Abstract
Due to the effects of climate changing, the importance of outdoor thermal comfort has been recognized, and has gained more and more research attentions. Unlike indoor space where air conditioning can be easily implemented, outdoor thermal comfort can only be achieved by localized thermal management. Using textile is a simple but energy-saving way to realize outdoor thermal comfort. Herein, we report the design of a smart thermal management film with the silver/vanadium dioxide/silver (Ag/VO2/Ag) sandwich structure prepared by one-dimensional (1 D) nanowires. It was found that the Ag/VO2/Ag sandwich film was able to lower the temperature by around 10 °C under intense infrared (IR) radiation. In addition, the Ag/VO2/Ag sandwich structure film showed a thermo-responsive electrical conductivity and an outstanding bending stability, due to network structure formed by nanowires. It was experimentally proved that this sandwich structure was superior to other layer structures in IR shielding performance and thermo-responsive electrical conductivity. The as-prepared Ag/VO2/Ag sandwich structure film has great potential for various applications such as wearable devices, flexible electronics, medical monitors and smart IR radiation management.
Introduction
Human exposure to excessive heat stress causes photo-ageing, heat stroke and even cancer when people performing outdoor activities [1]. Outdoor thermal management is therefore crucially needed in many situations for human health, thermal comfort and energy saving, but achieving it still remains a big challenge [2]. It is impractical and uneconomical to cool the enormous open space in the outdoor environment using energy intensive heating/cooling, ventilation, and air conditioning systems. On the contrary, textiles are able to provide a simple and energy-saving thermal management solution to realize localized outdoor thermal comfort [3,4]. Previous studies on thermal management textiles introduced forced liquid and air circulation for thermoregulation [5]. However, this kind of thermal management textile relies on the energy supply, making the textile cumbersome and energy consuming [6]. It has been a challenge for researchers to design and develop a textile that can self-regulate and efficiently modulate solar radiation (especially IR radiation, which causes thermal effect significantly) [7–9].
The intensive developments of smart materials and devices have opened up a new horizon to overcome this challenge. These smart materials and devices are able to respond to external stimuli by changing physical and chemical properties including colors, electrical conductivity, shapes and so on [10–13]. Particularly, thermo-responsive materials including thermo-responsive shape memory materials [14–16], thermo-responsive chromic materials [17], and thermo-responsive electrical conductive materials [18], are able to change their shape, color and electrical conductivity when experiencing temperature variations. Vanadium dioxide (VO2) is a kind of thermo-responsive material [19], which can be regarded as a smart phase change material. The IR reflection of VO2 changes significantly when the temperature is higher or lower than its phase transition temperature (Tc=68°C) [20]. Recently, VO2 has been applied to smart thermal management textiles for automatically modulating solar radiation at different temperatures [21,22]. For example, Lu et al [23] integrated VO2 with poly-dopamine or TiO2 to textile fibers. The resulting textiles had no obvious effects on temperature decrease at low-power light irradiation, while it could lower the temperature of about 17 °C under high-power light irradiation. Li et al [24] fabricated the smart functional fiber mats by electrospinning with a suspension of VO2 nanoparticles dispersed in polyvinyl pyrrolidone (PVP). The mats showed a significant difference in IR reflectance at low and high temperatures. In addition, Mao et al [25] coated W-doped VO2 on cotton fabric for thermo-responsive IR shielding with infrared emissivity of 0.851.
However, the IR reflection of VO2 is not adequate for IR protection purposes [26]. On the other hand, it is well known that silver (Ag) shows high IR reflectivity and is highly electrically conductive [27,28]. The sandwich structures with Ag, VO2 and other materials perform surprisingly in optical and electrical properties [29,30]. There is no study about silver/vanadium dioxide/silver (Ag/VO2/Ag) sandwich structure that has been reported. Hereby, we present a design of 1 D VO2 nanowires and Ag nanowires composites, whose structure is sandwich-like Ag/VO2/Ag layers on a porous Nylon film. Ag offers extremely high IR reflection to improve the IR shielding performance of the sandwich composite, while VO2 provides thermal-responsive smart function. This flexible and smart thermal management film is able to reflect IR radiation at high temperature and to be transparent to IR light at low temperature as shown in Figure 1. The Ag/VO2/Ag sandwich Nylon film showed outstanding IR shielding performance achieving a decrease of 10 °C under intense IR radiation and significant thermo-responsive electrical conductivity even after bending for 50 times. In addition, it can be prepared by using vacuum filtration, which provides a guide line for textile industry. This smart and flexible Ag/VO2/Ag sandwich film provides fast response to thermal stimuli for temperature-dependent IR shielding and electrical conductivity, which shows potential for thermal management, wearable electronics, energy generation and harvesting applications.
