Abstract
The need for alternative eco-friendly and sustainable materials has forced many researchers to focus on composites made using natural fibers and biodegradable polymer matrices. In addition to that, these natural fibers are low in cost, have good mechanical properties, biodegradability, and require less production energy. Therefore, this review paper will focus on one of the natural fibers known as Luffa cylindrica (LC) by understanding all the natural fibers' benefits. A detailed overview is provided for commonly used fiber surface treatments, surface treatment techniques, and processing techniques for various LC composites from numerous other researches. Furthermore, this paper will also discuss the characteristics, mechanical properties, and water absorption properties of LC composites before and after surface treatments, where a comprehensive understanding of LC reinforced polymer composites' behavior has been reviewed.
Keywords
Introduction
It is well known that the world is confronting a serious problem known as environmental pollution, which contributes to global warming. Researchers worldwide have been working on green materials to reduce specific areas of environmental pollution and, therefore, contribute to decreasing environmental pollution as a whole. This need has forced many researchers to focus on composites made from biodegradable materials. However, synthetic fibers such as glass fiber and carbon fiber have been utilized for several years in manufacturing composites. These synthetic fibers' enforced composites have been used significantly due to their remarkable mechanical properties. Synthetic fibers originate from petroleum; therefore, issues arise in recycling and biodegradability [1,2]. Synthetic fibers are not fully biodegradable; they are flammable and release huge smoke and toxic fumes. Therefore, to minimize environmental pollution, various researchers have started researching natural fibers because they are abundant, recyclable, and biodegradable [3,4].
Natural fibers can be considered as multipurpose materials because they have decent mechanical properties, good soundproofing, and high impact resistance [5,6]. Furthermore, these natural fibers are widely used due to their advantages; they are a renewable source, lightweight, possess good tensile strength, biodegradable, and abundant. In addition to that, being carbon dioxide neutral, low energy consumption, no skin irritation, and no residues when incinerated are some of the environmental advantages of natural fibers’ [2,7]. Various types of natural fibers are available, but this paper focuses on Luffa cylindrica (LC). This paper focuses on compiling and summarizing LC's research work in the past by various researchers. The paper discusses commonly used fiber surface treatments and their techniques, processing techniques for LC composites, mechanical properties, water absorption, scanning electron microscopy (SEM), Fourier transform infrared spectroscopy (FTIR) and thermal properties.
Luffa cylindrica
LC is commonly known as loofa or sponge gourd and belongs to a vegetable sponges family [8]. The plant is available abundantly in countries like China, India, and Japan. Young luffa plants can be consumed as a vegetable and for medical purposes [9]. LC comes under the category of cellulose/lignocellulose natural fibers, as shown in Figure 1. During the growth of the LC plant, cells elongate and form a fibrous structure. This fibrous structure consists of cellulose, hemicellulose, and lignin [10]. LC fibrous mat shown in Figure 2 possesses great mechanical properties such as high strength, stiffness, and energy absorption [11–13]. Considering LC's mechanical properties, several researchers have used LC to make composites, whereas other researchers are motivated to make composites for metal-ion removal [14]. Moreover, LC's other commonly used applications are packing mediums, mats, soundproof linings, and removal of heavy metals from wastewater. In addition to that, treatment for asthma, sinus, chronic bronchitis, inflammation, and jaundice are some of LC's medicinal uses [15,16].

Classification of cellulose/lignocellulose natural fibers.

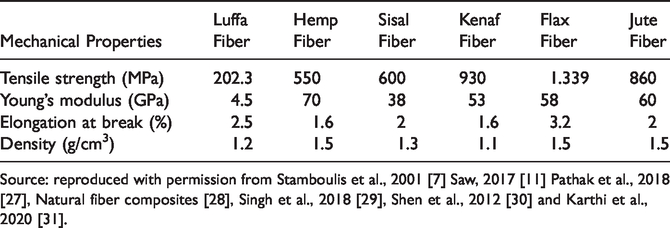
Luffa fiber.
As reported by various other researchers, LC contains high content of cellulose 55–70%, hemicellulose 8–22%, lignin 10–23%, extractives 3.2% and ash 0.4% [5,11,20–22]. From the findings mentioned, a comparison of chemical composition with other commonly used natural fibers can be seen in Table 1, and the molecular structure of cellulose, hemicellulose, and lignin can be seen in Figure 3. In addition to that, the density of LC lies between 0.82–0.92 g cm−3, which is not that high compared to other commonly used natural fibers like sisal or hemp [18,23,24]. Other than that, LC and other commonly used natural fibers’ mechanical properties can be seen in Table 2. Another advantage is that natural fibers such as LC have a higher fiber content than synthetic fibers; a small amount can provide equivalent performance, thus reducing the fiber's overall usage [25].
Comparison between luffa chemical composition and other commonly used natural fiber.


