on the surface for achieving high electrical conductivity by PPy-SNPs (P-S) nanocomposites. However, the textile substrate should be highly electrical conductive for many practical applications such as supercapacitors or solar cells. To overcome these limitations, here for the first time, surface of the cotton/lycra knitted fabric as a highly flexible textile was treated by a novel ternary structure including P-S and poly (3,4-ethylenedioxythiophene) polystyrene sulfonate (PEDOT:PSS). By introducing a conductive polymer PEDOT:PSS to the P-S nanocomposites, the fabric surface roughness was expected to be decreased. The PEDOT:PSS could fill the gaps between the fibers or parts which were not covered by P-S. Also, due to the electrical conductivity of PEDOT:PSS, it was expected that the electrical conductivity increase. Following this approach, the coating stability can also improve. During the in situ polymerization, silver ions, in addition to oxidizing pyrrole, were able to react with negatively charged cellulose hydroxyl groups. The ionic interactions increased the adhesion and stability of the coating on the fabric.
Materials and methods
Materials
All the reagents were used as received except pyrrole which was purified by vacuum distillation (60°C and 40 mbar pressure) and kept in dark at 5°C. Silver nitrate (99% purity), pyrrole monomer, absolute ethanol, isopropanol, and acetone were purchased from Merck Chemical Co., Germany. Poly (3,4-ethylenedioxythiophene) polystyrene sulfonate solution (5% wt.) was provided from Sigma Aldrich Co., Germany. The cotton/lycra rip 1*1 knitted fabric (230 g/m2) was supplied by Kavir Semnan (Iran).
Methods
In situ polymerization of P-S nanocomposites
PPy-SNPs nanocomposites were coated on the fabric through in situ polymerization using silver nitrate as oxidant for pyrrole monomers. In turn, the pyrrole acted as a reductant for silver ions reduced them to SNPs. Various molar ratios of silver nitrate and pyrrole were mixed in presence of cotton/lycra knitted fabric. First, pyrrole (0.3, 0.5, and 0.7 M) was added to 100 mL distillated water containing a piece of cotton/lycra fabric (3
For preparing the PPy coated cotton/lycra samples, all the procedure was similar to the in-situ polymerization of P-S, except using ferric chloride as the oxidant instead of silver nitrate.
Preparation of PPy-SNPs-PEDOT:PSS cotton/lycra fabric
The P-S coated fabric was covered by poly (3,4-ethylenedioxythiophene) poly (styrenesulfonate)(PEDOT: PSS) solution with different concentrations (0.3, 0.5, and 0.7 M) through dip-coating method for 3 h. Finally, the samples were washed 3 times with distilled water and dried in room temperature for 5 h.
Characterization
Four-probe instrument was employed for measuring the conductivity of pristine and treated cotton/lycra samples using Keithley 2400 source meter. For measuring the conductivity, the samples were cut in 2*0.5*0.01 cm (Figure 1) and a constant current was applied. The distance between each probe was also adjusted to 1.5 mm. For each sample, electrical conductivity was measured in three directions: course, wale, and diagonal, and the average values were reported. Measuring the electrical conductivity of samples by four-point probe method.
The morphology of fabrics and fibers cross-section were observed with a field emission scanning electron microscopy (FE-SEM; Mira3; Tescan; Czech Republic). This equipped with energy-dispersive X-ray spectroscopy (EDX) for elemental analysis. To investigate the chemical structure of the samples, Fourier transform infrared spectroscopy (FTIR) was performed by Nicolet Nexus 670 spectrophotometer. In order to investigate the crystalline structure, X-ray diffraction (XRD) patterns were collected on Rigaku Smart lab device using Cu Kα radiation at a wavelength of 0.154 nm in the range of 5 and 80°.
Stability of the coatings on cotton/lycra surface was investigated in water, acetone, and isopropyl alcohol (IPA) by immersing the samples in the solution at room temperature for 12 h. After immersion, leaching of the coating from the samples was assessed.