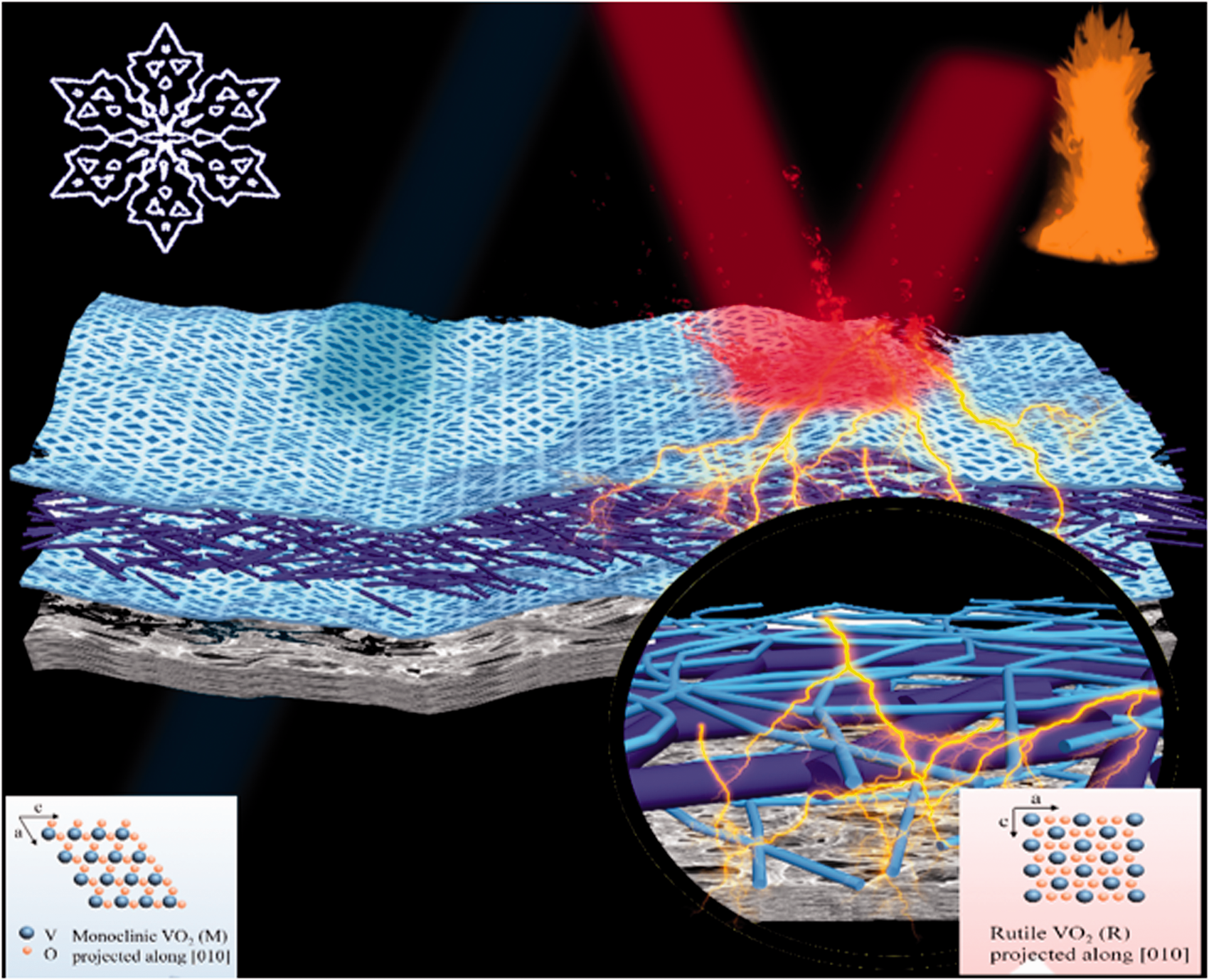
Schematic illustration of the Ag/VO2/Ag sandwich film for smart thermal radiation management.
Experiment
Materials
V2O5 (99.9%), hydrazine hydrate (N2H4·H2O, 78–82%), ammonia solution (NH3·H2O, 28%), oxalic acid dihydrate (99%), isopropanol (99.7%), polyvinylpyrrolidone (PVP, Mw ∼55,000), silver nitrate (AgNO3, 99%), sodium chloride (NaCl, 99.5%), ethylene glycol (EG, 99%) and ethanol (99.6%) were all purchased from Sigma (Australia). All of the chemicals were used as received without further treatment. 100% Nylon filter paper with pore size of 2 μm and the thickness of 170 μm was purchased from Shanghai Li Min Industrial Co., LTD.
Synthesis of the VO2 wires
In this study, the VO2 nanowires were prepared by a stepwise hydrothermal method with a few modifications [31]. The procedure is illustrated in Figure 2(a). First, the V3O7·H2O nanowires were prepared via the hydrothermal exfoliation and reduction of bulk V2O5 by oxalic acid dihydrate. In a typical reaction, 0.3 g of V2O5 was placed in a 45 mL PTFE cup with 16 mL of deionized water (DI) and 0.075 g of oxalic acid dihydrate. The cup was then placed in an autoclave and then sealed by following further heating at a temperature of 210 °C for 72 h. The resulting nanowires were filtered and washed with copious amounts of water and acetone in ultrasonic bath. After drying at 80 °C for 6 h under vacuum, 0.3 g of the V3O7·H2O nanowires were placed in a PTFE cup with 8 mL of DI water and 8 mL of isopropanol. The PTFE cup was then placed within a sealed autoclave and heated at a temperature of 210 °C for 72 h. The obtained VO2 (A) nanowires were washed with copious amounts of deionized water and acetone in ultrasonic bath for 15 min, successively. The collected nanowires were dried at 80 °C for 6 h under vacuum and were subsequently annealed at 550 °C under N2 atmosphere in a tube furnace with a heating speed of 10 °C min−1, while N2 flow of 300 sccm was maintained for 2 h. The VO2 nanowires crystallized in the M phase were obtained after cooling to room temperature.
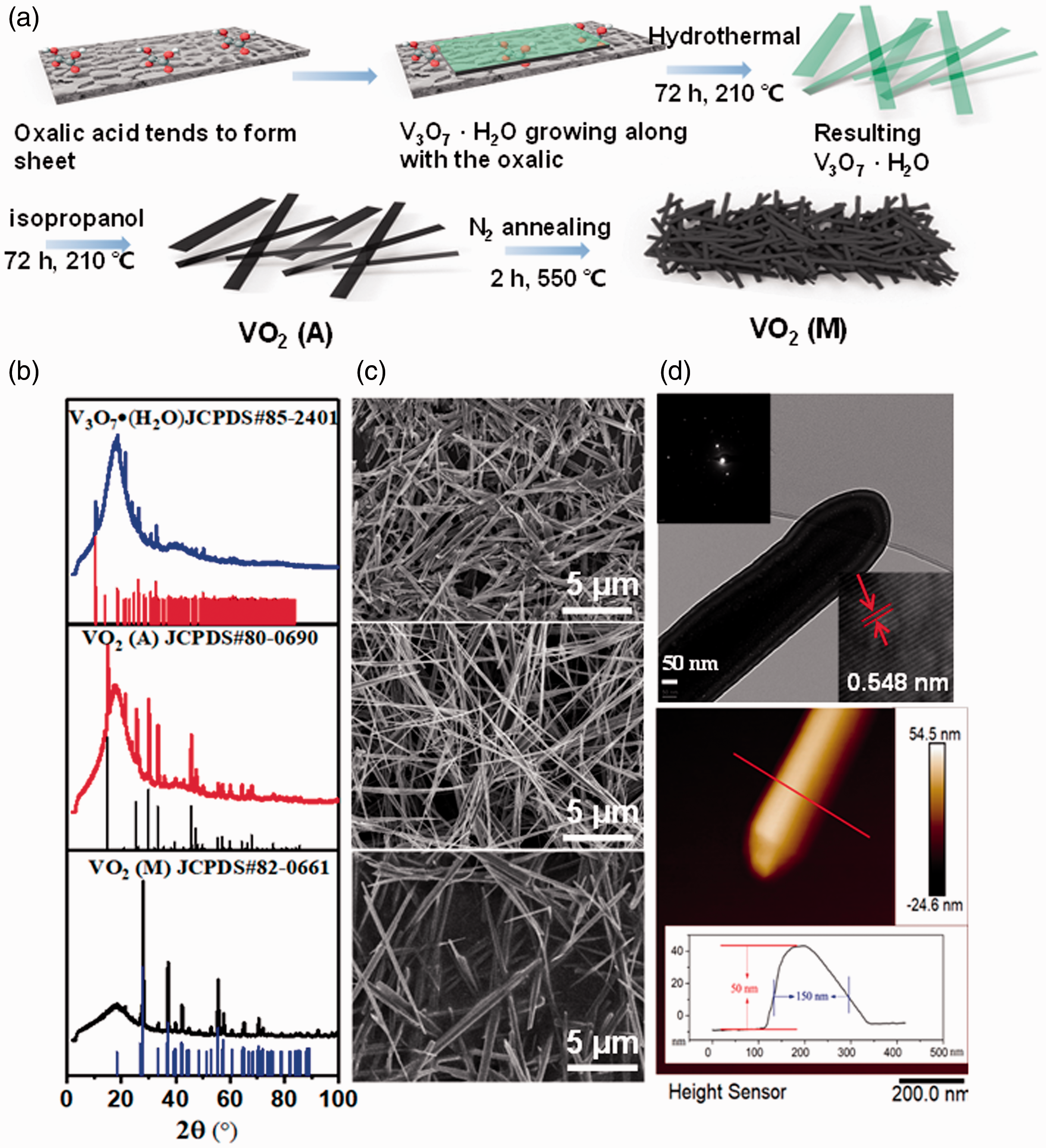
The synthesis and characterization of as-prepared VO2 wires. (a) Schematic diagram of synthesis of M phase VO2 wires, (b) XRD patterns and (c) SEM images and (d) TEM image and AFM image of the VO2 nanowires at different steps.
