Abstract
Tetraethoxysilane (TEOS),
Introduction
Cotton fabrics have been always popular because of good permeability, abrasion-resistance, softness and comfort properties.1,2 They are widely used in garment, decorative and industrial fabrics.3,4 However, once a cotton fabric end product is ignited as a first item and involved in a fire as a combustible, heat accumulates and dissipates quickly, flame emerges and spreads fast, 5 toxic and irritant smoke merges and transports everywhere. Also, continuous and repeatable smoldering processes may exist. Such fires will exist in the long term due to the inflammable nature of such fabrics. 6 Therefore, it is always necessary to enhance flame retardancy levels of cotton fabrics.
Aiming at this, many techniques including layer-by-layer assembly (LBL),7,8 chemical grafting, 9 plasma treatment 10 and sol-gel process11,12 have been used. A sol-gel process shows features of simple preparation steps, easy modifications, less environmental impacts and time saving operations.13–16 It can be used to prepare a homogeneous sol and create a physical barrier (gel coating) to protect the matrix from oxygen transport and heat transfer.17,18 At present, the sol-gel process has been widely used in modifying fabrics with antibacterial, UV proof, water repellent and flame retardant functions.19–22
In the last decades, many studies about flame retardant fabrics finished have been carried out. Among them, the silicon-based flame retardants have been applied on the cellulosic fibers to meet related requirements.23–27 And 3-(trimethoxysilyl) propyl methacrylate (KH570) as a typical type of silane can react with hydroxyl groups of a matrix to form stable covalent bonds.28,29 Furthermore, the boron-containing flame retardants have also fine application prospects and cases. The boron compounds have both good flame retardant effects and strong smoke suppression abilities. 30 It should be noted that inorganic borax and borate are not washing durable for finishing processes, while organic boron compounds have poor hydrolytic stabilities, so they are less investigated as flame retardants compared with nitrogen/phosphorus-based compounds. 31 In order to overcome the drawbacks, boron compounds have been combined with other elements such as phosphorous, nitrogen, titanium and silicon to obtain potential synergistic effect.32–35 Zhu et al. 33 designed a synergistic flame-retardant system including glycosyl cross-linking boron acid and phytic acid ammonium salt (r-GBAP) to enhance durable flame retardancy of cotton fabrics. The LOI and char residue increase to 37.4% and 38.4%, respectively. Bentis et al. 34 used titanium-based sol and boric acid as additives to prepare flame retardant and water resistance cotton fabrics. In particular, the cotton fabric treated with a molar ratio of 1.0:2.5 (Ti sol: Boric acid) shows good thermal stability and flame resistance. Its char residue rate at 600.0°C increase from 3.36% (original cotton fabric) to 11.46%. Li et al. 35 used TEOS, zinc borate (ZB) and 3-glycidoxypropyltrimethoxysilane (KH560) to prepare silica-based single, composite and hybrid sols via sol-gel processes and treated cotton fabrics with them via dipping-curing methods. It is found that the char residue rate of SiO2-KH560-ZB coated cotton fabric is as high as 28.43% and its peak heat release rate (PHRR) is reduced by 26.9% (83.7 W/g) compared with those of original cotton fabric. As can be seen from typical studies above, more attempts have been tried to introduce boron-containing flame retardants to a sol-gel system and coat them onto cotton fabric surfaces. Silica sols are mostly used as basic finishing reagents for cotton fabrics to constitute potential binary and ternary flame retardant systems. They are still ample rooms in selecting potential boron-based flame retardants, pursuing of higher flame efficiency levels and exploring corresponding synergistic effects for cotton fabrics. In this sense, ammonium pentaborate as boron-based inorganic flame retardants that have not been applied in this way are interested to be explored considering their chemical structures and functional natures (thermal stability, low toxicity and smoke suppression capability). To be more quantitative and precise, flame retardant effect can further be evaluated through a new derivative parameter of ΔLOI/Δm, 35 and interaction effects among components can be assessed through a synergistic effectiveness parameter (SE).36–38
Therefore, in this work, based on sol-gel processes, inorganic silica sol, inorganic-organic silica-KH570 composite sol and silica-KH570-AP doped sol were prepared by introducing TEOS as a precursor, KH570 as a coupling reagent and AP as a flame retardant additive. Such as-prepared silica-based sols were then treated onto cotton fabrics through dipping-curing processes. Surface microscopic morphologies, surface element contents and element distributions of all cotton fabrics were characterized by SEM-EDX and XPS. Internal chemical structures and crystal structures were determined by FTIR and XRD. Thermal stabilities and flame retardancy levels of original and treated cotton fabrics were investigated by TGA and LOI experiments and VFTs. The evolved gaseous products for original and SiO2-KH570-AP@COT during the thermal degradation were analyzed by TG-IR. The mechanical performances of the resultant cotton fabrics were performed in detail. Indices of ΔLOI/Δm and SE were calculated and analyzed. Based on the above results, the flame retardant mechanisms of treated cotton fabrics were generally deduced.
Experimental
Materials
The cotton fabric (weight: 111 g/m2, 13.3 tex × 7.2 tex) was supplied by Nanjing Wizards Beauty Textile Co., Ltd. (Jiangsu, China). TEOS (AR, ≥35.0%) and KH570 (IR, ≥99.0%, ρ:1.04 g/mL) were obtained from Sinopharm Chemical Reagent Co., Ltd (Shanghai, China). AP (CAS:12007-89-5, RG, ≥99.0%) was provided by Adamas Reagent Co., Ltd (Shanghai, China). Hydrochloric acid (HCl, AR, 37.0%) was purchased from Shanghai Pilot Chemical Co., Ltd (Shanghai, China). Anhydrous ethanol (C2H5OH, AR, ≥99.6%, ρ:0.79 g/mL) was provided by Wuxi Yasheng Chemical Co., Ltd (Jiangsu, China). Deionized water (H2O, IR, ≥99.9%, ρ:1.00 g/mL) was purchased from Nanjing Wanqing Chemical Glassware Instrument Co., Ltd (Jiangsu, China). The chemical structures of cotton cellulose, TEOS, KH570 and AP are presented in Figure 1.35,36 Usually, AP also combines four crystal water molecules that are not displayed in current chemical structure. Chemical structures of cotton cellulose, TEOS, KH570 and AP.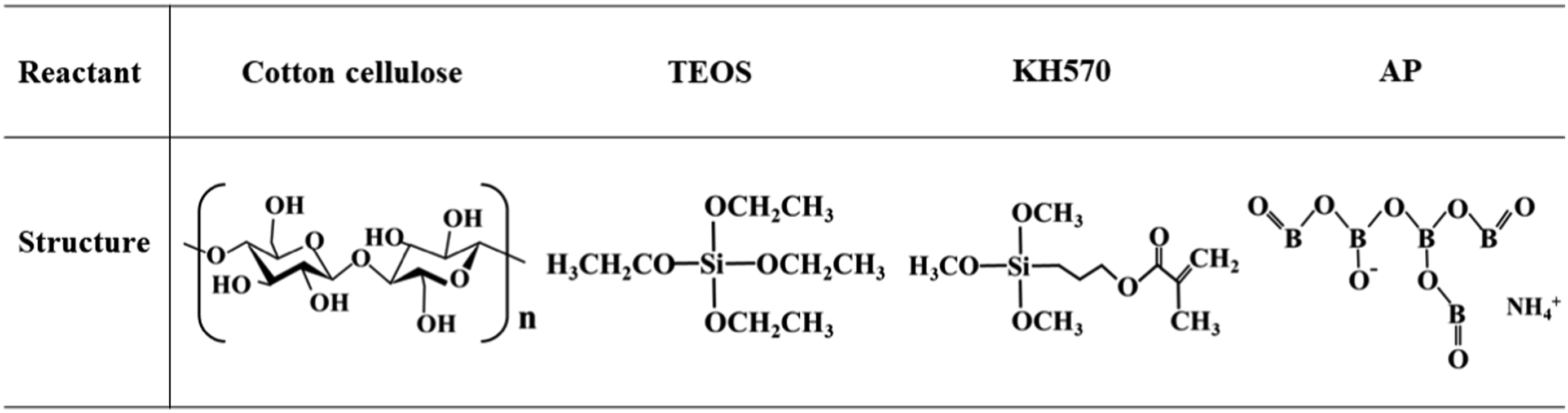
Preparation of single silica sol
TEOS (52.1 g, 0.25 mol), C2H5OH (23.0 g, 0.5 mol), and H2O (27.0 g, 1.5 mol) were mixed in a molar ratio of 1:2:6 and stirred by a magnetic stirrer (DF-101S, provided by Gongyi Yuhua Instrument Co., Ltd., China) at 60.0°C for 30 min. The mixture was adjusted by 10 wt% HCl to obtain a pH value of 6.0. After that, the whole solution was stirred at 60.0°C for 2 h to obtain a single original SiO2 sol. The hydrolysis and condensation reaction processes of TEOS are presented in Figure 2. The processes of TEOS hydrolysis (a) and condensation (b) reactions.
