Abstract
The main purpose of this paper is to develop a new manufacturing process leading to have antibacterial dyed non-absorbable braided polyethylene terephthalate (PET) sutures using biocompatible and non-toxic products. This manufacturing process allows better visibility of sutures in the surgical field and reduces the risk of infections and inflammatory reactions without affecting the mechanical properties while meeting the United States Pharmacopeia (USP) requirements. Plasma functionalization, acrylic acid (AA) grafting, and bioactive chitosan (CH) coating were used before the dyeing process with a biocompatible non-toxic acid dye, approved by the Food and Drug Administration (FDA). The influence of experimental parameters on the suture properties and the K/S values of the dyed sutures are investigated. Infrared spectroscopy confirms the presence of new bonds to immobilize chitosan on the surface of the suture. Mechanical tests confirm that the mechanical properties of sutures have not been affected. The in vitro antibacterial effect of dyed PET sutures showed an inhibition zone of 11 mm against S. aureus, 4 mm against P. aeruginosa, and 1 mm against E. coli. This study reveals that the new finishing process of sutures is a promising method to achieve an antibacterial effect with a uniform shade and smooth surfaces.
Keywords
Introduction
A suture is a biomedical device used to ligate blood vessels and bonding tissue that has been severed by incision or surgery.1–3 They are the most implanted materials in the human body.4,5 They have become the largest group of biomaterials with a huge market exceeding US$1.3 billion per year.2,6 Sutures are either monofilaments or multifilaments (twisted or braided) with quite different physical and chemical properties.3,4 Braided multifilament sutures are frequently used because of their flexibility and handling properties compared to monofilaments. 7 They are generally characterized by their colors. Indeed, the color makes it possible not only to distinguish the different types of sutures but also to help the surgical team or the attending physician to have better visibility of the suture in the blood field during the surgery. 8
The braided polyester suture is a non-absorbable ligature, made of polyethylene terephthalate (PET) having fluent aromatic rings as an integral component. 7 It has a very high tensile strength. It is used for permanent removable fixtures, such as prosthetic implant fixation with permanent support, cardiovascular surgery, and ophthalmic surgery. It is generally made colorless or green suture. Braided non-absorbable PET sutures are known for their high tenacity and their crystalline structure. However, their major weakness is the lack of functional groups on the surface, which makes it resistant to the uniform penetration of the dye solution by conventional dyeing techniques. Furthermore, their implantable characters require the use of non-toxic biocompatible dyes certified by the US Food and Drug Administration (FDA) named “D & C” (Drug & Cosmetics) or “D & F” (Drug & Food). These could be only acid or reactive dyes. Thus, there are limitations in the dyeing process and color auxiliaries. 1 Hutton and Dumican 8 were among the fewest researchers to successfully dye braided PET sutures. Indeed, they have mixed a biocompatible dye (authorized by the FDA) with an extrudable non-bioabsorbable thermoplastic resin. The melt blend has been extruded, stretched, and finally, braided into sutures.
On the other hand, several studies indicate that the presence of sutures significantly enhances susceptibility to infection, wound disruption, and chronic sinus formation in surrounding tissues.9,10 In fact, bacteria could be linked to medical devices during surgery, leading to a high risk of contamination.7,11 The frequency of infections could be reduced if bacteria were removed from surgical sites. The physical structure of sutures has been included in the susceptibility factors for surgical infections. Intrinsic surface, roughness, and capillarity of PET braided sutures increase risks of wounds infection. Some researchers have suggested coating the surface of the suture with antibacterial agents.11,12 This solution could prevent the development of bacterial colonies in the medical devices without altering the mechanical properties of sutures.5,13 Several papers have reported physical and chemical protocols to develop antibacterial sutures since the early 1980s. 9 Various studies had used silver metal, silver compounds, Triclosan, polyhexamethylene biguanide (PHMB), as well as antibiotics such as neomycin palmitate, penicillin, and sulfonamide to produce antimicrobial absorbable sutures.9,14,15 However, all these studies were performed on a laboratory scale; the developed sutures were not marketed. Furthermore, frequent use of antibiotics leads to ineffective long-term systemic antibiotics and increases the risk of microbial resistance generation. 16 In 2004, Ethicon marketed the first antibacterial sutures. These are absorbable sutures coated with triclosan named Vicryl Plus, Monocryl Plus, and PDS II Plus. Several research studies have confirmed the effectiveness of these sutures against the development of germs.17–19 However, they have several limitations, namely, that they could negatively affect immune functions and cell reproducibility.11,20