Molecular structure of (a) cellulose, (b) hemicellulose and (c) lignin
Comparison of luffa fiber mechanical properties with other commonly used natural fiber.
Fiber surface treatment
In order to enhance the properties of a composite, it is important to improve the adhesion between the fiber and the polymer matrix using fiber surface treatments. Natural fibers generally consist of a waxy surface and hydroxyl groups responsible for poor adhesion between the fiber and the polymer matrix. Therefore, it is important to thoroughly study various fiber surface treatments because each of the surface treatments has its own benefits [33,34]. Moreover, some of the most commonly used fiber surface treatments for LC are sodium hydroxide, silane, and acetylation surface treatments [26,35,36].
Sodium hydroxide surface treatment
One of the most commonly utilized fiber surface treatments is sodium hydroxide surface treatment. With the help of sodium hydroxide treatment, there is a disruption of hydrogen bonding, improving the surface roughness. Moreover, sodium hydroxide surface treatment causes depolymerization of cellulose, which then exposes short length crystallites [26]. There are many different ways through which this treatment can be performed to improve the adhesion [37]. Various researchers have shown various techniques to improve the adhesion using sodium hydroxide surface treatment in the past. Different values of concentrations, immersion times, immersion temperatures, and catalysts or coupling agents have been adapted by researchers [38]. The most commonly used techniques can be found in Table 3. Furthermore, the equation shown underneath shows the chemical reaction between the fiber and sodium hydroxide [21,39]
Shows various methods of using sodium hydroxide surface treatment.

Studies regarding different concentrations, immersion times, and immersion temperatures have been made by various researchers, which leads to the existence of various techniques to perform sodium hydroxide surface treatment. All of the magnitudes mentioned above can influence the mechanical properties of a composite in a slightly different way. Moreover, sodium hydroxide surface-treated fiber composites have always shown superior mechanical properties compared to untreated fiber composites. This insinuates that surface treatment using sodium hydroxide enhances the adhesion between a fiber and a polymer. In some studies where numerous concentrations of sodium hydroxide solutions were compared, the results had shown a decrease in composite strength when the concentration was increased. This result can be expected due to the concentration being high enough to destroy the fiber surface leading to poor adhesion. In addition to that, the increase in immersion time, such as 144 h, also leads to the destruction of the fiber surface. Furthermore, various studies showed fiber immersion at elevated temperature because better cleaning of fiber surface can be expected at elevated temperature compared to room temperature.
Silane surface treatment
Silane is a powerful coupling agent used to adhere to glass fiber with a polymer matrix. To show a reduction in cellulose hydroxyl groups in the fiber matrix, silane has been used to surface treat the natural fibers [26]. Silanol forms in the presence of moisture and, therefore, reacts with the hydroxyl groups, resulting in the formation of covalent bonds on the fiber's surface [26,39,54].
Several types of silane coupling agents are available for silane surface treatment. However, the most commonly used silane coupling agents are aminopropyltriethoxy silane, vinyltris (2-methoxyethoxy) silane, 3-aminopropyltrimethoxy silane, n-octyltrimethoxy silane, bis [3-(triethoxysilyl) propyl] tetrasulfide, and 3-trimethoxysilyl propyl mathacrylate [55]. Various researchers adapted different approaches to perform silane surface treatment [21]. These techniques were adapted to varying concentrations, coupling agents, immersion time, and immersion temperature. The most commonly used techniques to perform silane surface treatments are mentioned in Table 4
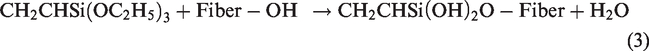
Shows various methods of using silane surface treatment.