Preparations of KH570 and AP hydrolysates
In order to strengthen potential chemical bonding forces among components, KH570 was selected as a silane coupling agent. C2H5OH (56.9 g, 72.0 vol%), deionized water (8.0 g, 8.0 vol%) and KH570 (10.4 g, 20.0 vol%) are mixed by magnetic stirring at room temperature. Certain amount of AP (4.0 g) was dissolved in deionized water (96.0 g) to form a 4.0 wt% AP hydrolysate.
Preparation of composite silica-based sol
KH570 (29.2 g, 0.125 mol) was added to the single SiO2 sol that consisted of TEOS (52.1 g, 0.25 mol), C2H5OH (23.0 g, 0.5 mol), H2O (27.0 g, 1.5 mol) and mixed by magnetic stirrer at 60.0°C for 30 min. Then, 10 wt% HCl was added to adjust a pH value of 6.0. After stirring for 2 h, a stable and transparent SiO2-KH570 composite sol was obtained. The hydrolysis of KH570 and the reaction between SiO2 and KH570 are shown in Figure 3. The hydrolysis of KH570 (c) and the reaction between SiO2 and KH570 (d).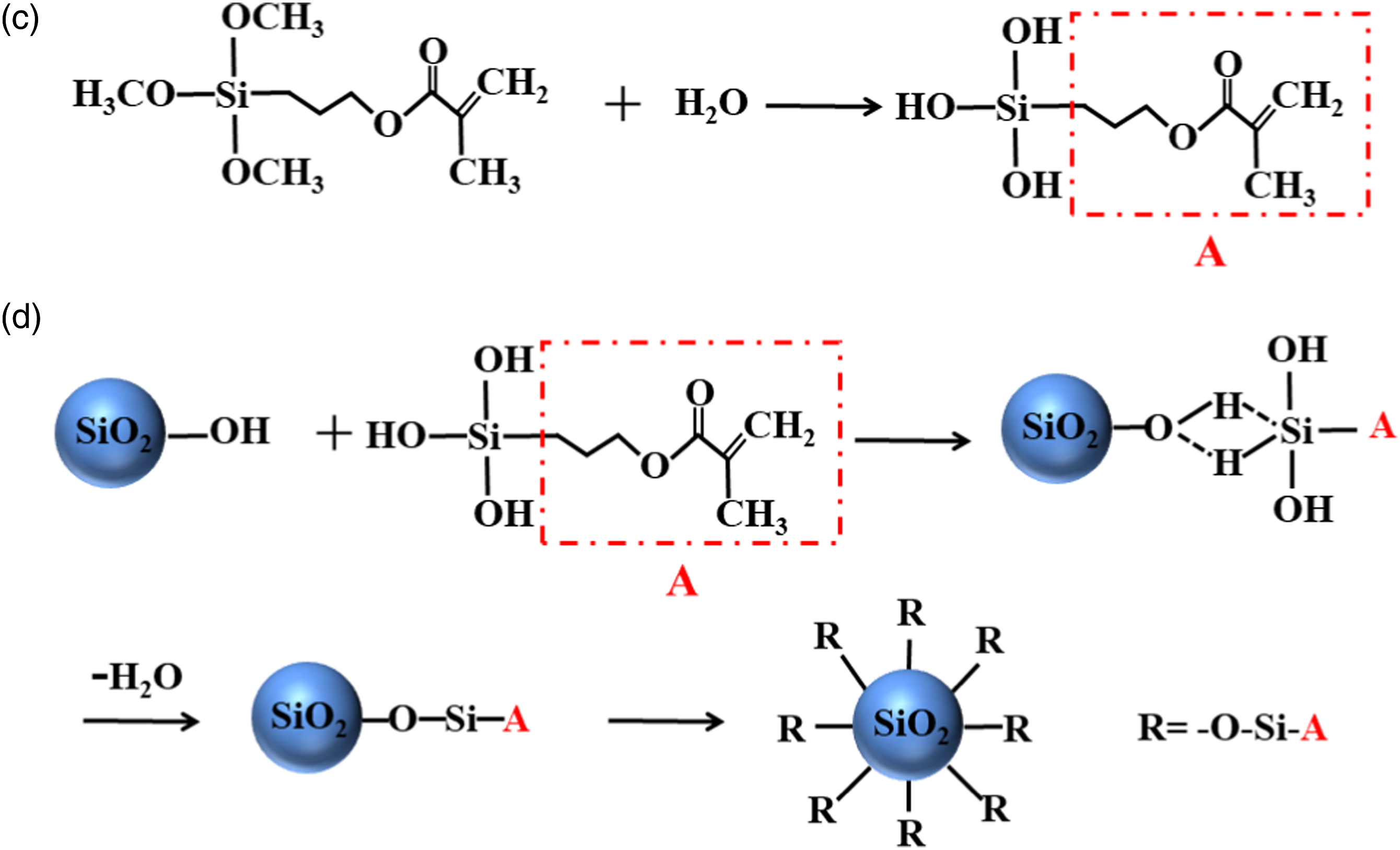
SiO2-KH570-AP doped sol was obtained by adding 4.0 wt% AP hydrolysate (100.0 g) to SiO2-KH570 sol (200.0 g) under the stirring condition.
Cotton fabrics treated by silica-based single, composite and doped sols
Information of prepared sol and hydrolysate types.


Schematic illustration of treatment process of the SiO2-KH570-AP@COT.
The add-on (%) of the treated fabrics is determined as follows.
The macro-surface appearances of the cotton fabrics finished by the series of sols are shown in Figure 5. Apparently, after observations and experiences, the shapes, colors and handles of treated cotton fabrics have not changed much as compared with original cotton fabrics. Appearances of cotton fabrics before and after finishing silica-based sols (the numbers represent add-on (%)).
Characterization techniques for treated (finished) cotton fabrics
Surface microscopic morphologies and surface elemental contents of cotton fabrics and their char residues after VFTs were observed by a SU8010 scanning electron microscope (SEM; Hitachi, Japan) along with a max20 EDX mapping. SEM was utilized with an accelerating voltage at 5 kV under vacuum condition. All the treated samples were coated with gold before SEM testing.
X-ray Photoelectron Spectroscopy (XPS) was carried out on a K-Alpha+ XPS spectrometer (Thermo Fisher Co., Ltd., USA) to perform surface elemental analyses. Cotton fabric samples (5×5 mm2) were cut and fixed to conductive carbon adhesive tapes.
Fourier transform infrared (FTIR) spectra of cotton fabric samples were recorded from 4000 cm-1 to 400 cm-1 by an IS5 FTIR spectrometer (Thermo Fisher Co., Ltd., USA) by using KBr pellet technique. Each sample was scanned 32 times at a resolution of 4 cm-1.
