Abstract
Fabric it is not an impermeable substrate because of fiber porosity. To study the solvent diffusion mechanism of coated fabric in the curing process, the drying model of PMMA/acetone coated glass fiber fabric was established. This drying model was verified by confocal Raman spectroscopy. Finally, the impact of fabric structure, thickness and porosity on the solvent diffusion process in coated fabrics was studied by the model. It was shown that the predicted solvent concentrations by the model were consistent with the experimental values. This model can be used to quantitatively calculate the solvent concentration at any position and at any time inside the coating film during the drying process. Moreover, it can also predict the curing time and residual solvent concentration of the coating fabric required to reach drying equilibrium. Compared with coated 3/1 twill, 5/3 satin and 2/1 twill, the solvent diffusion of coated plain fabric was faster during curing. Under the same environmental conditions, the thinner the fabric was and the greater the porosity was, the shorter the curing time was. The fitting equations for fabric thickness, fabric porosity and drying time were obtained, which can provide a theoretical guidance for the preparation, performance research and drying conditions optimization of PMMA coated textile materials.
Keywords
Introduction
Coated fabric refers to the composite fabric formed by coating a polymer or other materials on a fabric substrate. Many kinds of polymer solutions, such as silicone resin, acrylic resin, epoxy resin and polyurethane are commonly used. Among them, the acrylic resin film is transparent and does not susceptible yellow. The acrylic resin film has the advantages in terms of chemical stability, weather resistance, flame retardancy, low cost and good adhesion to glass fiber. It has been widely used in construction, decoration, transportation and military fields [1–4]. During the development of coated textile materials, experimental methods are often used as a way to optimize important parameters such as fabric structure, thickness and porosity and so on [5,6]. However, this approach requires repeated experiments with high costs and longer experimental times. Therefore, it is desirable to predict the change of solvent concentration in coated fabric under certain condition by means of numerical calculation.
Recently, researchers have made good progress to establish a numerical model for the solvent diffusion process of coated materials. For example, Raj Kumar Arya et al. [7] coated an impermeable steel plate using the coating solution prepared from a polymethylmethacrylate–tetrahydrofuran and polystyrene-p-xylene system. Based on the free volume theory, the drying model of the polymer film was established, and the concentration distribution of different solvent systems was predicted. It is concluded that the measured data in high volatile solvents are in good agreement with the predicted data, but the free volume model is not applicable in low volatile solvents. David Siebel et al. [8] applied a coating solution of polyvinyl acetate-toluene-methanol on the impermeable glass plate to study the drying behavior of the coating solution of three-phase system (polymer-solvent 1-solvent 2). The results show that the existence of high boiling point solvent will accelerate the evaporation of low boiling point solvent. He Sumin [9] coated an acrylic adhesive on a glass plate to simulate the influence of different temperatures on the adhesive drying process in hot air conditions, and the initial drying rate of adhesive thin layer was basically the same under the same drying temperature and different thin layer thickness. Bhargava et al. [10] established a solvent diffusion model for the two-phase system of poly (styrene) – tetrahydrofuran and poly (styrene) - xylene in the drying process of polymer film on impermeable substrate, studied the influence of coating thickness and solvent content on film foaming, and the thinner the coating and the higher the solvent concentration, the lower the foaming probability of the coating liquid during drying. However, the substrates of these coating material were mostly impermeable plate, only the solvent diffusion at the film-air interface was studied. Textiles are porous fiber materials, and the solvent diffusion mechanism on the fabric is different from that on the non-permeable substrate. The solvent diffusion occurs at both the film-air interface and the substrate-air interface. The solvent diffusion mechanism in the curing process of coated fabric has not been fully explored.
