Abstract
Electrospun nanofibers based on polysaccharides represent a consolidated approach in Tissue Engineering and Regenerative Medicine (TERM) and nanomedicine as a drug delivery system (DDS). In this work, two chemical derivatives of a low molecular weight gellan gum (96.7 kDa) with aliphatic pendant tails were processed by electrospinning technique into non-woven nanofibrous mats. In order to generate spinnable blends, it was necessary to associate poly vinyl alcohol (PVA). The relationships between the physicochemical properties and the processability via electrospinning technique of gellan gum alkyl derivatives (GG-C8 and GG-C12 having a degree of alkyl chain derivatization of 17 mol % and 18 mol %, respectively) were investigated. The deposition of nanometric fibers (212.4 nm ± 60.0) was achieved by using the blend GG-C8/PVA spinned at 5% w/v in water. The use of a binary solvent composed of water and ethanol in a volumetric ratio 95:5 improved further spinnability obtaining similar nanofiber diameters (218.0 nm ± 96.0). The rheological analysis has allowed to highlight the role of the alkyl portion (C8 and C12) on the spinnability of the blended polymers.
Introduction
Electrospinning (ES) is a versatile processing technique that enables the fabrication of a wide variety of non-woven fibrous mats, whose peculiar features, in terms of fiber mean diameter, porosity, aligned or random orientation of fibers, and 2D or 3D architecture of membranes can be designed by proper tuning of specific operating parameters and physicochemical properties of starting materials [1,2].
Concerning other spinning methods, ES allows the fabrication of membranes made of nanometric fibres, thus ensuring a high area-to-volume ratio [3–5]. Electrospun membranes (EM) mimic the extracellular matrix (ECM) architecture, with this feature being particularly suitable for tissue engineering applications [6]. Membranes can be biofunctionalized with therapeutic agents or covered with adhesion proteins to improve bio integration with the damaged tissue [7–11]. ES is widely used to produce devices in many wound healing applications [2,12–14]. Electrospun membranes are applied as haemostatic and permeable devices to permit the gaseous exchange, the absorption of exudates, or be employed as a delivery system to directly release bioactive molecules in the wound area [15–20]. Polysaccharides are often used to produce regenerative devices thanks to their high biocompatibility, in fact, several benefits in TERM field, such as dermal, cartilaginous, and bone regeneration, or more complex organs like liver or heart have already been achieved [21–24]. Among polysaccharides, cellulose, chitosan, hyaluronic acid and gellan gum and their derivatives are often used as biotemplates for these purposes [25,26]. However, the electrospinning of these natural polymers results tricky because most of them are polyelectrolytes showing high conductivity in water and low chain flexibility [27]. In light of these properties that make electrospinning difficult, polysaccharides are blended with synthetic polymers acting as carriers to improve spinnability. The carrier increases the entanglements among polymeric chains, thus increasing the solution viscosity and reducing the overall conductivity [28]. Polyvinyl alcohol (PVA), a hydrophilic biocompatible polymer, improves polysaccharides electrospinning efficiency [29]. It was recently found to improve chains entanglements and therefore the spinnability. For example, Hyaluronic acid (HA), a natural polysaccharide main constituent of ECM, is scarcely spinnable due to high conductivity and viscosity of its aqueous solutions. Blends of HA, or its derivatives, with PVA and hydroxypropyl-beta cyclodextrin (HPCD) permitted a satisfactory control of the viscoelastic properties of resulting blends improving spinnability [11,15]. Gellan Gum (GG) is an exopolysaccharide of microbial origin (Sphingomonas elodea) with tetrasaccharide repetitive units. GG and its derivatives are widely used in the biomedical field as drug delivery systems (DDS) or regenerative scaffolds in TERM applications [30–34]. The ionotropic behaviour of this polysaccharide is exploited in several therapeutic strategies [35]. In recent studies, native GG-based nanofibers were produced with PVA in a 1.5:10 w/w ratio and optimized at 1:1 w/w. The resulting mats were crosslinked following different protocols until the final goal is satisfactorily achieved, namely the generation of an ofloxacin drug delivery system [36–38]. Considering recent advancement in the applicative use of this polysaccharide, we herein report the standardization of electrospinning procedure of octyl and dodecyl derivatized GG, namely GG-C8 and GG-C12, aqueous dispersions blended with an equal amount of PVA (80 kDa). The relationship between the chemical structure of derivatives, i.e. the length of the inserted alkyl chain, and spinnability of investigated systems was elucidated by investigating rheological properties of such dispersions.