As mentioned earlier, several types of silane coupling agents are available; therefore, they affect the fiber differently, leading to slightly different mechanical properties. One of the most frequently used silanes coupling agents is 3-aminopropyltrimethoxy silane because it is easily and widely available. In addition to that, silane surface-treated fiber composites showed better mechanical properties than untreated fiber composites. It can be concluded that silane surface treatment improves the adhesion between the fiber and the polymer. From various techniques, as mentioned above, longer immersion times are adapted by various researchers. It is important to have a longer immersion time to ensure silane can diffuse properly with the fiber's cell walls. However, a higher immersion temperature value will eventually speed up the process of diffusion between silane and fiber cell walls.
Acetylation surface treatment
The whole purpose of acetylation surface treatment is to introduce acetyl functional groups. Acetylation surface treatment can be referred to as esterification of the cellulosic fiber as this surface treatment introduces acetyl functional groups in the microstructure of the fiber; therefore, making the fiber more hydrophobic [26]. The chemical reaction that takes place can be seen in the equation [21,26,39]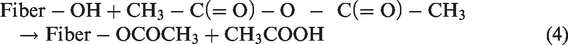
Acetylation surface treatment of natural fiber can be attained in several ways. Different values of concentration, temperature, immersion time, and catalyst can be used. No matter what the technique is, the end purpose is to make the fiber more hydrophobic. Commonly used techniques by various researchers can be seen in Table 5.
Shows various methods of using acetylation surface treatment.

Various techniques adapted by researchers have shown different immersion times, immersion temperatures, and concentration values. Acetylation surface-treated fiber composites show better mechanical properties when compared to untreated fiber composites. This can be due to better adhesion between the polymer and the fiber. This is because the acetylation surface treatment replaces hydroxyl functional groups with acetyl functional groups. Higher concentration values lead to poor mechanical properties because the fiber surface gets damaged. Furthermore, high immersion time leads to better bending strength but the deflection of the composite decreases.
Characteristics of LC polymer composites
Fourier transform infrared spectroscopy
FTIR is an analytical technique that is based on an interferometer. Spectrum bands for the IR usually range from 4000–400 cm−1 [68,69]. Various researchers follow the standards provided by the American Society for Testing and Materials (ASTM). In order to perform FTIR, researchers usually utilize ASTM E168-16 and ASTM E1252-98 standards to conduct qualitative and quantitative analysis. Furthermore, FTIR helps in understanding the functional groups which are present in the fiber. A comparison between the peaks of untreated, sodium hydroxide, silane treated, and acetylated LC can be seen in Figure 4.

A comparison between FTIR peaks using different chemical surface treatments.
FTIR results for silane treated LC samples, as shown in Table 4, indicate asymmetric stretching between Si-O-Si linkage and Si-O-cellulose bonds. This stretching is visible in the range of 950 and 1150 cm−1. Bands ranging from 936 and 1366 cm−1 confirm the bonding between silane and cellulose. In addition to that, Si-O-cellulose’s bond is also confirmed with the stretching of the bands. Bands ranging from 1200 and 1366 cm−1 also confirm the presence of Si-O-Si and Si-O-cellulose bonds [25].
Sodium hydroxide treated LC samples indicated in Table 3 have shown peaks in the range of 1500 and 1000 cm−1. This concluded the bending of CH2, O-H, and stretching of C-OR. Furthermore, the untreated LC sample showed a vibration of C = O at the range of 1735 cm−1. This can be due to the presence of carbonyl and acetyl groups in hemicellulose and lignin. However, sodium hydroxide treated LC samples showed no peak at 1735 cm−1. Therefore, successful removal of hemicellulose and lignin can be concluded [70].
Acetylated LC samples shown in Table 5 showed peaks in the range of 1735 and 1737 cm−1, which confirms the absorbance. Furthermore, the C-H bond present in lignin showed bending at the peak value of 1500 cm−1. Stretching and vibration of the carbonyl group C = O are caused due to the process known as esterification of hydroxyl groups; therefore, the stretching can be seen in the range of 1735 and 1737 cm−1. Furthermore, ester linkages between acetyl and hydroxyl groups are confirmed with the peak shown at 2372 cm−1 [39].
Thermogravimetric analysis
Thermogravimetric analysis (TGA) is a method adapted by various researchers to study composites' decomposition and thermal stability [71,72]. This method can be used to compare the composites' thermal decomposition with varying concentrations of chemicals used for chemical surface treatments, varying fiber volumes, and the addition of fillers or nanocomposites.
Thermal analysis was performed on thermoplastic starch/LC composites with varying fiber wt%. The processing technique can be seen in Table 6. The study concludes that the weight loss of 5 wt% LC composite started at 100–200°C, which could be attributed to the presence of moisture in the composite. Furthermore, the maximum amount of weight loss occurred at 333°C due to thermoplastic starch degradation. However, at the temperature value of 389°C, maximum weight loss occurred due to the cellulose and hemicellulose's thermal decomposition present in LC. In the case of 10 wt% LC composite, the initial weight loss was at a similar temperature of 100–200°C however, maximum weight loss occurred at 336°C. In addition to that, composite with 20 wt% LC showed similar weight loss initially at 100–200°C; however, maximum weight loss occurred at 343°C [71].
Shows various processing techniques used by researchers to manufacture luffa fiber composites.