Fabric structure parameters (such as fabric weave, yarn fineness, fabric density, etc.) can affect the pore shape, porosity and fabric thickness, and these parameters have a very important impact on the mass and heat transfer through the fabric. For instance, Zheng Xiaoqing et al. [11] designed and developed four different fabrics (pattern, 3/1 twill, 5/2 satin and 8/3 reinforced satin), analyzed the influence of fabric weave structure on the thermal and wet comfort of fabrics, and concluded that the thermal and wet comfort of 3/1 twill and 5/2 satin fabrics were better. Singh [12] and Jhanji [13] studied the effect of yarn density on the physiological properties of the fabric, and observed that the fabric made of high density fiber has high heat resistance and air permeability. Angelova et al. [14] studied the heat and moisture transfer process of single-layer and three-layer fabric system by experimental method, and the thicker layer system has the lower water vapor permeability, and the higher pore volume will increase the water vapor permeability. Therefore, it is necessary to explore the influence mechanism of fabric structure parameters on solvent diffusion during the coated fabrics curing process by numerical calculation. These parameters should then be optimized to achieve the purpose of energy saving and a reduction of emissions of organic solvent during the coating curing.
In this study, the solvent diffusion mechanism of PMMA/acetone coated glass fiber fabric during curing process has been investigated. By introducing the heat transfer equation of porous materials, Fick diffusion equation and the relationship between volume and time of coating liquid, the solvent diffusion model in coated porous media was established. The accuracy of the model was verified by confocal Raman spectroscopy. This model can be used to quantitatively calculate the solvent concentration at any position and at any time inside the coating film during the drying process, and it can also predict the curing time and residual solvent concentration of the coating material required to reach drying equilibrium. Finally, the effects of fabric structure, fabric thickness and porosity on solvent diffusion were discussed by the established drying model, which can provide theoretical guidance for the preparation, performance research and drying conditions optimization of coated textile materials.
Geometric model