Materials and methods
Chemicals
Gellan gum (Gelzan®; Mw 1000 kDa), sodium hydroxide, ion exchange cationic resin Dowex 50W-X8, tetrabutylammonium hydroxide (TBA-OH), bis(4-nitrophenyl) carbonate (4-NPBC), octylamine (C8-NH2), dodecylamine (C12-NH2), tetramethylammonium chloride (TMACl), ethanol (EtOH), acetone, and dimethyl sulfoxide anhydrous were purchased from Sigma Aldrich Italia. HCl 37% was acquired from Fluka. Merck supplied methanol. Polyvinyl Alcohol (PVA) 80 kDa was purchased from Serva Feinbiochemica.
DMEM and fetal bovine serum was obtained from Euroclone s.r.l. [3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium] (CellTiter 96® AQueous One Solution Cell Proliferation) was purchased from Promega.
Characterizations
1H-NMR spectra were recorded employing a Bruker Avance II 300 instrument, operating at 300.12 MHz. Size exclusion chromatography (SEC) analysis was performed using a multidetector SEC system equipped with a Waters 600 pump, a Waters 410 Refractive Index Detector, a PolySep-GFC-P 4000 column, and a guard column PolySep-GFC-P (35x8 mm) purchased from Phenomenex. The calibration curve was obtained by using pullulan standards purchased from Polymer Laboratories Inc. The elution medium was composed using an aqueous solution of TMACl 0.025 M and methanol 70:30 v/v with a flow rate of 0.6 ml/min at 35 ± 0.1°C. The rheological analyses were performed with an Ares G2 (TA Instruments). Electrospun mats were prepared using an instrument purchased from Linari Biomedical srl. SEM analyses were performed with the Phenom Pro X Desktop acquired from ThermoFisher Scientific. Analysis of fiber dimensional distribution and orientation was performed with ImageJ software and its plugins DiameterJ and OrientationJ. Attenuated total reflectance Fourier transform IR (ATR-FTIR) measurements of electrospun membranes were performed using a Bruker Alpha instrument.
Hydrolysis of gellan gum and synthesis of its alkyl derivatives
Hydrolysis of Gelzan® (1000 kDa) and the synthesis of alkyl derivatives GG-C8 and GG-C12 were performed according to previous procedures reported elsewhere [36,37,39]. The degree of derivatization of the materials, calculated via 1H NMR, was expressed as mean values ± standard deviation. 1H-NMR spectra (D2O) of GG-C8 and GG-C12 derivatives showed peaks at ∂ 0.7 and 0.8 (3H, of the -C
Size exclusion chromatography analysis
Powders of GG, GG-C8 and GG-C12 were dispersed in tetramethylammonium chloride (TMACl, 0.025 M in water) in the oven at 90°C for 4h. Then, the proper amount of methanol was added in each dispersion obtaining a mixture with the same solvent composition of the eluent phase, reaching a final concentration of 2.5% w/v at room temperature. The elution was performed on a Polysep P-4000 column at 35°C, using TMACl (0.025 M) as an eluent at a flow rate of 0.6 ml/min, using a calibration curve based on pullulan (range of Mw 5.9–212 kDa). All chromatograms were recorded after 30 min from injections.
