Abstract
The polylactic acid (PLA)/tea polyphenol (TP) nanofiber membranes were prepared by coaxial electrospinning. The physical properties, antibacterial agent release, degradation, and antibacterial properties were investigated. Results demonstrated that stepwise and controlled antibacterial agent release profiles were achieved based on the core-shell configuration and disparate degradation rate of PLA and TP. The mechanical performance decreased with the increase of the TP content in the shell layer. The cumulative antibacterial agent release rate of nanofiber membranes with different TP content was different, while the antibacterial agent release trend was the same. The antibacterial agent release rate of the sample was the fastest at the initial stage from 2 h to 8 h, and then gradually slowed down after 24 h. In addition, the antibacterial activity of the PLA/TP nanofiber membranes was confirmed by the inhibition zone method against both Gram-positive (
Keywords
Introduction
During the wound healing process, medical dressings can protect the wound surface, absorb exudation, prevent infection, and remove necrotic tissue. As moist healing products are widely used all over the world, the antibacterial effect of medical dressings is becoming more and more important and prompted the health care industry to develop medical dressings with antibacterial properties. 1
Due to the good biocompatibility, degradability, and excellent mechanical property, polylactic acid (PLA) has been widely used in medical fields of wound dressings, medical anti-adhesion films and controlled drug release.2–5 PLA fibrous membrane is often used as medical anti-adhesion membrane, which forms a barrier on the surgical wound and degrades after the formation period of surgical adhesions to prevent postoperative tissue adhesion. If PLA nanofiber membrane has antibacterial function, it will greatly reduce the infection at the implantation site.6–8 This paper aims to prepare a PLA fiber membrane with antibacterial property and the achievements are desired to expand the application field of PLA fiber membrane.
Improving the antibacterial property of PLA fiber membranes has been one of the research focuses currently. At present, there are essentially two methods which endow PLA fiber membrane with the antibacterial property. One method is to add the antibacterial agent to the polymer spinning solution and then consequently obtain a composite fiber with antibacterial property.9–12 For example, Zeng et al.
13
prepared an antibacterial agents-loaded nanofiber membrane by directly adding the hydrophobic antibacterial agent rifampicin into the PLA spinning solution in the process of blending electrospinning. The fiber membrane was loaded with a large amount of antibacterial agent and prolonged the release time of antibacterial agent. However, the blended antibacterial agent, which unevenly distributed within the fiber, would reduce the fiber strength and increase the burst rate of antibacterial agent. The finishing method is also widely used to impart antibacterial function to the PLA fiber membrane.
14
For instance, Cerkez et al.
15
coated a new heterocyclic N-halogenated amine ester homopolymer on a PLA nonwoven fabric and then dipped the PLA fabric into a dilute sodium hypochlorite solution to obtain the antibacterial property. The results showed that the modified fabric had excellent antibacterial effects against
In order to overcome the deterioration of the mechanical property of the PLA fiber caused by the antibacterial agent and the burst release of the antibacterial agent, we used the coaxial electrospinning method to prepare nanofibers with a special shell-core structure. The structure of the prepared nanofibers is shown in Figure 1. The nanofibers prepared by coaxial electrospinning technology have the characteristics of high specific surface area, uniform porosity and excellent mechanical properties, which provide an ideal environment for cell adhesion, growth and reproduction. In addition, PLA with good biocompatibility and biodegradability was selected as the raw material for electrospinning, which enabled nanofibers with high specific surface area to be used in the field of drug release. In this study, we used PLA as the core material to form the fiber skeleton and provide a mechanical support. The mixture of PLA and antimicrobial agent tea polyphenols (TP) was the shell material, which plays the role of loading antibacterial agent and slow release antibacterial agent. TP is an antibacterial agent which is separated and purified from tea. It has various pharmacological effects such as anti-oxidation, anti-tumor, anti-aging, and immune regulation. In addition, TP has a broad-spectrum and powerful antibacterial effect, which has a significant inhibitory effect on Gram-positive and Gram-negative bacteria.21–24 We will regulate the proportion of PLA and TP within the shell material to investigate the antibacterial property and antibacterial agent release rate. The structure and properties of PLA/TP nanofiber membranes, such as the surface morphology, chemical structure, mechanical properties, and antibacterial agent release performance and antibacterial property will be studied. As a new type of antibacterial dressing, it plays the role of slow release of drug and maintenance of drug activity, and has broad application prospects in the field of biomedicine. Schematic diagram of coaxial electrospinning.