In another study regarding TGA, untreated and surface-treated LC composites were compared. Sodium hydroxide surface treatment was utilized in this research, as shown in Table 3. Another surface treatment, known as FA-grafted composites, was utilized as well. The results can be seen in Figure 5. The study concluded that the FA-grafted composites showed maximum temperature value for the maximum rate of mass loss followed by sodium hydroxide treated composites and then by untreated composites [19].

TGA analysis of surface-treated luffa fiber epoxy composites.
Another study regarding TGA compared LC nanocomposites modified calcium carbonate and calcium phosphate. The result can be seen in Figure 6. Four samples were tested; B1 (calcium carbonate modified LC – resorcinol nanocomposites (1:1)) , B2 (calcium carbonate modified LC – resorcinol nanocomposites (2:1)), B3 (calcium phosphate modified LC – resorcinol nanocomposites (1:1)) and B4 (calcium phosphate modified LC – resorcinol nanocomposites (2:1)) [5].

Mass loss (%) using TGA.
Scanning electron microscopy
SEM analysis for sodium hydroxide surface treated samples, as shown in Table 3, concluded that the untreated fiber samples consist of impurities, as shown in Figure 7(a). These impurities will lead to poor fiber and polymer adhesion. In addition to that, a smooth surface was seen due to the presence of waxy impurities on the surface. Samples with an immersion time of 48 h shown in Figure 7(b) showed no sign of impurities, eventually leading to better adhesion between the fiber and the polymer. In addition to that, a rough surface was seen, confirming the removal of waxy impurities. However, samples immersed for 144 h indicated in Figure 7(c) showed complete annihilation of the fiber surface, and cracks were seen throughout the fiber's surface [40].

SEM samples: (a) untreated, (b) sodium hydroxide treated 48 h, and (c) sodium hydroxide treated 144 h.
Sodium hydroxide and silane surface-treated fiber samples were compared using SEM analysis. Surface treatment was performed using the techniques mentioned in Tables 3 and 4. With the help of Figure 8(a), it was witnessed that the surface of the fiber was smooth due to the presence of waxy impurities. In addition to that, linkages were present on the surface due to the fiber being rich in lignin. As shown in Figure 8(b), the fiber surface after the sodium hydroxide surface treatment was much rougher due to waxy impurities' removal. Another noticeable difference was the removal of linkages from the surface due to the removal of lignin. However, the silane surface-treated sample indicated in Figure 8(c) showed a smooth surface. This is due to silane sticking on the surface of the fiber [59].

SEM samples: (a) untreated, (b) sodium hydroxide treated samples, and (c) silane treated samples.
Another study regarding SEM analysis for untreated, sodium hydroxide, silane, and acetylated surface-treated fiber samples was conducted. As shown in Figure 9(a), untreated fiber samples showed impurities on the surface. These impurities were seen in the form of wax and lignin. Sodium hydroxide surface-treated samples indicated in Figure 9(b) showed a rough surface due to the removal of waxy impurities. This observation confirms that sodium hydroxide surface treatment was successful. Furthermore, the adhesion between the fiber and the polymer was improved due to the surface being rough. The silane surface-treated sample shown in Figure 9(c) did not show any specific changes because the fiber's surface was still smooth. This was due to the silane particles sticking on the surface of the fiber. However, the acetylated surface treated sample indicated in Figure 9(d) showed complete removal of impurities. In addition to that, the surface of the fiber was still smooth due to the replacement of hydroxyl groups with acetyl groups [64].

