Abstract
The sol-gel process has been applied to cotton fabrics to cover the fibers with a silica-based film, which can improve their thermal oxidation and combustion behaviors. Silica sol, silane coupling agent 3-glycidoxypropyltrimethoxysilane (KH560) and flame retardant zinc borate (ZB) are used together to prepare SiO2-KH560-ZB hybrid sol, which is then finished on the surface of cotton fabric through impregnation and baking. Fourier transform infrared spectroscopy (FTIR), thermogravimetric analysis (TGA), limiting oxygen index analysis (LOI) and microscale combustion calorimetry (MCC) are used to characterize functional groups, thermal stability and flammability properties of finished fabrics. Surface morphology of sol modified cotton fabrics are analyzed by scanning electron microscopy (SEM) and energy dispersive X-Ray spectroscopy (EDX). Results show the sols are successfully converted into gel coatings onto cotton fabrics, and continuous smoldering phenomenon of resulted fabrics disappears. ΔLOI/Δm of SiO2-KH560-ZB coated cotton fabric reaches the highest of 0.37%/g, char residue rate is as high as 28.43% and the peak heat release rate (PHRR) is reduced by 26.9% (83.7 W/g). KH560 has a significant coupling effect on combining components in the sol system and increases compatibility between sols and cotton fabrics. ZB (cooling, separation, dilution and suppression), silica sol (physical barrier) and KH560 show excellent synergistic effects in enhancing overall flame retardancy.
Keywords
Introduction
Cellulose is the most abundant biopolymer in nature and one of the most accessible natural resources, with 1.0 × 1010∼1.0 × 1011 tons produced annually [1]. Over the years cotton cellulose has been the main raw material resource for the textile industry with wide applications as decorative fabrics and household items (curtains, blankets, wall ornaments, cloth art furniture and bedding, etc.) due to its luster and comfort [2]. However, due to the highly flammable and flame-spreading properties of cotton fabrics [3,4], levoglucose moleculars will be produced during burning processes and inflammable combustion gaseous phases, it is often the source of ignition, the first combustible or the main combustible involved in fires of multiple end environments like buildings and warehouses. All the time, fire problems caused by cotton fabrics become a serious threat to people's lives and property [5]. Textile articles processed from natural fibers (cotton, kapok, hemp, flax and jute etc.) and man-made fibers (viscose, Lyocell, Modal and cellulose acetate fibers, etc.) in daily life usually have none or poor flame retardancy [6]. At the same time, during the burning process, cotton textiles are often accompanied by smoldering and thus pose hidden fire sources, smoking and toxic conditions. As a result, it is extremely urgent to prepare multifunctional composite decorative fabric materials on residential and public occasions with excellent flame-retardant properties through new chemical modification processes and techniques [7,8]. Based on the current scale of development, the relevant departments predict that the total transaction volume of flame-retardant textile articles in the world will reach about 5.8 billion dollars in 2022 [9].
In recent years, due to many realized and potential end applications [10] nanotechnology has become a rapidly growing research field of functional textile. Nanoparticles have been successfully used in conventional textiles to give new functions and improved properties [11]. In fact, they have high surface energies and large surface area/volume ratios, which makes them easy to connect to the treated substrate, thus increasing the durability of the functions assigned to the textile material [12]. In this case, the sol-gel method has some specific advantages due to its high efficiency, halogen-free, low toxicity, low smoke, low cost, green ecology, mild and convenient operation [13]. In recent years, sol-gel technology has been used for flame retarding, which creates a barrier to protect the polymer surface by creating a physical barrier [14,15], thereby improving the flame retardancy of the material being processed. Experiments show that due to the good coating properties of silica sol and the good physical properties of the three-dimensional carbonaceous silica skeleton formed during the reaction, the silica sol is prepared by sol-gel method and becomes a promising base for flame-retardant coating. REN et al. [16] studied the flame-retardant properties of polyacrylonitrile (PAN) fabrics treated with tetraethyl orthosilicate (TEOS) and polyphosphoric acid by using one-step sol-gel process. The LOI and char residue increase by 30.1% and 69.48%, respectively. The peak and total heat release of phosphorus-doped SiO2-coated PAN fabrics decrease significantly by 44.9% and 42.1% compared to those of untreated fabrics. In addition, the peak value of flue gas production and total smoke production are also significantly reduced. Grancaric et al. [17] added red phosphorus into the silica sol and finished the modified sol system to the cotton fabric. It is found that the combined effect of accelerated carbonization and heat shielding improves the thermo-oxidative stability of the cotton fabric. The LOI of the fabric reaches 29.0% and the fabric can self-extinguish after ignition. DEH et al. [18] used TEOS as a precursor to study the flame retardancy of cotton fabrics treated with phosphoric acid and urea phosphorylation via a sol-gel process. Experiments show that cotton fabrics have higher LOI (>25.0%) and can withstand 10 times of washes. The combination of phosphorus and silicon promote carbonization during pyrolysis, resulting in the formation of stable char up to 1100°C. So far, phosphorus- and nitrogen- containing compounds have been proved to be excellent flame retardants, and can form a synergistic effect with silica sol hybridization, which provides a reference for the research of high-efficiency flame retardants. However, most of the existing research results are limited to organophosphorous and nitrogenous flame retardants, and there are few studies on the compounding of inorganic flame retardants (such as zinc borate) and silicon-based flame retardants, surface modification of flame retardants, synergistic effects among triple (and above) system including both inorganic and organic components, particle super-refinement and nano crystallization, etc. At the same time, the current research on the selection of flame-retardant components does not involve the design of flame-retardant systems, and there is a problem that the flame-retardant mechanism is not explored deeply enough.
Zinc borate is a typical boron-based flame-retardant additive because of excellent thermal stability and smoke suppression [19,20]. Zinc borate degrades at 300.0°C to produce boric acid which promotes the formation of protective, honeycomb and glassy char layers. The char residual layer acts as a good insulating material and protects the underlying substrate from heat and oxygen [21]. The zinc species catalyzes the cross-linking reactions in the condensation phases of polymers, suppresses the smoke generated during the combustion processes, and improves the thermal stability levels of the carbonaceous compounds [17]. However, its large-scale application on fabric is rarely reported due to the presence of electron defects in boron atoms, which are poor in hydrolytic stability [22]. As an environmentally friendly non-halogen flame retardant, zinc borate is currently widely used in the processing of plastics and rubber. It is usually used alone or as a synergist for intumescent flame retardants, and has good flame-retardant effects.
In this paper, silica sol is modified with silane coupling agent KH560, and the modified sol is mixed with zinc borate (flame retardant) and coated on the surface of cotton fabric to prepare organic (KH560) - inorganic (Silica, ZB) hybrid material. Particle size distribution and specific surface area of silicon sol are measured by nano particle size laser analyzer. The internal chemical structure, surface microstructure and element distribution of cotton fabric treated by single and compound sol are characterized by FTIR, SEM and EDX. The thermal stability (pyrolysis characteristic), flame retardant performance and combustion condition (surface morphology) of the fabric are reflected by TG, LOI, MCC and SEM. Moreover, the flame-retardant mechanism of the hybrid gel coating is investigated comprehensively by analyzing the surface morphology, surface functional groups, pyrolysis characteristics and flame retardancy of cotton fabrics.
Experimental
Materials
Cotton fabrics with warp yarns of 133.0 yarns/cm and weft yarns of 72.0 yarns/cm are purchased from Nanjing Qicaimei Textile Co., Ltd. The fabrics are soaked in boiling water for at least 0.5 hours to remove all impurities and spinning oil from the fiber surface. Then they are washed in 2.0% non-ionic detergent (supplied by Nanjing Wanqing Chemical Instrument Co., Ltd.) at 40.0°C for 20 min and rinsed several times with deionized water (IR, supplied by Nanjing Wanqing Chemical Instrument Co., Ltd.), dried and placed in the oven (PHG-9036A, provided by Shanghai Jinghong Experimental Equipment Co., Ltd.) until the residual mass variation is within 1.0%. The sol-gel precursor TEOS (AR, ≥ 30.0%), silane coupling agent KH560 (IR, ≥ 98.0%), flame retardant zinc borate (AR, ≥ 99.0%) are supplied by Sinopharm Chemical Reagent Co., Ltd, and the chemical structure of them are presented in Table 1. The solvent anhydrous ethanol (EtOH, AR, ≥99.7%) is obtained from Wuxi Yasheng Chemical Co., Ltd. Catalyst hydrochloric acid (HCl, AR, 37.0%) is provided by Shanghai Pilot Chemical Corporation, and distilled water is purchased from Nanjing Wanqing Glass Instrument Co., Ltd.