Experimental
Materials
Polylactic acid (PLA, purity ≥99.5%) was the base material of fiber membrane from Jinan Daigang Bioengineering Co., Ltd. Tea polyphenol (TP, purity ≥98%) was an antibacterial agent from Shanghai Menghe Biotechnology Co., Ltd. N,N-dimethylformamide (DMF) and dichloromethane (DCM) were obtained from Tianjin Beichen Fangzheng Reagent Factory. Sodium Chloride (NaCl) was obtained from Tianjin Yongda Chemical Reagent Co., Ltd. Sodium hydroxide (NaOH) was obtained from Tianjin Benchmark Chemical Reagent Co., Ltd.
Preparation of PLA/TP nanofiber membranes
Different concentrations of TP and PLA mixed solutions as the shell spinning solution were prepared. 6 g of PLA was dissolved in a mixture of DCM and DMF as solvents at a concentration of 10 wt%. The mass ratio of DCM and DMF was 8:2. Then different mass fractions of TP powder (1 wt%, 2 wt%, 3 wt%, 4 wt%, 5 wt%, and 6 wt%) were added into the mixed solution, respectively. The shell spinning solution was prepared by adjusting the mass ratios of PLA and TP. The mixed solution was stirred at 500 r/min for 24 h at room temperature. 2 g of PLA was dissolved in a mixture of DCM and DMF and stirred st 500 r/min for 24 h to prepare a 10 wt% core spinning solution. The mass ratio of DCM and DMF was 8:2.
The shell and core spinning solution were pushed through the needle via a silicon feedline connected to a 5 mL syringe for coaxial electrospinning. PLA/TP nanofiber membrane was prepared by using a homemade electrospinning machine at a spinning voltage of 18 kV, a syringe needle specification of 18G, a spinning distance of 15 cm, a spinning rate of 2 mL/h, the ambient temperature of 20°C and the relative humidity of 45%.
Morphology and structure characterization
The morphology of the samples was examined by a field emission scanning electron microscope (SEM, JSM-6510LA, Japan). The samples were sputtered by gold with an accelerating voltage of 5.0 kV. Nano Measurer image analysis software was used to analyze the SEM image of the fiber sample, and 60 fibers were randomly selected for measurement to obtain the average diameter and diameter distribution of the fiber. The chemical structures of the PLA/TP nanofiber membranes were recorded by a Fourier Infrared Spectrometer (FTIR, Model TL-8000, USA) using the method of total reflection infrared spectrometry, with 4000–500 cm−1 of scanning rage. The X-ray diffractometer patterns of the samples were recorded by an X-ray diffractometer (XRD, Model TD-3700, Dandong Tongda Technology). The scans range of the diffractometer was from 10° to 50°(2θ) with a step of 4°/min.
Mechanical performance test
According to the ISO 1798:2008 standard, the mechanical performances of nanofiber membranes were measured using an Electronic Single Fiber Strength Machine (YG006, Ningbo Textile Instrument Factory). According to the working principle of constant speed elongation, a certain length of fiber membrane was stretched at a constant speed to break, and the tensile strength and elongation at break of the fiber membrane were recorded.