SEM samples: (a) untreated, (b) sodium hydroxide treated samples, (c) silane treated samples, and (d) acetylated treated samples.
Processing techniques
Various researchers have adopted numerous techniques in the past to produce LC composites. In some cases, several processing techniques were utilized to manufacture LC composites. Only a few techniques are used to produce LC composites with various types of polymer matrices. LC composites and their processing techniques with processing magnitudes are shown in Table 6.
Mechanical properties
Processing techniques and fiber surface modification techniques discussed earlier have different effects on mechanical properties. These effects are discussed in the following sections. Furthermore, Table 7 shows flexural strength, tensile strength, and hardness for all the mentioned LC composites and the type of fiber surface treatment utilized.
Shows the fiber treatment and the mechanical properties of various LC composites.

Flexural
In order to make sure that a natural fiber polymer composite is suitable for desired applications, it is important to determine flexural modulus, flexural strength, flexural load, and deflection at break [6,84]. There have been various studies on flexural strength, focusing on fiber volume, fiber length, and fiber surface treatment.
A study related to LC polyester composite showed a comparison between acetylation, combined process, and cyanoethylating treated and untreated LC polyester composites. The study results showed that the acetylated treated fiber recorded the highest amount of flexural strength, modulus, and elongation at break [49,79,80]. In a different study, LC and groundnut enforced epoxy composites were tested for flexural strength. This study focused on fiber volume in the composites. Different fiber volumes ranging from 10 to 50% were utilized to produce composites. The study showed a gradual increase in the strength as the fiber volume increased from 10 to 40%. However, an unexpected drop in flexural strength was observed when the fiber volume reached 50% [13,19,73]. In a study regarding LC/polypropylene composites, the composites were tested for flexural rigidity. LC was treated using sodium hydroxide surface treatment. Results from this research showed that with the addition of 20% LC in the composite, the flexural rigidity of the composite was almost doubled when compared to 100% polypropylene samples [82,85]. In another study, LC and polylactic acid (PLA) composites were tested for flexural strength. LC in this research was treated using sodium hydroxide surface treatment. A fiber volume of 2%, 5%, and 10% was used to produce composites with PLA. It was concluded in the research that the composite with 2% LC showed the highest value of flexural stress and flexural strain. However, composites prepared using 5% LC indicated the highest flexural modulus value [86].
A different approach of the study was conducted regarding bio nanocomposites of LC. Two samples were prepared in which the fiber was modified using calcium carbonate and resin with a weight proportion of 1:1 and 2:1, respectively. It was observed that the sample with 1:1 mixing had better flexural properties in comparison to 2:1 mixing composites [5].
As can be seen in the studies performed by various researchers, more than one factor can affect the flexural strength of the composite; it is possible to conclude that fiber surface treatment and a certain amount of fiber volume % in the composite lead to better flexural properties. However, different concentrations of chemicals in fiber surface treatments and different fiber % in the composites can lead to a different outcome. Therefore, it is recommended to analyze the composites and conditions thoroughly.
Hardness
A study showed a comparison between glass fiber epoxy composite, glass fiber/LC epoxy hybrid composite, ceramic epoxy composite, and ceramic/LC hybrid epoxy composite. The research concluded that the ceramic/LC epoxy hybrid composite showed the greatest value for hardness [81]. In a different study, micro fillers like calcium carbonate, aluminum oxide, and titanium dioxide were added to create LC polyester composites. Similar wt% of fillers were used; however, fiber wt% changed from 5 wt% to 10 wt%. The study concluded that polyester with 10 wt% LC and 5 wt% aluminum oxide showed the highest value of hardness, 12.9 Hv [75]. In another study regarding fillers, natural fillers like rice husk, wood powder, and groundnut were used. LC wt% of 30 was kept constant for all the samples; however, the wt% of 3%, 7%, and 11% was utilized for fillers. It was concluded in the study that as the wt% of rice husk and wood powder fillers increased, there was a decrease in hardness value. However, in the case of groundnut, the hardness value increased as the wt% of filler increased. The highest value of hardness, 13.3 Hv, was recorded when 11% of the groundnut was used [76]. In other research, the effects of fiber surface treatment were reviewed. LC was surface treated using a concentration of 10%. The treated and untreated LC polypropylene composite samples were tested for the Rockwell hardness number. The result concluded that treated LC polypropylene composites have better Rockwell hardness numbers than untreated LC polypropylene composites [82].
As shown in the studies performed by various researchers, more than one factor can affect the hardness value. Two main factors affecting the hardness value are different concentrations of the chemical and volume of the fiber. Micro fillers such as calcium carbonate, aluminum oxide, and titanium dioxide were added, and all of them showed better hardness value. However, when adding micro fillers, it is recommended to change the percentage of the fillers in the composite to study the effects that the fillers have on the hardness.
Tensile