Chemical structures of TEOS, KH560 and ZB.

Preparation of silica-based sols
TEOS, anhydrous ethanol and deionized water are firstly mixed in a beaker at room temperature at a molar ratio of 1.0:6.0:1.0 and stirred by a magnetic stirring device for 1.0 h. The pH value of the mixture is controlled from 5.0 to 6.0 with 10 wt% hydrochloric acid, and the mixture is stirred at 60.0°C for 2.0 h to obtain clear and stable SiO2 sol. The step-growth process of TEOS hydrolysis and condensation reaction is presented in Scheme 1.
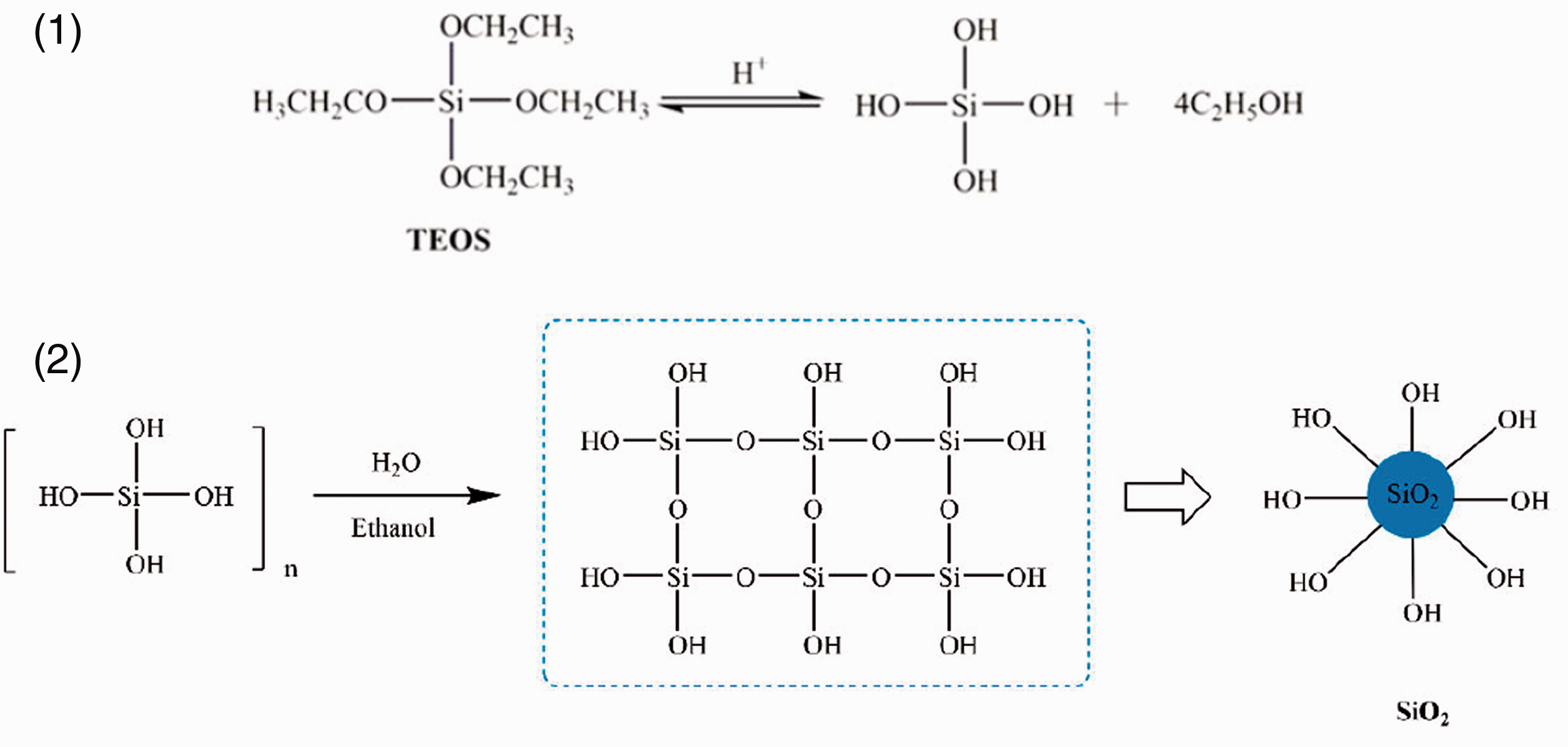
Hydrolysis of TEOS (1) and condensation of SiO2 (2).
In order to improve the agglomeration of silica molecules and increase the compatibility of SiO2 network with cotton fabrics, KH560 is selected as surface chemical modifier. KH560 (20.0 vol%), C2H5OH (72.0 vol%), deionized water (8.0 vol%) are mixed and conducted by magnetic stirring and hydrolyzed at room temperature. Then KH560 hydrolysate is added into the single SiO2 sol and the mixture is stirred at 60.0°C for 1.0 h to obtain SiO2-KH560 sol, the molar ratio of KH560 to TEOS is 1.0:1.0. At the same time, KH560 hydrolysate is retained as a control experiment. The hydrolysis of KH560 and modification of SiO2 by KH560 are shown in Scheme 2.

Hydrolysis of KH560 (3) and SiO2 modified by KH560 (4).
SiO2-KH560-ZB hybrid sol is prepared by adding 2.0 g zinc borate into 200 ml modified SiO2-KH560 sol and stirring at 60.0°C for 1.0 h in a constant temperature magnetic stirrer. Meanwhile, the concentration of 10 g/L zinc borate solution is prepared.
Finishing process of sols onto cotton fabrics
The pretreated fabrics are completely immersed in silica-based sols at room temperature. Then the fabric samples are dried at 80.0°C for 0.5 h and subsequently cured at 120.0°C for 1.0 h. The processes of preparing ZB hybrid silica sol-gel and finishing cotton fabric are shown in Figure 1.
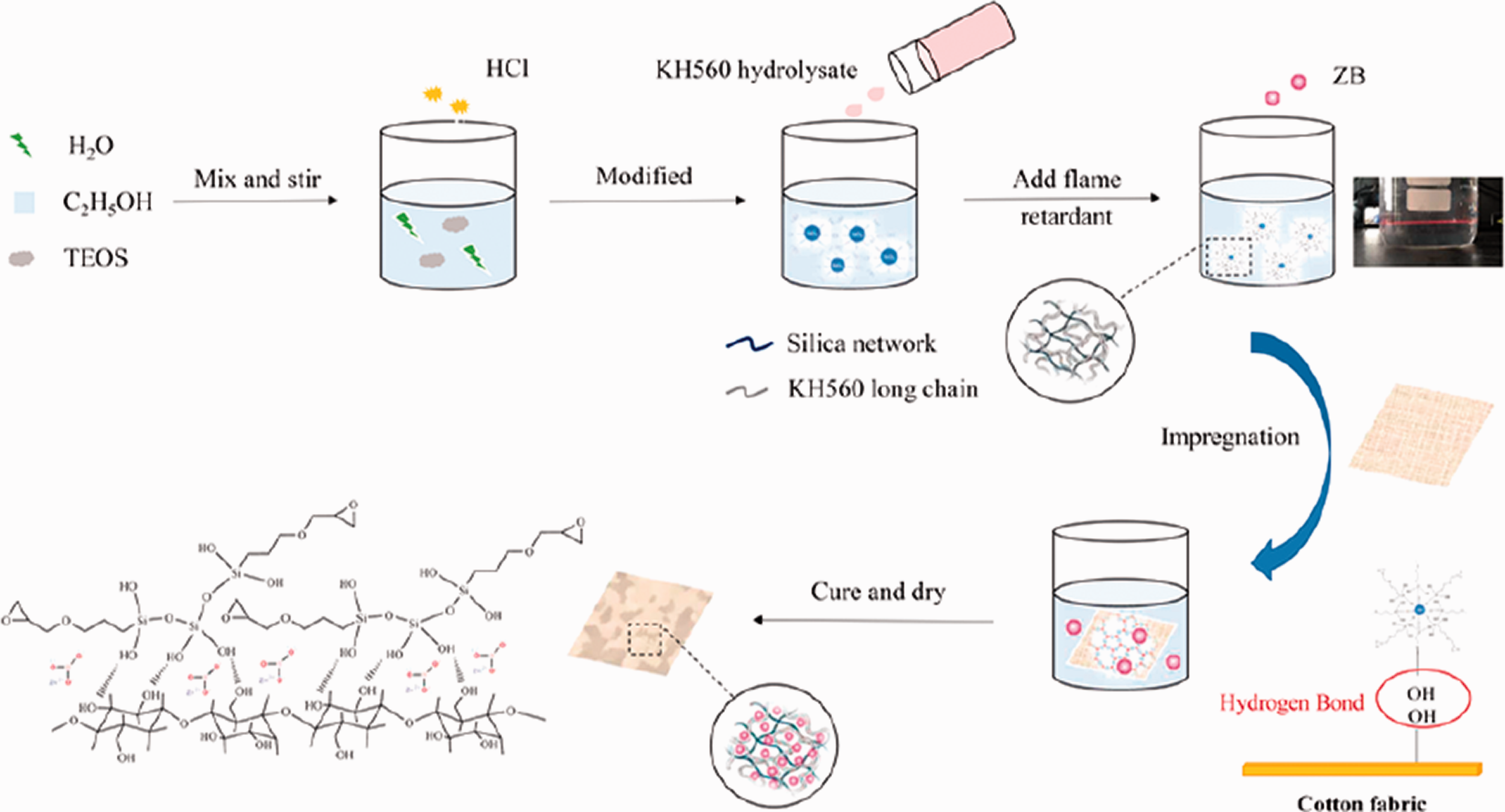
Preparation of ZB hybrid silica sol-gel and finishing process of cotton fabric.