Electro conductive stability of the treated cotton/lycra fabrics was measured by subjecting the samples to 100% stretching relative to their original length with several cycles (25, 50, 75, and 100). The specimens were being stretched and a certain number of loading cycles were applied then surface resistivity was measured by a digital multi meter (Thurlby 1503). The electrical conductivity was calculated according to equation (1)
Results and discussion
Formation of P-S coating on cotton/lycra
Figure 2 shows the FTIR spectra of raw and P-S coated cotton/lycra fabrics. For the raw fabric, peaks at 1645, 1431, and 3477 cm−1 can be attributed to deformation vibrations of H-O-H (absorbed water), CH2, and hydroxyl groups of the cellulose, respectively.
10
The peaks at 2922, 2852, 1220, and 1103 cm−1 can be related to lycra fibers.14,15 By increasing the pyrrole/silver nitrate concentrations in PPy-NPS coated samples, the intensity of related peaks to cellulose/lycra functional groups become lower due to more compact layer of P-S on the surface of fibers. The peaks at 1030–1050 cm−1 were assigned to C-H stretching of PPy.
10
At the highest concentration (0.7 M pyrrole), the peak at 1434 cm−1 can be related to C-N ring stretching vibrations of PPy. Also the peak at 1742 cm−1 can be assigned to carbonyl groups of PPy resulting from nucleophilic attack of water during polymerization.10,13 Further, the peaks at 1368–1397 cm−1 can be identified in plane deformation vibrations of N-H bond in PPy.
10
Hence, the FTIR spectroscopy confirmed polymerization of PPy on cotton/lycra and the action of silver nitrate as an efficient oxidant for pyrrole. Also the characteristic peaks of PPy are more prominent with increasing of pyrrole and silver nitrate during in situ polymerization. This indicates the formation of more compact layer of P-S on cotton/lycra fabric. ATR-FTIR spectra of raw (a) and P-S coated cotton/lycra fabric with 0.3 M (b), and 0.7 M (c) precursor concentrations.
Formation of P-S composites on cotton/lycra was further justified by XRD analysis (Figure 3). For all samples, the peak at 22° was assigned to the amorphous PPy.
10
This may also be attributed to (200) planes of the cellulose. However, superimposing the PPy peak with the cellulose confirmed successful covering of cotton/lycra with PPy. The other peaks located at 38, 44, 64, and 77° were related to (111), (200), (220), and (311) brag reflections of the face center cubic silver planes, respectively.
16