25
All membranes were cut into 2 mm × 40 mm. The thickness of each membrane was measured by the digital micrometer (0–12.77 mm, Henan Bangte Tools) for 10 times. Then, the average thickness and the cross-sectional area of the fiber membrane were calculated. The breaking strength of samples was recorded. The breaking strength could be calculated by the Formula (1). The test was carried out at room temperature and relative humidity of 40%–60% and the tensile speed was of 10 mm/min, each sample was tested at least five times, and the average value was calculated.
Antimicrobial agent release performance test
The absorbance of different mass fractions of TP solution was measured at λ = 540 nm by using an ultraviolet visible spectrophotometer (UV1800, Shanghai Meixi Instrument). The standard fitting line could be obtained as shown in Supplementary Figure S1. The fitting equation was y = 2467.93478 × −0.95924 (
The PLA nanofiber membranes loaded with different concentrations of TP were dried in a blast dry oven (BGZ, Shanghai Boxun Industrial Co., Ltd.). The fiber membranes were weighed with an electronic balance and the results were recorded as m1–m6. The PLA/TP nanofiber membranes were put into phosphate buffered solution in 37°C thermostat water bath. At set intervals, 3 mL of this solution was taken out and observed its absorbance. Moreover, under the condition that the volume of the solution remained unchanged, while a certain volume of solution was taken out, the same volume of pure phosphate buffered solution could be added.
25
The whole sampling time lasted for 48 h. According to the absorbance tests and the standard fitting line in Supplementary Figure S1, the concentration of antimicrobial agent in phosphate buffered solution at a specific time could be obtained. Finally, different PLA/TP mass ratios in the shell of the nanofiber membranes at different times were obtained through the cumulative release formula. The cumulative release rate of TP could be calculated by the Formula (2).
Antibacterial property test
According to GB/T 20944.1-2007 standard, the antibacterial property of the prepared membrane was investigated using the disk diffusion method which employed Gram-negative 
Results and discussion
Morphology of PLA/TP nanofiber membranes
The SEM images of the nanofiber membranes with different mass fractions of TP are shown in Figure 2. As shown in Figure 2(a) and (b), pure PLA nanofiber presented smooth surface, uniform fiber diameter without adhesion, and random alignment. The diameters of pure PLA nanofibers were approximately distributed uniformly with a narrow distribution from 450–500 nm, while the diameter of nanofiber with 2 wt% of TP was from 500 to 600 nm. The nanofibers with 4 wt% and 6 wt% of TP (Figure 2(c) and (d)) had agglomeration on the surface and uneven nodes. The fiber diameter of the nanofiber membranes with 4 wt% of TP were in a range of 650–800 nm. Moreover, with the increase of the mass fraction of TP, as shown in Figure 2(d), the agglomeration phenomenon is more apparent, causing an increase of diameter and consequently a poorer formation of the fiber membrane. SEM images of nanofiber membranes with different proportions of PLA/TP: (a) pure PLA nanofiber membrane, (b) PLA/TP nanofiber membrane with 2 wt% of TP, (c) PLA/TP nanofiber membrane with 4 wt% of TP, and (d) PLA/TP nanofiber membrane with 6 wt% of TP (×5000). SEM: scanning electron microscope; PLA: polylactic acid; TP: tea polyphenol.
Chemical structure of PLA/TP nanofiber membranes
The chemical structure of PLA/TP coaxial electrospun nanofiber membranes with different proportions was explored through FTIR as shown in Figure 3. Figure 3 shows that the nanofiber membrane with pure PLA had characteristic peaks, including the methyl vibration deformation absorption peak at 1375 cm−1. With the increase of the TP content of the shell layer in PLA/TP nanofiber membranes, the phenomenon of benzene ring substitution appeared in PLA. When the amount of TP in the PLA/TP nanofiber membrane was 4%, the benzene ring absorption peak appeared at 1632 cm−1. Additionally, when the content of TP was increased to 6 wt%, benzene ring absorption peaks also appeared at 820 cm−1, 1364 cm−1 and 1620 cm−1. This indicated that there was a conjugation effect between the methyl group on the PLA macromolecular chain and the benzene ring in the TP short molecular chain, resulting in the equalization of the electron cloud density and the shift of the absorption peak to the direction of low wavenumber. These results convinced that PLA and TP had an interaction between benzene ring and methyl group. IR spectrum of nanofiber membranes with different proportions of PLA/TP. PLA: polylactic acid; TP: tea polyphenol.
