Abstract
Poly(butylene succinate) (PBSU) is a biodegradable and biocompatible synthetic aliphatic polyester, which has been used extensively in packaging, catering and agriculture, and more recently in drug delivery and bone and cartilage repair. PBSU-based mats created by electrospinning show promise as wound dressing materials because of their good mechanical properties, high surface area-to-volume ratio and increased levels of porosity. In this work, we present the creation of antimicrobial PBSU fibrous mats through the incorporation of natural food grade agents via blend electrospinning. Three types of edible gums (namely arabic, karaya and tragacanth), two essential oils (coriander and lavender), and one free fatty acid (linoleic acid) were added to PBSU containing a chain extender and their effect on six clinically relevant pathogens was evaluated. Mats containing essential oils at the highest concentration studied (7% w/v) showed some antimicrobial behaviour against S. aureus, E. hirae and P. aeruginosa, whereas the incorporation of linoleic acid at both concentrations tested (3% and 5% w/v) gave a strong reaction against S. pyogenes. Gum arabic was the only gum that had a considerable impact on S. aureus. Furthermore, the three gums enhanced the mechanical properties of the polymer mats and brought them closer to those of the human skin, whilst all agents maintained the high biocompatibility of the PBSU mats when contacted with mouse fibroblasts. This work, for the first time, shows the great promise of PBSU blended fibres as a skin substitute and paves the way towards bioactive and cost effective wound dressings from renewable materials.
Keywords
Introduction
Poly(butylene succinate) (PBSU) is a synthetic aliphatic polyester, which can be produced from biomass-based raw materials, as well as the traditional petrochemical route. Since its commercial introduction in 1993, PBSU has gained popularity in various areas, including packaging, catering and agriculture [1–3]. More recently, PBSU has found application in the biomedical field, particularly in tissue engineering and drug delivery [4–6]. Although not as widely researched as other aliphatic polyesters, e.g. poly(lactic acid) (PLA), poly(glycolic acid) (PGL) and poly(ε-caprolactone) (PCL) [7–12], it is considered a very promising biomaterial candidate, because in addition to its biodegradability and biocompatibility, it can be easily tailored to suit different applications [4]. PBSU copolymer-based nanoparticles have been used as drug delivery vehicles, whereas various scaffold configurations have been employed for bone and cartilage repair [13,14], as well as in wound healing [15]. A comparison of some aliphatic polyesters used in wound dressing and other biomedical applications can be found in Table S1.
Although there are various methods available for the production of nano- and micro-fibres [16], electrospinning is one of the most favoured techniques for the creation of tissue engineered skin scaffolds [17,18]. It can be cost-effective, scalable, fast, ‘green’ (depending on the processing solvent(s) employed), and can produce a wide variety of materials with tuneable properties, compositions, shapes and dimensions using a large array of polymers (natural and synthetic) [7,17]. The generated nanofibrous constructs can have controllable porosity, and, with their high surface area-to-volume ratio, can closely resemble the architecture of the natural extracellular matrix (ECM). These properties allow for cell proliferation, moisture retention, haemostasis and removal of exudates [18]. The ‘ideal’ skin dressing, however, should also have mechanical properties which resemble those of the human skin, to ensure patient comfort and allow for its easy handling and application [19,20]. PBSU constructs suffer from poor mechanical properties, and low biodegradation rate due to the polymer’s high propensity to crystallise [4,5]. Although co-polymerisation with different monomers has been explored to alleviate these problems [21–23], Fabbri and co-workers [4] reported a reduction in strength of their synthesised polymers. The presence of small amounts of diisocyanates, typically diisocyanahexane (DCH), as chain extenders can, however, reduce brittleness and increase the elongation at break up to values comparable to those of polyolefins [4,5].
Current research in wound management aims to create ‘smart’ or ‘bioactive’ materials through the incorporation of therapeutic and/or antimicrobial agents, in order to fulfil multiple parts of the wound healing process and ultimately produce personalised bandages for different types of wounds and surfaces [17,24]. These agents include, but are not limited to, antibiotics, peptides, nanoparticles (e.g. iron oxide, titanium dioxide, silver, etc.) and natural substances (e.g. curcumin, henna, chitosan, etc.) and the reader is referred to some extensive reviews already published on the subject [10,25–27]. In this work, extended PBSU (PBSU-DCH) has been electrospun into fibrous mats suitable for skin wound regeneration for the first time. Through blend electrospinning, natural substances in the form of edible gums (arabic, karaya and tragacanth), essential oils (coriander and lavender) and a free fatty acid (linoleic acid) have been incorporated into the PBSU-DCH fabrics. All of these natural additives have been shown to possess antimicrobial effects against a broad spectrum of Gram positive and Gram negative bacteria [28–31]. Furthermore, and compared to other antimicrobial agents, are easy to process, inexpensive, abundant and pose no risk of antimicrobial resistance [25,32] The biocompatibility, antimicrobial performance and handling properties of the resulting constructs have been tested to evaluate our novel fabrics for their potential use in the treatment of burn and chronic wounds.