Preparation of electrospun nanofibers
GG, GG-C8, and GG-C12 powders were mixed with PVA powder keeping a ratio equal to 1:1 w/w. The blends were thermally treated in oven for 4 h at 90°C, and vigorously stirred prior to electrospinning. In the binary blended dispersions, ethanol was added after thermal treatment, to achieve a final water/ethanol ratio equal to 95:5 v/v. Experimental design was developed by changing one instrumental parameter each time. Flow rate was tuned from 0.130 ml/h to 0.625 ml/h, applied voltage was varied from 10 to 20 kV and needle-to-collector distance was progressively increased from 10 to 15 cm.
Rheological characterization
Rheological behaviour of pre-ES dispersions was investigated by a rotational rheometer (Ares G2, TA Instruments) in oscillatory frequency sweep mode using a 25-mm parallel-plate geometry. The measurements were carried out at 25 °C, within the angular frequency range 1–100 rad/s. The value of strain adopted was 1%, chosen after preliminary strain sweep tests.
In vitro cytocompatibility assay
Round pieces of GG-C8/PVA membranes were crosslinked in a CaCl2 1 M solution in triplicate. After 15 min, membranes were washed in PBS, placed into cellcrown inserts of 48 well plates and sterilized under UV light at 254 nm for 15 min. NIH 3T3 fibroblasts were cultured until passage 5, suspended in DMEM and loaded above the membranes, seeding 1 × 105 cells for membrane. After 30 min of incubation, inserts were moved to another well plate and medium refreshed every two days. Cell viability was evaluated after 1, 3 and 7 days of incubation using the MTS assay. Aliquots of culturing medium were incubated for 4 h at 37°C, and 5% CO2 with a 20% v/v solution of MTS in supplemented DMEM. The absorbances at 492 nm of the solutions were read with a multiplate spectrophotometer reader (Multiskan EX Thermo Scientific). Fibroblasts cultured alone on TCP (48 well plate) were used as a positive control.
Statistical analysis
Fiber diameter size distribution of the scaffolds was assessed from image analysis performed onto SEM micrographs. For each sample, six micrographs were taken and each was studied by counting at least fifty fibers per region (n = 300). The distribution diameter size and orientation of fibers was estimated with the aid of ImageJ software and its plugins. Mean diameter ± standard deviation (SD) were also provided.
Results and discussion
Hydrolysis of gellan gum and synthesis of its alkyl derivatives
The alkaline hydrolysis of Gelzan®[38], occurred at pH 13, and the product was involved in the generation of its tetrabutylammonium salt, GG-TBA. A previous activation step with 4-NPBC allowed the functionalization with different alkylamines on the polysaccharide backbone [39].
Here, the GG-alkyl functionalized derivatives GG-C8 and GG-C12 (obtained by reaction of GG with octylamine and dodecylamine, respectively) show a functionalization degree in C8 and C12 equal to 17 mol% and 18 mol%, respectively.
The derivatization with aliphatic chains affected the behaviour of the polymer, i.e. its ionotropic sensitivity, solubility, and processability. The effect was attributed to the intramolecular interactions created by the aliphatic chains, which interfered with the typical transition random coil-to-helix of GG, as already previously discussed [39]. The hydrophobic aggregation is related to the length of the aliphatic chains and the degree of derivatization (DD) of the polymer. SEC results showed a progressive Mw reduction of the derivatives (GG-C8, 94.2 kDa, PDI 1.73; GG-C12 91.5 kDa PDI 2.04) compared to GG sample (96.7 kDa, 2.6 PDI) [26], which depended to the length of the pendant alkyl chain. A reduced affinity for the aqueous environment was also noted for the derivatives. The chromatogram of the GG-C8 sample showed a broad peak of the polymer, and a shoulder peak, which suggested polymer could aggregate in the aqueous medium compared to GG and GG-C12, which showed single peaks (data not shown).