X-ray diffraction (XRD) patterns of cotton fabric samples were obtained by using a D8 Advance photoelectron spectrometer (Bruker Co., Ltd., German) with CuKa radiation generated at 36 kV and 20 mA at room temperature. The diffractogram scattering angles ranged from 10° to 50°.
Pyrolysis properties (thermal stability) of cotton fabric samples were conducted with a TGA/DSC3+ (Mettler-Todolly, USA). The samples with masses of 5–10 mg were heated from 30°C to 700°C at a heating rate of 10°C/min under nitrogen atmosphere.
According to the standard of GB/T 5454-1997, flammability levels of cotton fabric samples were evaluated by a ZY6155 A limiting oxygen index (LOI) apparatus (ZhongNuo Quality Inspection Equipment Co., Ltd., China). A fabric sample of 150×58 mm2 was fixed with a clip and placed in a glass cylinder. The sample was ignited from the top, and the ratio of nitrogen and oxygen was adjusted to obtain the oxygen concentration that the cotton fabric could just burn sustainably.
According to the standard of GB/T5455-2014, flame retardancy levels of the cotton fabric samples were evaluated by a CZF-5CD50 W vertical flammability tester (Tianjin Xunyu Technology Co., Ltd., China). A fabric sample of 300×89 mm2 was arranged vertically, and the bottom of the sample was exposed to a propane flame for 10 s. After removing the flame source, the after-flame time and after-glow time were measured. Each test was repeated 3 times and the average values were taken as the final results.
TG-IR analyses under nitrogen atmosphere of cotton fabrics (5–10 mg) were implemented on a TGA 4000 thermal analyzer (PerkinElmer Co., Ltd., USA), which was combined with a Spectrum Two FTIR spectrometer (PerkinElmer Co., Ltd., USA). The temperature range was from 30°C to 700°C, and the heating rate was 10°C/min. The recorded spectral range was from 4000 to 500 cm‐1.
The tensile strength data of the cotton fabrics were measured with a CMT6103 electronic universal testing machine (MTS System Corporation, China). The dimension of samples used for the test was 50×10 mm2. The tensile speed was 100 mm/min.
Results and discussion
Surface morphology analyses of original and treated cotton fabrics
The SEM images of original and treated samples are shown in Figure 6. In addition, the EDX probe acts on the surface of each sample to investigate the element contents in the coating. SEM images at × 5000 magnification, corresponding element mapping photos and EDX spectra of original and treated cotton fabrics.
As shown in Figure 6, obviously, there exist differences among micro-surface morphologies of original and treated cotton fabrics. But it can be seen from EDX mapping photos that all the elements (C, O, Si, N and B) are finely dispersed on the surfaces of cotton fabrics. For original cotton fabric, it is relatively smooth. Naturally distorted and clear striations can be observed. Besides, the cotton fabric surface contains only 61.7% C and 38.3% O elements. However, apart from C (51.7%) and O (40.9%), 7.3% Si is inspected for SiO2@COT. This proves that silica sol is successfully deposited on its surface. It has a smoother surface covered by a SiO2 gel coating after baking (Figure 6(a2) and (b2)). This is because the hollow interior and twisted porous surface of cotton fibers provide a good carrier for adsorbing fine particles (nano- and/or micro-scales). 35 And the hydroxyl groups (-OH) generated by dehydration and condensation reactions in the silica sols (Figure 2) can also react with the -OH groups on the surfaces of cotton fibers (Figure 1) to form hydrogen bonds and thus increase the adhesion of the inorganic coating to cotton fabrics. 15 For KH570@COT, concave and convex particles appear on its surface. KH570 as an organic silane coupling agent can form -Si-O-Si- skeleton through -OH groups among internal molecules. In addition, the -OH groups generated by dehydration reaction of KH570 can also form hydrogen bonds with the -OH groups on the cotton fabric surface, which promotes formation of a stable chemical structure (Figure 4). However, the organic structure of KH570 (Figure 1) shows certain space arrangement and makes the surface of treated cotton fabric coarser (Figure 6(a3) and (b3)). For SiO2-KH570@COT, its surface is relatively smooth since a dense gel coating is formed. The composite sol particles are connected by inorganic-organic -SiO2-KH570- (-Si-O-Si-A, Figure 3). 39 Spacing and orientations of cotton fibers are hidden due to strong coupling effect of KH570. As noted from the EDX spectra (Figure 6(b2)–(b4)), cotton fabrics treated with SiO2, KH570 and SiO2-KH570 contain C, O and Si elements. But the sample KH570@COT has only a Si content of 0.7%, so its flame retardant effect may be worse compared with the SiO2@COT (7.3%) and SiO2-KH570@COT (13.0%). For AP@COT, interstices and sunken areas of cotton fibers are filled by fine grains of AP (Figure 6(a5) and (b5)) attributed to the effect of physical adsorption and weak chemical hydrogen bond adsorption (crystal water). AP is stable at 0.0–90.0°C (checked at https://www.chemicalbook.com/). Therefore, AP may release a small amount of ammonia gas due to high temperature baking (above 90.0°C, Figure 4), and its surface is wrinkled and slightly rough. The sample AP@COT contains 8.5% B and 0.1% N due to the small amounts of additions and the release of ammonia gas. For SiO2-KH570-AP@COT, on its surface a dense layer with only a small number of particles is formed. This is assigned that the dispersion effect of AP ions is suppressed under the physical coverage effect of SiO2 gel and chemical coupling effect of KH570. This is favorable for potentially enhanced flame retardancy.
The SEM and EDX images demonstrate that AP doped silica-KH570 sol has been successfully adhered onto the cotton fabric surface. And the homogeneous distributions of elements in the EDX mapping photos manifest that there is good compatibility among AP, silica sol and KH570.
Surface chemical compositions of original and treated cotton fabrics
In order to determine the chemical compositions on the surfaces of all cotton fabrics, the global range XPS spectra are performed and illustrated in Figure 7. XPS spectra of original and treated cotton fabrics (a); enlarged view of AP@COT and SiO2-KH570-AP@COT (b).
