Abstract
Hybrid organic-silica material modified by amino groups has been investigated as adsorbent of radionuclides
Introduction
One of the most important challenges of the modern chemistry is search of agents for purification of potable and industrial water from radionuclide and toxic metals (Lujaniene et al., 2008; Myasoedova, 2005; Tarasevich and Dabrowski, 1999; Wu et al., 2008). The most appropriate ways to solve this problem is use of sorbents (Krasnopyorova et al., 2007, 2008, 2013; Mokhodoeva et al., 2011). Hybrid organic-silica materials which contain organic moieties are used in adsorption, catalysis, and chromatography (Boury, 2002; Laughlin et al., 2000; Morosanova et al., 1998; Shilova, 2002). The main idea of creating such materials is to combine their inorganic and organic parts, which allows to control their mechanical, thermal and structural stability as well as to provide specific material properties through chemical properties of organic modifiers (Shilova, 2002). The sol–gel technology of hybrid organic-silica materials production is referred as nanotechnology due to the possibility of regulating the material structure at the molecular level. The widespread sol–gel method allows to produce the materials with predictable properties and different morphology. These are organic mineral monoliths and films, sorbents, catalysts, fibers, ceramics, chemo- and biosensors and others (Boury, 2002; Laughlin et al., 2000; Morosanova et al., 1998; Shilova, 2002; Zub and Parish, 1996). In addition, among the materials obtained by the sol–gel method are widely adopted aminocontaining organic-silica materials (xerogels) (Dash et al., 2008; e.g. hybrid silica-organic; Tkachenko et al., 2013). The goal of this study is to investigate the sorption and concentration of radionuclides (
Experimental
Synthesis of hybrid organic-silica material
Amino containing hybrid silica-organic material was obtained by sol–gel method according to the procedure reported in (Morosanova et al., 1998). To the mixture of equal volumes of γ-aminopropyltriethoxysilane (C2H5O)3Si(CH2)3NH2 (APTES) and tetraethyl orthosilicate Si(OC2H5)4 (TEOS) (both Merck, 98%), ethanol ((Dubovyazivka Distillery, Ukraine, 96%) and aqueous solution of gelation catalyst (ammonium hexafluorosilicate, Reachim, Ukraine) were added. The ratio of volumes of the components in the mixture silane:ethanol:water was 2:5:2. The mixture was stirred until the gel formation and then