Chitosan is a polysaccharide known for its non-toxicity, biodegradability, biocompatibility, and antitumor properties.21–23 chitosan is considered to be an interesting antibacterial biomedical.10,24,25 It has been used as a functional agent to develop pharmaceuticals and cosmetics products or antimicrobial textile surfaces. Its antibacterial activity has been demonstrated against a broad spectrum of gram-positive and gram-negative bacteria.22,26,27 The antibacterial mechanism is generally obtained by an interaction between primary amine groups of chitosan, which provide positive charges on contact with acidic media. These positive charges can interact with negative charged groups on the surface of bacteria. Such interactions cause the leakage of intracellular substances.12,26,28
The application of biomaterials on PET sutures can reduce the risk of surgical infections. However, the absence of functional groups on PET sutures reduces the linkage with the chitosan biomolecules. For this reason, a surface grafting process seems necessary. The attractive feature of the grafting process is that the modification can be precisely controlled by the proper selection of the reaction conditions.29,30 However, the graft uselessness in biomaterials science is due to the fact that it modifies the bulk of polymers and leads to a loss of mechanical strength due to the high energy used in the grafting process.31,32 Thus, plasma processing was employed for the functionalization of the PET surface without any chemical modification of the polymeric materials.22,33–35 This reduces the consumption of reagents and their toxic effects. On the other hand, the low intensity of plasma energy limits surface changes to dimensions of a few nanometers without changing the bulk properties of the material.36-38 In fact, plasma processing uses different ionized gases to create the desired functionality on the surface.39–41 These active sites play an important role in the interaction of polymers with an organic surface. 42 This could activate desirable interaction with the biomolecules. The plasma grafting of an appropriate monomer followed by the immobilization of the bioactive molecules onto the modified surface would allow the development of a dyed suture that prevents bacterial infection.
In this study, we sought to develop a new manufacturing process allowing to have new antibacterial non-absorbable dyed PET sutures. for this purpose, low-pressure oxygen plasma was used to create hydroperoxides and to initiate acrylic acid (AA) grafting on PET surfaces. Thus, the immobilization of chitosan as a bioactive component on suture surfaces has been promoted. The chitosan would help the acid dye molecules bond and lead to develop the antibacterial effect.
In the present investigation, the influence of immobilization conditions on the suture surfaces will be studied. Chemical reactions and mechanical and antibacterial properties of developed sutures will, also, be investigated in order to determine the best manufacturing requirements.
Materials and Methods
Materials
Braided PET sutures made from 16 non-textured PET yarns with 49 dtex counts were fabricated using a HERZOG circular braiding machine (type 80, Germany). After braiding, they were cleaned by Soxhlet using ethanol and acetone as a solvent to remove all impurities. The fabricated sutures have an average diameter of 0.29 mm. According to USP 29–NF 24 monograph for non-absorbable sutures, 43 our PET sutures had an American Pharmacopeia (USP) number of 2–0.
Acrylic acid monomers (AA) were supplied by Merck, Germany. Acetic acid and chitosan with high molecular weight (HMW, 310–375 KDa) having a degree of deacetylation greater than 75% were purchased from Sigma Aldrich, Tunisia.
For the dye experiments, a mixture of two biocompatible, non-toxic FDA-cleared dyes was prepared to obtain the green dye. Acid yellow, D&C Yellow No10 (CI 47005), and acid blue, D&C Blue No2 (CI 73015), were used. Their chemical structures have been shown in Figure 1. UV-vis spectrum of acid green dye aqueous fractions and dyeing process of PET suture. PET: polyethylene terephthalate.
Preliminary tests have been carried out to define the appropriate proportion of each dyestuff. The UV-vis spectrum of the aqueous fraction of the dye was recorded using a spectrophotometer and was shown in Figure 1. This spectrum showed two major peaks in the visible region at 412 nm and 620 nm.
Manufacturing of PET grafted samples
In order to achieve a better reactivity of PET sutures with acid dyes, we performed surface activation using oxygen plasma treatment followed by AA grafting. The study of experimental parameters has been done in laboratory work and the optimal conditions are used for the chitosan immobilization and the dyeing procedure.