Glass fiber fabric is woven by ourselves using a rapier loom (model: ASL3000; manufacturer: Tianjin Longda Machinery Manufacturing Co., Ltd.). Fabric specification: yarn fineness is 280 tex, warp density is 120 ends/10 cm, and weft density is 100 picks/10 cm. First, the fabric structure parameters were obtained from an electron microscope images (see Table 1). The TexGen software was then used to establish a 3D geometric model of a fabric unit (Figure 1). The coating solution prepared by PMMA and acetone solution was then applied on the surface of the glass fiber fabric. The coating solution penetrated into the interior of the fabric. The penetration depth was 0.4 mm as obtained by the electron microscopy. The length and width of the geometric model for the coated fabric were 5.1 mm and 5.34 mm, respectively. During the curing process, the solvent moved from the inside of the film to the film-air interface (
Structural parameters of the glass fiber fabric.


Geometric model of the fabric. (a) Microscopic image of real fabric, and (b) geometric model of the fabric.
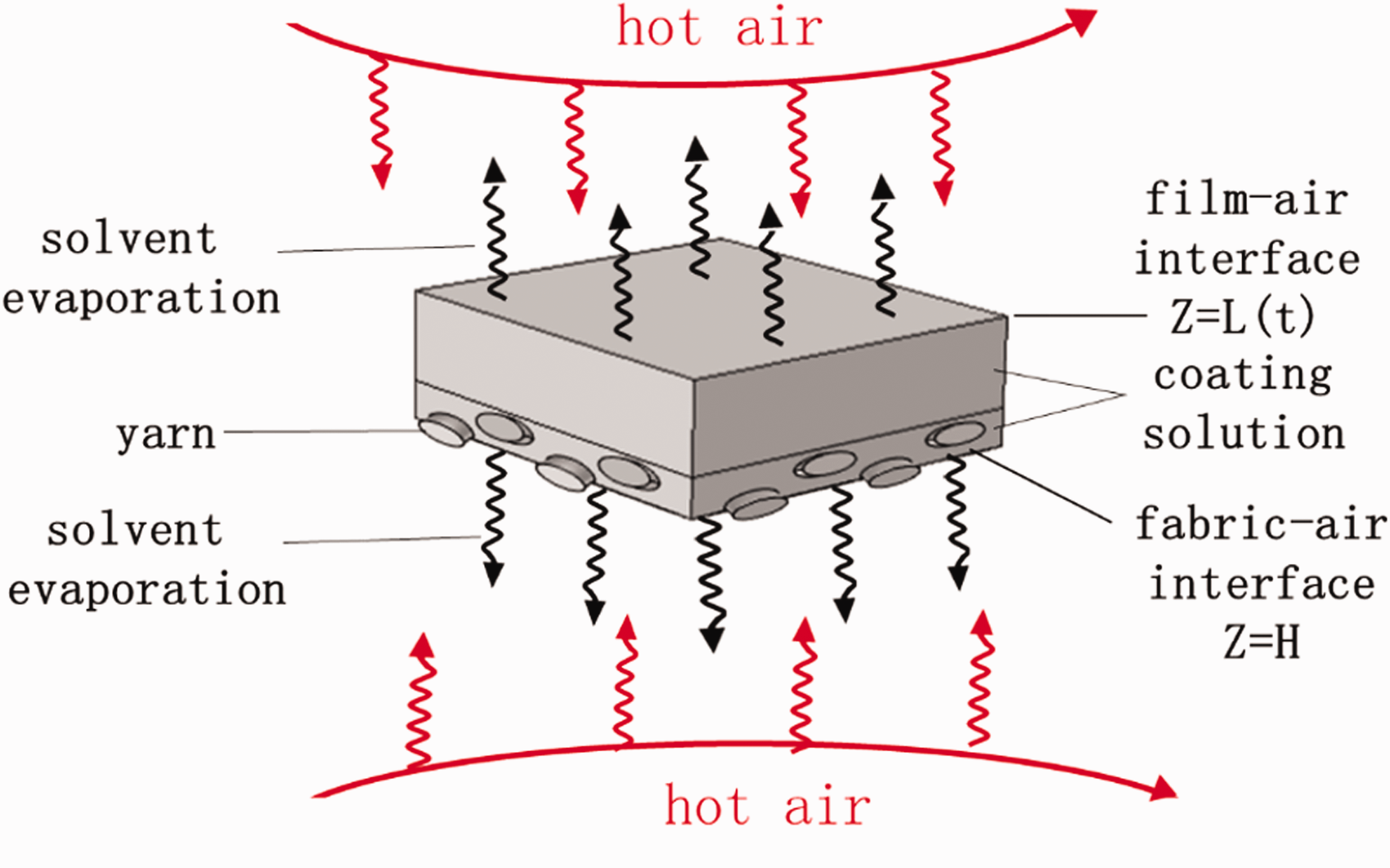
Schematic diagram of solvent diffusion during curing of the coated fabrics.
Mathematical model
The curing process of a film is a complex process in which heat transfer, mass transfer and resin volume shrinkage influence each other [15]. Heat is transferred from the environment to the interior of the film, which affects the mass transfer process by changing the internal temperature of the film. However, since the vapor pressure of the solvent at the gas-liquid interface is higher than that in the air, the solvent inside the film continuously diffuses into the atmosphere. As the solvent evaporates, the thickness of the resin coating decreases. When the vapor pressure of the solvent in the film is equal to that in the air, the diffusion reaches an equilibrium. Therefore, heat transfer, mass transfer and volume shrinkage all need to be considered during the curing process. To investigate the solvent diffusion mechanism during the curing process of the coated glass fiber fabrics, the following assumptions were proposed: The curing process of the film begins when the coating solution have penetrated into the fabric, and the penetration process of the coating solution is ignored; The coating solution is present above the yarns and in the pores between the yarns, and the capillary effect of the yarn on the coating solution is ignored; The environmental temperature and air speed are constant during the curing process; The thermal properties of the coating solution and yarns are constant during the curing process; The free volume parameters of the solvent-polymer system are constant.
Equation for heat transfer
Since the fabric is a porous material, the coating solution will penetrate into the pores of the fabric, therefore, solvent diffusion into the fabric pores should be considered. In porous fabrics, the thermal physical properties of the coating solution and the yarn are combined into effective characteristics, namely effective volume heat capacity and effective thermal conductivity. The conservation of energy equation in the fabric is as shown in equation (1) [15,16].