The appearances of the cotton fabrics finished by the series of sols are shown in Figure 2. The surfaces of the cotton fabrics are evenly coated after flame retardant finishing, and their colors, hardness and hand feels have not changed significantly compared with the original fabric.

Appearances of cotton fabrics before and after finishing sols (The samples from left to right are Cotton, SiO2@Cotton, KH560@ Cotton, ZB@Cotton, SiO2-KH560@Cotton and SiO2-KH560-ZB@Cotton).
The add on % (A) of the treated cotton fabrics are calculated by the weight (measured by a Mettler balance produced by Mettler-Todoli Co., Ltd., ±10−4 g) of the cotton before (Wi) and after the solution/sol impregnation treatment (Wf) with the following equation.
Testing and characterization
The solid content of single and hybrid sol/solution is calculated by mass fraction X, the value is expressed in % and determined as follows:
Where, m1 is the mass of the weighing bottle dried in an oven at (105 ± 2) °C for 1.5 h and cooled in a desiccator for 30 min, g; m is the weight of the single and hybrid sol (solution) before drying (accurate to 0.0001 g), g; and m2 is the mass of a dry bottle containing single and hybrid sol/solution, which is placed in an oven at (105 ± 2) °C, dried for 3.0 h, and placed in a desiccator to cool for 30 min, g.
The specific surface area and particle size of each sol-gel system and solution are obtained through BT-90 nanometer particle size laser analyzer (Dandong Baite Instrument Co., Ltd.).
FTIR is performed on transform infrared spectrometer (Nicolet Nexus 670, USA), with a test resolution of 4.0 cm−1 and a scanning range of 500–4000 cm−1.
The surface morphology of cotton fabric is observed through a high-resolution field emission scanning electron microscope (SEM, Hitachi S-4800, Japan). All the samples are required to be cut into 3.0 mm × 3.0 mm. Before the test, the cotton fabric samples should be fixed on a black conductive tape and coated with the gold to increase the electrical conductivity. The magnification of the optical microscope to observe the samples is 800 times. The element content of selected area for cotton fabric is measured with a Xplore-30 energy dispersive X-Ray spectrometer (OXFORD, UK).
The TGA tests are carried out on a TG209F1 instrument (NETZSCH, Germany). Air flow of 50 mL/min is used as the pyrolysis atmosphere during the test. The heating rate is controlled at 10.0°C/min and the pyrolysis temperature range is set at 30.0–700.0°C.
The LOI experiments of pristine and treated cotton fabrics are measured using ZY6155A Digital Oxygen Index Instrument (Dongguan Zhongnuo Quality Inspection Equipment Co., Ltd.). The samples with the size of 160.0 mm × 40.0 mm are placed on a U-shaped sample frame (inner frame size 140.0 mm × 38.0 mm) according to Chinese standard GB/T5454-1997 “Textile Burning Performance Test Oxygen Index Method”.
A microscale combustion calorimeter (MCC) is used to measure the heat release rates of cotton fabrics. Approximately 5.0 mg samples are heated in MCC-2 (Govmark, USA) from 90 to 700°C at a heating rate of 1°C/s in a stream of N2 flowing at 80 mL/min.
Results and discussion
Apparent characterizations of the sols/solution
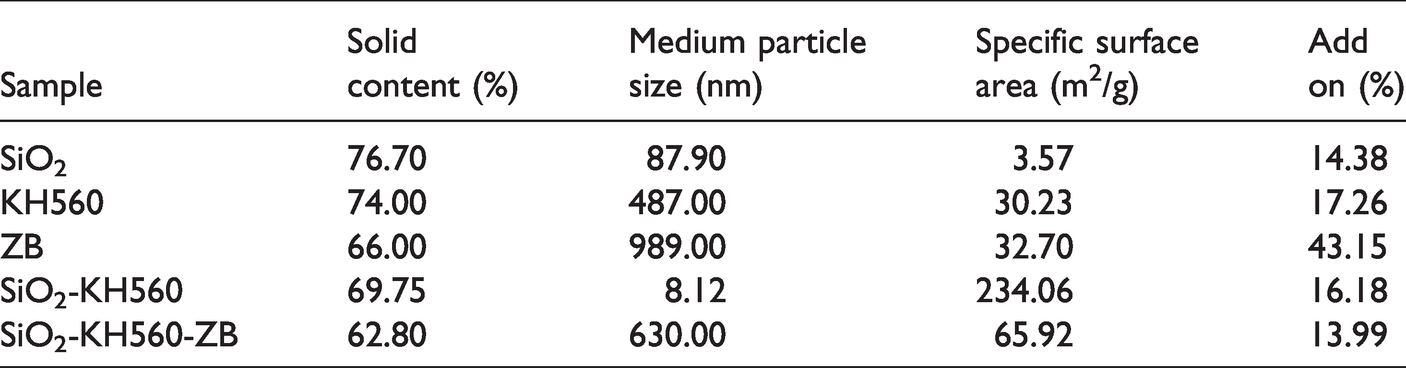
The results of solid content, particle size distribution, specific surface area and increment in weight of the single, composite and hybrid sol/solution systems are shown in Table 2. It is found that the solid content of the sol decreases slightly with the addition of coupling agent KH560 and flame retardant ZB, but they all belong to high solid content sol (more than 60.00%), especially the sample ZB@Cotton has the largest increase in weight. This is mainly due to the steric hindrance effect of branched molecular structure in KH560 and the limited physical adsorption binding point of zinc borate, resulting in the decrease of aggregation of colloidal particles. Generally speaking, high solid content will lead to excessive viscosity of the sol, poor stability and short gel time, while the soft organic segment in KH560 structure can further improve the flexibility and elasticity of the film and increase the crack resistance. The medium particle size of single SiO2 sol is about 87.90 nm, which lies in the nanoscale (1.0–100.0 nm) range, the specific surface area is 3.57 m2/g, and the particle sizes of the composite SiO2-KH560 and hybrid SiO2-KH560-ZB are 8.12 nm and 630.00 nm. The particle sizes of KH560 and zinc borate sol/solution system both increase to varying degrees. The particle size of KH560 modified silica sol is much reduced. Refer to Scheme 2 (4), SiO2 sol will combine with KH560 by hydrogen bonding. The positional hindrance effect of KH560 causes difficult polymerization of hydrolyzed TEOS to form a large continuous network, while a single SiO2 sol can form a three-dimensional continuous network. In addition, according to the numerical judgment, the integration between hydrolyzed KH560 and silica sol particles is limited, or the spatial configuration of KH560 prevents their further aggregations [23]. Zinc borate molecules are adsorbed into the three-dimensional framework of silica sol by van der Waals forces, but due to the limited effective binding points, and the zinc borate itself has a larger particle size (989.00 nm) and a smaller specific surface area (32.70 m2/g), as a result, the particle size and specific surface area of hybrid SiO2-KH560-ZB sol are increased compared with silica and KH560 sol systems, and the particle roughness is high. These parameters play potential roles in flame retardancy levels of treated cotton fabrics as revealed later.