Materials and methods
Materials
Poly(butylene succinate) extended with 1,6-diisocyanatohexane (PBSU-DCH), gum arabic, gum karaya, gum tragacanth, linoleic acid, coriander oil, lavender oil, Luria Bertani (LB), Mueller-Hinton agar medium, Dulbecco's Modified Eagle’s Medium (DMEM), Fetal Bovine Serum (FBS, γ-irradiated, sterile filtered), trypsin (0.25%)-EDTA and Trypan Blue, were purchased from Sigma-Aldrich Ltd (Gillingham, UK). Chloroform, dichloromethane, methanol and ethanol were acquired from Fisher Scientific (Loughborough, UK). 3T3-Swiss albino (ATCC® CCL-92™) mouse fibroblasts were obtained from the American Type Culture Collection (Manassas, VA, USA). Staphylococcus aureus, Methicillin resistant Staphylococcus aureus (MRSA), Escherichia coli, Pseudomonas aeruginosa, Enterococcus hirae and Streptococcus pyogenes, were isolated and classified at Aston University (Birmingham, UK).
Preparation of antimicrobial PBSU-DCH mats
Prior to incorporating the antimicrobial agents, a series of optimisation experiments was carried out to determine the best combination of polymer concentration, solvent mixture and electrospinning conditions. Samples of PBSU-DCH solutions (final concentrations of 12% and 14% w/w) were transferred to a 5 mL standard syringe fitted with a 20 G blunt stainless steel needle. High voltage (10, 15 and 20 kV) was applied (Genvolt, Bridgnorth, UK) and the fibres were collected on a grounded stainless steel plate covered with aluminium foil. Three different flow rates (1, 1.5 and 2 mL h−1) and tip to collector distances (TCD; 10, 15 and 20 cm) were investigated in order to identify optimum preparation conditions. The ambient temperature was ∼20°C and RH30%. The solvent mixtures tested were chloroform: methanol, chloroform: ethanol and dichloromethane: methanol, at 90:10, 80:20 and 70:30 solvent ratios.
Six natural antimicrobial agents were incorporated into the PBSU-DCH fibres during the electrospinning process, namely gum arabic, gum karaya, gum tragacanth, coriander oil, lavender oil and linoleic acid. For the edible gums (arabic, karaya and tragacanth), 6 g were dissolved in 60 mL of methanol (under continuous stirring for 16 h at 40°C), filtered to remove any residual solids and then placed in a fume hood until most of the methanol was evaporated, leaving 4 mL of concentrated gum solutions. Each solution was subsequently mixed with chloroform (90:10 chloroform: gum solution) and then used to dissolve PBSU-DCH (14% w/w) prior to electrospinning. In the case of linoleic acid and essential oils (coriander and lavender), they were added directly to the PBSU-DCH solution (14% w/w in 90:10 chloroform: methanol) at various concentrations (3, 5 and 7% w/v), stirred at 40°C until dissolved completely and then used for electrospinning. Following optimisation experiments, the electrospinning conditions for all antimicrobial fibrous mats were: 15 kV voltage; 1 mL h−1 flowrate; 20 cm tip to collector distance. The fibrous mats were subsequently placed in a convection oven at 60°C for 20-30 min (SciQuip Oven-80 HT, Newtown, UK) to evaporate the solvent residues, before being transferred to a desiccator (Nalgene™, ThermoFisher Scientific, Loughborough, UK) to cool down. Controls, in the form of PBSU-DCH films containing antimicrobial agents, were prepared by casting the final mixtures into glass Petri dishes and drying at RT. The fibrous mats and films were cut into squares (8 x 8 mm2), sterilized by UV illumination (365 nm, 15 Watts) using a Chromato-vue C71 light box and UVX radiometer (UV-P Inc., Upland, CA, USA) for 2 h and then placed in sterile closed containers prior to the antimicrobial and cytotoxicity assays.
Characterisation of fibrous mats
Fourier transform infrared (FTIR) spectroscopy was performed with a PerkinElmer Frontier™ FT-IR spectrometer (PerkinElmer Inc., Waltham, MA, USA) over the range of 4000–600 cm−1 with a resolution of 4 cm−1 within 16 scans.