The coating solution above the fabric is not porous material, so fluid heat transfer is adopted, and the energy conservation equation of the coating film during curing process is as follows:
The left side of the equation represents the increased energy per unit time for the coating solution system, while the first and second formulae on the right side of the equation represent the heat transferred by thermal conduction and the heat carried by solvent diffusion in the coating solution system, respectively.
Equation for mass transfer
In the coated fabric system, solvent molecule diffusion conforms to the second law of Fick diffusion [17], which is an unsteady process changing with time. The equation for mass transfer is shown in equation (6).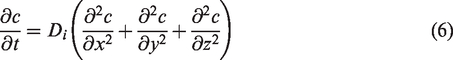


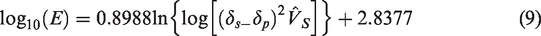
Relationship equation of coating solution volume with time
During the coated fabric drying process, the continuous solvent evaporation causes the coating film to gradually thin. Based on the solvent mass balance equation, the equation for coating thickness change with time was established [20], as shown in equation (10):


Solution conditions of solvent diffusion model
Initial conditions and model parameters
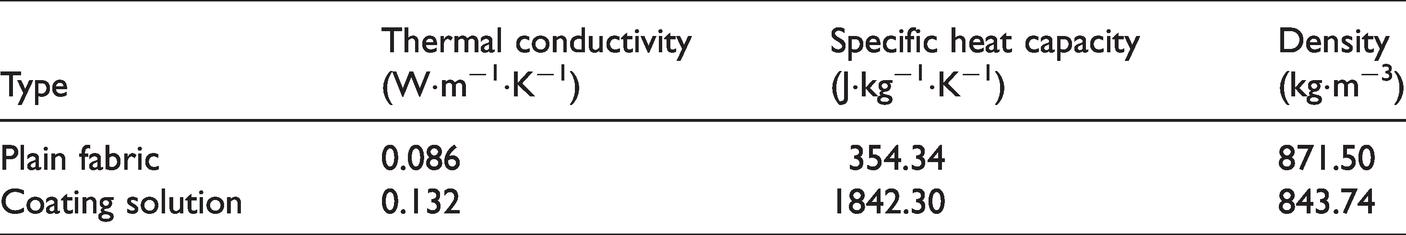
The air speed and temperature of the environment were defined as 0.095 m·s−1 and 298.15 K, respectively. The initial mass fraction of the acetone in the PMMA coating solution was 80% while the initial thickness of the coating film on the glass fiber fabric was 960 μm. The free volume parameters required for the calculation were as shown in Table 2 while the material properties of the fabric and coating solution were as shown in Table 3.
Free volume parameters of the solvent-polymer system.

Material properties of the fabrics and coating solution.
Setting of the boundary conditions
During the curing process of the coated fabric, due to the existence of fabric porosity, the coating solution will penetrate into the fabric pores, therefore, the fabric-air interface and film-air interface will exchange heat and mass with the surroundings.
At position 
While the mass transfer boundary equation is:
At position 
While the mass transfer boundary equation is:
Calculation results and verification
Model calculation results
Based on the above-mentioned solvent diffusion model in the coated fabric curing process, the distribution of solvent concentration at any position inside the coating and the variation curve for the solvent interdiffusion coefficient with time in the coating solution system were predicted. The results were as follows:
As shown in Figure 3(a), solvent concentration at different positions inside the film gradually decreased with time extension, and tended to be constant at the 2,000 s mark, indicating that the curing process was basically completed under this condition. At this time, the average residual solvent concentration in the film was 51.528 kg·m−3. In addition, after the same drying time, solvent concentration at 455 μm away from the fabric surface decreased by more than that at 273 μm. This is attributed to the fact that the driving force for diffusion is the solvent pressure difference and concentration gradient. Solvent molecules move from the inside of the film to the film-air interface, and solvent evaporation occurs at the film-air interface. The closer the solvent is to the air side, the greater the concentration gradient and the faster the diffusion.