Solid content, particle size and specific surface area of single, composite and hybrid sol/solution.
Intrinsic chemical compositions of the cotton fabrics
In order to explore the degree of integration of cotton fabrics with ingredients, FTIR is utilized to study the changes in internal structure of cotton fabric before and after different flame-retardant treatments. Figure 3 shows the corresponding FTIR spectra of the cotton fabrics. Some characteristic peaks of cellulose at 718, 1047, 1240, 1350, 1700 cm−1 are presented in cotton samples, which are attributed to C-H out-of-plane bending vibration, C-O-C asymmetric stretching, C-O symmetric stretching, C-H deformation vibration and -CHO stretching vibration [24]. The weak absorption peaks at 2896 cm−1 and 3410 cm−1 are caused by C-H stretching vibration and -OH. Compared to Cotton, SiO2-Cotton displays three new characteristic peaks in the low wavenumber range. These bands at 966, 790 and 1070 cm−1 are assigned to the Si−OH stretching vibration, Si−O−Si bending vibrations and Si−O stretching vibration, respectively [25]. This indicates that the silica sol has been successfully prepared and applied to the surface of cotton fabric. Furthermore, the intensities of characteristic peaks at 718, 1240, 1700, 2896 cm−1 increase to varying degrees, which indicates that the exposed -OH on the fabric backbone and the -OH in the silica sol form hydrogen bonds (see Figure 3). The absorption peak of Si-O-C and -CH(O)CH- appear at 1050 cm−1 and 913 cm−1 on KH560 modified silica flame retardant fabric, and the intensities of -OH and Si-OH peaks are significantly weakened [26]. Nano-SiO2 and the silane coupling agent KH560 molecules complete the condensation polymerization. The addition of KH560 effectively reduces the hydroxyl on the silica sol surface and hinders the polycondensation of TEOS, thus reducing the molecular agglomeration (reducing the particle size), which can be verified by the analyses in 3.1. This confirms the data of medium particle sizes of KH560 modified silica sol. The new B-O absorption peak at 1450 cm−1 of the cotton fabric is the characteristic peak of zinc borate [27]. At the same time, the strength of -OH bond at 3410 cm−1 increases and its position moves to high wavenumber. The phenomenon indicates that the zinc borate molecules have been attached to the cotton fabric through the intermolecular force and the hydrogen bond of the chemically bound water (3.5H2O) in the zinc borate crystals, which changes the stretching frequency of the chemical bond on the surface of the cotton fabric.

FTIR patterns of the pristine and silica-based gel-coated cotton fabrics.
Microstructure analyses of the cotton fabrics before burning tests
The surface morphologies of the pristine and coated cotton fabrics are analyzed by SEM, as shown in Figure 4. In addition, the EDX probe acts on the surface of each sample to confirm the element compositions and evaluate their distribution in the coating. For untreated cotton fabric (Figure 4(a)), the fibers are interlaced independently with natural gullies and deformations, smooth surfaces and no obvious adhesion. The cotton fabric treated with SiO2 sol has a rough surface and a distinct material layer (Figure 4(b)). 49.25% C element, 39.68% O element and 11.08% Si element can be inspected and are the main components of the original cotton fabric and SiO2 sol. Silica sol interacts with cellulose and is deposited tightly on the cellulose grid to form a uniform distribution of silica coating. This is because cotton fabric fibers belong to single-cell seed cellulose, and the hollow interior and twisted porous surface provide a good carrier for adsorbing fine particles. In addition, a large amount of -OH on the surface of the cellulose forms a hydrogen bond with the -OH generated by the dehydration reaction in the silica sol, which provides support for the formation of a stable chemical structure. Each cotton fiber can still be identified clearly due to the nanoscale sizes (Table 2) and inorganic nature of silica sol particles. Although there is a protective layer on the surface of the cotton fabric treated by KH560 (Figure 4(c)), the twisted texture and direction of the fabric can still be roughly seen, but the single fiber is not obvious enough and becomes wider (apparently connected) and thicker. The coating is integrally wrapped and continuously distributed, reflecting the organic nature, good physical adsorption (the specific surface area is 30.23 m2/g) and chemical bonding of KH560. In contrast, the sample KH560@Cotton only has 4.41% Si content, so the flame retardant effect may be worse. After ZB coating (Figure 4(d)), the single cotton fiber can still be identified, but it turns into a cylinder, which is similar to the effect of silica sol coating. This indicates that ZB and silica sol have similar actions, that is, they have physical attachment and chemical hydrogen bond adsorption (induced by chemically absorbed water in the salt) at the same time, but ZB shows larger particle sizes in its solution than those of silica sol, therefore the latter has stronger effect obviously. The sample ZB@Cotton contains 7.06% Zn element and 1.57% B element, which indicates that ZB is attached to the surface of cotton fabric through finishing, but the contents of elements detected are low due to the small amount of additions. Unlike the texture of the SiO2 sol coated fabric, the surface of SiO2-KH560 coated fabric seems to be more natural and forms a uniform and dense protective film (Figure 4(e)). After adding KH560, the coupling effect of silica sol on the fabric surface is significantly enhanced. The composite sol particles are connected by organic-inorganic KH560-SiO2-(-R-Si-O-Si-) [28], as shown in Scheme 2. The SiO2-KH560-ZB@Cotton sample shows a certain surface roughness (medium size of 630.00 nm and a specific area of only 65.92 m2/g) because of the presence of zinc borate particles attached on cotton substrate fibers (Figure 4(f)). The morphologies of Figure 4(f) and (e) are similar, indicating that the common/double chemical bond forces of organic-inorganic hybrid -KH560-ZB- and -KH560-SiO2- are dominant in the hybrid sol particles. The thickness and continuity of coating layer increase, and the shape and direction of cotton fiber are not that visible. The Si contents in samples SiO2-KH560@Cotton and SiO2-KH560-ZB@Cotton are 16.35% and 13.48% respectively, reflecting the coupling effect of SiO2 and KH560.
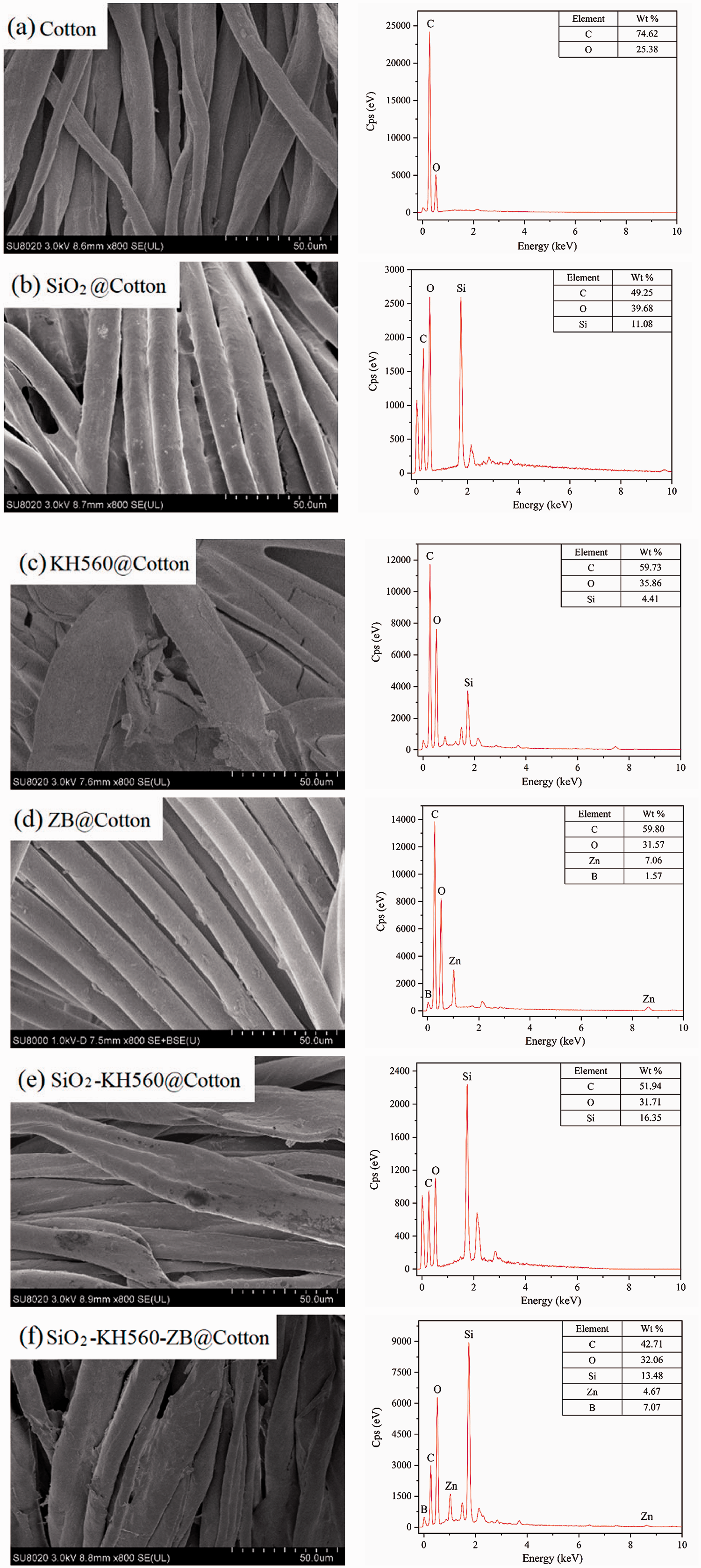
SEM and EDX images of the pristine and coated cotton fabrics before burning tests.