For comparison, VO2 nanoparticles were prepared by the hydrothermal method [32]. Briefly, 0.36 g of V2O5 was dissolved in 60 °C DI water (30 mL) containing 0.85 g of 28% NH3·H2O. Hydrazine hydrate (120 µL) was added into the solution under vigorous stirring after the solution was cooled down. Then, the solution was placed into a PTFE autoclave and kept at 220 °C for 24 h. The products were washed successively by centrifuging and re-dispersing in ultrasonic bath with DI water and dehydrated ethanol for several times. The VO2 was dried under vacuum at 60 °C for 4 h. Then, a pre-annealing was conducted at 180 °C for 2 h in the atmosphere. Next, the synthesized VO2 was put into a tube furnace under 500 sccm N2 atmosphere at 600 °C for 2 h with a heating speed of 10 °C min−1.
In this study, the VO2 nanowires and nanoparticles with Tc of ∼68°C was used to clearly demonstrate thermo-responsive IR shielding and electrical conductivity of the Ag/VO2/Ag sandwich film. In addition, Tc of VO2 can be decreased (like 36 °C, which closes to human skin’s surface temperature) by doping elements for practical applications.
Synthesis of the Ag nanowires
Ag nanowires were synthesized by a common polyol method with some modifications [33]. Firstly, 1 g of polyvinylpyrrolidone (PVP, Mw ∼55,000 Sigma-Aldrich) and 0.014 g of NaCl were dissolved in 20 mL ethylene glycol (EG) and denoted as PVP-NaCl-EG. Then, 0.204 g of silver nitrate (AgNO3) was dissolved in 10 mL EG (denoted as AgNO3-EG). The PVP-NaCl-EG was heated to 170 °C and then the AgNO3-EG was added into the system with speed of 5 mL h−1 under stirring. After that, the reaction was kept at 170 °C for 1 h. When the product was cooled down, 70 mL acetone was added into the system. The mixture was put in the ultrasonic bath for 30 min. The mixture was centrifuged with speed of 7000 rpm. Then, a small amount of acetone was added and the washing procedure was repeated for several times. The final product was dried in an oven under vacuum at 80 °C for 6 h.
Preparation of the Ag/VO2/Ag sandwich structure film
The Ag/VO2/Ag sandwich structure film was prepared by using the vacuum filtration method. Briefly, 0.15 g Ag and 0.15 g VO2 nanowires were dispersed in 100 mL of 10 g L−1 PVP ethanol solution separately in the ultrasonic bath for 30 min (denoted as Ag-PVP and VO2-PVP). Ag-PVP ethanol solution (10 mL), 10 mL of VO2-PVP ethanol solution and 10 mL of Ag-PVP ethanol solution were successively filtered on the round shape Nylon filter paper (3 cm in diameter) under vacuum. The Ag/VO2/Ag sandwich structure film was air dried at room temperature.
The other structures of the Ag, VO2 and Ag/VO2 composites films were all prepared by vacuum filtration method with the same amount of Ag and VO2 as can be found in Figure S1.