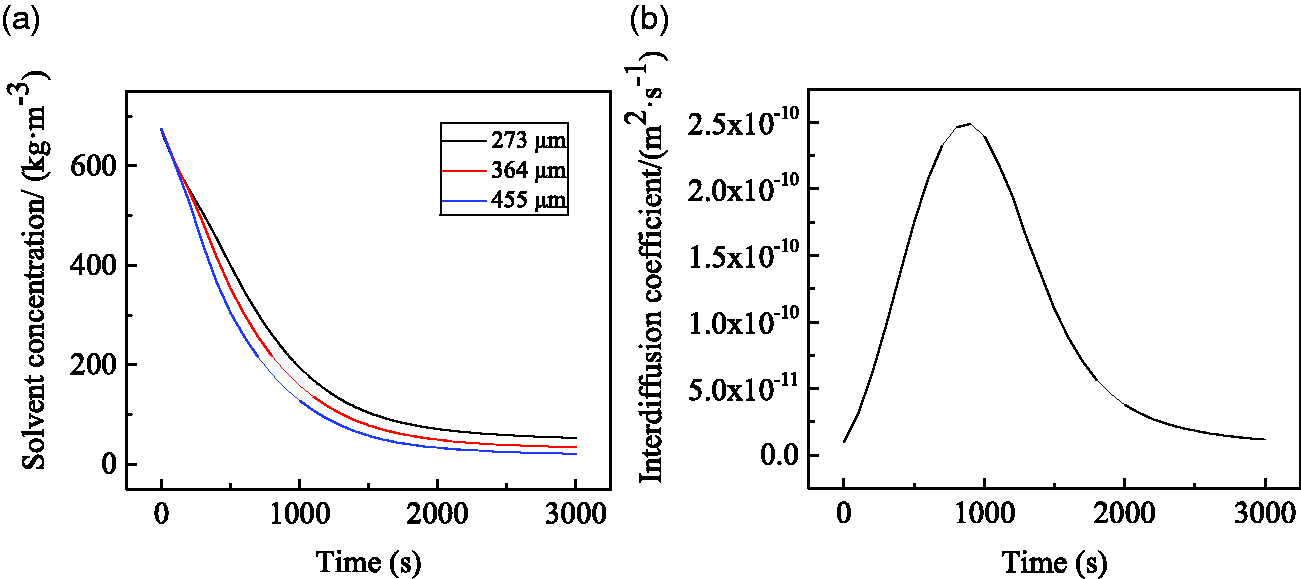
Solvent diffusion during curing of the coated fabric. (a) Distribution of solvent concentration at 273, 364 and 455 μm away from the surface of the fabric, and (b) time dependence curve for the interdiffusion coefficient.
As shown in Figure 3(b), the interdiffusion coefficient in the solvent-polymer system first increased and then decreased with time extension, and reached the maximum value after about 1,000 s. At the initial diffusion stage, the solvent concentration gradient was higher, and the difference between solvent vapor pressure and ambient vapor pressure was larger, therefore, solvent diffusion was faster. As the diffusion progresses, there is less solvent in the system, and the viscosity of the coating solution and the diffusion resistance are both larger.
Because the three-dimensional model is based on the real pore structure, the coating solution will penetrate into the fabric. In order to clearly and intuitively observe the concentration distribution in the coated fabric at different times during the drying process, we selected the distribution diagram of solvent concentration in the coated fabric at 100 s, 300 s, 600 s, 900 s, 1500 s and 3000 s, as shown in Figure 4.

Distribution of solvent concentration in the coating at 100 s, 300 s, 600 s, 900 s, 1500 s and 3000 s.
It can be seen from Figure 4 that when the drying time was 100 s at Z = 0 (the interface between fabric and coating solution), the color of solvent concentration in the coating fabric system was dark red, and it can be seen from the legend that the solvent concentration in the system was larger. With the extension of drying time, the solvent volatilized and the color of the system became lighter. When the drying time was 3000 s, the color of solvent concentration in the coated fabric system changed to orange, and the solvent concentration in the system was small. The distribution of the solvent concentration in the coating film can be clearly and intuitively understood by the cross section of the coating fabric system at different positions and at different times, which can provide a theoretical basis for the follow-up study of the film performance and film quality.