Cleaned PET braided sutures were treated with oxygen plasma for 5 min under a cold RF plasma system (Diener electronic, Germany) at low pressure operating at a frequency of 13.56 MHz. The oxygen was introduced into the chamber at a flow rate of 100 cm3/min. Immediately after the plasma treatment, PET samples were placed in a flask containing an aqueous solution of AA at a concentration of 100% for 10 min. After the grafting reaction, AA grafted sutures (PET-AA) were removed from the flask, were dried in an oven at 50°C, and were extracted with water by Soxhlet to remove any homopolymer adhering to its surface. Following the grafting process of AA and surface activation by oxygen plasma, the concentration of carboxylic groups generated on the surface and the grafting rate were measured. Results were found at 2.60 μmol/g and 48.8%, respectively.
Immobilization of chitosan on the PET sutures grafted with AA (PET-AA-Ch) was carried out by the dip-coating method, as described by Gupta et al. 36 PET-AA sutures were immersed in a chitosan solution, obtained by dissolving a determined concentration of chitosan in an acetic acid solution of 1%, for different periods at room temperature (Table 2). Sutures were then removed and dried for 30 min in an oven at 100°C. After heat setting, the suture immobilized by chitosan was washed. They were immersed in acidified deionized water at room temperature for 1 min under constant stirring to remove excess chitosan from the sutures.36,44 Samples were dried at 60°C in an oven. The PET-AA-Ch sutures were subsequently dyed.
Experimental plan of chitosan immobilization treatment.

General full experimental plan.

Dyeing procedure
The chitosan-immobilized PET sutures dyeing experiments were performed in an AHIBA dyeing machine. According to preliminary tests, dyeing of PET-AA-Ch sutures should be done in a dye bath containing 3% acid dyestuff at a liquor ratio of 1/100. The dyed samples were then rinsed with deionized water and dried at room temperature. The dyeing process at a pH value of 4 is shown in Figure 1.
Developed suture characterization
The relative color strength (K/S values) of the dyed samples was evaluated by a Datacolor 400 spectrophotometer. It was evaluated using the following Kubelka–Munk equation (1)
The immobilization yield of chitosan on the suture surface was calculated using the following equation (2)
The surface morphologies of all samples were examined using SEM scanning electron microscope operating in a low vacuum mode. The SEM used was a TM3000 Tabletop Microscope (Hitachi, Japan).
Infrared spectroscopy of PET-AA-Ch sutures was performed in order to prove the presence of newly immobilized chitosan on the surface of the suture. ATR-FTIR spectra were recorded using FTIR-ATR spectrophotometer (Varian 3100, Germany). Each spectrum is the average of 32 scans from 650 to 4000 cm−1 at a resolution of 4 cm−1.
Thermal analyzes were performed using a thermogravimetric analysis of the STA 2500 Regulus type (NETZSchitosan, Germany). 8 mg samples, weighed with a precision balance (±0.0001 g), were placed in an aluminum crucible and heated to 600°C under an argon flow rate of 20 mL/min and a heat flow of 10°C/min.
In order to determine suture diameter, we have used the united states pharmacopeia (USP) convention based on USP32-NF27 S2 < 861 > method for non-absorbable sutures. 45 The measure was done using a deadweight mechanical thickness gauge (SODEMAT-Troyes, France) with a grading precision of 0.02 mm divisions.
Tensile strength (TS) and knot pull strength (KPS) tests were performed using the Instron tester method. Suture samples were subjected to longitudinal traction to failure at a strain rate of 300 mm/min with a load cell of 100 N and an initial gauge length of 150 mm using a Universal Testing Machine (Lloyd, England).46,47 A square knot was used for knot pull tests. Ten samples were tested and the average value of breaking load (N) was determined.
Friction coefficient difference (FCD) simulates the configuration of the sutures during surgical knot tying. It was determined to study the friction properties of treated sutures. It affects the knot security and handling properties of suture materials. Two sutures were warped together and placed on upper pulleys. Specific weights (T0 = 0.2N) were attached at the ends of each suture. The upper pulleys were undergoing a vertical displacement of 30 mm at a rate of 12.7 mm/min. The difference between two successive extremums (the maximum friction and the minimum friction (μs-μk)) was calculated. The average of this difference represents the FCD. 47
All tests were carried in a controlled environment of 20°C ± 1°C temperature and 65% ± 2% relative humidity according to ASTM D1776-04. 48
Antibacterial efficiency
In order to evaluate the antibacterial effect of treated and non-treated PET sutures, agar diffusion plate tests were used according to the standard test method for the determination of antibacterial activity of textiles (ISO 20645: 2004). 49 The bacteria of Staphylococcus aureus (ATCC 6538), Escherichia coli (ATCC 11229), and Pseudomonas aeruginosa (PAO1) were selected for the antibacterial tests.