These peaks showed the incorporation of SNPs in the PPy matrix. It should be noted that, the XRD pattern of coated sample with 0.7 M P-S was presented due to the similarity. XRD pattern of the P-S coated cotton/lycra fabric.
Changes on the fabrics weight, before and after coating with P-S or P-S-P nanocomposites (samples dimensions: 1*1*0.05 cm).
Morphological observations of P-S-P coated cotton/lycra fabric
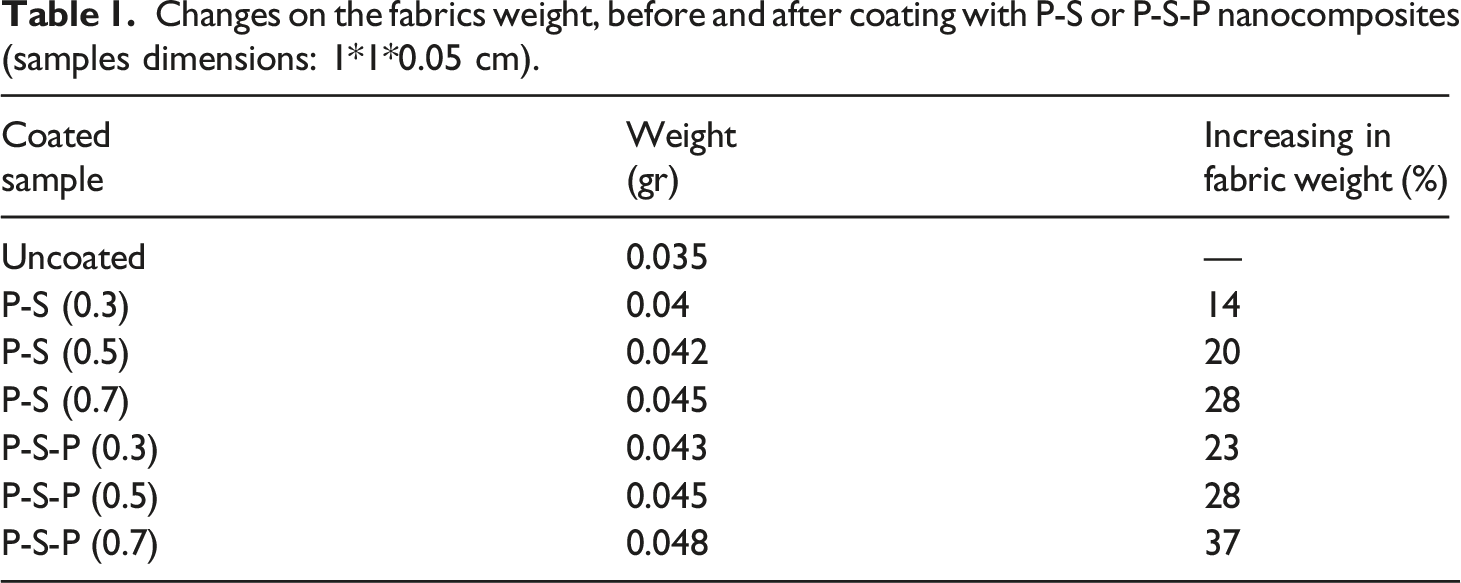
Formation of P-S-P composites on cotton/lycra fibers was confirmed by FESEM images (Figure 4). The neat and smooth surface of cotton/lycra fibers with fibril structure was clear for the raw sample (part (a)). In case of PPy coated sample, a thin and uncompact layer formed (part (b)) and the samples coated with P-S-P nanocomposites, rougher fiber surface can be observed. By comparing the raw and P-S-P coated samples at lower magnification (parts (c) and (d)), the lower porosity can be seen. The raw sample is porous with lots of space between the fibers. In the case of P-S-P coated sample, in addition to the surface, the nanocomposite was also penetrated into the spaces between fibers resulted in lower fabric porosity. In case of P-S-P coated samples, more pyrrole and silver nitrate during in situ polymerization, led to the denser coating. As can be seen in part (h), the fiber surface was completely covered by an even layer of the nanocomposites (at 0.7 M). During in situ polymerization, bounded pyrrole monomer on cotton/lycra surface was polymerized and formed a thin layer on the fibers. The SNPs in the following step deposited on the PPy layer through absorption or electrostatic interactions. As the concentration of pyrrole and silver nitrate increased through in situ polymerization, formation of the silver clusters on the PPy increased as well. This could also be seen in the EDAX elemental analysis (Table 2 and Figure 5). Increasing in pyrrole and silver nitrate from 0.3 to 0.7 resulted to 57% increase in silver content. The morphology and shape of PPy oxidized with iron chloride had no difference with those samples in situ polymerized with the silver nitrate. According to cross-sectional image (Figure 4, part (h)), it was clear that samples coated with P-S-P, in addition to the surface, silver also penetrated into the fibers (white dots represent penetration of silver into the fiber). This was due to electrostatic interactions between silver ions and hydroxyl groups of cellulose. It was reported that cellulose improves generation of SNPs due to extensive presence of hydroxyl groups. The cellulose hydroxyl groups serve as nucleation points for SNPs through ion-dipole interactions.