Characterization
Powder X-ray diffraction (XRD) characterization of the prepared materials was performed using a Rigaku Miniflex 600 diffractometer (Japan) with Cu Kα radiation (λ¼ = 1.5418 Å, voltage and current of 40 kV and 15 mA, respectively). The JADE 8.5 was used for pattern fitting and phase identification. The morphology of the obtained nanowires was examined by scanning electron microscopy (SEM, FEI Magellan America, at an acceleration voltage of 5 kV) and transmission electron microscopy (TEM, FEI Tecnai T20 America, operated at an accelerating voltage of 200 kV). TEM samples were prepared by dispersing nanowires in 2-propanol by ultrasonication, and casting onto 400 mesh copper grids coated with a Formvar/carbon film. The thickness of the VO2 nanowires was characterized by an atomic force microscope (AFM, Bruker Dimension iCon AFM, America). Fourier Transform Infrared Spectroscopy (FTIR) spectra were characterized by PerkinElmer Spectrum Two spectrometer (America). IR shielding of the Ag/VO2/Ag sandwich structure film was tested by the heat-insulating film tester (LS300 Linshang Co., LTD, Shenzhen, China). The equipment used for evaluating the smart IR shielding performance was shown in Figure 5(a). The equipment is a metallic box with two ventilation devices. Two IR lamps (150 W) were fixed at the top of the box. The samples and thermocouples were fixed at the middle and the bottom of the box, respectively. The temperature before and after IR radiation was recorded. For weak radiation, an IR filter glass was fixed under the lamp, while removed for intense radiation. The surface temperature of the testing film was recorded by an IR gun. The electrical conductivity of the Ag/VO2/Ag sandwich structure film was measured by the CHI660E electrochemical workstation (Chenhua, shanghai, China). The two parallel copper tapes with length of 2 cm and width of 0.8 cm were stuck on the surface of the Ag/VO2/Ag sandwich structure film. The distance between two copper tapes was kept at 0.5 cm. The voltage applied on the Ag/VO2/Ag sandwich structure film was scanned from -0.4 to 0.4 V with a scan rate of 50 mVs−1. The measurement was repeated for 3 times at 3 different places for averaging. The flexibility test of the Ag/VO2/Ag sandwich structure film was conducted by a linear translation stage with integrated controller (LTS150/M, Thorlabs, United States). The moving speed was 20 cm min−1. The moving distance of the holder was 1.5 cm (Figure S2). The digital images were taken by a camera.
Results and discussions
Morphology and structure of the VO2 nanowires and the Ag/VO2/Ag sandwich structure
To fabricate the Ag/VO2/Ag sandwich smart thermal management film, the key step is to synthesize monoclinic (M) phase VO2 nanowires. The VO2 (M) nanowires were prepared by three steps in a hydrothermal system as shown in Figure 2(a). Firstly, the V3O7·H2O nanowires were synthesized by reaction of V2O5 with oxalic acid using the hydrothermal method, yielding dark green phase-pure V3O7·H2O nanowires (reaction 1):
In this step, the oxalic acid was used as the reductant as well as a template to direct the growth of the V3O7·H2O nanowires. In the synthesis system of VO2, the oxalic acid tends to form sheet shape and serves as a template for VOx growth. The products were then centrifuged and dried for the next step. In the second step, the hydrothermal reduction of V3O7·H2O took place using a mixture of isopropanol and water as the reductant and solvent, respectively, as shown in the following equation:
Finally, the resulting products with black color were centrifuged and dried for the annealing procedure. After annealing with N2 protection at 550 °C for 2 h, the M-phase VO2 nanowires were successfully synthesized.
The XRD pattern confirms the crystal phase of the resultant V3O7·H2O nanowires as shown at the top of Figure 2(b). The broad peak at around 20°(θ) is attributed to the gel that was used to fix the samples onto the holder. The other sharp characteristic peaks closely match PDF No.85-2401, indicating the presence of pure phase of the V3O7·H2O. The middle pattern belongs to the product of the second step, which closely matches PDF No.80-0690 of VO2(A) (a metastable crystal phase). The final step is to transform VO2 (A) into VO2 (M) in N2 atmosphere (annealing) for good thermo-responsive IR shielding and electrical conductivity. After annealing, the XRD pattern closely matches PDF No. 82-0661 of VO2 (M) (bottom of Figure 2(b)) and shows that the product has been completely changed from VO2 (A) to VO2 (M). It can be seen from the top of Figure 2(c) that the V3O7·H2O is in nanowire shape with length of ∼15 μm and diameter of ∼150 nm. However, in the middle of Figure 2(c), the diameter of the VO2 (A) nanowires decreases to ∼100 nm and the length grows to ∼25 μm, indicating that the V3O7·H2O nanowires were etched by isopropanol during the hydrothermal reduction, while the VO2 evolved into the longer nanowires. The products from the last step are shown in the bottom of Figure 2(c). The VO2 (M) has length of ∼10 μm and diameter of ∼150 nm after annealing. The high temperature may cause melting and recrystallization of the nanowires, yielding short length of nanowires.
The VO2 (M) nanowires were furthered characterized by TEM and images are shown at the top of Figure 2(d). The end of VO2 (M) nanowires is not sharp but smooth and round. The spot diffraction pattern at the top inset is bright and regular, showing that VO2 (M) nanowire is well-crystallized. Some blurt spots are from other crystal faces due to the thickness of the wire. From the high resolution TEM image in the right bottom inset, the interplanar crystal spacing is around 0.548 nm. In order to further confirm the diameter of the VO2 (M) nanowires, we used AFM to detect their profile. As shown at the bottom of Figure 2(d), the thickness of the nanowire is around 50 nm while the width is ∼150 nm, indicating that the nanowire is not round but elliptical. In the synthesis system of VO2, the oxalic acid tends to form sheet shape and serves as a template for VOx growth. Upon crystal growth, an oriental attachment is a simultaneous process, which has a tendency to minimize the most energetically unstable surface facets by fast and preferential growth perpendicular to the correspondent facet [34]. Thus in the first step, short and narrow nanowires with cross section of rectangular shape were formed. In the following steps, the VO2 nanowires were oxidized by isopropanol as well as recrystallized during annealing, yielding etched edges with elliptical shape. These results confirm the successful synthesis of VO2 (M) nanowires.
The XRD pattern (Figure S3) of Ag nanowires shows that a strong characteristic peak at 2θ of 38.1° is attributed to the Ag crystal face of (111) plane. Weak characteristic peaks at 2θ of 44.2°, 64.4°, 77.3° and 82.3° are assigned to the (200), (220), (311) and (222) reflection lines of Ag, respectively. The XRD pattern identified by the JCPDS card (file No 89–3722) reveals that the Ag wires are polycrystalline with a characteristic face-centered cubic structure.
To prepare the Ag/VO2/Ag sandwich structure as shown in Figure 3(a), the Ag nanowires with 20 μm long were filtered on Nylon filter paper with 2 μm pore size under vacuum pressure, turning filter paper from white to gray. Then, the VO2 nanowires with 10 μm long were filtered on Ag deposited Nylon filter paper under vacuum pressure, resulting in black color. The top layer of Ag nanowires was deposited by the same procedure as the first step, resulting in brown color in appearance (as shown in Figure 3(b)). It can be seen that Nylon filter paper is the 3 D porous network structure with ∼2 μm even pore size (Figure 3(c)). Therefore, Ag and VO2 nanowires can remain on the filter paper instead of passing through. The diameter and length distributions of VO2 and Ag nanowires were shown in Figure S4.