Settings of electrospinning parameters
Settings from aqueous dispersions
ES parameters from aqueous polymeric dispersions are reported in Table 1. The achievement of a uniform charged jet required a critical polymeric concentration, so initial PVA concentration was the principal value to set. The lowest processable concentration of PVA (80 kDa) in an aqueous solution was 6% w/v as limit value to allow the generation of proper entanglements and a continuously charged jet [40]. The first goal was to produce blends of GG or its derivatives (C8 and C12) with PVA in a weight ratio equal to 1:1. Concentration of polymeric blend GG/PVA higher than 5% w/v do not produce membranes due to their high viscosity (data not showed). Considering the ability of GG-C12/PVA based aqueous blend to generate Taylor’s cone, the polysaccharide concentration was further reduced to 4% w/v. Distance, flow rate, and applied voltage were properly tuned until the reported conditions (Table 1) for each blend.
Optimized electrospinning conditions in deionized water.

The inability to process the GG/PVA mixture was highlighted; in fact, the SEM micrograph relating to the stated parameters showed globular stains coming from the dripping needle (Figure 1(a)). The morphology was irregular and short fibers with blobs were deposited randomly on the collector.

SEM micrographs of the electrospun mats from aqueous solutions. (a) GG/PVA 5% w/v; (b) GG-C8/PVA 5% w/v; and c) GG-C12/PVA 4% w/v. Red arrows indicated globular stains from dripping needle.
A similar study was carried out for the dispersion with the alkyl derivatives of GG. The achievement of optimal spinning conditions from GG-C8/PVA blend (Table 1) was confirmed by SEM micrographs (Figure 1(b)) showing a pore size of 1.5 ± 0.6 µm (ImageJ software). It displayed a homogeneous matrix composed by randomly deposited long nanofibers instead of unregular and segmented structures obtained at greater distances, voltages, and flow rate in other tests. The main parameter which governed the integrity and the diameter of these fibers was supposed to be the applied voltage. Figure 2 provides the fiber diameter distribution of the mats achieved by electrospinning aqueous solutions of GG-C8 (5% w/v) at 15 and 20 kV. Although both levels of applied voltage gave rise to nanofibrous mats characterized by unimodal distributions with maxima centered at 200 nm, when the applied tension was increased to 20 kV, a narrower and symmetric distribution was observed (Figure 2 blue bars), with 30% fibers displaying diameter of 200 nm. Notably, under this condition it is possible to achieve mats having more than 50% fibers with diameters below 200 nm, with obvious advantages in terms of specific area. Moreover, 66% fibers displayed diameters between 175 and 225 nm, with this exceptional uniformity being particularly suitable for all of those applications that require a high control of materials structure and architecture, such as in the case of device design for a predictable, controlled drug release. By contrast, at lower values of applied voltage a wider range was detected, with half fibers displaying diameters in the range 200-350 nm. In the case of GG-C12/PVA 4% w/v, all tests, including those performed by using the optimized conditions reported in Table 1, did not result in any efficient deposition of nanofibrous matrices (Figure 1(c)).

Dimensional distribution of nanofibers for electrospun mats of GG-C8/PVA 5% w/v in deionized water at 20 kV (blue bars) and 15 kV (red bars).
Settings from binary solvent/non-solvent dispersions
The use of 95:5 v/v deionized water/ethanol mixture increased spinnability of the gellan gum derivatives as reported previously in a similar work for other polysaccharides [6]. The mixture, relying on a vapor pressure higher than water, is able to quickly evaporate during ES. However, the volume ratio was selected to avoid the complete gelation of the polymers in the dispersion. On this light, it is a common strategy to promote the formation of stable entanglements despite a reduction of blend conductivity. Optimized instrumental parameters are reported in Table 2. SEM micrographs showed no improvements in terms of spinnability, and no fibers could be detected for GG/PVA and GG-C12/PVA blends in stated conditions (Figure 3(a) to (c)). In contrast, GG-C8/PVA 5% w/v dispersion (Figure 3(b)) generated a dense fibrillar matrix with a pore size of 1.8 ± 0.7 µm. The parameters reported in Table 2 for this latter, enabled the complete evaporation of both solvents for sample tested. The size distribution of nanofibers, provided in Figure 4, showed larger diameters of nanofibers if compared with those obtained from aqueous polymeric blend.