For original cotton fabric, C1s (284.6 eV) and O1s (532.2 eV) peaks are detected. The C1s peak at 284.6 eV is contributed by C-H and C-C bonds in cotton fibers. For SiO2@COT, KH570@COT and SiO2-KH570@COT, their XPS spectra exhibit new peaks at 103.5 and 152.6 eV that can be assigned to Si2p and Si2s, respectively.39,40 The results manifest that the SiO2 gel, KH570 hydrolysate and SiO2-KH570 gel coatings have been successfully incorporated onto the surfaces of cotton fibers with chemically covalent connections (hydrogen bonds). From the Figure 6(c2)–(c4), the EDX spectra of SiO2@COT, KH570@COT and SiO2-KH570@COT also show existences of Si element, which is consistent with the EDX results. In addition, the apparent peak of Si2p is further divided into two peaks for SiO2@COT. The peak at approximately 102.1 eV is related to the Si-O-C bond (Figure 4) and the other peak emerging at 103.0 eV corresponds to the Si-O-Si bond (Figure 2). This reflects as-prepared silica sol is both physically and chemically absorbed onto cotton fabrics. For AP@COT, the emerging signals about N1s and B1s demonstrate that AP is successfully entrapped on the cotton fabric substrate. The binding energy peaks of B1s and N1s are located at 190.4 and 398.0 eV, respectively. For SiO2-KH570-AP@COT, Si2p, Si2s, N1s and B1s peaks can be detected, which can prove that the AP doped SiO2-KH570 gel coating is adsorbed to cotton fabric surface. The Si2p and Si2s peaks are derived from SiO2 and KH570, while the N1s and B1s peaks are derived from AP. Moreover, the intensities of element binding energy peaks have changed for different samples. It can be seen from Figure 7(a) that the intensity of C1s peak for SiO2@COT is decreased compared with that of original cotton fabric. EDX results show that C elements account for 61.7% (Figure 6(c1)) and 51.7% (Figure 6(c2)) for original cotton fabric and SiO2@COT, respectively. It can be speculated that the SiO2 gel has largely covered the fabric surface, thereby reducing the content and intensity of C element of SiO2@COT. The intensity of C1s peak is slightly reduced for SiO2-KH570@COT compared with that for KH570@COT, indicating that chemical bonds are formed between the SiO2 gel and the KH570 hydrolysate coatings. It is found from Figure 7(a) that the intensity of O1s (532.2 eV) peak increases significantly after treating with SiO2-KH570 gel as compared with original cotton fabric, which can be assigned to the strong silane coupling agent effect of KH570. By comparing Figure 6(c1), (c3) and (c4), the O content of SiO2-KH570@COT is only higher than that of original cotton fabric, and lower than that of KH570@COT. It shows that KH570 has a specific spatial arrangement as an organic molecule (Figure 1), and tends to lift the O atoms (O1s) after combining with silica sol (Figure 6(a3) and (b3)). Similarly, combined with the content in Figure 6 and the intensity in Figure 7, it shows that KH570 also tends to lift Si atoms (Si2s and Si2p) after combining with silica sol, and fix Si atoms in the shallow layer of the cotton fabric surface. The above results comprehensively show that the coupling effect of KH570 is significant. The intensities of Si2p and Si2s are also the highest for SiO2-KH570@COT from Figure 7(a), which indicates the firm covalent bond linkage is formed between SiO2 and KH570. For AP@COT, the intensities of C1s and O1s peaks have no significant changes in contrast to original cotton fabric. This is due to the small addition (loading) amount of AP (Figure 6(c5)) and relatively weak binding force formed between AP and the cotton fabric. However, for SiO2-KH570-AP@COT, the intensity of C1s peak decreases and the intensity of O1s peak increases as compared with AP@COT. This can be attributed to the combination of O and Si via -SiO2-SiO2- (-Si-O-Si-) and -SiO2-KH570- (-Si-O-Si-A-) (Figures 3 and 4). The above results are consistent with the EDX results.
Functional group analyses of original and treated cotton fabrics
Figure 8 depicts FTIR spectra of original and treated cotton fabrics. For original cotton fabric, the broad peak at around 3300 cm-1 is assigned to the stretching vibration of -OH group, which is caused by the structure of cellulose (Figure 1 cellulose) and absorbed water. The peak at about 2915 cm-1 is associated with the stretching vibrations of C-H structures.
5
The peak near 1161 cm-1 is accordance with the C-O-C stretching vibration of the glucose ring (Figure 1 cellulose).
41
FTIR spectra of original and treated cotton fabrics.
For the treated samples, they still contain these groups, indicating that the sol-gel systems do not damage the original molecular structures of cotton fabrics. Meanwhile, spectra of treated cotton fabrics show some new adsorption peaks. For SiO2@COT, its spectrum displays new bands at 521 cm-1 and 790 cm-1 that belong to the Si-O-Si bending and Si-O-Si symmetric stretching. The absorption peaks between 1060 and 1160 cm-1 are caused by Si-O stretching vibrations that come not only from the internal structures of SiO2, but also from the cross-linking structures between cotton fabric and silica sol. The existence of Si-O-Si and Si-O groups indicate that SiO2 sol has been successfully applied to the cotton fabric surface. This is consistent with the results of EDX and XPS. For KH570@COT, its spectrum shows bands at 1720 cm-1 and 1638 cm-1 that belong to C=O and C=C vibrations (Figure 1 KH570). 42 And the peak at 1050 cm-1 indicates the presence of Si-O-C groups, which confirms that KH570 is adhered to the cotton fabric surface. For SiO2-KH570@COT, its spectrum also shows absorption peaks of Si-O-C and C=O vibrations. However, the intensity of Si-O absorption peak between 1060 and 1160 cm-1 becomes weaker, which is attributed to the condensation reaction between SiO2 and KH570 (Figure 3(d)). For AP@COT, the intensity of -OH absorption peak at 3300 cm-1 is enhanced by anti-symmetric stretching vibrations of a large number of -OH groups on the surface of cotton fabric. Also, the 1147, 1091 and 665 cm-1 signals are associated with the -NH, B4-O and B3-O vibration, respectively. For SiO2-KH570-AP@COT, there are absorption peaks of typical groups from SiO2, KH570 and AP at the same time, which indicates the AP doped silica-KH570 sol has been coated onto the cotton fabric successfully, both physically and chemically. As in the EDX analysis above, SiO2-KH570-AP@COT contains C, O, Si, N and B; and in the XPS analysis, SiO2-KH570-AP@COT detects C1s, O1s, Si2p, Si2s, N1s and B1s simultaneously. These facts agree well with each other.
Microscopic (crystal) structure analyses of original and treated cotton fabrics
The internal crystal structures of original and treated cotton fabrics reflected by XRD patterns are shown in Figure 9. For original cotton fabric, its pattern exhibits both crystalline and amorphous areas. The characteristic peaks at 14.8°, 16.8°, 22.8° and 34.7° correspond to the crystal faces (1-10), (110), (200) and (004) of cellulose I.
43
It can be seen from Figure 5 that SiO2@COT, KH570@COT, SiO2-KH570@COT, AP@COT and SiO2-KH570-AP@COT show high add-on (%) values. In addition, EDX, XPS and FTIR results also indicate that the coatings are successfully adhered to the cotton fabric surfaces. This suggests there should be detectable components in coatings for treated cotton fabrics. It is known that XRD patterns of SiO2 and KH570 show peaks at 21.0 and 22.0°, respectively.42,44 And AP has high intensity and sharp crystalline diffraction peaks at 14.7°, 16.0°, 25.1°, 26.3°, 31.6°, 41.3° and 47.6°, respectively.
45
However, there are no significant differences between original cotton fabric and SiO2@COT, KH570@COT, SiO2-KH570@COT, AP@COT, SiO2-KH570-AP@COT. It indicates that the treatments do not affect the main crystal structures of cotton fabrics. The above XPS and FTIR results show that -OH groups on the cotton fabric surface and SiO2 sol are combined, and -OH groups of SiO2 sol and KH570 are also combined. As a result, the original hydrogen atoms on the surface are replaced by "impurity" atoms carried by silica sol and KH570. The reaction is mainly atomic occupancy, so the crystal structures barely change. There are small changes in peak positions or intensities, but no new peaks are generated. Besides, for SiO2-KH570-AP@COT, this phenomenon is assigned to the low amounts of AP and the good compatibility of silica sol and AP (Figure 6(b6)). Although there are no new peaks, the intensities of diffraction peaks of treated cotton fibers are slightly lower than that of original cotton fabric. This should be caused by relatively high treatment temperature (Figure 4, “dry at 90.0°C, cure at 120.0°C”) of dipping-curing processes. Another reason is that cotton fibers are chemically bonded and embedded by SiO2-KH570 composite sol and SiO2-KH570-AP doped sol, the original crystal structures cannot be fully displayed. In addition, it is inferred from hand feeling tests that the amorphous states of gel coatings and the lack of some ordered crystal structures will help to maintain the softness of cotton fabrics. XRD patterns of original and treated cotton fabrics.