Preparation of the Ag/VO2/Ag sandwich structure: (a) schematic of vacuum filtration of three layers of the sandwich structure, (b) the digital image of the Ag/VO2/Ag sandwich film, SEM images of (c) Nylon filter paper, (d) M phase VO2 wires, (e) Ag nanowires, (f) top surface, (g) high magnification, and (h) the cross-section of the Ag/VO2/Ag sandwich film.
From Figure 3(d) and (e), we can find that the diameter of the VO2 nanowires (∼150 nm) is larger than that of Ag nanowires (∼90 nm). The thinner and longer nanowires of VO2 benefit to form a strong packing network by entanglements. However, the electrical conductivity of VO2 nanowires increases with increasing diameters of VO2 as reported in the literature [35]. Actually, thinner diameter of Ag nanowires than that of VO2 nanowires benefits surface contacting between the Ag and the VO2 layers for sandwich structure. Therefore, in this study, to balance the film forming and electrical conductivity, we used the VO2 and Ag nanowires with diameter of 150 nm and 90 nm, respectively. Top surface of the Ag/VO2/Ag sandwich structure is observed by SEM and presented in Figure 3(f). It can be found that the top layer is the loose and fluffy Ag nanowires with network structure. From high magnification image (Figure 3(g)), only the Ag nanowires can be seen, which means that the top Ag layer is thick enough to fully cover the bottom VO2 layer. To further confirm the Ag/VO2/Ag sandwich structure, the cross-sectional SEM image is presented in Figure 3(h). It clearly shows the boundaries between the VO2 and the Ag layers due to different color contrast, which are marked with red lines. We can find that the thicknesses of the top Ag, the middle VO2 and the bottom Ag layers are around 15 μm, 50 μm and 1 μm, respectively (Elemental mapping also confirms the sandwich structure as shown in Figure S5). Thus the total thickness of the sandwich film was around 235 μm (including Nylon film). The amount of the two Ag layers is kept the same, but the bottom Ag layer is thinner than the top layer due to high vacuum pressure at the beginning of filtration and compression of the top VO2 and Ag layers. Therefore, the density of the bottom Ag layer is higher than that of the top Ag layer. The smart responsive properties are based on the thermal phase change material of VO2. Therefore, we prepared a thicker VO2 function layer than Ag layers in the middle. The above results confirm successful preparation of the Ag/VO2/Ag sandwich structure on Nylon porous film.
Mechanism and performance of smart IR shielding and electrical conductivity of the Ag/VO2/Ag sandwich structure
The mechanism of thermo-responsive IR shielding of the Ag/VO2/Ag sandwich structure is illustrated in Figure 4. In the sandwich structure, the VO2 layer isolates Ag layers when the temperature is lower than T c of the VO2 nanowires. The VO2 layer acts as a bridge to interconnect the top and the bottom Ag layer when the temperature is higher (Figure 4(a) and (b)).

Mechanism of thermo-responsive IR reflection (electrical conductivity) of the Ag/VO2/Ag sandwich structure: (a) VO2 is insulating at low temperature, and (b) conductive at high temperature.
At low temperature, the VO2 in its monoclinic phase (M phase) is insulating and IR transparent. Two layers of Ag nanowires are separated and isolated by the VO2 (M) layer (Figure 4(b)). The continuous and smooth Ag surface shows 98% IR reflection. However, if the surface of Ag is rough and not continuous, the IR reflection and electrical conductivity decrease significantly [36], resulting in high IR transmittance and adsorption. The separated Ag layers are not connected and too thin to show high IR reflection. In this case, free electrons are not moving from the Ag layer to the VO2 layer and not forming resonant oscillation of the conduction electrons for IR reflection, as illustrated in Figure 4(a).
On the contrary, at high temperature, VO2 in its Rutile phase (R phase) is conductive and IR reflective. The top, the middle and the bottom layers of Ag and VO2 are electrically connected, and free electrons are moving from the Ag layer to the VO2 layer, as shown in Figure 4(b). The resonant oscillation of the conduction electrons at the interface stimulated by incident IR light occurs on the surface of the Ag/VO2/Ag sandwich structure, which increase the IR reflection of the surface significantly [32].
In summary, in this sandwich design, the isolated Ag layers and VO2 (M) layer possess high IR transmittance at low temperature, while electrically connected Ag and VO2 (R) layers show high IR shielding at high temperature. The Ag layers provide more free electrons for the connection of the Ag and VO2 layers to reflect IR when VO2 changes to R phase. VO2 offers thermos-responsive solar regulating function to the sandwich film. This synergistic effect of VO2 and Ag was investigated in our previous study [32].
To confirm this hypothesis, we evaluated the IR shielding performance of the Ag/VO2/Ag sandwich film under IR radiation (as well as FTIR analysis presented in Figure S6). The equipment used for evaluating the smart IR shielding performance is shown in Figure 5(a). It can be seen from Figure 5(b) that the recorded temperatures of Nylon and the Ag/VO2/Ag sandwich film are close (all stay at ∼20°C), no matter it is before or after the weak radiation for 1 min (light blue and pink columns). The result means that the IR transmission of Nylon and the Ag/VO2/Ag sandwich structure is almost the same under weak IR radiation. This phenomenon can be explained by that the temperature is lower than Tc of VO2 under weak radiation, yielding VO2 in IR transparent and insulating state. The Ag layers are sitting on insulating VO2 layer and Nylon substrate, where free electrons are not easily moving to form resonant oscillation. Therefore, the Ag/VO2/Ag sandwich structure shows close IR transmission with Nylon under weak IR radiation.

Smart IR shielding performance of the Ag/VO2/Ag sandwich structure film: (a) the equipment for evaluating IR shielding performance, (b) recorded temperature values of Nylon and sandwich film under weak and intense radiation, (c) surface temperature of samples under weak and intense radiation, and (d) Vis-NIR transmittance spectra of the VO2 nanowires at 30 °C and 90 °C.