The mechanical properties of the antimicrobial PBSU-DCH mats (8 mm gauge length; 0.13 mm average thickness; n = 5) were measured using a Hounsfield 10 K Tensometer (Tool and Gauge Co, Birmingham, UK) at room temperature with a stress range (50 MPa), an extension range (300%) and a test speed of 20 mm min−1 with samples taken to failure. PBSU-DCH fibrous mats without antimicrobial agents were used as controls.
The morphology of the PBSU-DCH mats, as well as those containing gum arabic and linoleic acid 3% (w/v), was examined by a Philips XL-30 FEG Environmental Scanning Electron Microscope (FEI Company, Hillsboro, OR, USA) operated at 10.0 kV. Prior to imaging, the fibrous mats were sputter coated in gold or platinum. Fibre diameters were measured using ImageJ software (National Institutes of Health, Bethesda, MD, USA) and reported as an average value of ∼100 independent measurements. ImageJ was also used to determine the average porosity of the mats containing gum arabic and linoleic acid (i.e. the mats with the best antimicrobial behaviour). Grayscale SEM images (see Figure S1) of different magnifications (1500×, 5000× and 10,000×) were analysed and the average value was calculated from 4 measurements [24,33].
Antimicrobial activity assay
The agar disc diffusion method was employed for the determination of the antimicrobial properties of the PBSU-DCH fibrous mats and films. First, the bacterial isolates (S. aureus, MRSA, E. coli, S. pyogenes, P. aeruginosa, and E. hirae) were transferred from frozen glycerol stock solutions to Trypticase Soy Agar (TSA) plates (supplemented with 5% v/v horse blood for S. pyogenes) and incubated for 24 h at 37°C in aerobic conditions. A single colony was then transferred to 5 mL LB broth medium and incubated for 24 h at 37°C. Mueller-Hinton agar plates were inoculated with the bacterial suspension (∼1 x108 CFU mL−1) and left to dry for 5 min before adding the sterile mats. The plates were incubated for 24 h at 37°C in aerobic conditions, before measuring the zone of inhibition around each sample.
Cytotoxicity assay
3T3-Swiss albino mouse fibroblasts were cultured in Dulbecco’s Modified Eagle’s Medium supplemented with 10% FBS, up to 50% confluence in a Heracell 150i CO2 incubator (Thermo Scientific, Fisher Scientific, Waltham, MA, USA) in a controlled environment (5% CO2, 37°C) prior to initiating the cytotoxicity assay.
The cytotoxic effect of the antimicrobial fibrous mats was assessed using the ‘direct contact’ method, as well as the ‘extract’ (or ‘indirect’) test. The former method evaluates the effects of the physical interaction between the support and the cells, whereas the latter looks at the influence of potential leachable components. Cells cultured in the absence of fibrous mats were used as a negative control, and addition of 70% (v/v) ethanol acted as a positive cytotoxic response. Cells were seeded in sterile 24-well plates at a concentration of 6 × 105 cells mL−1 and incubated at 37°C and 5% CO2 for 24 h until a cell monolayer was formed. For the direct contact test, and following monolayer formation, the medium was discarded and replaced with fresh medium (2 mL), and the fibrous mats were placed on top of the cells and left to incubate for 24 h. For the indirect test, fibrous mats were added to sterile 24-well plates containing 2 mL culture medium and left for 24 h. This liquid was then used to replace the growth medium over the cell monolayer, followed by a further 24 h incubation. After incubation, 1 mL of trypsin-EDTA was added in each well to aid cell detachment. Trypsin was then inactivated by the addition of 2 mL of fresh culture media. The cell suspension was centrifuged at 220 g for 5 min, the supernatant discarded and the pellet re-suspended in 1 mL of culture medium. Cell viability was checked with Trypan blue exclusion assay and cells were counted using a haemocytometer and an Olympus CK2 Inverted Microscope (Olympus Corporation, Tokyo, Japan).