Pyrolysis characteristic analyses of original and treated cotton fabrics
The thermal stability of original and treated cotton fabrics is characterized by TGA in nitrogen atmosphere. TG and derivative thermogravimetric (DTG) curves of cotton fabrics are shown in Figure 10, and main pyrolysis data are listed in Table 2. The onset decomposition temperature (T10%, defined as the temperature of 10 wt% mass loss), the temperature of maximum mass loss rate (Tmax) and char residue rate at 700.0°C are included. TG (a) and DTG (b) curves of original and treated cotton fabrics. TGA data of original and treated cotton fabrics.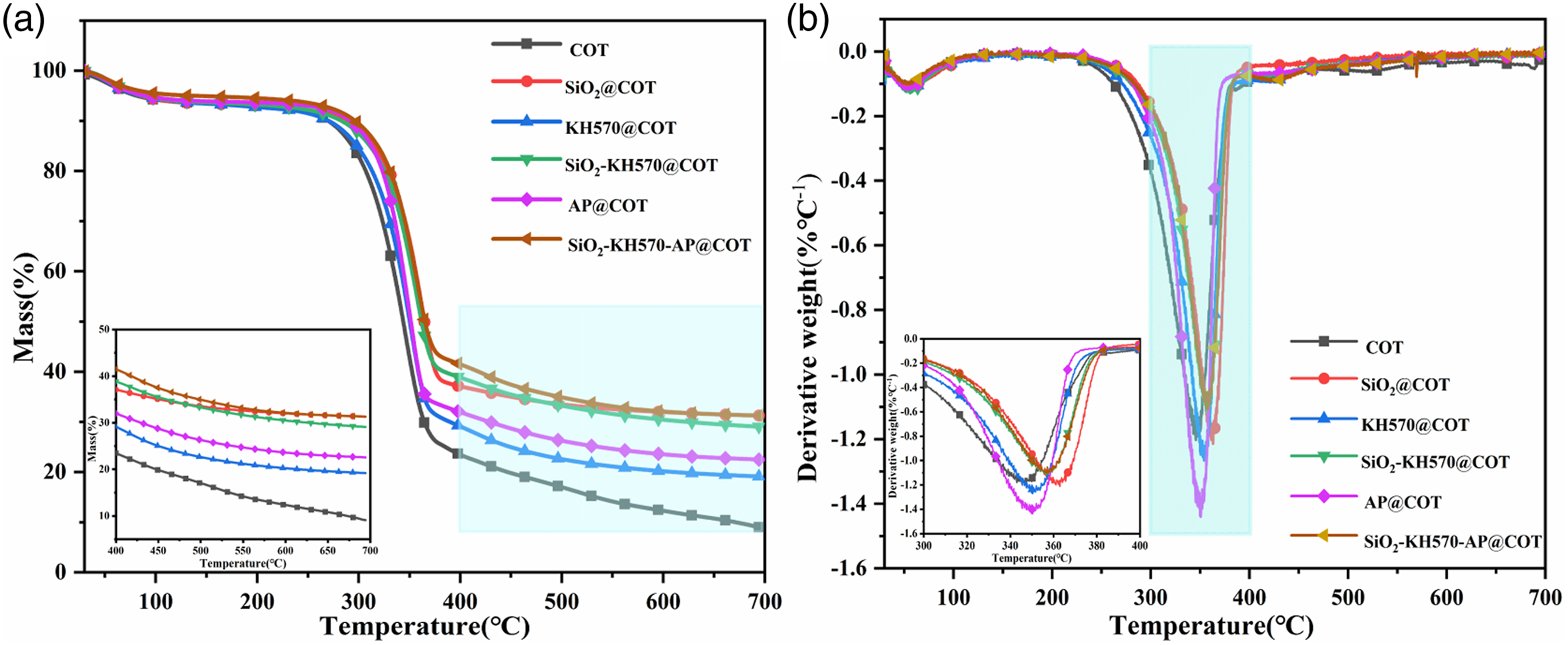

For original cotton fabric, a slight weight loss before 100.0°C occurs due to the physical property changes and loss of physically absorbed water. The thermal degradation mainly occurs between 260.0 and 400.0°C, and the weight loss is about 64.0%. During this stage, the glycosyl units (Figure 1 cellulose) depolymerizes to produce volatile species and the main chain dehydrates to form thermally stable char. 46 Upon 400.0°C, the formed char further oxidizes and a steady char residue is obtained at nearly 700.0°C. Finally, the char residue rate is 9.0%. Besides, TG data shows that the T10% is around 270.0°C and the Tmax is around 345.0°C.
As shown in Figure 10, all the treated cotton fabrics show similar thermal degradation behaviors as compared with original cotton fabric. However, the T10% and Tmax are all increased to different extents. For SiO2@COT, its T10% is 288.3°C, 18.4°C higher than that of original sample. Its Tmax occurs at about 357.6°C, 12.0°C higher than that of original sample, and 54.8% weight is lost in primary pyrolysis stage (64.0% for original cotton fabric). Its char residue rate is 31.3% at 700.0°C. The reason for these changes is that a layer of SiO2 gel coating is formed and connected by Si-O-Si bonds (Figure 2) inside its bulk under high temperature (dry at 90.0°C and cure at 120.0°C). Such coating covers cotton fabric substrate via hydrogen bonds formed through dehydration and condensation processes of Si-OH groups in the SiO2 sol and the -OH groups on the cotton fiber surfaces. This has been validated in above XPS and FTIR results regarding successful loading of SiO2 gel coating on cotton fabric surfaces. In the pyrolysis process, water is released to absorb a lot of heat and dilute concentrations of intermediate pyrolysis products. Also, such coating protects the fabric surfaces from heat transfers, oxygen transport and exchanges of intermediate pyrolysis products. Whereas for KH570@COT, its T10% is around 270.0°C and similar to that of original cotton fabric. Its Tmax is at 351.9°C, which is 6.3°C ahead than that of original sample. Its char residue rate is 19.1% at 700.0°C and lower than that of SiO2@COT (31.3%). On one hand, the add-on (%) value of KH570@COT is lower than that of SiO2@COT. On the other hand, KH570 is organic in nature (Figure 1 KH570) and more easily be pyrolyzed. For SiO2-KH570@COT, its T10% and Tmax (281.8°C and 355.3°C) are higher than those of KH570@COT (270.0°C, 351.9°C) but slightly lower than those of SiO2@COT (288.3°C, 357.6°C). Its add-on (%) value (12.7%) is lower than those of both SiO2@COT (17.2%) and KH570@COT (13.8%). Its char residue rate (29.0%) is slightly lower than that of SiO2@COT (31.3%) but much higher than that of KH570@COT (19.1%). First, this shows the strong coupling effect of KH570 to reinforce connections among SiO2, KH570 and cotton fibers via hydrogen bonds (Figure 3(d)). Second, its thermal stability is also guaranteed and contributed by strong physical barrier effect of SiO2 gel coating. Finally formed chars are hence maintained. Third, KH570 solely is easy to decompose completely at high temperature of 700.0°C compared with SiO2-KH570@COT (Table 2), and its coupling effect is somewhat limited when embedded and blended with silica gel coating. Fourth, KH570 shows its steric effect and spacial arrangement due to its branched chemical structure (Figure 1 KH570) when combining SiO2 gel. This is especially proved by increased peak intensities of O1s, Si2s and Si2p in its XPS spectrum (Figure 7). For AP@COT, its T10% is clearly delayed to 291.7°C, 21.8°C higher than that of original cotton fabric. It is mainly due to the removal of crystal water from AP at 100.0–200.0°C, which can absorb certain amount of heat and reduce the temperature of the burning area. 45 However, its Tmax is only increased by about 5.2°C as compared with original cotton fabric. This is due to the release of certain amount of NH3 gas during pyrolysis. Its char residue rate at 700.0°C is 22.5%. This is caused by the formation of B2O3 at a temperature of Tmax. For SiO2-KH570-AP@COT, it has the highest T10% (296.4°C), the highest char residue rate (31.5%) and the second highest Tmax (356.6°C). They are close to those of AP@COT and SiO2@COT, SiO2@COT and SiO2-KH570@COT, SiO2@COT and SiO2-KH570@COT in value, respectively. These facts clearly reflect primary contributions of AP, SiO2 gel and KH570 both individually and jointly. Here, SiO2 gel shows its strong physical shielding effect (enhancing T10.0% and Tmax in pyrolysis) and structural support effect (keeping dimensional stability and maintaining char residue amounts); KH570 plays its strong coupling effect (enhancing Tmax in pyrolysis and char formations); AP shows its strong dehydration capability (enhancing T10.0% and Tmax in pyrolysis) and certain char formation capability (maintaining char formation). In detail, for AP, it is converted to B2O3 with a small amount of ammonia at 390.0–470.0°C, taking away the excess heat and reducing the combustible gas content. 45 Also, a small amount of boric acid will be produced when AP is hydrolyzed, which can be used as an acid source to catalyze the dehydration and carbonization of cotton fibers. Such facts also indicate that three components can interact with each other well and shows obvious synergistic effects. Therefore, SiO2-KH570-AP@COT shows the best thermal stability.