On the other hand, the recorded temperature of the Ag/VO2/Ag sandwich film under intense radiation for 1 min is 21.5 °C, which is around 2.5 °C lower than that of Nylon (24 °C). In this case, the surface temperature of samples increases to higher than Tc under intense IR radiation. Then VO2 turns to be electrically conductive and IR reflective, providing a pathway for free electrons to interconnect Ag nanowires from top and bottom layers [32]. The free electrons move from the Ag layer to the VO2 layer, forming resonant oscillation for high IR reflection. Actually, the Ag/VO2/Ag sandwich film can lower the temperature by ≈10°C under intense IR radiation from 31 to 21.5 °C, comparing with that without any coverage as shown in Figure S7. This result indicates that this smart thermal management film shows great potential for IR shielding in outdoor summer. Compared with other studies like Peng et al [2], it shows significant smart function, which is high IR shielding at high temperature while high IR transmission at low temperature. In addition, the sandwich film also shows higher IR shielding efficiency comparing with other shielding textiles based on other materials [1].
In order to verify the above temperature-dependent phase change of VO2, the surface temperatures of Nylon porous film and the Ag/VO2/Ag sandwich film under weak and intense radiation were measured (Figure 5(c)). In this study, Tc of used VO2 nanowires is 68 °C without any modification, while it can be further decreased (to around 36 °C) by doping other elements for practical applications. The VO2 with lower Tc can be applied to textile for practical use, yielding lower surface temperature. Under weak radiation, the surface temperatures of both Nylon and the Ag/VO2/Ag sandwich film increase from 20 to 30 °C, which are still lower than Tc of VO2. In this situation, the Ag/VO2/Ag sandwich film performs high IR transmittance. Therefore, the recorded temperature of the Ag/VO2/Ag sandwich film under weak radiation is around 20 °C, which is close to that of Nylon. However, the surface temperature of the Ag/VO2/Ag sandwich film increases to 105 °C under intense radiation, while for Nylon film, it is only 50 °C. In this situation, the surface temperature is higher than Tc of VO2, resulting in phase transition of VO2 to reflect IR. Therefore, the Ag/VO2/Ag sandwich film can lower around 10 °C, showing better IR shielding comparing with Nylon. The high surface temperature is probably caused by that rough surfaces of the Ag and VO2 nanowires absorb IR light to heat themselves. This smart function (temperature-dependent IR reflection) can also be approved by the Vis-NIR spectra of the VO2 film under high and low temperatures, as shown in Figure 5(d). The transmittance of IR light at 90 °C (red) is around 15% lower than that at 30 °C (black) due to phase transition of VO2. The results confirm the smart IR shielding property of the Ag/VO2/Ag sandwich film.
Moreover, the Ag/VO2/Ag sandwich film also possesses thermo-responsive electrical conductivity due to phase transition characters of VO2 as shown in Figure 6. The electrochemical workstation and the hotplate were used to test the thermo-responsive electrical conductivity of the Ag/VO2/Ag sandwich film, as shown in Figure 6(a). The temperature of the hot plate was set at the required temperature. The sample was put closely on the surface of the hot plate. The two isolated copper tapes were parallelly pasted onto the surface of the Ag/VO2/Ag sandwich film with 0.5 cm gap. The currents at various voltages were measured by the electrochemical workstation. In Figure 6(b), the current of the Ag/VO2/Ag sandwich film increases with the increasing temperature, but it is lower than 5 × 10−6 A at −0.4 V when the temperature is below 60 °C. At this stage, VO2 is in its M phase, which is the insulating state. The current increases with the rise of temperature, because some imperfect crystals change to R phase at lower temperature. In fact, the current increases significantly when the temperature is higher than 70 °C (Tc=68°C), and reaches 2 × 10−5 A at −0.4 V when temperature is 90 °C. At this stage, VO2 changes to R phase, which is a conductive state. These results show that the Ag/VO2/Ag sandwich structure films possess thermo-responsive electrical conductivity.
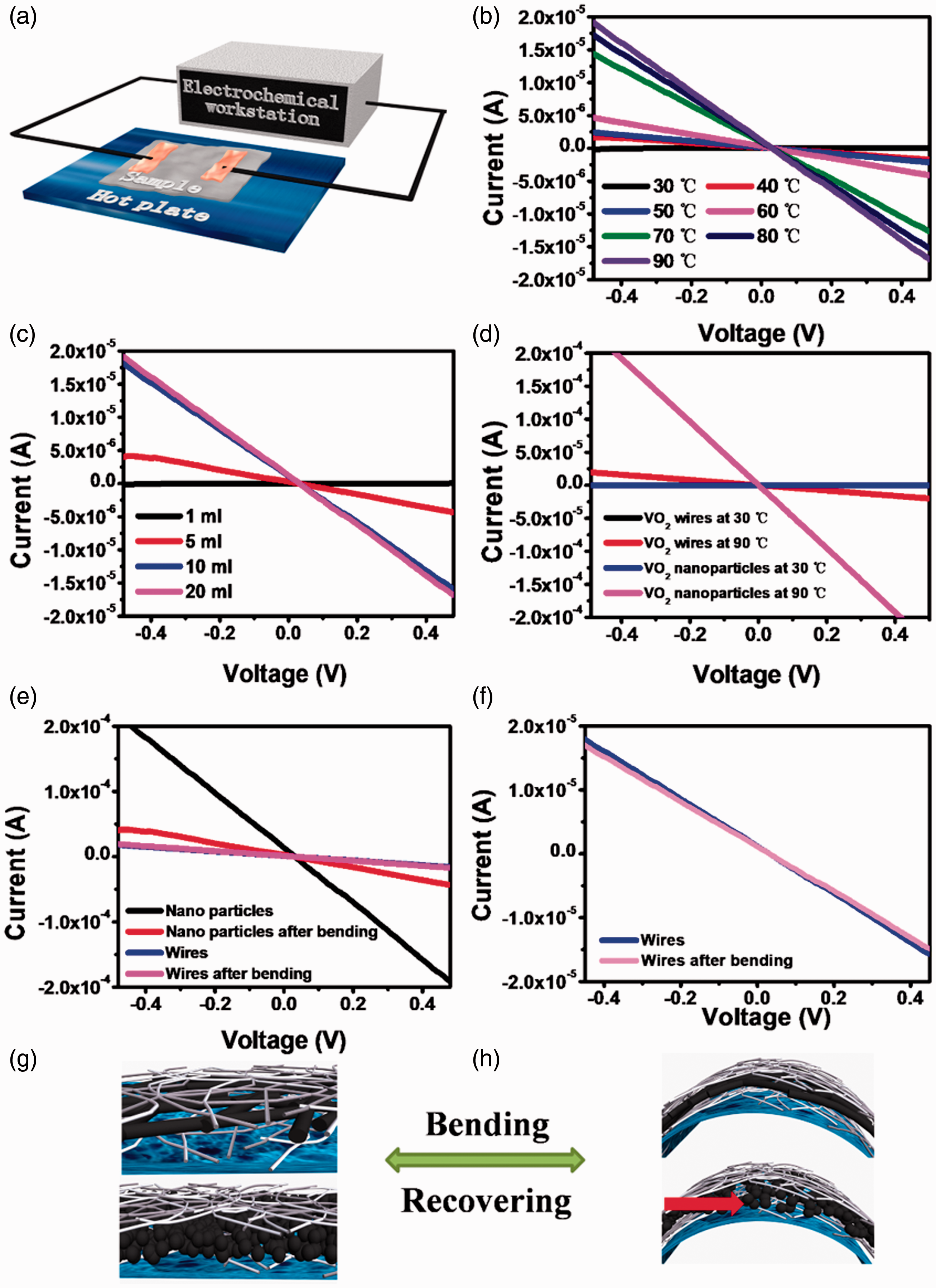
Electrical conductivity of the Ag/VO2/Ag sandwich structure film: (a) the equipment for evaluating electrical conductivity, (b) thermo-responsive electrical conductivity at various temperatures, (c) different thickness of VO2 nanowires, (d) VO2 nanowires and nanoparticles at 30 and 90 °C, (e) the Ag/VO2/Ag sandwich structure with VO2 nanowires and nanoparticles after bending 50 times, (f) the Ag/VO2/Ag sandwich structure with VO2 nanowires at high magnification, and schematic diagrams of magnifying the Ag/VO2/Ag sandwich structure with VO2 nanowires and nanoparticles (g) before and (h) after bending.