Results and discussion
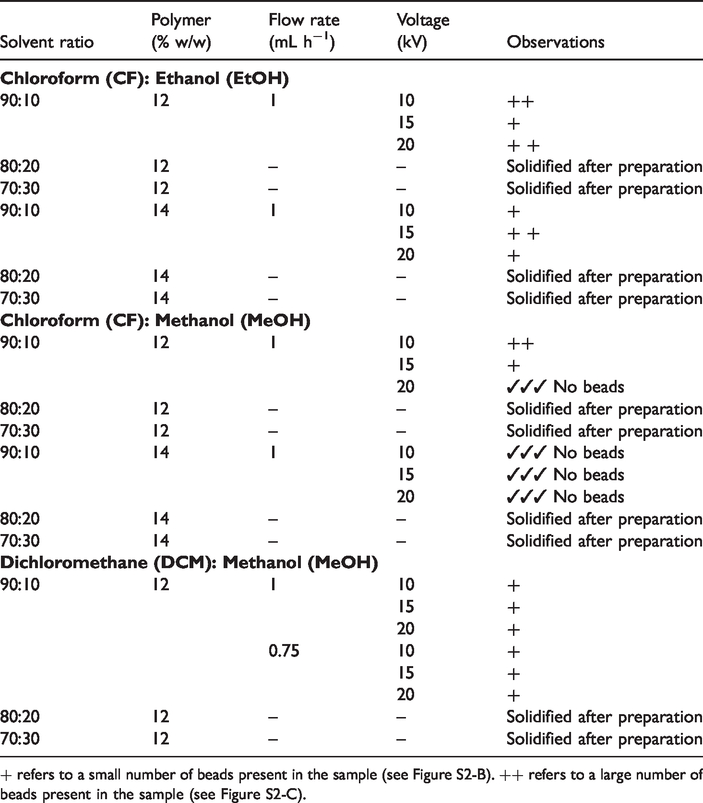
Following an initial screening experiment, the optimum combination of solvent and electrospinning conditions for the production of bead-free PBSU-DCH mats (in the absence of antimicrobial agents) was identified (see Table 1 and Figure 1), and subsequently used for the creation of the antimicrobial fibres (see Figure 2).
Optimization of the PBSU-DCH electrospinning parameters.
+ refers to a small number of beads present in the sample (see Figure S2-B). ++ refers to a large number of beads present in the sample (see Figure S2-C).

Scanning electron microscopy images and corresponding fibre diameter distributions for electrospun PBSU-DCH mats (from chloroform: methanol 90:10) using different tip-to-collector distance (cm), flow rate (mL h−1), and voltage (kV). (a) 20 cm, 1 mL h−1, 10 kV; (b) 15 cm, 1 mL h−1, 10 kV; (c) 10 cm, 1 mL h−1, 10 kV; (d) 20 cm, 1.5 mL h−1, 10 kV; (e) 20 cm, 2 mL h−1, 10 kV; (f) 20 cm, 1 mL h−1, 15 kV.

Schematic of the preparation process of PBSU-DCH antimicrobial fibrous materials.
From Table 1, it can be seen that PBSU-DCH dissolved in 90:10 chloroform: methanol at a 14% (w/w) concentration, was easy to electrospin and gave consistently bead-free fibres [34,35], whereas lowering the concentration to 12% (w/w), resulted in a less reliable process. It was also noted that the other two solvent systems tested led to either bead formation or non-spinnable solutions due to solidification of the sample prior to electrospinning.
Additional investigation into the effect of flow rate, voltage and tip to collector distance on the fibre diameter showed that increasing the flowrate and/or reducing the TCD has a negative impact on fibre homogeneity (Figure 1) [36,37]. Therefore, the optimum conditions for this study were revealed as: 1 mL h−1 flow rate; 20 cm TCD; 15 kV; chloroform: methanol (90:10) as solvent; PBSU-DCH concentration 14% (w/w). Further process optimisation involving the use of, for example, a greener solvent system is currently under way in our laboratories.
Chemical composition of antimicrobial PBSU-DCH fibrous mats
FTIR spectroscopy has been used to elucidate the presence of the functional groups in the mats, and to reveal the effect of the preparation procedures and electrospinning on the chemical functionality of the antimicrobial agents.
FTIR spectra showed clear differences in the concentration of expected functional groups between the PBSU-DCH control and the mats containing linoleic acid, gum arabic and essential oils (7% w/v) (see Figure 3), whereas these differences are far less prominent in the remaining mats (see Figure S3). The differences arise from the active components present in the natural antimicrobials (see Figures S4-S9).

FTIR spectra for PBSU-DCH mat containing (a) linoleic acid (5% w/v), (b) gum arabic, (c) coriander oil (7% w/v), (d) lavender oil (7% w/v) (red line), compared to the control PBSU-DCH mat (black line).
FTIR spectra for the mats containing edible gums revealed an increase in the absorption bands. Most notably, increases were observed between 2900-3000 cm−1 related to C-H stretching (methylene group); at 1720 cm−1 due to C = O stretching (carbonyl, acetyl and carboxylic groups); between 1300-1500 cm−1 due to C-H bending (methyl, methylene and aldehyde groups), O-H bending (carboxylic acids) and COO asymmetric stretching (carboxylate); between 1160-1210 cm−1 due to C-O stretching (ester); at 1050 cm−1 is a complex bands due to C-O stretching (starch content) and C-O-C asymmetric bridge stretching; and below 900 cm−1 due to the vibration of the pyranose ring (Figure S8; [38–40]).