Ultimate electrospinning conditions in mixture water: ethanol 95:5 v/v.


SEM micrographs of the electrospun mats from water:ethanol 95:5 v/v mixture. (a)GG/PVA 5% w/v; (b) GG-C8/PVA 5% w/v; and (c) GG-C12/PVA 4% w/v. Red arrows indicated globular stains.
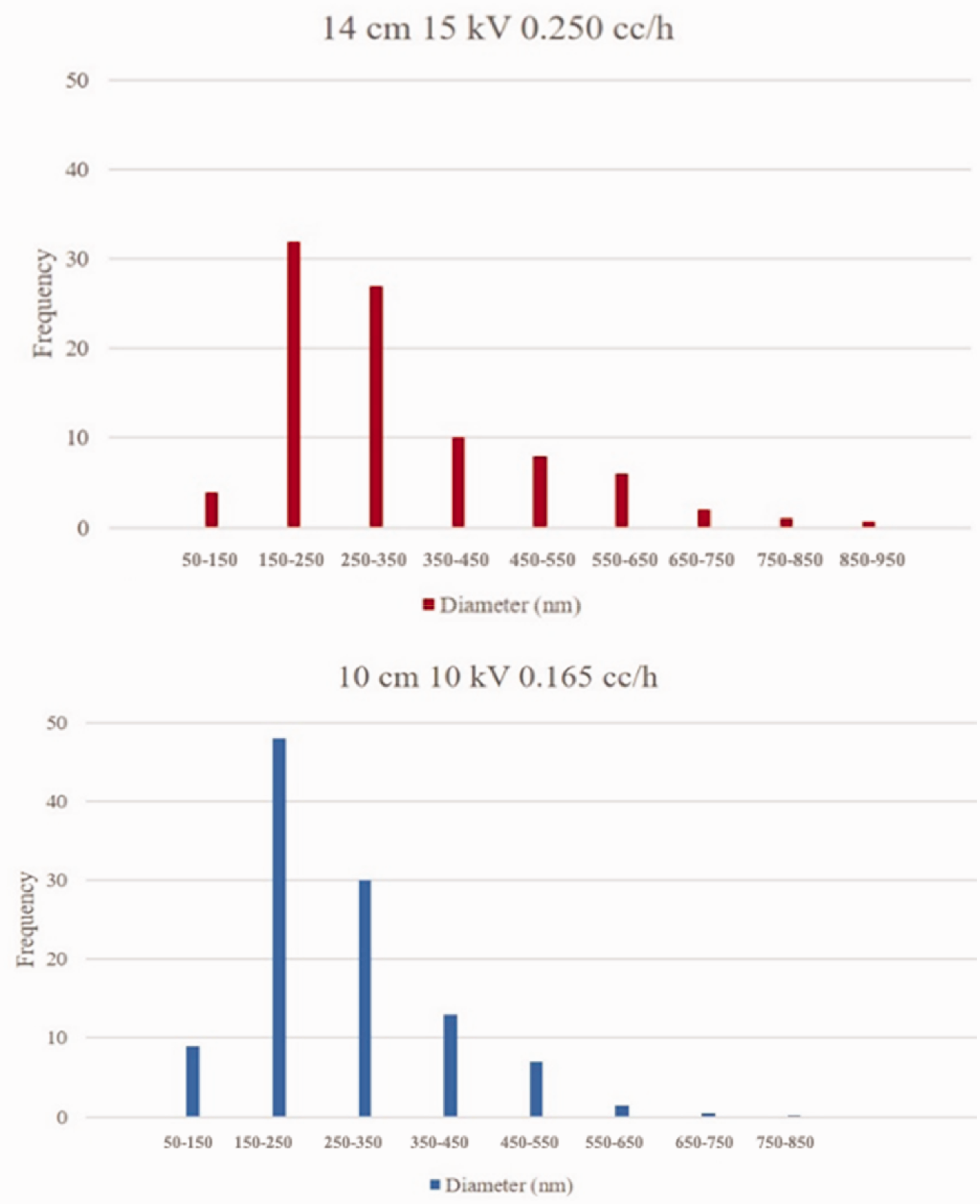
Dimensional distribution of nanofibers for electrospun mats of GG-C8/PVA 5% w/v from aqueous (red bars) and the binary mixture deionized water: ethanol 95:5 v/v (blue bars).