17
During the in situ polymerization, silver ions, in addition to oxidize pyrrole monomers, possibly form ionic interactions with negatively charged hydroxyl groups of cellulose and penetrated into the fibers. FE-SEM images of raw (a) and PPy (b) or P-S-P coated cotton/lycra with different concentrations: 0.3 M (c), 0.5 M (d), and 0.7 M (e). Cross-section of P-S-P coated fiber (f) EDX spectra of the coated textiles with 0.3, 0.5, and 0.7 M P-S-P nanocomposite. Elemental analysis based on EDX for cotton/lycra fabrics coated with P-S-P with different concentrations. Elemental mapping of P-S-P coated fabrics with different concentrations for carbon (blue), silver (red), and sulfur (orange).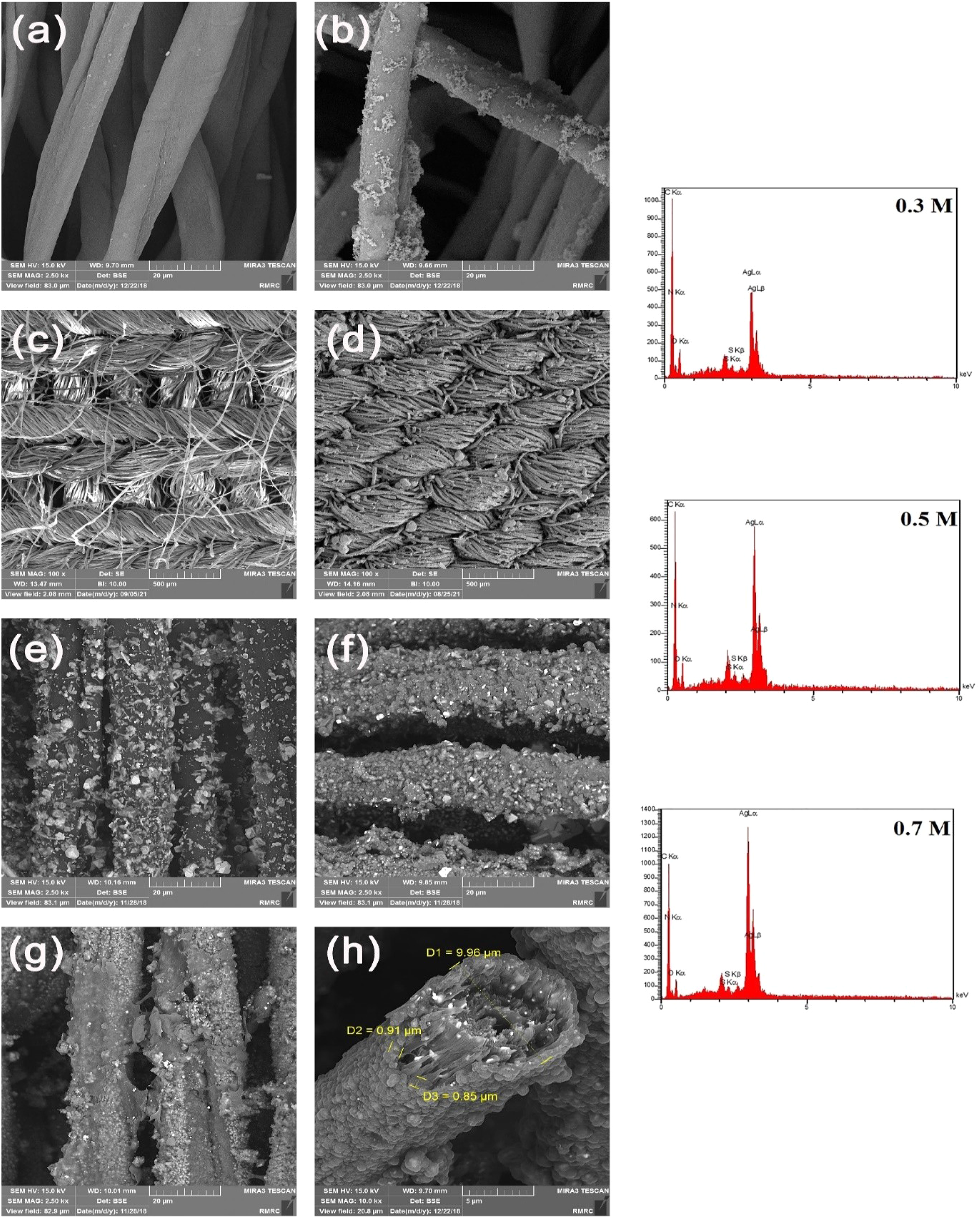


Mapping images in Figure 5 proved the coating of cotton/lycra fibers with PEDOT:PSS due to the sulfur atoms (orange dots). After treating the P-S coated cotton/lycra with PEDOT:PSS, the polymer can fill the gaps between the fibers. Also, by increasing in concentration of the precursors to 0.7 M, the distribution of sulfur atoms, related to PEDOT:PSS, between the fibers increased and filled the empty spaces between the fibers with PEDOT:PSS. These images also showed that the compactness and distribution of the silver atoms were higher than that of nitrogen (PPy) indicated location of the SNPs on the PPy surface. Further, increasing the concentration of precursors during in situ polymerization led to the more distribution of silver atoms (red dots) on the surface. Hence, the sample coated with P-S-P at 0.7 M showed the denser silver NPs on the surface according to the EDAX results.