Flame-retardant performances of original and treated cotton fabrics
Related parameters of LOI experiments and VFTs.

Note: ▵m is the increased mass of the sample, g. ▵m = Mf -Mi (Mf: the mass of a treated cotton fabric, g; Mi: the mass of original cotton fabric, g).

Appearances of char residues of original and treated samples after VFTs (a): COT, (b): SiO2@COT, (c): KH570@COT, (d): SiO2-KH570@COT, (e): AP@COT, (f): SiO2-KH570-AP@COT).
LOI values can be used to judge the difficulty of burning when a cotton fabric is in contact with a flame. Fabrics having LOI value below 21.0% ignite easily and burn rapidly in the air, belong to the inflammable levels. Those with LOI values above 21.0% and below 27.0% ignite and burn more slowly, belong to the combustible levels. 47 The LOI increment (ΔLOI/Δm) is used to further measure the inherent flame retarding capability of each component, which means the increment of LOI value caused by unit increasing mass. 35
From Table 3, the LOI values of all treated cotton fabrics have generally been improved to varying degrees. The LOI value of original cotton fabric is 18.3%, which shows a high flammability. For SiO2@COT, its LOI value (20.2%) increases by 1.9% than that of original cotton fabric, belonging inflammable level. Its ΔLOI/Δm (1.73%/g) is the third highest except SiO2-KH570-AP@COT (4.75%/g) and SiO2-KH570@COT (4.63%/g). This suggests that a smooth and dense SiO2 gel coating (inter-connected by Si-O-Si bonds) deposits on the surface of the cotton fabric by hydrogen bonding (Si2s and Si2p in XPS spectrum, Figure 7; Si-O vibrations in FTIR spectrum, Figure 8) and physical adsorption effectively (Figure 6(a2)). The SiO2 gel coating itself provides basic guarantee for improved thermal stability and flame retardancy. It can act as a physical barrier (conceal cotton fibers) and block oxygen transport, heat exchange and oxidative decompositions. In addition, the silica coating evolves during pyrolysis and combustion to produce high-temperature resistant Si-C compounds, which are further transformed into a char layer on cotton fibers. These analyses have been validated in TG results. SiO2@COT shows the third highest T10% (288.3°C), the highest Tmax (357.6°C) and the second highest char residue rate (31.3%). However, brittle fracture occurs at high temperature when silica sol is used alone, so its flame retardant effect is limited. For KH570@COT, its LOI value (19.9%) increases by 1.6% than that of original sample but is still at inflammable level. This reflects limitation of KH570 alone since it is organic in nature and easy to decompose at high temperature (Table 2). During pyrolysis, KH570@COT shows the second lowest T10% (270.4°C, similar to that of original cotton fabric), the third lowest Tmax (351.9°C) and the second lowest char residue rate (19.1%). For SiO2-KH570@COT, its LOI (22.0%) and ΔLOI/Δm (4.63%/g) values are both the second highest besides SiO2-KH570-AP@COT. This clearly illustrates the strong physical barrier effect of the SiO2 gel coating, coupling effect of KH570 and their synergistic interactions. It is found from FTIR analysis that the absorption peak of Si-O stretching vibration between 1060 and 1160 cm-1 becomes weaker, which proves that the condensation reaction between SiO2 and KH570 occurs as shown in Figure 3(d). For AP@COT, its LOI value (19.3%) only increases by 1.0% as compared with original cotton fabric, its ΔLOI/Δm value (1.67%/g) is the second lowest besides KH570@COT. On one hand, it is due to the low loading amounts of AP hydrolysate. On the other hand, it can be attributed to the deposition of AP on the surface of cotton fabrics mainly by physical adsorption. For SiO2-KH570-AP@COT, it shows both the highest LOI (24.0%) and ΔLOI/Δm (4.75%/g) values. Each component shows its disadvantage as analyzed above. This poses certain unexpected competitiveness among them in improving general performance. Fortunately, when they combine together, more synergistic effects occur. As proved by the TG results, SiO2 gel shows its strong physical barrier and structural support effects in enhancing thermal stability and providing char frame, KH570 plays its strong coupling effect in combining components and reinforcing interface properties between components and cotton fibers, AP plays a role in dehydration (release of water molecules for cooling burning zone, diluting and isolating oxygen and gaseous products) and promoting carbonization (char formation).
Generally, increased LOI and ΔLOI/Δm values represent lower combustibility and better flame retardancy levels. This can also be proved by VFTs. For original cotton fabric, it burns violently (an after-flame time (AFT) of 23.0 s) and causes obvious smoldering after removing the ignition source (an after-glow time (AGT) of 30.0 s), leaving only a small amount of char residue (Figure 11(a)). This reflects the inflammability nature of natural cotton fibers. This is consistent with the char residue rate (9.0%) of original cotton fabric in TG results (Table 2). For SiO2@COT, its AFT (16.0 s) and AGT (13.0 s) decrease clearly by 30.4% and 56.7% compared with those of original cotton fabric. This shows the strong barrier effect of an inorganic SiO2 gel coating as previously analyzed. Its char length (CL, 30.0 cm) is the same as that of original cotton fabric. This shows that SiO2 gel coating can act as a structural support to promote mechanical strength and dimensional stability of cotton fabrics. Such effects also remain in other treated cotton fabrics including SiO2 components. For KH570@COT, its AFT is slightly reduced by 11.9% and AGT is much elongated by 127.7% as compared with SiO2@COT. This suggests the organic nature of KH570 clearly. It solely is more easily decomposed at high temperature with the lowest T10% (270.4°C, Table 2) and the second lowest Tmax (351.9°C, Table 2) among treated cotton fabrics. Thus, it still can maintain a smoldering process reflected by a relatively high AGT. Moreover, it shows a less strength in maintaining structural integrity (large crevice, Figure 11(c)) as compared with inorganic SiO2 gel coating (Figure 11(b)). For SiO2-KH570@COT, its AGT decreases to 0.0 s and a whole char residue is observed with only a narrow crevice left (Figure 11(d)). A smoldering process disappears. This again proves that KH570 can well connect SiO2 sol particles and cotton fibers via hydrogen bonds (formations of -Si-O-Si- and -Si-O-C- bonds) and thus reinforce connections among them. Certainly, its AFT (17.0 s) is higher than both SiO2@COT (16.0 s) and KH570@COT (14.1 s) since SiO2 gel still plays its rigid structural support role (a CL of 30.0 cm) although KH570 is easily decomposed earlier. For AP@COT, its AFT is the highest among treated cotton fabrics but AGT is much decreased by 73.0% as compared with KH570@COT. Also, its char residue is more complete (Figure 11(e)) than that of KH570@COT. Clearly, AP alone shows effectively comprehensive flame retardancy capabilities and keeps dimensional stability as an inorganic additive. However, it will also pyrolyze at a high temperature (see Table 2) and can therefore still sustain a short time smoldering process (an AGT of 8.0 s). AP is mainly deposited on cotton fabric by physical adsorptions, and the add-on (%) of AP@COT is low (9.4%); AP will lose crystal water and release ammonia gas at high temperature during the combustion process. For SiO2-KH570-AP@COT, its AFT (14.1 s) and AGT (0.0 s) are both the lowest among treated cotton fabrics. Its char residue is relatively complete with small area of visible crevices (Figure 11(f)). This further proves that the synergistic roles among three components play both physically and chemically although they each show disadvantages, that is, they can compensate each other in general. Each of them is important in improving thermal stabilities and flame retardancy capabilities overall as analyzed previously (analyses of TG results).