Results verification
Raman spectra of single PMMA solid, acetone solution and coating solution with different solvent mass fraction were obtained using laser confocal Raman spectroscopy (model: xplora plus; manufacturer: Horiba scientific instruments, Japan) as shown in Figures 5 and 6. The test steps were as follows: sample preparation; instrument calibration; setting test conditions (laser wavelength and laser intensity at 785 nm and 10%, grating at 600 Gr/mm, objective lens at 20 times, laser slit at 100, scanning spectrum range from 200 cm−1 to 2000 cm−1); sample focusing and testing (the laser can detect the Raman signals at different positions inside the sample by adjusting the three-dimensional XYZ lifting platform of the instrument, and set the laser scanning time to 20 s, integrate twice, and start the test); record the test data, and analyze the collected spectra. The characteristic peaks of acetone and PMMA in the coating solution are 786.6 and 811.4 cm−1, respectively. The spectrometer directly obtained the spectrogram of the sample rather than the concentration value, so it was necessary to establish the relationship between the peak intensity ratio and the mass ratio of PMMA/acetone at the characteristic peak, and draw the concentration standard curve, as shown in Figure 7.

Raman spectra: (a) acetone solution and (b) PMMA polymer.

Local spectra of coating solution with different solvent mass fraction.

The relation curve between
The concentration calibration equation was established as follows: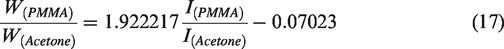
The linear correlation coefficient was

Comparison of simulated concentration and experimental concentration at (a) 273, (b) 364 and (c) 455 μm away from fabric surface.
As shown in Figure 8, before the 2,000 s mark, both simulated and experimental values of solvent concentration decreased rapidly with time. However, after the 2,000 s mark, solvent concentration in the coating solution remained nearly constant, indicating that it took no more than 2,000 s to complete the curing of the coating solution under this condition. In addition, the experimental values of solvent concentration were consistent with the simulated values. The average relative errors between the simulated values and the experimental values at 273, 364 and 455 μm away from the fabric surface were 6.9%, 10.8% and 12.1%, respectively. In the latter drying stage, the simulated values of solvent concentration were slightly higher than the experimental values, indicating that the solvent diffusion as predicted by the model was slightly slower. This is because the free volume parameter of the solvent-polymer system was assumed to be constant in the model calculation, and the influence of the change in solvent concentration on the free volume parameter was ignored.
Influence of fabric structure parameters on solvent diffusion
Fabric weave
Four types of fabrics, plain, 2/1 twill weave, 3/1 twill and 5/3 satin, were woven with glass fiber with yarn fineness of 280 tex (The electron microscope and model of plain fabric are shown in Figure 1, and the remaining three kinds of weave are shown in Figure 9). Their warp density was 120 ends/10 cm and weft density was 100 picks/10 cm. The weave structure parameters of different fabrics were shown in Table 4. Under the drying temperature (80 °C), air speed (0.7514 m·s−1; using a DGP-9057B-2 blast drying oven) solvent mass fraction (80%) and coating thickness (960 μm) conditions, the curing process of the plain coated fabric was simulated, and the effects of different fabric structures on solvent diffusion were studied. The calculation results are shown in Figure 10.

Different fabric types. (a) microscopic images of real fabrics, (b) geometric model, (c) warp cross-section, and (d) weft cross-section.
Different fabric weave parameters.