Linoleic acid-based mats (3% and 5% w/v) showed the most significant changes among the antimicrobial mats when compared to the control. Similar differences in the FTIR data were observed to those of the edible gums, in addition to a new peak at 800 cm−1 and an increase in the bands between 600-100 cm−1 due to C = C bending [41].
Moving to essential oils, the main components of their chemical structure include linalool, linalyl acetate, aliphatic aldehydes and aromatic compounds. (see FiguresS6 and S7). Accordingly, the following differences in their FTIR spectra were observed at: 2900–3000 cm−1 due to C-H stretching (alkene); 1700–1800 cm−1 due to aromatic compounds; 1300–1400 cm−1 due to O-H bending (phenol groups); 1160–1300 cm−1 due to C-O stretching (aliphatic and aromatic esters) and 800–1000 cm−1 due to C = C bending (Figure S9; [42,43]). These changes increased as the oil concentration was increased in the fibrous mats.
Aside from the additional bands arising from the presence of the functional groups of the edible gums and essential oils in the FTIR spectra, it is notable that the broad band in the region of 3000-3700 cm−1 relating to O-H stretching (observed in the FTIR spectra of the antimicrobial agents alone; see Figures S8 and S9) is not present in any of the samples. This is attributed to esterification occurring between the carboxylic acid functionality in the antimicrobial agents and the methanol used in the preparation of the PBSU-DCH spinning solution. This was further confirmed by the increase in the absorption bands at 1180 cm−1 due to C-O stretching (aliphatic and aromatic esters).
Mechanical properties
An important requirement for the ‘ideal’ wound dressing is possessing mechanical properties that are similar to those of the human skin [19,20]. The dressing can then aid effective wound healing, but also be easily handled and manipulated.
Figure 4 shows the mechanical properties of the PBSU-DCH mats with and without antimicrobial agents, and representative stress-strain curves can be found in Figures S10 and S11. Typical Young’s modulus values reported in the literature for normal human skin are 4.6-20 MPa, based on tensile tests at quasistatic speeds (6–54 mm min−1) and are highly dependent on the body part, age, skin colour and genetic heritage [44,45]. The Young’s moduli for all of our materials (measured at 20 mm min−1) were between 1.2 and 3.11 MPa, which indicates that they are less stiff than human skin, with less resistance to deformation.
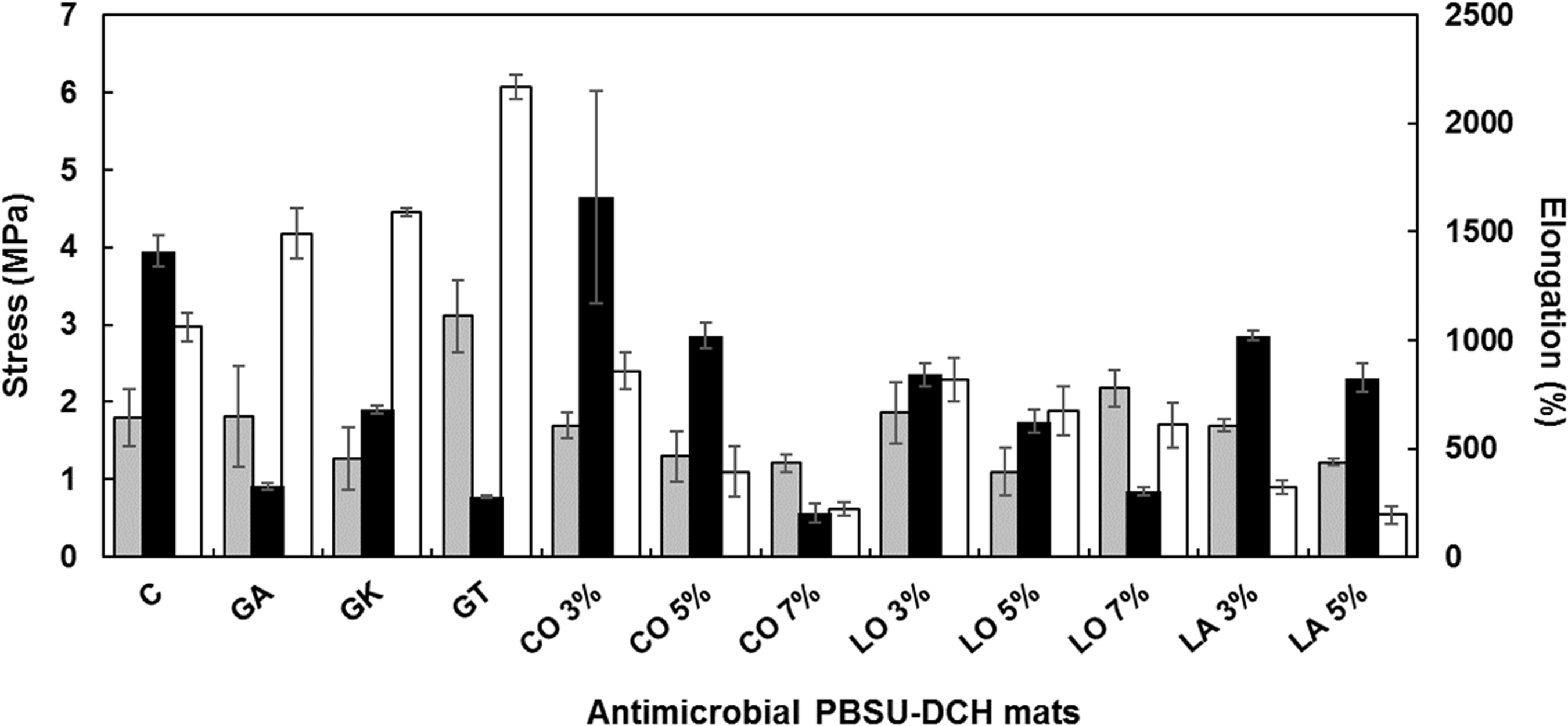
Mechanical properties of the antimicrobial PBSU-DCH fibrous mats (± 1 standard deviation). Grey bars – Young’s modulus; Black bars – Elongation at break; White bars – Tensile strength. Key: C = Control (PBSU-DCH mat); GA = gum arabic; GK = gum karaya; GT = gum tragacanth; CO = coriander oil; LO = lavender oil; LA = linoleic acid.