Crystalline structure of PLA/TP nanofiber membranes
Figure 4 shows the XRD pattern of different PLA/TP nanofiber membranes. In the XRD pattern of PLA/TP nanofiber membrane, diffraction peaks at 2θ values of 17.5° ∼ 21.5° showed the typical structure of nanofiber membranes. The peak strength of pure PLA nanofiber membrane was significantly higher than that of PLA nanofiber membrane with TP.26–28 With the increase of TP addition, the main crystallization peak of the fiber membrane shifted, and the peak intensity and crystallinity showed a decreasing trend. When TP was 6 wt%, the crystallinity of the fiber membrane was the smallest, and the main crystallization peak appeared in 22.5° ∼ 25°, there was a slight deviation compared with the pure PLA fiber membrane. The main reason was that the addition of TP leaded to the destruction of the regularity of the original PLA molecule, thereby reducing the crystallinity. XRD pattern of nanofiber membranes with different proportions of PLA/TP. PLA: polylactic acid; TP: tea polyphenol; XRD: X-ray diffractometer.
Mechanical properties of PLA/TP nanofiber membranes
The mechanical properties in terms of the tensile strength of nanofiber membranes with different proportions of PLA/TP mass ratio were evaluated and the results were summarized in Figure 5(a). The stress strain curves of PLA/TP nanofiber membranes were shown in Figure 5(b). (a) The mechanical property of nanofiber membranes with different proportions of PLA/TP, (b) Tensile stress–strain curves. PLA: polylactic acid; TP: tea polyphenol.
Figure 5(a) shows that the tensile strength of the nanofiber membrane was relatively small. With the increase of the mass fraction of the shell layer TP, the tensile strength of the fiber membrane decreased significantly. This was mainly due to the breakage and slippage of the fibers in the fiber membrane. The PLA macromolecular segment was longer, which was the main load-bearing molecular segment when the fiber was broken. Moreover, TP was the short molecular segment. As the content of TP was increased, the PLA macromolecular segment decreased. At the same time, due to the interspersed TP short molecular segments, the PLA macromolecular segments cannot be well straightened and elongated during the stretching process. Therefore, the tensile strength of the composite fiber membrane decreased. Noted that when the content of TP was changed from 2 wt% to 3 wt%, the tensile strength of the fiber membrane decreased fastest. When the content of TP was increased to 3 wt%, the PLA macromolecular chain content would be reduced and the TP segment would be increased further. Moreover, the TP short molecular segment could embed the gap between the PLA macromolecular segments, which reduced the intermolecular force, and finally resulting in a decrease in the strength of the fiber membrane.
The stress–strain curve in the Figure 5(b) generally was showed the relationship between the mechanical properties and the change of TP introduced into the PLA fiber membrane. When TP was introduced into the fiber, its dispersion in the fiber could have a significant impact on the mechanical properties of the fiber. With the increase of TP content, the tensile failure stress of the nanofiber membrane was gradually decreased, and the tensile failure strain was increased first after reducing.
Antimicrobial agent release performance of PLA/TP nanofiber membranes
The cumulative antibacterial agent release rate curves of PLA/TP nanofiber membranes with different contents of TP are shown in Figure 6. The accumulated rate of antibacterial agent release of nanofiber membrane with different proportions of PLA/TP. PLA: polylactic acid; TP: tea polyphenol.