The formation of P-S-P composites on cotton/lycra surface can be proved by the morphological observations with FESEM. The silver NPs are mostly formed over PPy layer however they may have penetrated inside fibers as a result of ionic interactions between silver and cellulose hydroxyl groups which may increase the strength of the coating. In addition, FESEM images confirmed formation and attachment of PEDOT:PSS over P-S layer.
Electrical conductivity
Conductivity of cotton/lycra fabrics coated with P-S and P-S-P with different concentrations.

The P-S nanocomposites formed on cotton/lycra through redox reaction between pyrrole and silver nitrate. In this process, pyrrole as reducing agent diffused into the solution or on the substrate and the polymerization started after introducing the silver nitrate as the oxidizing agent. Due to proximity of pyrrole standard oxidation potential with silver standard reduction potential (0.8 V vs NHE), silver can act as a strong oxidizing agent for pyrrole.
10
During PPy polymerization, the polymer will be doped by nitrate ions (NO3−) of silver nitrate, which became a conductive material. Moreover, the SNPs were intrinsically had strong conductivity; thus, the P-S coating acted as a very efficient conductive covering to turn insulating cotton/lycra fabric into a highly electro conductive substrate. High conductivity of the P-S coated textile indicated effective oxidation of PPy by silver nitrate and also efficient polymer doping with NO3− ions. For these samples, increasing in pyrrole and silver nitrate concentrations caused the higher electrical conductivity up to 116%. Further increase of precursors had no effect on the electrical conductivity. The data related to the samples coated with P-S nanocomposite at higher pyrrole and silver nitrate concentrations were not reported due to the same electrical conductivity values. One of the most important limiting factors in increasing the electrical conductivity is the fabric surface roughness.
22
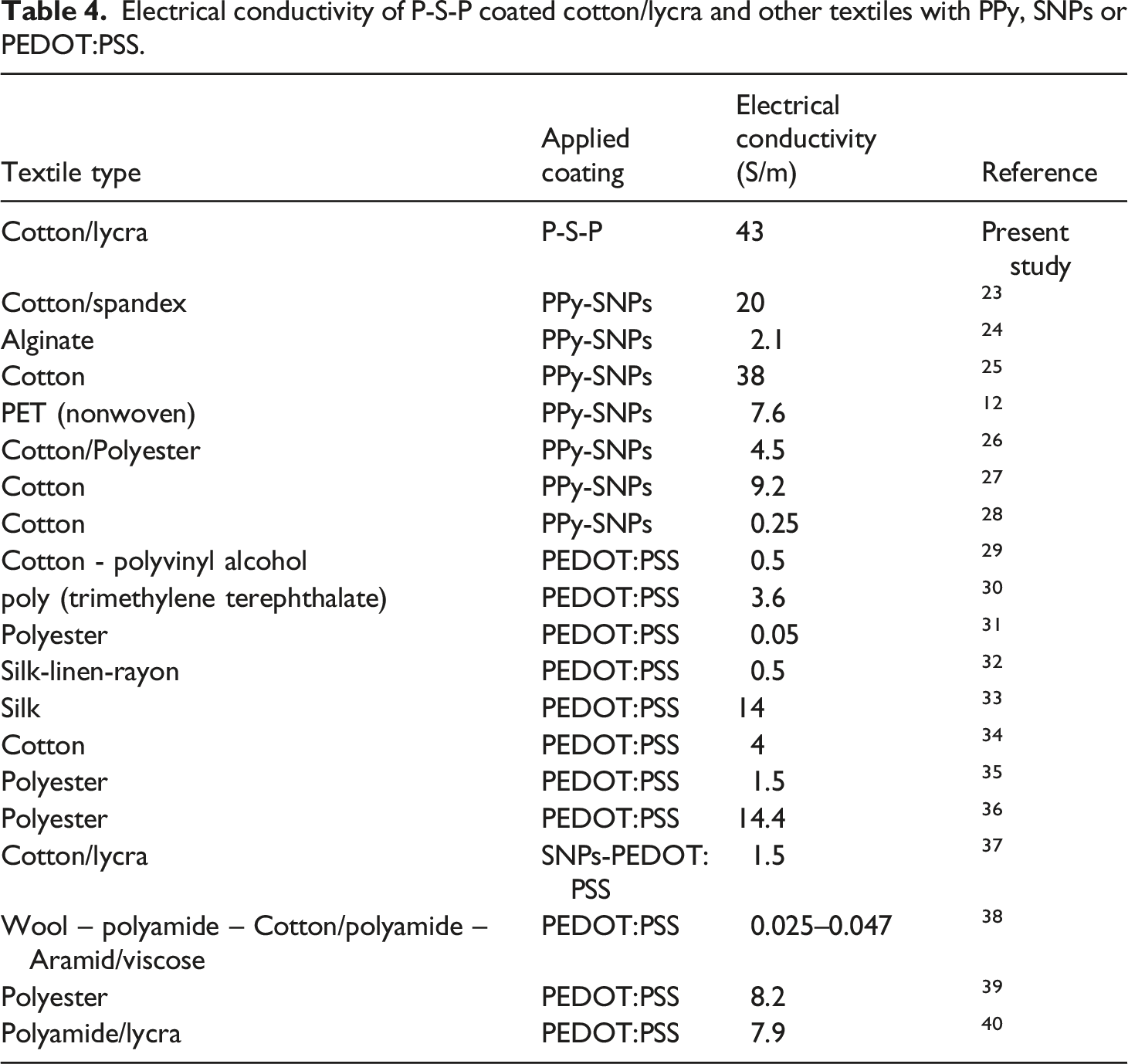
On this base, a uniform and continuous layer could not be formed on the rough surface of cotton/lycra after coating with the P-S nanocomposite, hence increasing the precursors during the in situ polymerization had no significant effect on the electrical conductivity. To overcome the mentioned problem and improve the electrical conductivity, PEDOT:PSS was used as the second coating layer. This filled the uncovered parts of P-S treated fibers with PEDOT:PSS led to the better continuity of the coating on surface. In addition, due to π–π interactions between the heterocyclic rings of PEDOT:PSS and PPy, the number of conductive pathways