Analysis of pyrolysis characteristics of the cotton fabrics
The pyrolysis processes of pristine cotton fabric samples after single, composite and hybrid sol/solution finishing in air atmosphere are shown in Figure 5. The main parameters in the pyrolysis processes are shown in Table 3.
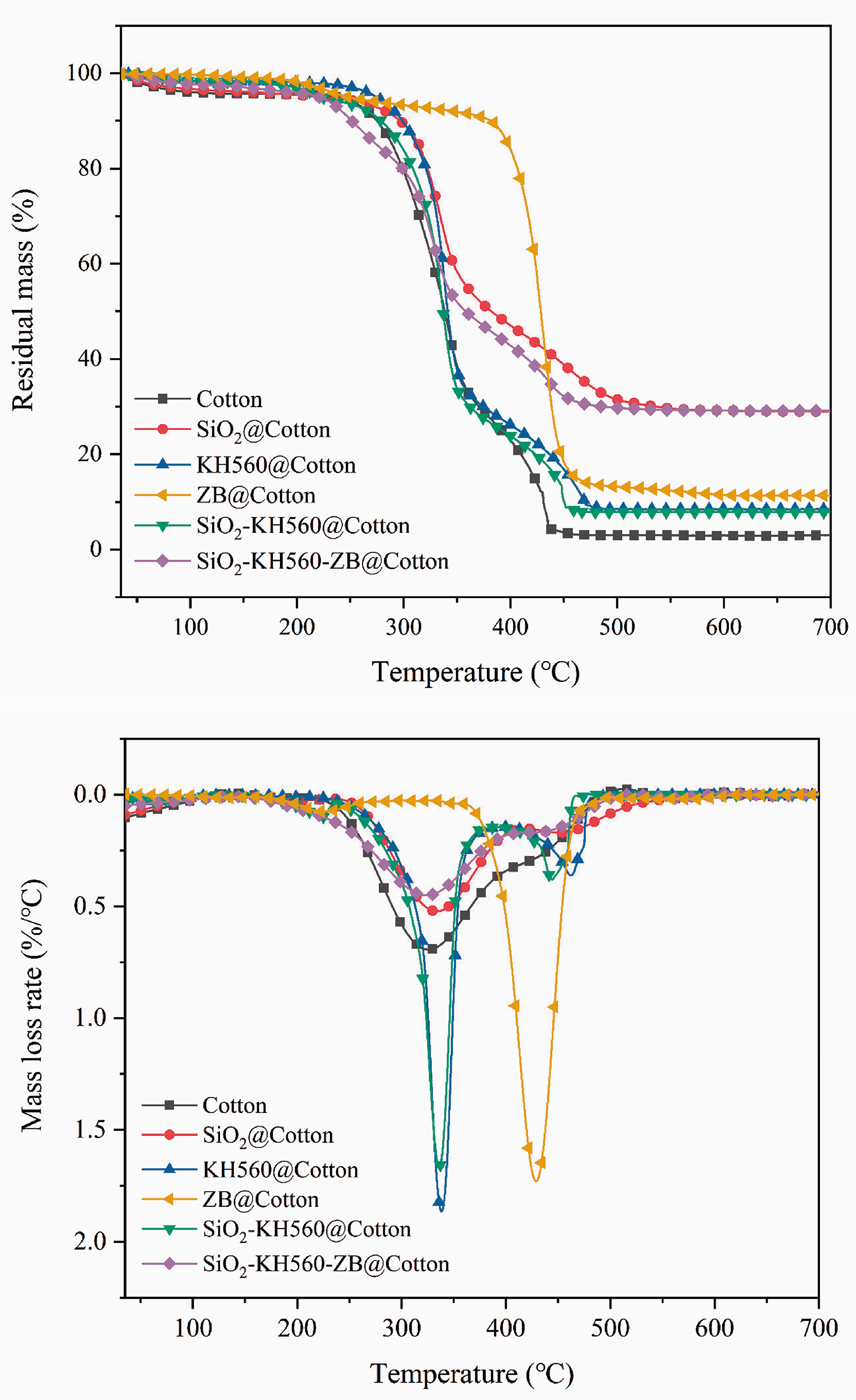
TG and DTG (First-order derivative of TG) curve of the pristine and coated cotton fabrics.
Main parameters of TG of the pristine and coated cotton fabrics.
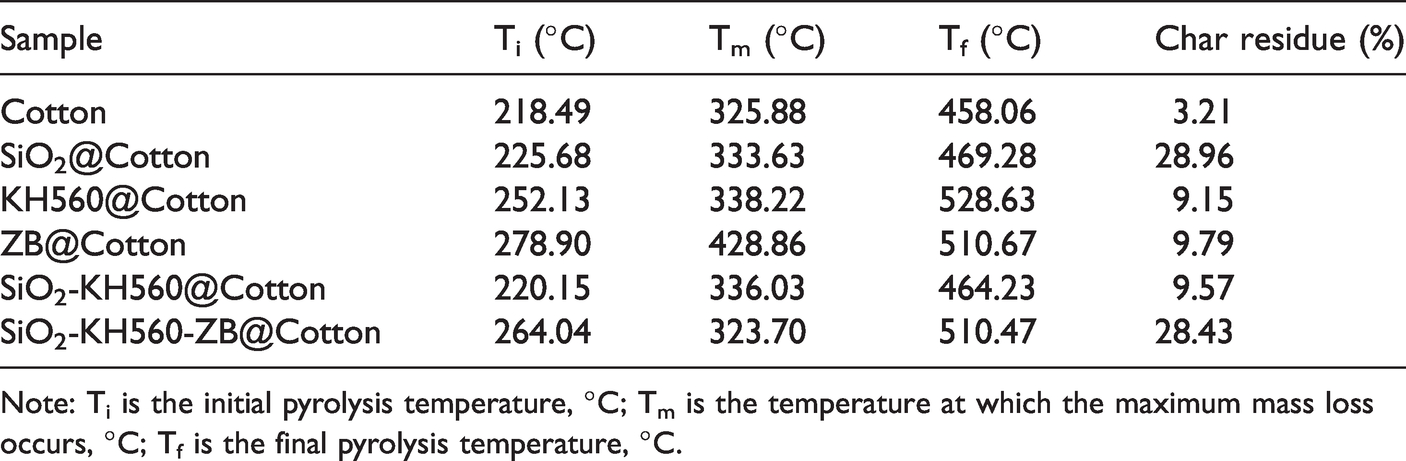
Note: Ti is the initial pyrolysis temperature, °C; Tm is the temperature at which the maximum mass loss occurs, °C; Tf is the final pyrolysis temperature, °C.