Figure 6 shows that the cumulative release percentage of the sample gradually increased with time. The antibacterial agent release rate of the sample was the fastest when the antibacterial agent release time was 2–8 h and then began to slow down after 24 h. The main reason for this phenomenon is the influence of the concentration gradient of TP. When the antibacterial agents released for 2–8 h, the TP concentration on the surface of the nanofiber membrane was higher than the TP concentration in the solution, that is, there is a difference in TP concentration gradient. The greater the concentration gradient between solutions, the faster the rate of diffusion is. Since TP has a tendency of diffusing from the nanofiber membrane into the solution, the drug release rate of the sample gradually decreased over time. Due to the effect of TP content on the diameter of nanofibers, the antibacterial agent release performance was different. The smaller the fiber diameter, the larger the specific surface area of the fiber, and the larger the contact area with the solution medium, so that the release of TP in the nanofiber membranes was more.
Antibacterial performances of PLA/TP nanofiber membranes
The antibacterial test results of pure PLA and PLA/TP nanofiber membrane against Test results of inhibition zone of nanofiber membrane with different proportions of PLA/TP. (a) The inhibition zone of nanofiber membrane with different proportions of PLA/TP. PLA: polylactic acid; TP: tea polyphenol.

It can be seen from Figure 7 that the pure PLA nanofiber membrane had no formation of a bacteriostatic zone. A large number of bacteria multiply under the test sample, indicating that the pure PLA nanofiber membrane had no antibacterial ability. When TP was added to the nanofiber membrane, the sample inhibition circles of different sizes appeared around. According to GB/T 20944.1-2007 standard, when the inhibition zone of the sample is greater than 1 mm, the sample is considered to have good antibacterial property. Table 1 shows that the PLA/TP nanofiber membrane had a wider inhibition zone against Antibacterial mechanism of PLA/TP nanofiber membranes. PLA: polylactic acid; TP: tea polyphenol.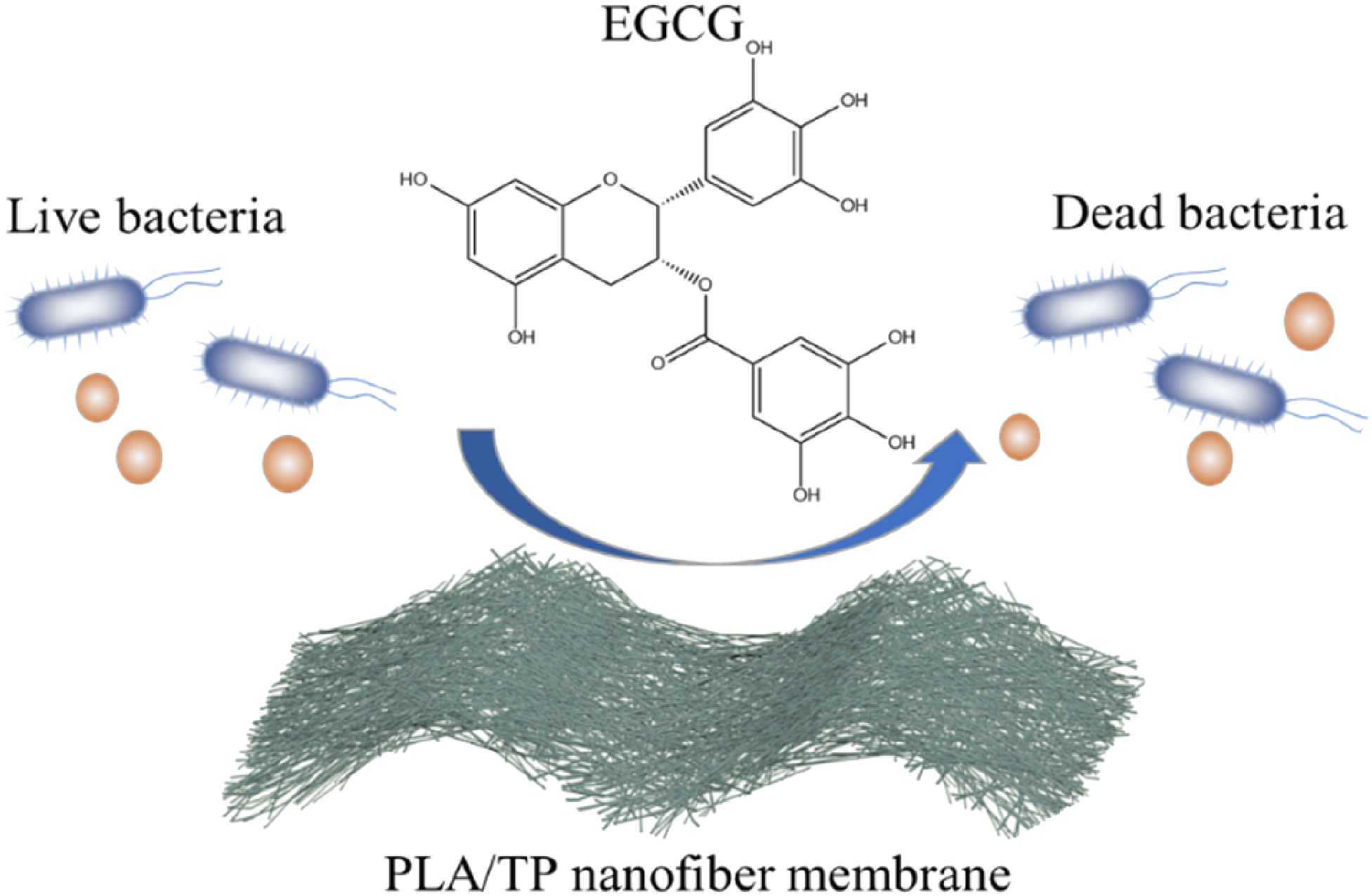
Conclusion