was dried in the microwave at 70 W (10 min) and 150 W (5 min) (Figure 1). The details of structure and properties of hybrid organic-silica were described earlier in Baraban et al. (2012). The specific surface area of the material was 70 ± 5 m2/g, while specific concentration of amino groups was 4.02 mmol/g was calculated from the reagent mixing conditions. This value was in agreement with the one which is obtained by the maximal absorption of H+ ions (4.04 ± 0.02 mmol/g).
Amino containing xerogel formation scheme.
Sorption of radionuclides
and
on amino containing xerogel
Sorption ability of amino containing xerogel to absorb radionuclides
Results and discussion
Distribution coefficients Kd and sorption coefficients Ks values for amino containing xerogel are different for various radionuclides

Analysis of the kinetic curves of the radionuclides sorption on the xerogel at pH 7 showed that the sorption equilibrium in the system radionuclide sorbent for Kinetic curves of radionuclides sorption on amino containing xerogel at pH 7.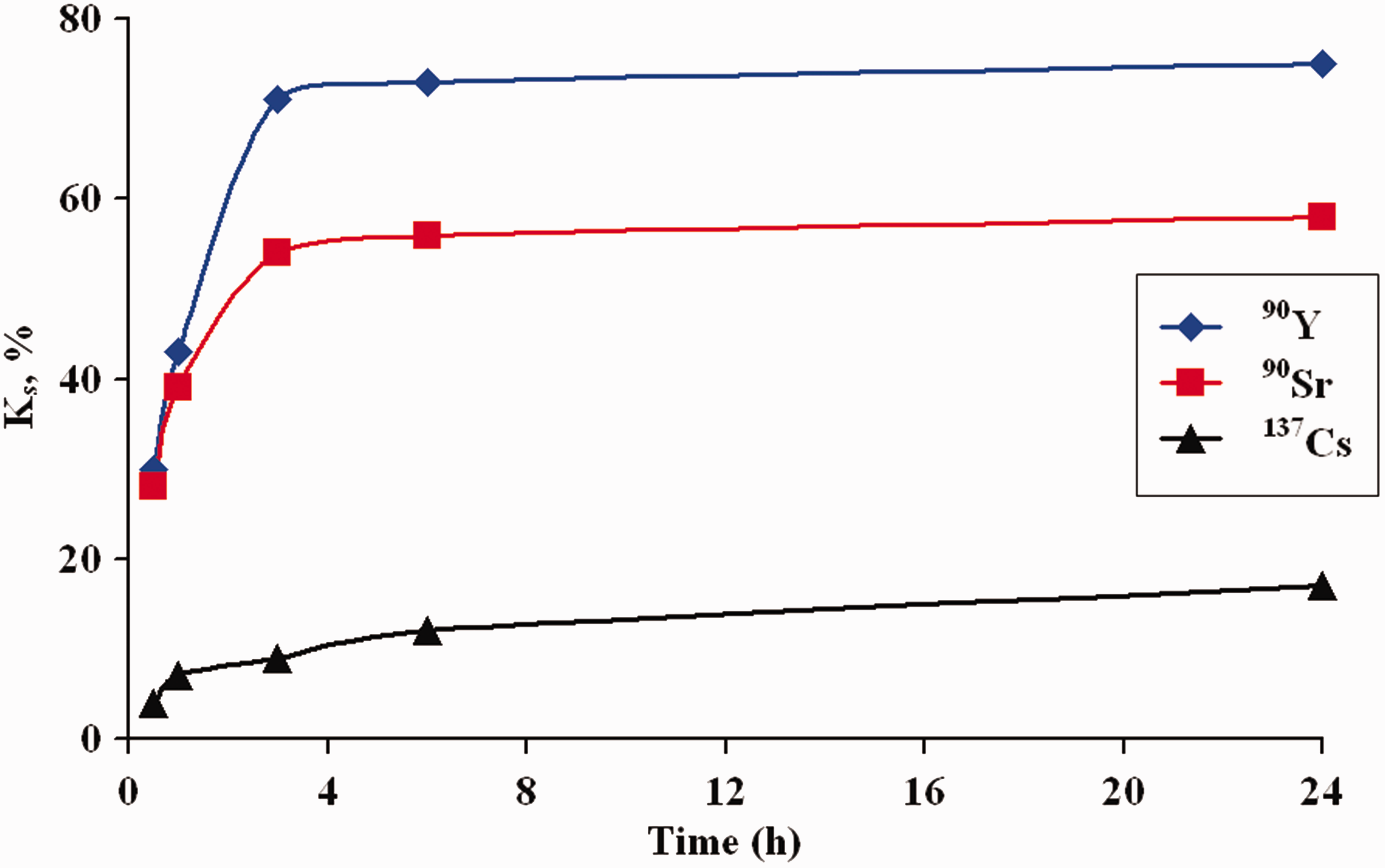
The mechanism of the sorption process is rather complicated. Diffusion in the solution makes an impact on initial speed of the sorption process. The literature analysis shows that the main contribution to sorption of radionuclides on the hybrid material surface gives complexation (Myasoedova and Nikashina, 2006). Different functional groups of available sorbent lead to slower diffusion on its surface, and the absorption of radionuclides is achieved due to the formation of complexes on the surface (1) and ions exchange (2).


The pH of an environment significantly affects the quantitative sorption characteristics. Sorption of radionuclides is minimum at pH = 2. With increasing pH, the sorption increases and reaches the maximum at pH 8–9 (Figure 3).
Sorption coefficients Ks values for amino containing xerogel are different for various radionuclides 
Such dependence of the sorption from pH is typical for weakly acidic ion-exchangers, which is modified xerogel, due to the presence of silanol groups. It should be noted that the sorption
Conclusion
The distribution of radionuclides
Footnotes
Acknowledgements
This study has previously been presented at 15th Ukrainian-Polish Symposium on Theoretical and Experimental Studies of Interfacial Phenomena and Their Technological Applications, Lviv, Ukraine, 12–15 September 2016.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