Briefly, the bacteria strains were taken up in non-inhibitory nutrient agar in order to obtain isolated colonies. After 24 h of incubation at 37°C ± 1°C, 4–5 colonies were isolated and impregnated in a buffered saline solution. After adjustment, a swab was performed on Mueller Hinton medium and the bacterial surface was allowed to dry for at least 30 min. Sutures were sterilized in an autoclave at the temperature of 100°C for 20 min then they were placed on the bacterial lawn using sterile forceps. 4 cm length of dyed PET sutures samples immobilized with chitosan were used. The non-treated sutures were tested as a reference.
The inhibition zone after incubation at 37°C for 24 h showed the recorded result. 7
Results and Discussion
The main purpose of this paper was to develop antibacterial dyed polyester sutures using biocompatible non-toxic acid dyestuff and chitosan as an antibacterial biomaterial agent. However, the biocompatible dyes used were anionic dyestuffs and could not be linked to raw PET surface. Hence, the proposed process required surface activation of the braided sutures, grafting of acrylic acid, and then the immobilization of chitosan to the surface. Figure 2 shows the different steps in the development of dyed antibacterial PET sutures. Schematic representation for the development process of antibacterial dyed PET sutures. PET: polyethylene terephthalate.
Suture surface investigation by ATR-FTIR
The ATR-FTIR spectra of the treated sutures are investigated in order to confirm the immobilization of chitosan on the PET-AA sutures and eventually to determine the possible reactions.
The ATR-FTIR spectra of different PET sutures are shown in Figure 3. The raw PET spectrum showed that characteristic bands at 2960 cm−1 corresponding to the aliphatic chitosan2 stretching. The origin of the band at 1713 cm−1 may be assigned to carbonyl stretching vibrations of –C = O, while the band at 1240 cm−1 may be attributed to C-O stretching vibrations of ester groups.36,50 ATR-FTIR spectra of PET sutures: (a) virgin PET; (b) PET-AA; (c) PET-AA-Ch-4; (d) PET-AA-Ch-7; and (e) PET-AA-Ch-5. PET: polyethylene terephthalate.
PET-AA spectra showed two new bonds at 2360 and 2340 cm−1 proving that AA was successfully grafted onto the PET polymer.51–53 From these spectra, it can also be noticed that the peak intensity of carbonyl groups at 1713 cm−1 is increased due to the presence of the carbonyl groups of AA.54–56 This is the indication of the grafted acrylic acid on the braided suture surface.
For the chitosan-immobilized surface, the functional groups of pure chitosan were observed in a broad peak between 3300 and 3600 cm−1 for the presence of –OH, –NH, and NH2 stretching vibrations. The infrared absorbance of carbonyl in carboxylic acid found at 1713 cm−1 shows lower intensity compared to PET-AA. This can be an indication of the loss of some free carboxyl groups due to the formation of an amide bond with deacetylated sites of chitosan molecules. The chitosan-immobilized surface also shows new peaks at 1650 and 1545 cm−1 were, respectively, related to amide I C = O in O = C-NH and amide II NH in O = C-NH.36,57 All these observations are sufficient indications of chitosan immobilization on the PET braided suture.
Based on spectra analyzes and referring to the literature reviews, the mechanism of interaction between chitosan molecules and PET sutures after oxygen plasma treatment and AA grafting is presented in Figure 4. Interaction mechanism of chitosan molecules and acrylic acid grafted on plasma-treated PET sutures. PET: polyethylene terephthalate.
According to results given by infrared spectra, chitosan was well immobilized on the grafted PET suture surface. This can be explained by four potential interactions that can occur between chitosan molecules and AA grafted onto the plasma-treated PET surface. These interactions are illustrated in Figure 4 and detailed as follows: 1. A: Hydrogen bond between the –NH2 groups of chitosan and carboxylic groups of AA grafted onto the PET surface. 2. B: Hydrogen bond between the –OH groups of chitosan and carboxylic groups of AA grafted onto the PET surface. 3. C: Ester group between the –OH groups of chitosan and the carboxylic groups of AA grafted onto the PET surface. 4. D: Ionic bond between the –NH2 groups of chitosan and –COOH groups of AA grafted onto the PET surface.