In this paper, the PLA/TP nanofiber membrane was successfully prepared by coaxial electrospinning technology. Due to the large specific surface area, coaxial electrospun nanofiber was chosen as the substrate to obtain a desired load and release of TP. The methyl group in PLA and the benzene ring in TP produced a chemical effect and affected the crystalline structure of the nanofiber membrane. Moreover, the addition of TP had a greater impact on the mechanical performance of the fiber membrane. The tensile strength of the fiber membrane gradually decreased, and the elongation at break first increased and then decreased. Through in vitro antibacterial agent experiments, it was found that the burst release rate of the antibacterial agent carrier prepared in this way was significantly reduced and the antibacterial agent release time was effectively prolonged. The PLA/TP nanofiber membranes showed an obvious antibacterial property against both E. coil (Gram-negative) and
Supplemental Material
sj-pdf-1-jit-10.1177_15280837211054219 – Supplemental Material for Coaxial electrospinning preparation and antibacterial property of polylactic acid/tea polyphenol nanofiber membrane
Supplemental Material, sj-pdf-1-jit-10.1177_15280837211054219 for Coaxial electrospinning preparation and antibacterial property of polylactic acid/tea polyphenol nanofiber membrane by Jie Wu, Shuqiang Liu, Man Zhang, Gaihong Wu, Haidan Yu, Huimin Li, Fu Li and Lu Jia in Journal of Industrial Textiles
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the MOE (Ministry of Education in China) Project of Humanities and Social Sciences (No. 18YJC760051), Transformation of Scientific and Technological Achievements Programs of Higher Education Institutions in Shanxi (TSTAP) (No. 2020CG014), the Natural Science Foundation of Shanxi (No. 20210302123114), the Shanxi Province Philosophy and Social Science Planning Project (No. 2020YJ037), the Postgraduate Education Innovation Project in Shanxi (No. 2020SY466), the Students Innovation and Entrepreneurship Training Program Project of Taiyuan University of Technology (No. 202085) and the Program for the Philosophy and Social Sciences Research of Higher Learning Institutions of Shanxi (PSSR) (No. 201803060).
Supplementary Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