The influence of the thickness of VO2 layer on electrical conductivity is presented in Figure 6(c). The amount of the Ag layer keeps the same. The current of the Ag/VO2/Ag sandwich film is close to 0, when a VO2 dispersion of 1 mL is applied, yielding a thin middle VO2 layer in sandwich structure. The reason may be that VO2 is too thin to entangle with each other to form a network. As expected, the current increases when the amount of VO2 increases. It can be found that the current reaches 2 × 10−5 A at −0.4 V, when the VO2 dispersion more than 10 mL is applied. Even if the amount of VO2 is further increased, the current at −0.4 V remains around 2 × 10−5 A, but doesn’t further increase. At the beginning stage, increasing the amount of the VO2 nanowires benefits to form a continuous VO2 film. However, when the amount of VO2 is enough, the electrical conductivity of the sandwich film doesn’t further increase because the excess thickness of VO2 nanowires does not contribute to the overall electrical conductivity. Therefore, the amount of VO2 dispersion is fixed at 10 mL for the Ag/VO2/Ag sandwich structure film.
We also investigated the influence of VO2 shape on electrical conductivity of the Ag/VO2/Ag sandwich structure, and the results are compared in Figure 6(d). VO2 nanoparticles and nanowires were applied to the Ag/VO2/Ag sandwich structure film. Both nanowires and nanoparticles are insulating at 30 °C; therefore, the current is close to 0 in those two samples. The currents of both shapes increase significantly at 90 °C. It can be seen that VO2 nanoparticles in the sandwich structure show better electrical conductivity (the current at −0.4 V is higher than 2 × 10−4 A) than VO2 wires (the current at −0.4 V is around 2.5 × 10−5 A) at 90 °C. The contact between VO2 wires is not as good as that between densely packed nanoparticles due to steric hindrance. In addition, they form network with porous structure, yielding poor electrical conductivity. On the other hand, the VO2 nanoparticles are in very small size and densely packed, resulting in better electrical conductivity. In fact, for a single VO2 nanowire, it exhibits almost 4 orders of magnitude abrupt (within 1 °C) metal−insulator transitions [1]. Therefore, the electrical conductivity of sandwich structure can be further improved by preparing uniform VO2 nanowires. In addition, VO2 nanowires show their advantages in flexible films and it will be discussed in the following part.
As shown in Figure 6(e), VO2 nanoparticles in sandwich structure show poor bending stability as compared with VO2 nanowires. The current of the Ag/VO2/Ag sandwich film with VO2 nanoparticles decreases from 2.5 × 10−4 A to 5 × 10−5 A at −0.4 V after bending for 50 cycles. In contrast, the current of the Ag/VO2/Ag sandwich film with VO2 nanowires remains 2 × 10−5 A at −0.4 V after bending (Figure 6(f)). As illustrated in Figure 6(g) and (h), the flexible Ag/VO2/Ag sandwich structure film is bent and recovered repeatedly for 50 times. The VO2 nanowires and nanoparticles are densely packed and well contacted with each other in the sandwich structure when the film is flat. When the film is bent, the VO2 nanowires are entangled with each other, and form a flexible network. Therefore, the VO2 nanowires network wouldn’t crack after bending cycles due to larger contact surface, resulting in good bending stability. However, VO2 nanoparticles in the sandwich structure are not well connected because of small contact surface, yielding cracks in the film during bending cycles. Therefore, the electrical conductivity decreases significantly. These results indicate that the Ag/VO2/Ag sandwich structure film with VO2 nanoparticles can be applied to situations where high electrical conductivity is required but bending is not frequent. On the other hand, the Ag/VO2/Ag sandwich structure film with VO2 nanowires is more suitable for situations in which frequent bending is required, including textile, flexible film, wearable devices and so on.
Comparison of IR shielding and electrical conductivity of different structures
To verify that the Ag/VO2/Ag sandwich structure shows superior thermo-responsive property than other layer structures, we also prepared other five layer structures, as shown in Figure S1. Five different layer structures, namely sandwich, Ag only, VO2 only, Ag/VO2, VO2/Ag, and VO2/Ag mixture structures, all with the same amount of materials are compared. The IR shielding of the different structures was determined by the heat-insulating film tester, which was shown in Figure 5(a). The temperature differences before and after intense IR radiation are shown in Figure 7(a). It can be seen that without coverage, the temperature difference before and after intense radiation rises around 11 °C, while with coverage of Nylon film, the temperature difference is 4 °C. For Ag only and VO2 only samples, the temperature difference is also around 4 °C, which is quite close to that of Nylon film. This result may be explained by the fact that the thickness of the Ag layer is too thin for shielding intense IR radiation, while the IR reflection rate of the VO2 layer is limited due to its intrinsic characteristics.