Analysis of dyed antibacterial sutures
Figure 5 shows the main effect plots of chitosan concentration and impregnation time on chitosan immobilization yield (a) and color strength (b). It can be seen that the variation of chitosan concentration from 0.5% to 1% does not affect the immobilization yield, while beyond 1% of chitosan concentration, a significant decrease in the immobilization yield is observed. Indeed, when the chitosan concentration increases, the viscosity of the grafting medium is increased and hinders its diffusion toward the active sites of PET-AA. Gupta et al.
58
have found that beyond 40% of the monomer concentration the grafting yield decreases. They explained that the increase in viscosity prevents the diffusion of monomers to functional groups. On the other hand, it can be observed that an increase in the impregnation time leads to a remarkable increase in the immobilization yield. Indeed, PET-AA initially treated with oxygen plasma and grafted with AA has a negatively charged surface due to the presence of carboxylic groups. Consequently, these groups interact with the positively charged amino groups of chitosan to form crosslink by ionic bonds. For this reason, by increasing the impregnation time in the chitosan solution, the number of active sites will be increased. These sites will be occupied and the immobilization yield will increase to reach a maximum of 10 min. Thus, the increase in immobilization yields is achieved through the increase of links to active sites. However, for more than 5 min, the immobilization rate becomes less important. This is because it is more difficult to interact with the deeper sites where they need more time to react. Main effect plot of the chitosan concentration and the impregnation time on (a) the chitosan immobilization yield and (b) the color strength.
The main effect plot of the chitosan concentration and the impregnation time on the color strength values is presented in Figure 5(b), It can be seen from this figure that both experimental parameters have a significant effect on color strength. An increase in the chitosan concentration from 0.5% to 1% leads to an increase in the K/S values. Due to the positively charged nature of the protonated amino groups of chitosan, the anionic dye will selectively react with these chitosan groups and not with PET groups. In fact, the intensity of the color of treated sutures indicates a specific interaction between chitosan and the dye molecules. Therefore, at a higher chitosan concentration (2%), a slight decrease has been observed (Figure 6) Interaction mechanism between the color-stuff and chitosan immobilized PET suture. PET: polyethylene terephthalate.
On the other hand, an increase in the impregnation time from 1 to 10 min induces a dip in the color strength around 5 min to express a decrease in the dye affinity between 1 and 5 min then an increase between 5 and 10 min, these results coincide with those found in the study of the effect of the impregnation time on the immobilization rate of chitosan sutures. These results could be explained by the chronological evolution of reactions expected between chitosan and the raw material.
Response surface contour plots of chitosan immobilization yield and color strength are given in Figure 7. Contour plots illustrate the behavior of the measured characteristic as a function of the two based factors. Indeed, the response surface is considered as a two-dimensional surface where all points that have the same response are connected to produce the contour lines of constant responses.
59
According to plot (a) variations in immobilization yield can be seen because of the selection of different values of chitosan concentration and impregnation time. It can also be noted that the best immobilization yield was obtained by the simultaneous increase of chitosan concentration and impregnation time of sutures in chitosan solution. Contour plot of (a) the immobilization yield and (b) color strength.
Figure 7 b shows the contour plot of the manufacturing parameters on color strength. It can be seen in this figure that better results are obtained under two conditions: when the chitosan concentration is increased (>0.75%) during a short impregnation time (>2 min); otherwise. when the chitosan concentration is decreased (<1.75%) for a longer impregnation time (>8.5 min).
An example of dyed sutures after chitosan immobilization at 2% of chitosan concentration for 1 min is presented in Figure 8. Dyed PET sutures after AA grafting and chitosan immobilization at 2% chitosan concentration for 1min. PET: polyethylene terephthalate.
Characterization of the developed sutures
Antimicrobial properties of treated PET sutures
Antibacterial properties of treated PET sutures.