It can be seen from Figure 5 that the pyrolysis processes of the fabrics are roughly divided into three stages: initial pyrolysis stage (30.0∼300.0°C), main pyrolysis stage (300.0∼500.0°C) and end pyrolysis stage (500.0∼700.0°C). In the initial pyrolysis stage, the physically adsorbed water and small molecule auxiliaries in the fabric are released as the temperature rises, and the mass loss of the sample at this stage does not exceed 10.0% of its own mass. The main pyrolysis stage is accompanied by severe mass loss. The mass loss during this period is mainly because the temperature continues to rise and the fiber material and its protective gel coatings derived from composite/hybrid sols begin to decompose and crack. At this stage, the mass loss of the fabric reaches 80.0%∼90.0% of its own mass. In the end pyrolysis stage, the mass losses of cotton fabrics are relatively stable, and the mass loss is caused by dehydration and carbonization. The mass loss is less than 5.0% and the final tendency is unchanged.
The data in Table 3 shows that the initial pyrolysis temperature of cotton fabrics treated by SiO2 sol is from 218.49°C to 225.68°C, and the char residue reached 28.96%, which is attributed to the polycondensation reaction of the three-dimensional network of silicon dioxide (Si-O-Si) under high temperature, forming a dense protective layer and effectively isolating heat and oxygen. It is worth noting that the initial pyrolysis temperature of ZB coated cotton fabrics is significantly delayed to 278.90°C, mainly due to the removal of crystalline water by zinc borate at around 300.0°C, a process that absorbs a large amount of heat. Compared with pristine cotton, the sample SiO2-KH560-ZB@Cotton has a higher initial pyrolysis temperature and termination pyrolysis temperature. In addition, the char residue content of cotton fabric treated by SiO2-KH560-ZB@Cotton is significantly higher than that of Cotton fabric and ZB@Cotton fabric at 700.0°C. Meanwhile, by comparing the samples of SiO2@Cotton, KH560@Cotton, ZB@Cotton and SiO2-KH560@Cotton, it can be found that zinc borate plays a leading role in delaying the thermal oxidation of the fabric, while SiO2 plays an irreplaceable role in the formation of strong and coherent carbon layers. At high temperatures, zinc borate is decomposed into B2O3 and ZnO and hydrolyzed to produce boric acid, which is attached to the fabric surface to form a coating through hydrogen bonding and physical adsorption [29]. The coating inhibits the formation of flammable gases and also prevents oxidation and pyrolysis. About 38.0% of the zinc in zinc borate enters the gas phase in the form of zinc oxide or zinc hydroxide and dilutes the combustible gas and reduces its combustion rate [30]. From Table 3, it is known single SiO2, KH560 and ZB mainly contributes to high char residue and high terminal pyrolysis temperatures (for ZB, also high primary pyrolysis temperature) in pyrolysis processes of cotton fabrics. KH560 plays its organic effect and strong coupling role in attracting each component together. When they are combined, such features are well maintained for the SiO2-KH560-ZB@Cotton sample. In this case, it well reflects that silica gel, KH560 and zinc borate show excellent synergism of blocking, coupling, denaturing, inertness and other effects respectively, which are of great value in improving the thermal stability and flame-retardant ability of cotton fabrics.
Analyses of flame-retardant properties of the cotton fabrics
The LOI values of the single, composite and hybrid sol systems and each single solution finished fabric sample are recorded as shown in Table 4 and Figure 6. The LOI values of all modified cotton fabrics have generally been improved to varying degrees. They rely on the coexistence forms of each component. Compared with the pristine cotton, the LOI value of the sample after the silica sol treatment has increased by at least 2.5%. However, the sample SiO2-KH560@Cotton has no significant improvement compared with the sample SiO2@Cotton, mainly because SiO2 is connected to the cotton fabric through physical adsorption and hydrogen bond before fire. After fire, the process of oxygen transport, heat exchange and oxidation decomposition could be inhibited. However, as the temperature continues to rise, the brittle SiO2 coating will be cracked. Although the addition of KH560 has a certain coupling effect, it can bond with SiO2 and the surface of the fabric, but it is easy to decompose completely at high temperature of 700.0°C (Table 3), and its role of coupling is limited when embedded and blended with silica coating. Therefore, pure silica sol is limited in improving the flame retardancy of cotton fabrics. In contrast, the LOI values of KH560@Cotton, ZB@Cotton and SiO2-KH560-ZB@Cotton increase notably (ΔLOI = 5.0%), and the flammability level of cotton fabric has been raised from inflammable to combustible, which corresponds to the much delayed initial pyrolysis temperature by 26.94–53.61°C (see Table 3). In particular, the sample SiO2-KH560-ZB@Cotton has the highest LOI and the lowest maximum thermal oxidation rate, which demonstrates the mutual competitiveness and synergy between SiO2, KH560 and ZB. The strong physical covering effect of silica sol and its bonding with zinc borate trigger strong flame-retardant effect and high thermal stability. However, although silica sol (gel) has strong covering ability, it is fragile when heated and has limited fire resistance. The poor performance of silica gel at high temperature can be made up by strong coupling effect of KH560 and prominent pyrolysis delaying capability of ZB. To further confirm the inherent flame retarding capability of each component, a new concept is introduced here, namely the LOI increment (ΔLOI/Δm) brought by unit weight gain, which means the flame-retardant efficiency of the material, it can eliminate the influence of materials to the LOI. The sample SiO2-KH560-ZB@Cotton has less material attached to the surface (Δm = 13.51 g), but the ΔLOI/Δm (0.37%/g) is the highest, 0.25%/g higher than ZB@Cotton, which shows that SiO2-KH560-ZB actually has a high flame-retardant efficiency. This also verifies the excellent synergy of silica sol and zinc borate. Zinc borate melts at high temperature to form a glassy coating and then dehydrated. The evaporation heat of chemically bound water, dilution and cooling effects of water vapor help to make the burning self-extinguish. Both the dehydrated zinc borate and the gelled silica sol can promote the formation of the inorganic carbon layer, which is not only impedes ignition and burning but also insulate transportation of oxygen, combustion products and heat. The high ΔLOI/Δm (0.30%/g) of KH560 reflects its single important role in coupling each component and improving flame retardancy all the time.
LOI values of the pristine and coated cotton fabrics.

Note: Δm is the increase of the sample, g.

ΔLOI and ΔLOI/Δm of the pristine and coated cotton fabrics.
Analyses of the combustion behaviors of the cotton fabrics
In LOI experiments, the combustion behaviors of the fabrics finished by different sol-gel systems are recorded, as shown in Figure 7. After the untreated cotton fabric samples burn, the residue collapse. All the samples after sol/solution modification do not collapse after burning. The flame of cotton fabrics treated with silica sol is small, so silicon-based flame retardants have a positive control effect on the flame sizes during the combustion process of original cotton fabrics. The deposited single and hybrid SiO2 coatings can still provide certain protection for cellulose (see Figure 4), prevent and slow down the flame propagation. Meanwhile, the smoke amount of the fabrics treated with SiO2-KH560-ZB is less in the combustion process, which may be due to the fact that zinc borate is a good smoke suppressant basically similar to that of a certain metal oxide. In the burning process, it strongly promotes the carbonization of material, thus reducing the contents of combustible substances in the gas phase and the smoky property of the material [31]. According to the morphology of the burnt residues in Figure 7, it can be found that SiO2-KH560 and ZB have a synergistic effect on the formation of a complete carbon layer (the sample SiO2-KH560-ZB@Cotton in Table 3 has nearly the highest char residue of 28.43%). This is due to the thermal decomposition of zinc borate to produce a B2O3 glass-like film, while the silica sol is heated to produce Si-C compounds and inorganic silicon, which cover the substrate and act as a heat and oxygen barrier. Another issue worthy of attention is that the smoldering phenomenon of the sample SiO2-KH560-ZB@Cotton has basically disappeared and greatly improves the fire safety performance of textile, which is consistent with the good afterglow suppression effect of boron.
Combustion behaviors in the LOI experiments of the pristine and coated cotton fabrics.