The analysis of the size distribution of the fibers in the samples obtained under these conditions still shows unimodal behaviour, although wider and with generally larger fiber diameters than those electrospun from aqueous solutions. Notably, using different combinations of solution concentration, applied voltage, and needle-to-collector distance, while ensuring electrospinnability, allows tuning maximum frequency of distribution, mean diameter and degree of homogeneity/heterogeneity of the fibrous mats, thus expanding the range of possible applications of such materials.
Viscoelastic behaviour of the polymeric dispersion in the binary mixture
The viscosity and gelation of GG are influenced by its molecular weight, temperature and ionic strength of the solution. The presence of divalent cations must be taken into account. The alkyl derivatives of gellan gum are characterized by an additional associative effect, whose extent monotonically increases as a function of the length of the pendant chain, as demonstrable by pyrene assays [41]. The hydrophobic aggregation interfered with the coil-to-helix transition of the GG, also influencing the aggregation of coils in junction areas. Rheological properties of the polymeric dispersion in water/ethanol mixture are reported in Figure 5. The graph highlights differences between samples that can be related to their respective ES performances. The blends can be classified in terms of descending viscosity order as follows: GG-C8/PVA 5% w/v > GG-C12/PVA 4% w/v > GG/PVA 5% w/v, considering the whole frequency range. GG/PVA 5% w/v dispersion (black squares) showed an almost Newtonian behaviour (at the lowest frequencies analyzed), suggesting a lower density of interchain entanglements, that may have determined the observed poor processability of this biomaterial. Among the samples, GG-C12/PVA 4% w/v in water/ethanol (red dots) showed complex viscosity values higher than those of GG/PVA 5% w/v but still lower than those of GG-C8/PVA in the whole range of frequency detected. These data can be explained considering the stronger hydrophobic aggregation of the GG-C12 derivative in the GG-C12/PVA blend, exalted by the non-solvent, which causes a more intense chains aggregation and a reduced interchains entanglement with respect to the blend with the GG-C8 derivative. In fact, this latter (blue triangles) showed the highest value of complex viscosity (Figure 5). The addition of ethanol favored tangles between different polymer chains with positive effects on processability, as discussed before. The addition of non-solvent has been shown to positively influence the properties and morphology of the mats, compared to samples treated with water, and allowed to modulate the architecture of the fibers, intended as size distribution and orientation, of the mats.

Flow curves of gellan gum and its derivatives in water:ethanol 95:5 v/v at 25°C.
Physical and biological characterization of GG-C8 based mats
A comparison of fiber orientation obtained from GG-C8/PVA 5% w/v in water and water:ethanol 95:5 v/v mixture was obtained from SEM micrographs and reported in Figure 6.


ATR-FTIR spectra: comparison of electrospun GG-C8/PVA membranes obtained from deionized water (black line) or water:ethanol 95:5 v/v mixture (red line).
From Figure 6, we can conclude that the small addition of ethanol to the electrospinning blend GG-C8/PVA (Figure 6(b)) allowed a more homogeneous random deposition of fibers on the collector than the sample obtained from the deionized water alone. ATR-FTIR spectra of electrospun membranes based on GG-C8/PVA from both investigated solutions resulted similar, as clearly visible in Figure 7.