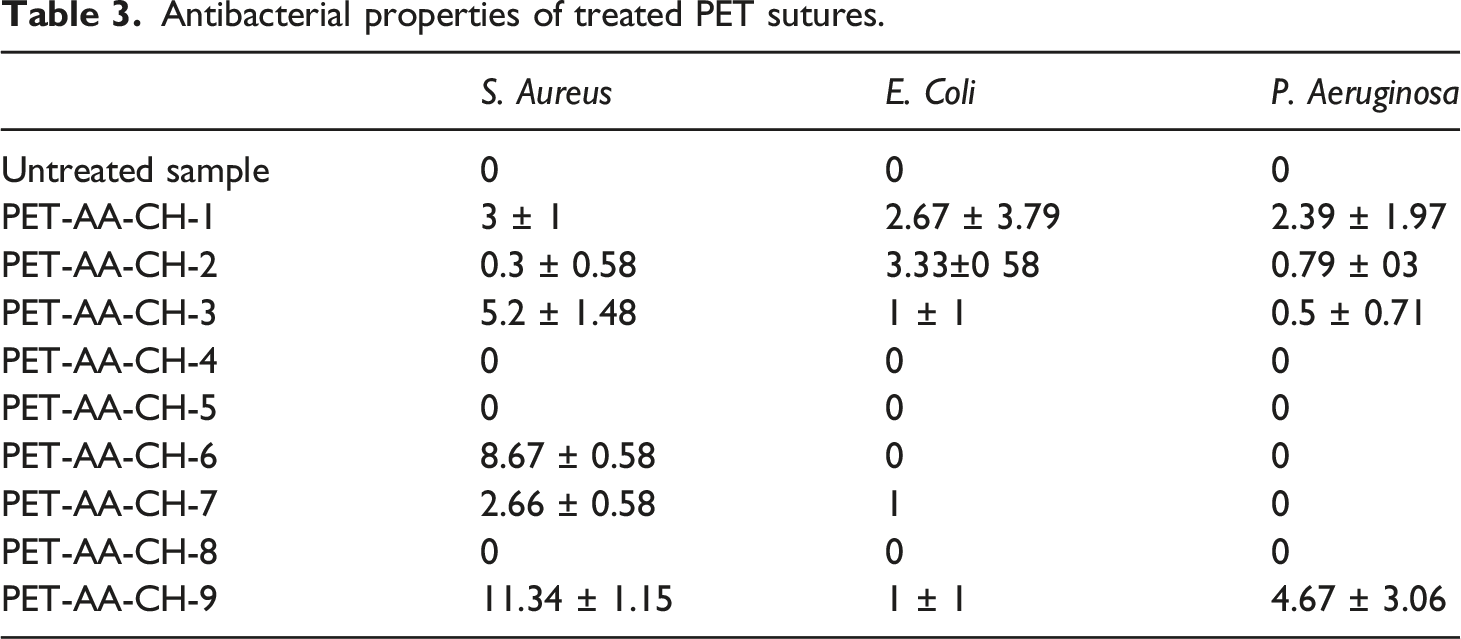
On the other hand, when the impregnation time is increased, the interaction between the positive groups of chitosan and the negative groups of acrylic acid grafted on the PET increases. Subsequently, most of the amino groups of chitosan are covalently disclosed to the negatively charged acidic groups of acrylic acid grafted onto PET. Then, chitosan might not be released out in the medium to form an inhibition zone.22,63,64
Thermogravimetric analysis of chitosan immobilized suture
Thermogravimetric curve of the optimal treated sutures determined previously was analyzed by TGA and shown in Figure 9. In this figure, it can be seen that both treated and untreated PET show initial weight loss at around 160°C due to polymer dehydration. Then, the polymer begins its decomposition at 400°C for raw material and at about 360°C for optimal chitosan-immobilized suture. After the main drop in weight loss, it can be seen that the sample continues to decompose smoothly up to 600°C. These results are in agreement with previous studies which have shown that the decomposition of PET is between 350 and 550°C.50,65 The incorporation of chitosan molecules on PET sutures did not imply any change in the thermal behavior of the PET. This coincides with results reported by Bonilla et al.
66
This is because the decomposition of PET occurs when the main bonds in the chain are broken. In the case of the optimum treated suture (PET-AA-CH-9), the bonds developed between chitosan molecules and AA grafted PET sutures are relatively weak. Thus, the decomposition of AA-CH links begins before the main chain bonds of PET and leads to a decrease in the onset temperature. TGA curves of chitosan immobilized PET-AA sutures. PET: polyethylene terephthalate.
Mechanical properties
Mechanical properties of chitosan immobilized dyed PET sutures.