To quantitatively assess synergy among components, Lewin's Synergistic Effectiveness parameter (SE) is defined as:36–38
TG-IR analyses of original and treated cotton fabrics
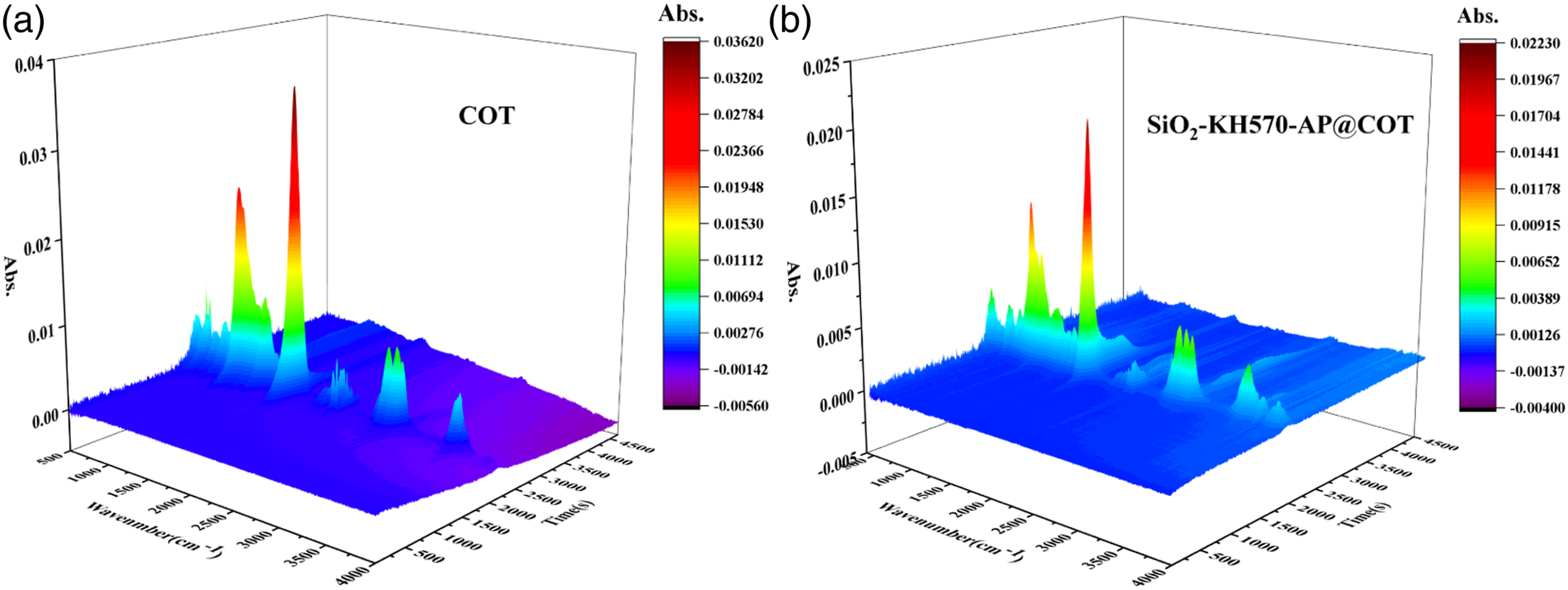
In order to disclose gas-phase flame-retardant mechanism, compositions of pyrolysis gaseous products from COT and SiO2-KH570-AP@COT were analyzed by TG-IR. The corresponding FT-IR spectra of the released volatiles at selected temperatures are plotted in Figure 12(a) and (b). Three-dimensional (3D) TG-IR diagrams of pyrolysis volatiles are shown in Figure 13(a) and (b), respectively. For original cotton fabric, typical absorptions of H2O, hydrocarbons, CO2, C=O and ethers appear at 3567, 2926, 2357, 1746 and 1110 cm‐1, respectively. Among these volatiles, H2O and CO2 as nonflammable gases are from the dehydration process of cellulose fibers. Hydrocarbons, C=O and ethers as flammable gases generate from the depolymerization of cellulose fibers during pyrolysis processes.
48
For SiO2-KH570-AP@COT, the treatment remarkably reduces the absorption intensities of hydrocarbons, C=O and ethers. This suggests that the presence of SiO2-KH570-AP significantly decrease the release of flammable volatiles, thus suppressing the combustion processes. The peaks at about 957 cm‐1 detected in the TG-IR spectra of SiO2-KH570-AP@COT are assigned to typical absorption of NH3. The decomposition of AP is responsible for the formation of NH3. The nonflammable gases like NH3 can dilute oxygen and flammable volatiles during combustion processes. All these data verify that SiO2-KH570-AP plays flame retardancy effects via suppressing the release of flammable volatiles and releasing nonflammable gases in the gas phase. FT-IR spectra of the released gases of COT and SiO2-KH570-AP@COT at selected temperatures. 3D TG-IR diagrams of COT and SiO2-KH570-AP@COT during pyrolysis processes.
SEM images of char residues after VFTs
The surface microscopic morphologies of char residues after VFTs are shown in Figure 14. For original cotton fabric, its char residue shows a cracking surface with some holes (see also Figure 11(a)). This is attributed that cotton fibers are decomposed and fully carbonized during combustion. The produced char layer is naturally formed by the dehydration of the D-anhydroglucose ring units in cotton fibers. For SiO2@COT, the interspace between cotton fiber bundles in its char residues after burning are covered with a rough and broken silica gel coating formed by gelation and polycondensation processes of SiO2 sol at high temperature. In such processes increasingly strengthened surface tensions, capillary effects and bearing stresses make the outer layer of inorganic SiO2 gel coating first crack. Fortunately, this gel coating still maintains dimensional stabilities of internal cotton fibers (Figure 11(b)). But these fibers are exposed to flame gradually and directly when such inorganic silica gel coating is cracked until it loses protection role as the temperature increases (357.6°C in Table 2). Then the substrate cotton fabric is gradually pyrolyzed and fully charred. For KH570@COT, its char residue is relatively dense, but there are also fractures. This can be attributed that the hydrolysate of KH570 contains hydroxyl (-OH) groups which can combine the -OH groups of β-1-4 glycosidic bonds in the molecules of cotton fibers through Si-OH. However, KH570 belongs to compliant organic molecules, it will be pyrolyzed at higher temperature. According to the TG results (Table 2), the T10% and Tmax of KH570 are similar to those of original cotton fabric. Hence, unlike the silica gel, the structural strength of its coating is not strong enough to play a role of a physical barrier. Eventually, a layer of char particulates (Figure 12) from both KH570 and cotton fibers are left. In this way, its T10%, Tmax and char residue rate (Table 2), ΔLOI/Δm (the lowest, Table 2), AFT and AGT (Table 3) and structural integrity of char residue (Figure 11(c)) are generally poorer than those of SiO2@COT. For SiO2-KH570@COT, thanks to strong physical barrier and structural support effects of SiO2 gel coating, strong coupling effect of KH570 and their positive interactions, it is pyrolyzed more steadily (relatively higher T10%, Tmax and char residue rate, Table 2), its inflammability level (the second highest ΔLOI/Δm, Table 3) and flame retardancy level (no AGT, Table 3) are clearly bettered. Therefore, its char residues are largely complete (Figure 11(d) and Figure 12). For AP@COT, there are some bubbles on the surfaces of cotton fibers. Combined with LOI and especially ΔLOI/Δm results (1.7%/g, Table 3), it can be seen that AP has certain flame-retardant effects in both condensed phase and gas phase as noted from the following four aspects: (1) When AP is heated to above 90.0°C, it releases ammonia to dilute the oxygen in the surrounding environment. (2) When the temperature reaches 100.0°C, crystal water of AP is lost, which can absorb a large amount of heat and reduce the temperature of the combustion surface. (3) At the high temperature, the converted B2O3 glass-like film is covered on the cotton fabric to act as a heat and oxygen barrier. (4) A small amount of boric acid will be produced when AP is hydrolyzed. Boric acid can be used as an acid source to catalyze the dehydration and carbonization of cotton fiber. Although the flame retardant effect of AP is multiple, its individual effect is limited. This is attributed to the fact that AP is mainly deposited by physical adsorption on the surface of cotton fabric. For SiO2-KH570-AP, its dense and smooth char layer can be seen, which is consistent with VFTs result (Figure 11(f)). It shows the highest T10.0% and char residue rate (Table 2), the second highest Tmax (Table 2), the highest ΔLOI/Δm (Table 3) and the most beneficial AFT and AGT (Table 3). Three components in the ternary system plays their synergistic roles to the largest degree. Actually, as analyzed elsewhere, due to the inorganic-organic coexistence conditions, there are some competitive but more synergistic effects among different components: silica gel can provide strong physical shielding and structural support effects, but it is easily broken at high temperature and loses its protective effect, while the strong coupling effect of KH570 can compensate these disadvantages and improve general chemical stabilities. AP is thermally dehydrated to release water molecules that can cool combustion environment, dilute intermediate pyrolysis gaseous products and isolate transported oxygen. Also, AP is thermally decomposed to generate boric acid to accelerate the dehydration and carbonization degrees of cotton fibers and thus improve more through formations and chemical stabilities of the char layers. SEM images of char residues for original and treated cotton fabrics at × 2000, ×5000 magnification after VFTs.