for electron passage increases. As a result, the fabric electrical conductivity was improved (Schematic 1(a)). As can be seen in Table 2, the sample coated with P-S-P had an excellent electrical conductivity (43 S/m), increased by 65% compared to samples coated with P-S. In Table 4, the electrical conductivity of the P-S-P coated cotton/lycra was compared with other P-S composite or PEDOT:PSS especially used on textiles. Electro conductive stability of P-S-P (0.5 M) coated cotton/lycra fabrics at 100% tension in different cycles. Electrical conductivity of P-S-P coated cotton/lycra and other textiles with PPy, SNPs or PEDOT:PSS.
Stability of the coatings and effect of strain on the electrical conductivity
Here, the effect of tension on the electrical conductivity of P-S-P coated cotton/lycra under tension was studied since a flexible electronic device may be subjected to successive tension and bending during usages. According to Figure 6, by increasing tension, the electrical conductivity of samples increased due to fiber reorientations or knit structure of the fabric. During the tension, the loops get closer to each other and the entanglement of the fibers and their connection points increases that leads to the lower surface resistivity. Another test was performed by using the coated fabrics inside an electronic circuit. One end of the textile was connected to a current source (battery) and the other end to a LED lamp. After tension, the light intensity of lamp increased confirming the more current passed through by applying tension to the fabric due to the closer loops and more connections between the fibers. (a) Formation of P-S and P-S-P nano composites on the fibers; (b) reactions between silver, pyrrole, and hydroxyl groups of cotton and PEDOT:PSS with silver ions and PPy.
The silver ions, in addition to oxidize pyrrole, react with the hydroxyl groups of cellulose and penetrate into the fibers. On the other hand, π–π interactions between heterocyclic rings of PEDOT:PSS and PPy and additionally ionic interactions between sulfonate groups of PEDOT:PSS and silver ions could attach PEDOT:PSS to P-S (Schematic 1(b)). All of these interactions may improve the stability of coating on the fibers. For investigating the stability of the P-S-P coating, the P-S-P, PPy, and P-S treated samples were immersed in water, IPA, and acetone for 24 h. For the P-S-P and P-S coated samples, no leaching was observed indicating the proper stability of the coating on the fabrics. For the sample coated with PPy, some parts of the coating separated from the fabric. It can be concluded that ionic interactions had a significant effect on the high stability of coating.