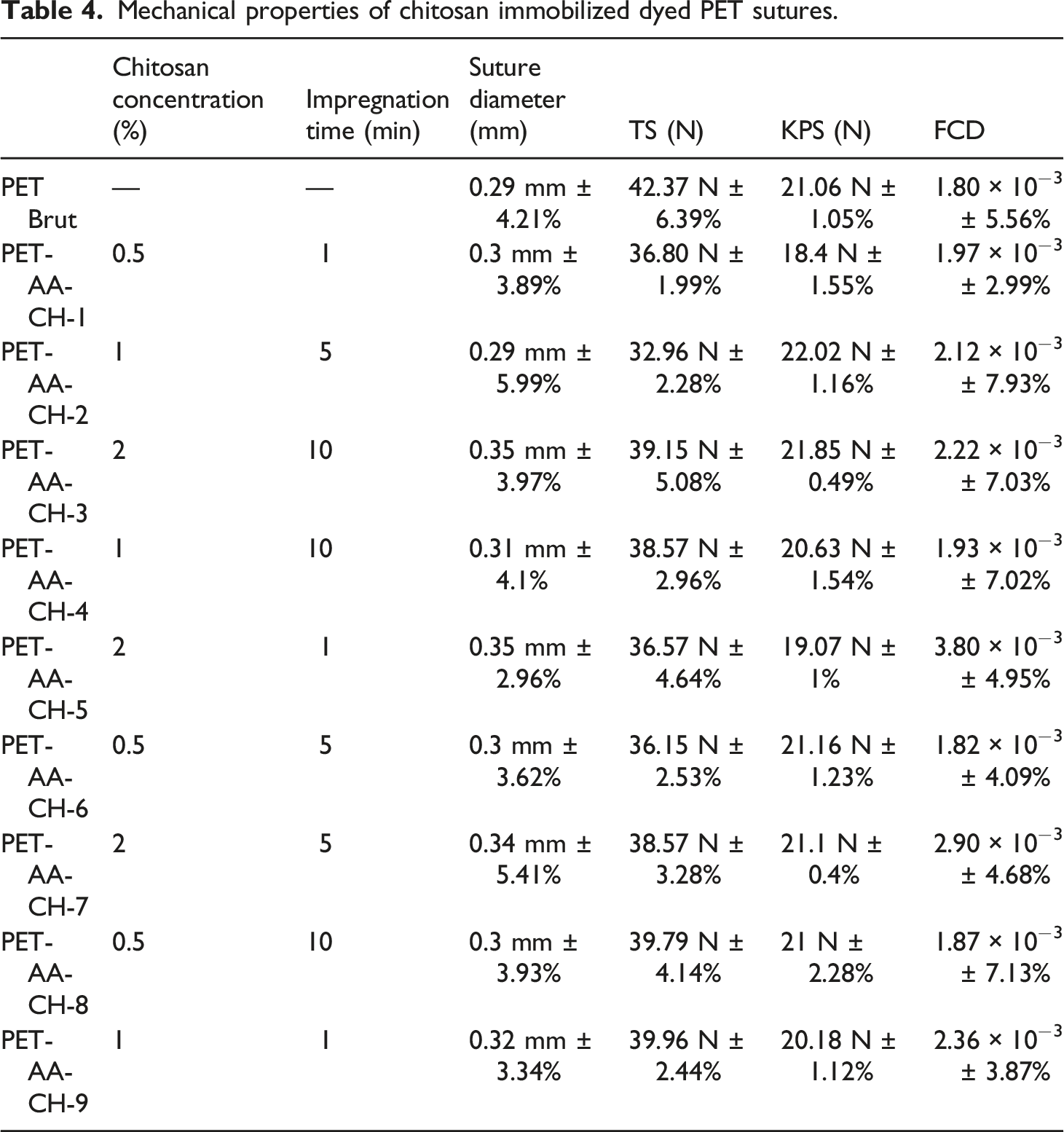
From Table 4, it can be seen that suture diameter has increased from 0.29 mm ± 4.21% for untreated sutures to a maximum of 0.35 mm ± 3.97% for developed dyed sutures. This thickening is due to the swelling of the PET fiber during the surface functionalization and dying in the acid bath (pH = 4). Indeed, to fill the intermolecular spaces, steric hindrance took place at the graft sites which increased the diameter of the yarn. According to the USP requirements, treated suture diameter remains in the intervals of USP number 2–0.