The presence of the gums increased the strength of the PBSU-DCH mats (stress at failure of 3.3-7.8 MPa for all gums, compared to 0.2 MPa for the PBSU-DCH control), and made them more resistant to failure (as indicated by the increase in ultimate tensile strength from 2.97 MPa for the control, to 4.18, 4.4 and 6.07 MPa for the gum arabic, gum karaya and gum tragacanth, respectively). Incorporation of the gums also led to a considerable reduction in their elongation at break, thereby resulting in less elastic materials, whereas gum tragacanth enhanced the stiffness of the mat, measuring the highest Young’s modulus amongst all samples tested, producing a fabric with mechanical properties much closer to the most elastic types of human skin. Arabic, karaya and tragacanth gums are well known thickening agents in the food industry and have been used extensively to modify the rheology of different foods [46]. The addition of these highly branched hydrocolloids (see Figure S4) therefore led to the strengthening of the mat structure, whilst simultaneously restricted their ability for plastic deformation due to the enhanced non-specific entanglement of the disordered polymer chains [46,47].
The incorporation of the essential oils or free fatty acid did not appear to have a major impact on the stiffness or hardness of the mats, but did affect their ductility. In all but one case, the elongation at break was less than the PBSU-DCH mat without additives. The coriander oil at 3% (w/v) concentration was the only sample that showed a tendency to undergo larger plastic deformation before breakage than the control, however it is not possible to draw definitive conclusions without further investigations, due to the high standard deviation associated with these measurements (see Figure 4).
Antibacterial activity of the fibrous mats against common skin pathogens
The effect of the natural antimicrobial components on pathogens commonly found in wound infections, was evaluated by agar disc diffusion. The six different antimicrobial mats (gum arabic, gum karaya, gum tragacanth, coriander oil, lavender oil and linoleic acid) were tested against Gram positive S. aureus, MRSA, S. pyogenes and E. hirae, and Gram negative E.coli and P. aeruginosa, and the results are summarised in Table 2. The above bacteria were chosen because they are routinely isolated from burn wounds. More specifically, Streptococcus, Staphylococcus and Enterococci spp. colonise the wound very soon following injury, whereas E. coli and Pseudomonas spp. are associated with secondary infections following hospitalisation [48,49].
Antimicrobial efficacy of PBSU-DCH fibrous mats against six clinically relevant pathogens, expressed as sizes of zone of inhibition (radii in mm) observed following direct application.
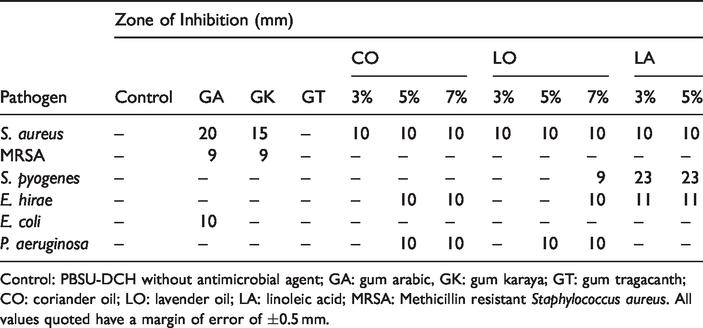
Control: PBSU-DCH without antimicrobial agent; GA: gum arabic, GK: gum karaya; GT: gum tragacanth; CO: coriander oil; LO: lavender oil; LA: linoleic acid; MRSA: Methicillin resistant Staphylococcus aureus. All values quoted have a margin of error of ±0.5 mm.