Tensile strengths and elongations at break of original and treated cotton fabrics
Figure 15 shows the tensile strengths and elongations at break data of original and treated cotton fabrics. The tensile strength of the original cotton fabric in the weft and warp directions were 23.74 MPa and 24.77 MPa, respectively. In comparison, the tensile strengths of SiO2-KH570-AP@COT cotton fabric in the weft and warp directions remain at 19.54 MPa and 23.01 MPa, decrease by 17.69% and 7.11% as compared to those of the original cotton fabric. Meanwhile, the elongations at break in the weft and warp directions for the treated cotton fabrics are 18.66% and 21.54%, decrease by 35.36% and 23.37% compared to the original cotton fabric (28.87% and 28.11%). It can be seen that the presences of flame retardant systems result in tensile strength and elongation at break losses of treated cotton fabrics. Also, the acidic and high temperature conditions trigger the degradation of cellulose macromolecule chain, resulting in the decrease of tensile strength.
49
The above results indicate that the tensile strengths and elongations at break of SiO2-KH570-AP@COT are lower, but this hardly affects the applications of cotton fabrics especially those for decoration cases. Tensile strengths and elongations at break for the original and treated cotton fabrics.
Explorations of comprehensive flame retardant mechanisms
In summary, the flame retardant mechanism of SiO2-KH570-AP@COT is illustrated in Figure 16. When the hydroxyl-rich cotton fiber is exposed to a fire, it is dehydrated and carbonized, and produce water, carbon dioxide and char residues.
50
Due to different properties, the components exhibit their respective acting effects, as well as competitive and synergistic effects. First, the silica sol connected to the surface of cotton fabrics via hydrogen bonds is converted to a gel coating after baking. This helps to protect the substrate by creating a physical barrier to oxygen transport and heat transfer, hindering the formation of volatile products that fuel the further degradation. In addition, silica gel coatings evolve to produce high temperature resistant Si-C compounds during pyrolysis and combustion, which are further converted into char layers on cotton fibers.
51
Silica gel coatings show structural support effect in keeping dimensional stability and maintaining the amounts of nonflammable char residue to reinforce the protective effect at higher temperatures. Second, the KH570 as a silane coupling agent enhances this barrier effect and char formations, which is essentially derived from strengthening of physical coverage and chemical bonding. Third, AP will release ammonia when the temperature above 90.0°C, which can dilute the oxygen in the air to reduce the burning rates of cotton fabrics. Besides, the water also plays the contribution in terms of dilution in the gas phase. When it is higher than 100.0°C, the AP will lose crystal water, which can absorb a large amount of heat and reduce the temperature of the burning surface. As the temperature increases, the B2O3 glass-like film converted by AP is covered on the cotton fabric to isolate heat and oxygen.
52
In addition, a small amount of boric acid will be produced when AP is hydrolyzed. Boric acid as an acid source can catalyze the dehydration and carbonization of cotton fiber, thus improve the stability of carbon layer. Therefore, flame-retardant effect of cotton fabric finished by SiO2-KH570-AP sol-gel system is mainly promoted by both individually positive and jointly synergistic actions of silica sol (strong physical barrier and structural support effects in enhancing thermal stability and providing char frame), KH570 (strong coupling effect in combining components and reinforcing interface properties between components and cotton fibers) and AP (release of water molecules for cooling burning zone, diluting and isolating oxygen and gaseous products and promoting char formation) Schematic illustration of the flame-retardant mechanism of SiO2-KH570-AP@COT.
Conclusions
In this paper, sol-gel systems using TEOS as a precursor, KH570 as a silane coupling agent and AP as a flame retardant additive were successfully prepared via sol-gel processes and deposited on cotton fabrics via dipping-curing methods.
General results show that with introductions of SiO2 gel, KH570 and AP in SiO2-KH570 composite and SiO2-KH570-AP doped sol-gel systems, their finished cotton fabrics show improved chemical stabilities, thermal stabilities, dimensional stabilities, char residue integrities and rates, and improved flame retardancy capabilities. These findings are deduced from and well supported by results of multiple characterization techniques and experimental methods including SEM-EDX, XPS, FTIR, XRD, TGA, LOI and VFTs. SiO2-KH570-AP@COT shows the greatest thermal stability and flame retardancy. In detail, it has the highest T10% (296.4°C), the highest char residue rate (31.5%), the second highest Tmax (356.6°C), the highest LOI (24.0%) and ΔLOI/Δm (4.75%/g), the most beneficial AFT (14.1 s) and AGT (0.0 s), and the most complete char residue (30.0 cm) with small area of visible crevices after a VFT. The tensile strength and elongation at break of the SiO2-KH570-AP treated cotton fabric are lowered as compared to those of original cotton fabric, but this hardly affects its applications especially when it is used for decoration products.
Three components in the final ternary sol-gel system of SiO2-KH570-AP (or the so-called AP doped SiO2-KH570 sol-gel) exert certain competitive but more synergistic effects in different ways for flame retarding the cotton fabric. SiO2 gel shows its strong physical barrier and structural support effects in enhancing thermal stability and maintaining a char frame, KH570 plays its strong coupling effect in combining components and reinforcing interface properties between components and cotton fibers, AP plays a role in dehydration (release of water molecules for cooling burning zone, diluting and isolating oxygen and gaseous products) and promoting carbonization (char formation). Such synergistic effect is fully reflected via synergistic effectiveness parameter (SE).
Footnotes
Acknowledgements
Thank Ms Jing Ma for her assistance in preparing some raw materials.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Fundings
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study is financially supported by the National Natural Science Foundation of China (Grant No. 51376089 and 50906039) and the 15th Six Talent Peaks Project in Jiangsu Province in Renewable Energy and Energy Internet Industry (XNY-016), and the 2019 Key Project of The Natural Science Foundation of the Jiangsu Higher Education Institutions of China (19KJA520007, A Class).