In a study regarding LC enforced epoxy composites, the comparison was shown between treated and untreated LC with varying wt%. The results revealed that treated LC composites had higher tensile strength relative to untreated LC composites. Furthermore, 40% of fiber volume composites showed the highest tensile strength compared to 30% and 50% fiber volume composites [24,74]. In another study, LC filled poly (butylene succinate-co-lactate)/starch blend composites were compared with kenaf fiber-filled poly (butylene succinate-co-lactate)/starch blend composites. Both the fibers were surface treated using the same concentration of sodium hydroxide. Furthermore, the mixing ratio was unchanged, as well. The result showed that the tensile strength of kenaf fiber enforced composite was higher than LC enforced composite. Moreover, the difference between the tensile strength was less than 4 MPa [70,87]. In another study, LC polyester composites were tested before and after adding calcium carbonate, aluminum oxide, and titanium dioxide as micro fillers. Fiber volume of 5 wt% and 10 wt% were used in this study, and the micro fillers were used at 5 wt%. The result showed that there was a huge increase in tensile strength. However, calcium carbonate fillers and 5 wt% fiber showed the overall highest tensile strength [75]. In a different study, natural fillers such as rice husk, groundnut, and wood powder were used to increase the tensile strength of luffa fiber enforced polyester composites. Additionally, 30 wt% of LC was used throughout all the samples, whereas wt% of 3, 7, and 11 were used for fillers. The results showed a decrease in tensile strength in the case of rice husk and wood fiber as the wt% went from 3 to 11. However, there was an increase in tensile strength when groundnut wt% went from 3 to 11. It was concluded that this was due to the difference in the fillers' size [76]. The effects of stacking patterns were tested in another study. LC and coir fiber were used to make epoxy enforced composites. Both of the fibers were surface treated using the same concentration of sodium hydroxide. Tri-stacking (Luffa/Coir/Luffa), tri-stacking (Coir/Luffa/Coir), and bi-stacking (Luffa/Coir) were tested in this study. The results showed the highest tensile strength for the tri-stacking (Luffa/Coir/Luffa) method. However, tri-stacking (Coir/Luffa/Coir) showed the lowest tensile strength [11].
As shown in the studies performed by various researchers, more than one factor can affect the tensile strength. Different concentrations of chemicals and the volume of the fiber are two main factors affecting the tensile strength. Furthermore, different methods of stacking can affect tensile strength as well. Another approach adopted by researchers of adding nanofillers can also affect the tensile strength. However, when adding fillers, it is recommended to change the wt% of nanofillers in the composite to study the effects on tensile strength thoroughly.
Water absorption properties
It is important to have a study related to water absorption because these studies help researchers to discover the impacts due to the moisture on natural fiber composites. Moisture leads to debonding between the fiber and the polymer matrix, deforms the composite's shape, and eventually leads to poor mechanical strength [6,88]. In addition to that, natural fibers like LC and cotton fiber have poor hydrophobicity [89,90]. Therefore, it is important to make sure that natural fibers and their composites have good water repellence.
LC composites had been tested for water absorption on various occasions. In a study, sodium hydroxide treated, and untreated LC enforced epoxy composites were tested according to the American Society for Testing and Materials (ASTM) D570 standard. Specimens were initially oven-dried to obliterate the moisture. This assessment procedure took 48 days, during which the weight and the dimensions of the immersed specimens were recorded every 24 h. The results showed that the treated composites absorbed 13.8% less water than the untreated composites [24]. In another study, fiber bundles of LC were tested. These samples were surface treated using a varying percentage of sodium hydroxide. According to the American Society for Testing and Materials (ASTM) D 2654-89a standard, the samples were tested. Thirty samples of each type of treatment were placed in an oven for 4 h at 105°C to completely eliminate the moisture. The samples were weighed and measured before being placed in the humidity chamber at 65% relative humidity at 24°C for 24 h. The results concluded that the untreated fiber absorbed the highest amount of moisture; however, sodium hydroxide treated fiber showed a decrease of 29%. Furthermore, as the percentage of sodium hydroxide increased in the solution, a higher amount of moisture absorption was recorded [51,91]. Another study was conducted on polypropylene/LC composites were tested before and after silane and MAPP surface treatment for water absorption. The samples were immersed in distilled water at room temperature, where the measurements and differences in weight were recorded frequently. The untreated fiber samples showed a 2.8 wt% increase; however, a decrease of 39 and 28.4% was seen for silane and MAPP treated fiber samples, respectively [20,25]. LC in another research was tested for water absorption after sodium hydroxide, grafted, and hydrolyzed surface treatments. The samples were immersed in 500 ml of de-ionized water, which was heated from 10°C to 60°C. The researchers were monitoring all kinds of changes in the samples' weight after a certain amount of time. The result indicated that for sodium hydroxide and hydrolyzed treated samples, water absorption increased as the temperature increased. However, grafted samples indicated that water absorption decreased as the temperature increased [92,93].
According to other researchers' findings, there are numerous ways to assess water absorption in the sample. One of the approaches is to utilize moisture regain %, and the other is to find uptake % [25,51]