The PBSU-DCH mats containing gum arabic and gum karaya showed a significant antimicrobial effect against S. aureus and to a lesser extend MRSA, but had no impact on S. pyogenes, E. hirae and P. aeruginosa. Gum arabic also inhibited the growth of E. coli. Our results partially agree with other studies, which have demonstrated activity of gum arabic and gum karaya extracts against some Gram positive and Gram negative bacteria [50–53]. At the same time, we observed no effect in the presence of gum tragacanth for all bacteria species tested, which contradicts what has been claimed by Padil et al. [28] and Ranjbar-Mohammadi et al. [54]. Discrepancies between our results and others’ can be attributed to the difference in the gums’ final composition, which is influenced by the country of origin, plant age, plant part (e.g. leaves, bark, etc.) and process used to obtain the extractants, i.e. the use of aqueous or organic solvents [55]. Although combinations of edible tree gums with polymeric materials or metal/metal oxide nanoparticles have been used successfully for biomedical applications, including wound dressings [28,54], their usefulness as antimicrobial agents hasn’t been firmly established yet, which highlights the importance of additional investigations such as our work.
The incorporation of coriander and lavender oils equipped the mats with very similar antimicrobial characteristics, which can be attributed to the presence of common compounds such as linalool, camphor, borneol and limonene (see Table 3). The increase in essential oil concentration did not seem to have an effect in the case of S. aureus, but there was an indication of improved antimicrobial properties against E. hirae. Furthermore, coriander essential oil failed to produce any antimicrobial effect against S. pyogenes, which is in agreement with Silva and co-workers who found that most Gram-positive bacteria are less sensitive to coriander essential oils than Gram-negative bacteria [58]. In contrast, lavender oil is more potent against streptococci [59], as demonstrated by the 9 mm inhibition zone around S. pyogenes. Neither coriander nor lavender essential oils supressed the growth of MRSA and E. coli, despite contrasting reports in the literature [29,30,57,60]. One possible reason for this unexpected result could be the source of the oils. Similar to the edible gums, the chemical composition of the essential oils is highly dependent on the part of the plant they are extracted from (e.g. for coriander it can be the leaves, the immature plant (cilantro), or the seeds), and varies enormously between the different geographical locations (see Table 3 and associated references). This, combined with the fact that they are complex mixtures of more than 300 compounds in some cases [61], makes it difficult to carry out exact comparisons between reported data across the world.
The addition of linoleic acid to the PBSU-DCH fibrous mats exerted highly potent activity against S. pyogenes (23 mm), and moderate suppression of S. aureus (10 mm) and E. hirae (11 mm). Medium and long chain polyunsaturated fatty acids have been shown to be more active against Gram positive bacteria than Gram negative bacteria and their antibacterial properties are influenced by their structure and shape [62,63]. In general, it appears to be a direct correlation between the number of double bonds and their isomerism (preferably cis) in the carbon chain of an unsaturated fatty acid and its antibacterial efficacy, and therefore fatty acids, such as γ-linolenic, eicosapentaenoic and the linoleic used in this study (an 18-carbon chain with two double bonds in cis configuration; see Figure S5), are more potent compared to, for example, elaidic acid [31,62].
Nanoparticles are currently considered the most promising alternatives to antibiotics and have already entered the market within various sectors, such as healthcare, consumer goods, food and construction (e.g. Biomaster Antimicrobial technology, Microban, etc.). Although most natural compounds are not as effective, they can elicit a considerable antimicrobial response, as we demonstrated in our work, with the additional lack of potential toxicity frequently associated with nanoparticle use [25,32].
Finally, and in order to investigate the influence of the morphology of the fibrous mats on their efficacy in releasing the antimicrobial agents, antimicrobial PBSU-DCH films were also tested with the six pathogenic bacteria, using the agar disc diffusion method. None of the films showed antimicrobial behaviour towards the pathogens. This is more probably due to the diffusion limitations associated with the film configuration. Electrospun mats have an open structure and therefore higher porosity compared to films, which makes them ideal supports for wound dressings and facile release of bioactive substances, such as anti-inflammatory and antimicrobial agents [17].