Performance comparisons of different structures: (a) IR shielding performance, (b) long-term IR shielding performance, (c) IR shielding performance after bending 50 cycles, and (d) electrical conductivity.
However, we found that the IR shielding performance is improved when Ag is integrated into the structure with VO2. The temperature differences of the VO2/Ag composites (including Ag/VO2, VO2/Ag, Ag/VO2 mixture and Ag/VO2/Ag sandwich structures) are only around 1.5 °C under intense IR radiation. In these cases, the VO2 layer offers a dense substrate and 3 D framework for thermos-responsively bridging the Ag layers, while Ag layers provide more free electrons for the composites film to reflect IR. As a result, the VO2/Ag composite structures show 2.5 °C lower temperature than Ag only or VO2 only structures.
To further compare the long term IR shielding of the Ag/VO2 sandwich and the Ag/VO2 mixture structures, the temperature differences between before and after IR radiation for 10 min are shown in Figure 7(b). It can be seen that the temperature difference increases in all samples with increasing IR radiation time. The temperature difference of no coverage increases from ∼10 to ∼15°C due to heat accumulation. For the Nylon film, at the beginning, the temperature difference just increases slightly because Nylon absorbs IR at the first place, however, the temperature difference increases significantly with increase of time for Nylon emitting IR in return. The temperature differences of the Ag/VO2/Ag sandwich and mixture structures increase slightly (within 4 °C) even after IR radiation for 10 min. This phenomenon can be explained by the synergistic effect of the VO2 and Ag for IR reflection.
For bending stability, as shown in Figure 7(c), the temperature difference of the Ag/VO2 mixture before and after IR radiation increases a little (0.3 °C) from 1.7 (first bending) to 2.0 °C (50th bending), while for the Ag/VO2/Ag sandwich structure, the temperature difference is kept at around 1.5 °C (±0.1 °C). This result indicates the super advantage of the Ag/VO2/Ag sandwich structure on bending stability. Defects emerge in the Ag/VO2 mixture structure during bending due to size difference between the VO2 and Ag nanowires. These defects decrease IR reflection of the Ag/VO2 mixture. However, the VO2 layer and Ag layers are deposited layer by layer, forming sandwich structure. The nanowires with same size are cross linked to form a network within the layer on the Nylon, which is not easily broken. Therefore, the bending stability of the Ag/VO2/Ag sandwich structure film is in favor of long-term IR shielding.
In addition, among all of these composite structures, the Ag/VO2/Ag sandwich structure shows better performance in electrical conductivity than other structures as shown in Figure 7(d). The currents of Ag only, VO2 only and Ag/VO2 mixture structures are around zero even at 90 °C (black line, red line and yellow line, respectively). In Ag only case, Ag nanowires are too thin to form a well-connected network on the rough surface of the Nylon film, yielding poor electrical conductivity (the current is too low to be detected). For VO2 only structure, the size of VO2 nanowires is big and not densely packed, resulting in poor connections between each other. In addition, the electrical conductivity of the VO2 is not as good as that of Ag. Therefore, VO2 only structure shows poor electrical conductivity. It is interesting to note that Ag/VO2 composite structures show better electrical conductivity, including Ag/VO2, VO2/Ag, and Ag/VO2/Ag sandwich structures as shown in blue, green and purple lines, respectively. The currents of the Ag/VO2, VO2/Ag, Ag/VO2/Ag sandwich structures are 1 × 10−5, 1.5 × 10−5 and 2 × 10−5 A at −0.4 V and 90 °C, respectively. These results are attributed to the facts that Ag layer provides free electrons for VO2 layer, while VO2 layer offers a smooth and interconnected buffer substrate for thin Ag layers. For the sandwich structure, both top and bottom Ag layers provide free electrons, acting as electrodes. On the other hand, the VO2 layer in the middle also provides transport channels for free electrons, and the relatively smoother substrate for the Ag layer. Synergistic effect of Ag and VO2 is beneficial for electrical conductivity of the Ag/VO2 composite structures. However, the current of the Ag/VO2 mixture structure is close to zero, which doesn’t show synergistic effect of Ag and VO2. This phenomenon may be caused by poor mixture and network continuity between Ag nanowires and VO2 nanowires due to size difference.
The above results indicate that the Ag/VO2/Ag sandwich structure constructed by 1 D nanowires shows excellent smart IR shielding, thermo-responsive electrical conductivity and bending stability, as compared with other structures.
Conclusions
In summary, we have designed a flexible Ag/VO2/Ag sandwich structure film for smart IR shielding with thermo-responsive electrical conductivity. This film combines VO2 and Ag nanowires into sandwich networks, performing thermo-responsive IR shielding and electrical conductivity. The Ag/VO2/Ag sandwich film can lower the temperature by ≈10°C under intense IR radiation from 31 to 21.5 °C. In addition, the Ag/VO2/Ag sandwich film shows significant electrical conductivity difference under temperature variations while maintaining good flexibility and bending stability. The performance of the sandwich structure was compared with other layer structures and it showed better IR shielding and electrical conductivity. This new Ag/VO2/Ag sandwich film thus has great potential for various applications including smart thermal management for energy saving films, thermal-responsive sensors, wearable devices, and smart electronics.
Supplemental Material
sj-pdf-1-jit-10.1177_1528083720986542 - Supplemental material for Ag/VO2/Ag sandwich nylon film for smart thermal management and thermo-responsive electrical conductivity
Supplemental material, sj-pdf-1-jit-10.1177_1528083720986542 for Ag/VO2/Ag sandwich nylon film for smart thermal management and thermo-responsive electrical conductivity by Linghui Peng, Lingling Shen, Weiren Fan, Zichuan Liu, Hongbo Qiu, Aibing Yu and Xuchuan Jiang in Journal of Industrial Textiles
Footnotes
Acknowledgements
The authors acknowledge the use of instruments and scientific and technical assistance at the Monash Centre for Electron Microscopy, a Node of Microscopy Australia. This work was performed in part at the Melbourne Centre for Nanofabrication (MCN) in the Victorian Node of the Australian National Fabrication Facility (ANFF).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We gratefully acknowledge the financial support of the Australian Research Council (ARC) projects (DP160104456, LP140100786, IH140100035) and China Scholarship Council.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