On multiple occasions, bio-nano composites reinforced with LC were tested for water absorption as well [23]. The experiment was implemented at 25°C using a temperature-controlled incubator. Water activity was set in the range of 0 to 0.95. Samples weighing approximately 20 mg were exposed to nitrogen flux inside the chamber. The readings were taken until there were no further changes [94]. In another research, an LC sponge was used to test the absorption of oil. The fiber was surface treated using 2 wt% aminopropylisobutyl POSS with hexane as a solvent and 2 wt% trisilanophenyl POSS with ethanol as a solvent. These samples were immersed in motor oil, pump oil, olive oil, vegetable oil, and diesel oil. The result showed that 2 wt% aminopropylisobutyl POSS with hexane as a solvent treated LC sponge had better absorption capacity (mL/g) when compared to 2 wt% trisilanophenyl POSS with ethanol as a solvent treated LC sponge [95]. Absorption capacity was calculated using the formula below
where, W1 and W2 represent the fiber's weight before and after the absorption of oil, and
Conclusion
There are various types of materials available to make composites; natural fibers are among those materials. Besides being biodegradable and eco-friendly, natural fibers hold advantages like low cost and good mechanical properties. This paper reviewed one of the natural fibers known as Luffa cylindrica (LC), with the aim to compile and summarize the important work of various other researchers on LC and its composites. This paper has shed light on commonly used chemical surface treatments and their techniques to modify LC's characteristics, for example, sodium hydroxide, silane, and acetylation surface treatments. Furthermore, various types of matrices were reinforced using LC, such as epoxy, thermoplastic starch, polyester, polypropylene, and polylactic acid. In addition to that, two of the most commonly utilized techniques to prepare LC composites were hand lay-up and compression molding. Using scanning electron microscopy (SEM), LC's surface was observed to be flaky and waxy. With the help of chemical surface treatments, the LC's surface was modified to improve its structure and remove the waxy impurities. Moreover, with the addition of LC in matrices, overall mechanical properties were improved. However, the highest number of mechanical properties was seen for LC reinforced epoxy composites, where the tensile strength of 192.70 Mpa, the flexural strength of 106.67 Mpa, and hardness of 50 Hv was achieved. Furthermore, chemical surface-treated LC composites showed slightly better mechanical properties when compared to untreated LC composites. With the addition of LC in polymer matrices, there was an increase in overall water absorption. However, with the help of chemical surface treatments, this issue was tackled; for example, silane surface-treated LC composites showed a 39% decrease in water absorption when compared to untreated LC composites. Different natural and synthetic fibers were utilized as well to hybridize LC composites, such as glass, groundnut, and coir fibers. Hybridizing LC composites exhibited better mechanical properties when compared to pure LC composites.
Future research on LC and LC composites should focus on thermal stability as not enough research has been conducted in this area. Furthermore, more research is required if LC composites are to be used in automotive, aerospace, sports, and other industries. Thus, it is demonstrated in this paper that further research should be undertaken in terms of analytical analysis using Hirsch’s, Halpin-Tsai models, and Fourier's heat conduction equation to predict tensile strength, young's modulus, and thermal characteristics, respectively. Using analytical analysis, one can save both time and resources. Finally, more research needs to be done when considering recycled polymer matrices to make composites with LC. Thus, environmental pollution can be mitigated with the utilization of natural fibers like Luffa cylindrica (LC), where research provides a solution through the sustainable use of green composites and the incorporation of natural fibers in them.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