Biocompatibility of the fibrous mats
The effect of the chain extended PBSU, as well as the toxic solvents (i.e. chloroform and methanol) employed in the electrospinning of PBSU-DCH on cell viability was assessed, in order to confirm the biocompatibility of the fibrous mats [5,14] (see Table S2). In addition, it has been shown that essential oils at high concentrations can be cytotoxic, due to their lipophilic nature, which causes damage to the plasma membrane, and therefore have been used to kill cancer cells [64,65]. Indirect cytotoxicity tests, using the extraction media, following 24 h of contact with the scaffolds, showed 100% biocompatibility with mouse fibroblasts in all cases. Direct cytotoxicity studies also confirmed the creation of cell-friendly surfaces, resulting in 94-100% viable cells for the PBSU-DCH and antimicrobial mats. These results are in agreement with the well documented biocompatibility of the edible gums [28], whereas the concentration of the essential oils employed in this study, is far below the cytotoxic thresholds reported elsewhere [66].
Effect of antimicrobial additives on PBSU-DCH fibre morphology
Surface topography, and in particular pore size, of tissue engineering scaffolds is a very important factor in the promotion of cellular adherence, differentiation and proliferation [67], and therefore the effect of the addition of gum arabic and linoleic acid (the two compounds that gave the most promising antimicrobial responses) on the PBSU-DCH fibre morphology was further investigated.
Incorporating gum arabic into the electrospun mats significantly reduced fibre diameter and also led to better homogeneity (see Figures 1(f) and 5(a)). This was an expected outcome, since gum arabic is a negatively charged gum [68] and its presence in the mixture leads to increased charge on the surface of the droplet to form Taylor cone and a decrease in fibre diameter [69]. In contrast, the linoleic acid-containing PBSU-DCH fibres were less uniform compared to the control, resulting in a significantly wider diameter range (see Figures 1(f) and 5(b)).

SEM & fibre diameters for fibrous mats created from: (A) PBSU-DCH with gum arabic; (B) PBSU-DCH with 3% (w/v) linoleic acid. Electrospinning conditions: 1 mL h−1 flow rate; 20 cm TCD; 15 kV; chloroform: methanol (90:10 ratio) solvent; PBSU-DCH concentration 14% (w/w).
Similar results in terms of the effect of the incorporation of natural compounds on polymer fibre morphology have been reported elsewhere. For example, through the addition of cinnamon extract to PCL fibres, Ahmed and co-workers created antifungal bandages comprising thinner and more homogeneous fibres compared to ‘plain’ PCL [26]. It is worth noting however, that the increase in fibre diameter for the mats containing linoleic acid was mirrored by enhanced porosity (32.6 ± 1.5% c.f. 21.7 ± 1.3% for those containing gum arabic), reaching values which are closer to the open structures (>30%) suitable for wound healing applications [15,24].
Conclusions
Six different food grade antimicrobial agents were successfully incorporated into PBSU-DCH mats using blend electrospinning. The addition of edible gums rendered the mats mechanical properties, in terms of Young’s modulus and ultimate tensile strength, closer to human skin but compromised their ductility. Of the different food grade agents, linoleic acid and to a lesser extend gum arabic, demonstrated the highest antimicrobial activity against three out of the six pathogens tested, whereas gum tragacanth and low concentrations of the two essential oils showed no effect. The gum arabic also improved the homogeneity of the fibres, which in turn contributed towards the creation of stronger fibrous mats. Furthermore, the presence of the additives didn’t compromise the biocompatibility of the PBSU-DCH mats.
Based on our results, a combination of two or more of the natural agents tested has the potential to fulfil the different requirements of the wound healing process and therefore merits further investigation, as is the use of other fibre configurations, such as core-sheath, to aid the controlled delivery of the active components [70].
Supplemental Material
sj-doc-1-jit-10.1177_1528083720987209 – Supplemental Material for Poly(butylene succinate) fibrous dressings containing natural antimicrobial agents
Supplemental Material, sj-doc-1-jit-10.1177_1528083720987209 for Poly(butylene succinate) fibrous dressings containing natural antimicrobial agents by Kinana Aliko, Mohamad Basel Aldakhlalla, Laura J Leslie, Tony Worthington, Paul D Topham and Eirini Theodosiou in Journal of Industrial Textiles
Footnotes
Acknowledgement
We thank Mr Joe Homer for the FTIR spectroscopy of the edible gums and the essential oils.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the UK Engineering and Physical Sciences Research Council: Bridging the Gaps between Engineering and Physical Sciences in Antimicrobial Resistance as part of the UK Cross-Research Council Initiative on Antimicrobial Resistance Grant No. EP/M02735X/1.
Supplemental material
Supplemental material for this article is available online.