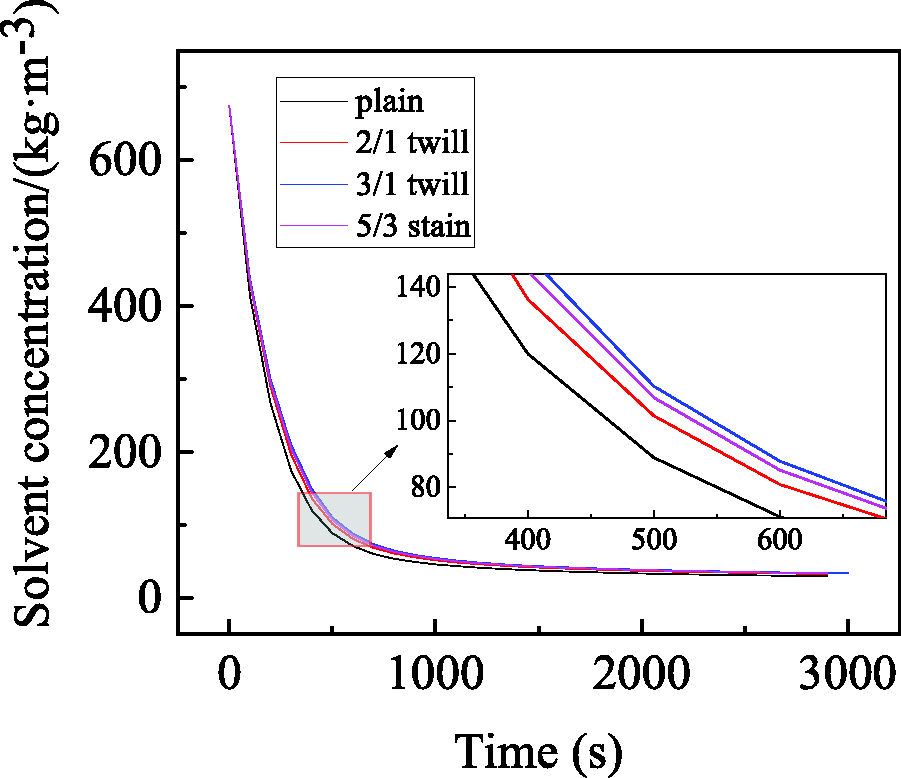
Influence of different fabric weave on diffusion.
As shown in Figure 10, the solvent concentration in the coating film after curing for the same time was in the following order: 3/1 twill > 5/3 satin > 2/1 twill > plain. It reflects that the order of solvent diffusion rate was as follows: 3/1 twill < 5/3 satin < 2/1 twill < plain, that is, the solvent diffused faster in the plain fabric. For the same fineness of yarn and warp/weft density of fabric, plain fabrics were the thinnest. It can be seen from Table 4 that the porosity of the fabric was 5/3 satin > 2/1 twill, but Figure 10 shows that the solvent diffusion rate was 5/3 satin < 2/1 twill. The reason for this phenomenon may be that both the fabric thickness and porosity will affect the solvent diffusion. However, when the porosity was similar, the thickness of the fabric was the main factor affecting the solvent diffusion, so we need to discuss the influence of the fabric thickness and porosity on the diffusion.
Fabric thickness
Under the conditions of temperature 80 °C, wind speed 0.7514 m·s−1, solvent mass fraction 80% and coating thickness 960 μm, the curing process of coated plain fabric with porosity of 52.76% was simulated. The solvent diffusion in the coating system with fabric thickness (0.2-1.1 mm) was studied. The calculation results were as shown in Figures 11 and 12. When the solvent diffusion reaches equilibrium in the coating solution, the time required was referred to as the equilibrium drying time, which was represented by

Solvent concentration under different fabric thickness.