The electrical conductivity of P-S-P coated samples immersed in the solvents was re-examined under tension (Figure 6). All samples were stretched 10–100% relative to their original length and then their electrical conductivity was evaluated. The electrical conductivity increased with more elongation compared to the pristine sample, despite the lower electrical conductivity. Among the solvents, the samples immersed in water had less reduction in electrical conductivity. Some ionic bonds could be broken with placing the treated fabric in the solvents possibly form new hydrogen or ionic bonds between silver ions, cellulose hydroxyl groups, or even sulfonate groups of PEDOT:PSS with the solvents that may affect the stability of coating. All of these parameters may lead to a decrease in electrical conductivity. Despite the reduction in the electrical conductivity to some extent, the treated samples immersed in solvents for 24 h still showed good electrical conductivity, indicating proper stability of the coatings on fabrics.
As textiles are subjected to tension during the usages, maintaining the electrical conductivity is very crucial for their use as wearable electronic device. Another experiment was performed to evaluate the electro conductive stability of treated cotton/lycra fabric. The samples were subjected to 100% stretching (relative to their original length) with several cycles (25, 50, 75, and 100) and their electrical conductivity was evaluated (Figure 7). For applying 100% tension to the samples, they were stretched 100% relative to their original length by a homemade instrument as shown in Figure 7. The instrument had two holding clamps that were connected to each other by a spring. To apply 100% tension to the fabric, after attaching the fabric to the holders, it was stretched 100% of its original length by stretching the spring. As the number of loading cycles increased, the electrical conductivity decreased to some extent. By increasing in loading cycles, cracks may form, resulting in lower electrical conductivity due to the defects in coating. However, despite the decrease in their conductance, the samples still had reasonable electro conductivity. In the present study, the cotton/lycra fabric was used according to its very high stretch-ability due to lycra as elastane fibers and also the knitting structure. When the fabric is under tension or stretched the coating might crack and become defective. During tension or stretching, the yarn which are in the form of loops get closer to each other. The proximity of the loops to each other ensures the continuity of the coating on the surface of the fabric thus the electrical conductivity of the fabric is not significantly reduced. If the substrate does not have high elasticity, the coating will crack and the cohesion will decrease after applying stress; however, in the case of cotton/lycra fabric, due to its high elasticity and knitting texture, the cohesion of the coating will not decrease significantly during stretching or tension. As a result, the electrical conductivity of the fabric preserves. Changes in the electrical conductivity of P-S and P-S-P coated samples after immersion in different solvents and extended 10–100% relative to their original length.
Conclusion
In this study, highly electro conductive cotton/lycra knitted textile was fabricated using the ternary composites of PPy-SNPs-PEDOT:PSS. Cotton/lycra was used due to the extremely elastic properties of lycra, which is very important for production of flexible and highly stretchable electronic device. The coated samples showed a high electrical conductance (43 S/m). The electrical conductivity of cotton/lycra coated with PPy-SNPs-PEDOT:PSS enhanced to 760 and 65% compared to the samples coated with PPy or P-S composites. Hence, covering P-Ss nanocomposites with PEDOT:PSS led to the better connections of coating on surface of the fibers and the surface resistivity decreased significantly. Due to π–π interactions between the heterocyclic rings of PPy and PEDOT:PSS and additionally the ionic interactions between the sulfonate groups of PEDOT:PSS with silver ions, uniformity and cohesion on the surface of the fibers improved possibly enhanced the continuity provided various conductive paths on surface of the fibers. The electrical conductivity of treated fabrics reduced to some extent after immersion in different solvents; however, the fabrics still had a very high electrical conductivity and water showed the least effect. The most important feature of the treated fabrics was higher electrical conductivity after tension, either in the normal state or immersion in water or organic solvents for 24 h. In addition, those samples subjected to 100% tension for maxium 100 cycles still showed high electrical conductivity. This unique feature makes it possible to wide usages of fabricated fabric in highly flexible sensors and electronic equipment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