Analysis of combustion properties of the cotton fabrics
The microscale combustion calorimetry is an effective method to evaluate the combustion performances (flammability levels) of raw and finished cotton fabrics on a microscale with outputs like Peak Heat Release Rate (PHRR) and Total Heat Release (THR). Figure 8 shows the relationship between heat release rate (HRR) and temperature of the cotton fabrics. Peak heat release rate (PHRR), total heat release (THR) and the temperature to PHRR are listed in Table 5. The results show that the pristine cotton fabric burn quickly, and its PHRR and THR are 310.6 W/g and 14.0 kJ/g, respectively. The PHRR and THR of SiO2@Cotton decrease by 37.1% and 32.1% (195.4 W/g and 9.5 kJ/g) when compared to pristine cotton, respectively. This significant decrease in PHRR may be attributed to the protective structure formed after the prepared coating is burned, which can act as a barrier to release the combustible pyrolysis products of cotton fibers [32]. The PHRR values of samples KH560@Cotton and ZB@Cotton are both higher than the PHRR of SiO2 finishing cotton fabric, indicating that the heat insulation effect of KH560 and ZB in solid phase is worse than that of SiO2. Also, KH560 is an organic agent and increases combustible pyrolysis products. ZB significantly reduces the THR of the coated cotton fabric, thanks to the catalytic ability of ZB to form carbon, which can effectively reduce the content of combustible substances in the gas phase. Compared with SiO2@Cotton, the sample SiO2-KH560 @Cotton and SiO2-KH560-ZB@Cotton show higher PHRR and THR. This just approves the both competitive and synergistic effects among components of SiO2 gel, KH560 and ZB, that is, SiO2 sol is important to provide a basis physical barrier coating, while KH560 and ZB destroy the connected coating to some extent due to their pyrolysis behaviors under flame conditions. However, KH560 and ZB do show their synergism in intensifying coating strength in solid phase and disturbing radical reactions in gas phase. Certainly, due to very limited loading ratios (The concentration of KH560 hydrolyzate is 20%, the addition amount of this hydrolyzate is 50%, and the addition amount of ZB is 10 g/L), their roles are also restrained. Whatever, compared to the pristine cotton fabric, the PHRR, THR values of sample SiO2-KH560-ZB@Cotton have decreased by 26.9% and 22.9%. This just reflects that SiO2-KH560 and ZB exhibit considerable synergistic flame retardant effect. Moreover, all coated cotton fabrics have different degrees of advancement of temperature to PHRR. This is understandable since all additives play their flame retardant roles earlier (in their functional stage via decomposition) and reduce the PHRR values meanwhile (see Figure 5 for reference). ZB@Cotton has the largest advancement (ΔT = 28.2°C) with a PHRR decrease of 34.3%. From the viewpoint of exothermic behavior, the flame retardant finishing will make the finished cotton fabric have a smaller fire hazard. The results are generally consistent with those of LOI experiments (see Table 4, Figures 6 and 7). Certainly, they show deviations since their operation principles are different. Therefore, to judge flame retardant effect of a system reasonably, it is necessary to use multiple flammability experiments comprehensively.
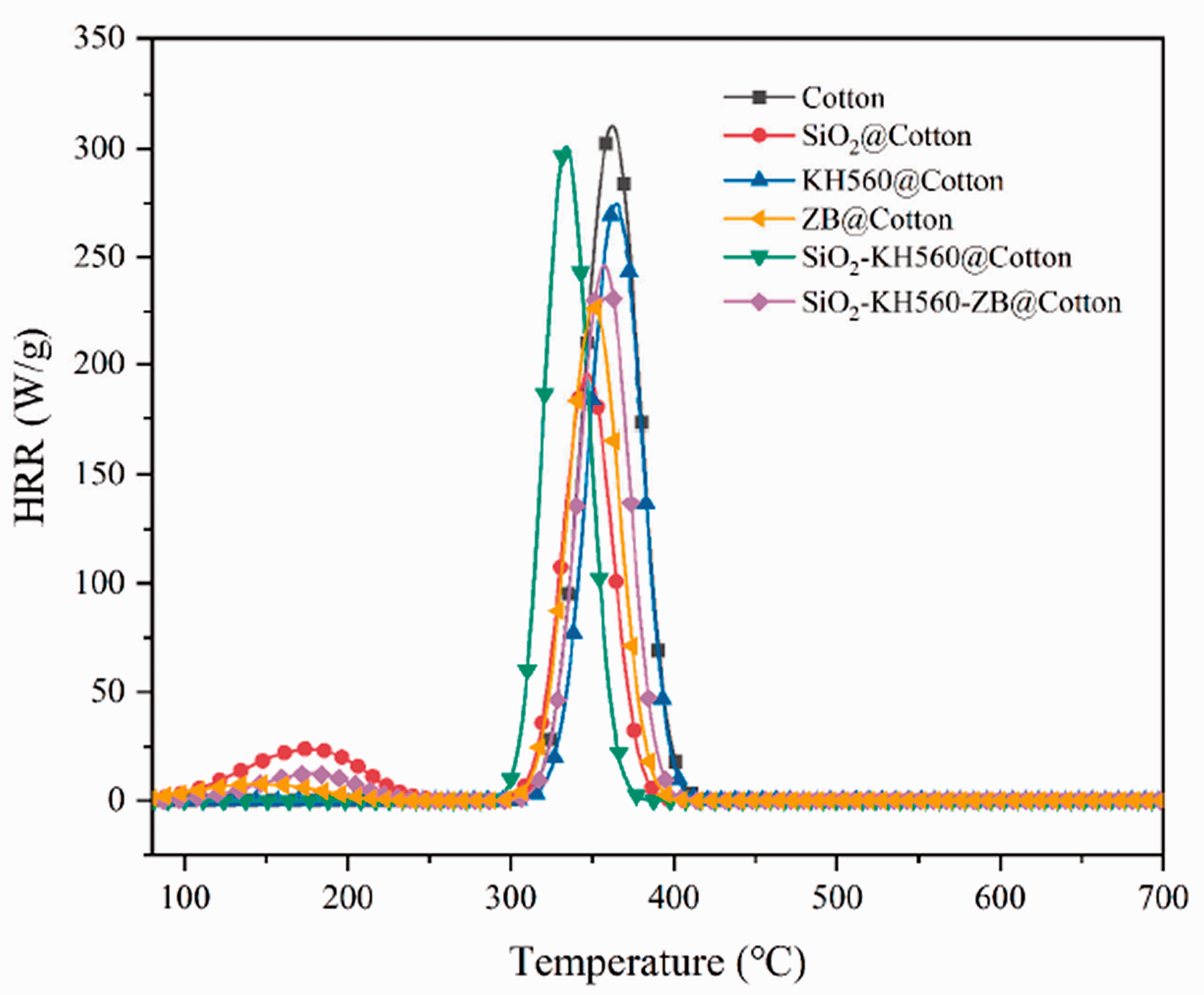
Relationship between HRR and temperature for the pristine and coated cotton fabrics.
Flammability values of the pristine and coated cotton fabrics via MCC.