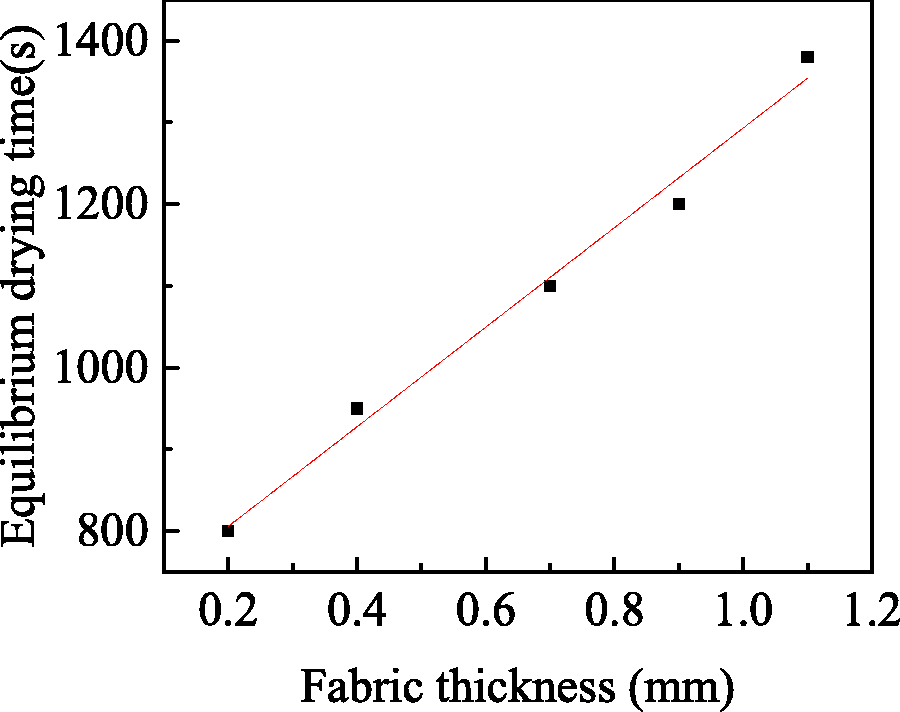
Relationship between equilibrium drying time and fabric thickness.
As shown in Figure 11, at the same drying time, the thicker the fabric was, the smaller the decrease of solvent concentration was and the slower the solvent diffusion rate was. Because in thick fabrics, the distance that the solvent molecules moved from the coating solution to the fabric- air interface was relatively longer than that of the thin fabrics. With the increase of fabric thickness from 0.2 mm to 1.1 mm, the equilibrium drying time gradually increased from 800 s to 1380 s. The relationship between fabric thickness and equilibrium drying time was shown in Figure 12. The fitting equation of them was as follows:
Fabric porosity
Under the conditions of temperature 80 °C, wind speed 0.7514 m·s−1, solvent mass fraction 80% and coating thickness 960 μm, the curing process of coated plain fabric with fabric thickness 0.7 mm was simulated. The solvent diffusion in the coating fabric system with porosity (0-1) was studied. The calculation results are as follows:
As shown in Figure 13, when the coating solution was coated on the impervious substrate (porosity was 0), the time for solvent diffusion to reach equilibrium was 1700 s;when the coating solution was free diffusion in air (porosity is 1), the solvent diffusion was fast and the equilibrium time was 900 s. It can be concluded that the effect of porosity on solvent diffusion was significant. Because the larger the porosity of the fabric was, the larger the contact area between coating solution and air was. The faster the solvent diffusion is, and the shorter time it took to reach diffusion equilibrium. When the substrate was replaced with glass fiber fabric with porosity range of (0.30-0.75), the relationship between equilibrium drying time and porosity was shown in Figure 14, and the fitting equation of them was as follows: Solvent concentration under different porosity. Relationship between equilibrium drying time and porosity.


Conclusion
By establishing the solvent diffusion model in the coated fabric drying process, the solvent diffusion internal mechanism in porous media was explored and the average relative errors between the simulated concentration and the experimental concentration were 6.9%, 10.8% and 12.1% at 273, 364 and 455 μm away from the surface of the fabric, respectively. The solvent concentration curves of numerical simulation were consistent with that of the experiment and could be used to predict the distribution of solvent concentration during the drying process of coated fabrics. Both fabric thickness and porosity can affect solvent diffusion, but when the porosity is similar, the fabric thickness is the main factor affecting solvent diffusion, and the thicker the fabric is, the slower the solvent diffusion is. The relationship between fabric thickness and drying time is
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Natural Science Foundation of Tianjin (18JCYBJC86600).