Tensile strength and KPS of developed sutures exhibit slight fluctuations and do not differ significantly from untreated sutures. Moreover, sutures should meet the USP requirements of KPS as so not to break unexpectedly during use. Obtained results prove that KPS values still meet the USP requirements. All values are higher than 17.6 N, for a suture having USP number 2–0 which is a limit on the average minimum required by USP.43,45 It can be concluded that the treatment took place on the surface and did not affect the suture material.
The friction coefficient difference (FCD) was also measured and the obtained results are shown in Table 4. Obtained results show a slight increase in the FCD-mean for developed sutures. It can also be noticed that FCD increases when increasing the chitosan concentration. These results can be related to the chitosan accumulation on the suture surface leading to surface roughness, which is in agreement with other researches that have shown that the FCD increases when increasing the surface roughness,4,72 since it is well known that the friction force is related to the surface roughness. 73 Thus, the results of FCD indicate that an increase in the impregnation time induces a decrease in FCD.
SEM analysis
Figure 10 shows SEM pictures of chitosan-immobilized PET sutures. The morphological structure of PET sutures treated with chitosan during 10 min using 0.5% (Figure 10(b)), 1% (Figure 10(c)), and 2% (Figure 10(d)) of concentration are compared to the morphological structure of untreated PET sutures. SEM pictures show the presence of chitosan on the surface of the sutures and in the interfibrous pores. Figure 10(a) and Figure 10(b) notice smooth surfaces, Figure 10(c) show less smooth surfaces, and Figure 10(d) show striated zones. It can also be seen that surfaces are not always uniform. Indeed, at a concentration of 0.5% chitosan (PET-AA-Ch-4), smooth and uniform surfaces are shown. By increasing concentration to 1% (PET-AA-Ch-7), surfaces become less smooth but remain uniform. For a concentration of 2% (PET-AA-Ch-5), agglomeration zones at the surfaces appear. (a) SEM photograph of untreated PET suture, (b) SEM photograph of 0.5% chitosan concentration/10 min, (c) SEM photograph of 1% chitosan concentration/10 min and (d) SEM photograph of 2% chitosan concentration/10 min. PET: polyethylene terephthalate.
Conclusion
PET braided non-absorbable sutures are frequently used because of their mechanical and handling properties. They are generally green in color to improve their visibility in the surgical field. However, they are responsible for surgical wound infections.
The present paper proposes a new manufacturing process that provides at the same time antibacterial and dyed PET sutures. For this purpose, we started by plasma-induced graft polymerization of AA on sutures surfaces. These treatment conditions, previously verified in the laboratory, give the sutures carboxylic groups on the surface and allow the covalent bond of chitosan molecules. Then, the immobilization process of chitosan was investigated. ATR-IR spectroscopy was used to examine potential reactions. Many possible bonds between chitosan molecules and carboxylic groups present in the grafted surface of PET sutures could be made. These bonds prove the immobilization of chitosan on the suture surfaces. Subsequently, the immobilized sutures were dyed using biocompatible non-toxic mixtures of dyestuffs approved by the USP requirement. Color strengths of dyed sutures were evaluated as a function of chitosan immobilization conditions. Better results were found under two conditions when the chitosan concentration increased for shorter impregnation times or when the chitosan concentration decreased for longer impregnation times. To check the antibacterial activity of dyed sutures, we have used the agar diffusion method against three colonies: S. aureus, a gram-positive bacterium, E. coli a gram-negative bacterium, and P. aeruginosa, a gram-negative bacterium. Best results have been found in the case of treated sutures at a chitosan concentration of 2% and an impregnation time of 1 min. This suture shows the inhibition zone of 11.34 mm against S. aureus, the inhibition zone of 1 mm against E. coli, and the inhibition zone of 4.63 mm against P. aeruginosa.
We also found that the developed process did not affect the mechanical properties and that the sutures diameter and the KPS values still meet USP requirements. Finally, the suture surface was examined using friction coefficient difference test and SEM image analysis. Morphologic analysis showed that uniform surfaces were obtained at a chitosan concentration of 1% for 10 min.
Thus, this paper revealed the best experimental conditions to find antibacterial suture dyed with an FDA-approved biocompatible green dye using a new process that combines physical activation and chemical grafting to bind the natural dye to the suture surface.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