In particular, PVA absorptions cannot be distinguished from GG-C8 absorptions due to chemical similarity of functional groups. Bands at ∼3300 cm−1 can be assigned to OH stretching, peaks at ∼2936 and ∼2903 cm−1 are relative to symmetric stretching of CH2, peak at ∼1630 cm−1 can be attributed to C=O stretching of amide I of GG-C8, signals at ∼1416 cm−1 are attributed to bending of CH2 and at ∼1025 cm−1 resulted from C-O-C symmetric stretching vibration. A mild peak at ∼1142 cm−1 can be attributed to the coupled C-N and C-O stretching vibrations due to urethane linkage with the aliphatic amine.
Cell viability of fibroblasts cultured above the GG-C8/PVA membrane was reported in Table 3. After seeding and incubation (30 min), fibroblasts which did not attach to the membrane, were found in the culture plate; thus, in order to detect viability of fibroblasts growing in the GG-C8/PVA membranes, the cell-crown inserts were moved to new wells. Fibroblasts that were able to attach to the GG-C8/PVA membrane were viable and able to proliferate as demonstrated by the fact that after 7 days, the metabolic activity of NIH 3T3 cells reached values comparable to the control (cells growing on plate of culture).
Metabolic activity of NIH 3T3 fibroblasts growing on GG-C8/PVA membranes after 1, 3 and 7 days of culture.
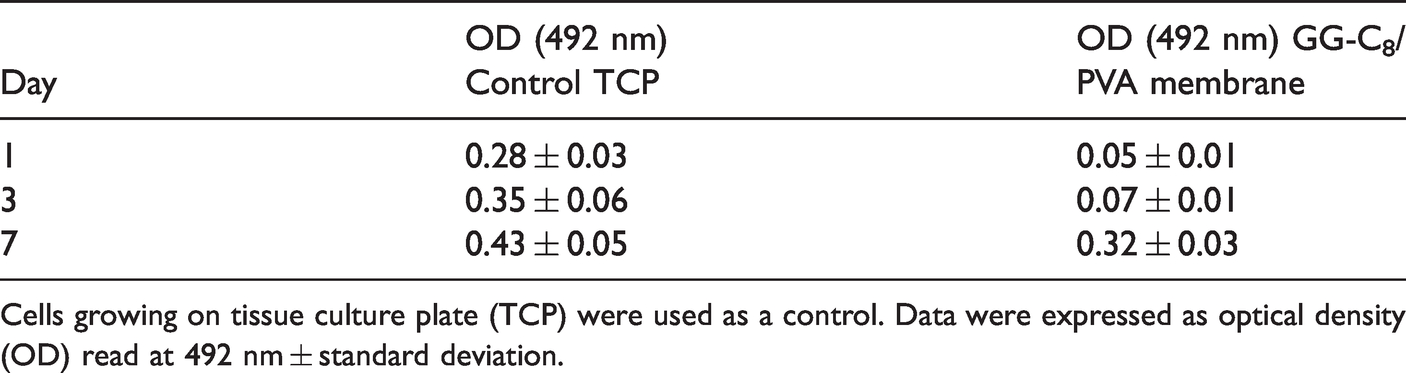
Cells growing on tissue culture plate (TCP) were used as a control. Data were expressed as optical density (OD) read at 492 nm ± standard deviation.
Conclusions
In this study, we prepared nanofibrous scaffolds based on alkyl derivatives of gellan gum which could find applications as biomedical devices or drug delivery systems. The influence of pendant aliphatic chains on gellan gum backbone on spinnability was investigated. In particular, the derivative GG-C8, blended with an equal amount of PVA, was more easily processed if compared to the GG-C12 derivative, obtaining fibers of 200 nm about. The use of a water:ethanol mixture, instead of water alone, improved spinnability and quality of GG-C8/PVA fibers. The spinnability of GG-C12 derivative was affected by more robust hydrophobic interactions which reduced polymer flexibility, thus exerting a negative effect. The MTS assay suggests that GG-C8/PVA membrane have optimal cytocompatibility.
The relationship between structural properties and processing conditions, elucidated in this work, could provide guidance for future advances in the fabrication of a new generation of fibrous polysaccharide-based biomaterials.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