Microstructure analyses of the cotton fabrics after LOI experiments
To further reveal the mechanism of flame-retardant effect, the morphologies of char residues after the LOI experiments are characterized by SEM, and the results are displayed in Figure 9. The cotton fabric exhibits a porous microstructure after combustion, and the char layer is sparse and loose (Figure 9(a)). The fiber structure is decomposed and fully carbonized during combustion; the char layer is formed naturally by dehydration of D-anhydroglucose ring units of cotton. The cotton fabric treated with SiO2 sol has a rough and dense film on the surface after burning (Figure 9(b)), which is a vitreous coating layer formed by polycondensation of silica network at high temperature [33]. This layer can maintain the complete structure of the internal fibers during the combustion process, and functions as heat insulation, oxygen barrier and smoke suppression. However, such inorganic silica gel is easy to become brittle, cracked and disordered until losing its protection role as the temperature increases (333.63°C in Table 3). After that, the substrate cotton fabric is exposed to start the second round of pyrolysis and carbonization. Figure 9(c) and (d) display the surface of the cotton fabric treated by KH560 and ZB after burning. There is obvious connected coating (induced by chemical coupling of KH560 and physical covering of ZB pyrolyzate) on the surface under which cotton fibers are embedded. Combined with LOI and especially ΔLOI/Δm results, it can be seen that both KH560 and ZB have certain flame-retardant effects. Zinc borate can play a flame-retardant effect in the condensed phase and gas phase at the same time, which is mainly reflected in the following three aspects: 1) When zinc borate is heated to above 300.0°C, it releases crystal water, absorbs a large amount of heat energy, and reduces the temperature of the combustion surface. 2) The released water and zinc oxide enter the gas phase to dilute the oxygen in the air and inhibit the burning reaction. 3) The finally produced B2O3 glass-like film is covered on the polymer to act as a heat and oxygen barrier. Moreover, zinc borate has strong charcoal properties, therefore, the char layer on the surface of ZB@Cotton sample is intact. A more pronounced and complete fiber structure can be seen from the cotton fabric treated by SiO2-KH560 (Figure 9(e)) and SiO2-KH560-ZB (Figure 9(f)), and the surface of the char layer is dense and smooth. Due to the inorganic-organic coexistence conditions of components, there shows competing interactions among them: the silica gel inclines to crack into pieces while KH560 and ZB pyrolyzate just makes up this. As a result, each charred cotton fiber can be separated and identified. This further indicates that the organic KH560 has a significant coupling effect and plays an observable role in maintaining structural stability. In Figure 9(f), a large number of distinct particulate solids adhere to the surface of the silica network, while fine pores exist outside the char layer due to the decomposition reaction of zinc borate and cracked silica gel particles. This kind of pore structure will clearly elongate the winding path lengths and numbers of exchanged and emitted air, oxygen and combustion products. Such a difficulty is beneficial to improve flame retardancy. It can be verified in the TG test. The silica gel coating modified by KH560 can improve the adhesion, sealing and crack resistance of cotton fabric when heated. After adding zinc borate, it can further improve the char forming ability of cotton fabric (the amount of char residue increased by 18.86% and the layer is complete and dense). The flame retardant efficiency (ΔLOI/Δm = 0.37%/g) produces a smoke suppression effect and achieves a significant synergistic effect.

SEM images of the fabrics and char residuals after LOI burning tests.
Explorations of flame-retardant mechanisms of silica-based sol systems
To figure out the potential and comprehensive flame-retardant mechanisms of silica-based sol systems in this work, inductions will be done in this section solely. The flame-retardant mechanism of SiO2-KH560-ZB@Cotton system is illustrated in Figure 10. In general, the components show complex interactions in flame retarding processes due to their different nature and function stages. They show more synergistic effects than competitive roles. When exposed to fire, cotton fibers with abundant hydroxyl groups become carbon sources in the flame-retardant system. As the temperature increases, the cellulose is dehydrated and carbonized, producing water, carbon dioxide and coke residues [34] in Figure 9(a). The silicon dioxide connected to the surface of cotton fabric by hydrogen bonds is thermally decomposed into inorganic silicon dioxide and high-temperature thermally stable Si-C compound, and then further converted into graphite or glassy carbon on the surface of cotton fiber (Figure 10(a)) [35]. As a result, a dense expanded barrier layer is formed on the surface of the fiber which effectively protects the internal fibers from heat and flame by impeding heat transfer, diluting oxygen and isolating flammable volatiles [36], plays the role of char layer isolation and porous isolation (reasonable improvement in LOI). The coupling effect of KH560 strengthens this isolation effect, which is essentially derived from the strengthening of physical coverage and chemical bonding (hydrogen bonds among different substances and those between components and fabrics). However, the organic nature of KH560 makes it easier to pyrolyze. As a flame retardant, zinc borate will lose crystal water when it is higher than 300.0°C, absorb a lot of heat energy and reduce the combustion surface temperature. Meanwhile, the released water can dilute the oxygen in the air, take heat away and cool down the burning sites, and thus inhibit the burning reaction. At high temperatures, ZB is decomposed into B2O3 and ZnO and brings vitreous coating on the surface of fabric. Part of ZnO flushes into the gaseous phase and dilutes the combustible gas, lowers the cotton fabrics burning rate and further increases its flame retardancy. ZB helps to reduce the release of flammable volatile decomposition components and slow release of heat (Figure 10(b)). In fact, ZB has also been shown to change the thermal decomposition pathway of some combustible substances and inhibit the formation of combustible gases. B2O3 has an inhibitory effect on the oxidation reaction of carbon compounds, thus controlling R+O2→RO2· free radical generation and accelerating the termination of chain reaction. In addition, a small amount of boric acid [37] will be produced when zinc borate is hydrolyzed. Boric acid can be used as an acid source to promote the formation of char layer, produce synergistic effect with silica sol, and reduce the amount of smoke produced by cotton fabric (the maximum pyrolysis temperature of the sample SiO2-KH560-ZB@Cotton in Table 3 is advanced, and the char residue increases). In general, the flame retardant effects of cotton fabrics finished by sol-gel gel coating are primarily contributed by the following aspects: SiO2 sol builds special folding three-dimensional space carrier and provides a framework shielding layer; KH560 and ZB reduce the burning surface temperature, strengthening the stability of the shielding and isolation effect, accelerate the termination of the chain reaction through physical covering and chemical coupling [38–40]. The indispensable individual advantages and obvious synergistic effect are shown especially by the ΔLOI/Δm index.

The schematic diagram of flame-retardant mechanism of SiO2-KH560-ZB@Cotton system.
Conclusions
This work mainly focuses on the flame-retardant needs of cotton decorative fabrics and products. The advanced sol-gel technology is used to prepare a ternary sol-gel system mainly including silica sol, KH560 and ZB. The system uses the impregnation baking method to modify and finish pure cotton fabrics to achieve cost-controllable and easily degradable flame-retardant properties. Experiments show that distributions of particle sizes, surface microscopic morphology and functional groups in sols picturing the overall existence states of condensed matters on the surfaces of cotton fabrics. They can play important and interactive roles of affecting pyrolysis properties and flame retardancy levels of treated cotton fabrics.
The SiO2-KH560-ZB hybrid sol is produced on a nano-micro scale (with a particle size of 630.00 nm). The transformed gel shows a special three-dimensional skeleton structure (as shown in Scheme 1), and is coupled through KH560 to strengthen the chemical bond (as shown in Scheme 2). This can maintain the structure of the gel coating (as shown in Figure 4) and ensure that the components are tightly connected to each other and the cotton fabric through physical adsorption and hydrogen bonding force (as shown in Figure 3). The fabric treated with SiO2-KH560-ZB has acceptable flame retardant properties, and the value of ΔLOI/Δm reaches the highest value of 0.37%/g. Meanwhile, the hybrid gel coating delays the initial pyrolysis of the cotton fabric, reduces PHRR by 26.9% and THR by 22.9%, reduces the amount of smoke generated (as shown in Figure 7), and improves the ability to form char. At 700.0°C, the char residue of the fabric is increased from 3.21% (Cotton) to 28.43% (SiO2-KH560-ZB@Cotton).
In fact, the silane coupling agent KH560 contributes the most to the final flame-retardant level, and the SiO2 sol has the most significant impact on the amount of char residue, which can be reflected by the ΔLOI and TG data of the finished cotton fabric. Clearly, the cooling, separation, dilution and inhibition effects of zinc borate, the physical barrier effect of silica sol and the coupling effect of KH560 play an excellent synergistic effect, which is fully reflected via ΔLOI/Δm. The special spatial arrangement and hydrogen bonding force of the gel coat adhered to the surface of the cotton fabric provide a framework and connection for the function of each component. Zinc borate as a flame-retardant synergist reduces the surface temperature of the fabric to form a glassy package. Its covering layer of pyrolyzates and combustion products isolates oxygen and heat, generates zinc compounds and boron compounds to promote carbon formation, accelerates the termination of the chain reaction, and simultaneously exerts a flame-retardant effect in both the condensed phase and the gas phase. Therefore, the inorganic-organic combination among a traditional flame retardant, a coupling agent and silica sol systems can give full play to the flame-retardant advantages of all components, and provides a new direction for the development of flame-retardant functional products in the field of decorative textiles. In the next steps, to further enhance the general flame retardancy, alternative ratios, coupling agents and even whole ternary systems can be explored.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study is financially supported by the National Natural Science Foundation of China (Grant No. 51376089 and 50906039), the 15th Six Talent Peaks Project in Jiangsu Province in Renewable Energy and Energy Internet Industry (XNY-016), the 2019 Key Project of The Natural Science Foundation of the Jiangsu Higher Education Institutions of China (19KJA520007, A Class), and the Postgraduate Research & Practice Innovation Program of Jiangsu Province (SJCX20_0372).
