Abstract
Recent crisis in the form of COVID-19 has rendered wearing of mask mandatory for patients, health care workers and members of public worldwide. This has caused a sudden shift of focus on availability, effectiveness, re-use and development of face masks/respirators. In the current pandemic situation, the shortage of masks has also led to rethinking on strategies of reuse of masks after due sterilization. This work discusses a quick laboratory methodology to test/determine the particle filtration efficiency of face masks/respirators. The testing parameters include the particle capture efficiency of the mask material/full mask, pressure drop and the fit factor. Two different, simple, make-shift set-ups have been adopted for the present context. The first is used to measure the intrinsic particle capture efficiency and pressure drop of the filter material and the second as a ‘full mask sampler’ to assess the leakages through seams and joints of the mask. Experiments conducted with atomized NaCl test particles on three types of mask viz. commercial N-95 respirator, surgical mask and cloth mask have been used for evolving the methodology. The differences in terms of capture efficiency of aerosol particles for the filter material and for the full mask in face fix/sealed fixture have been linked to improvement of the mask design in development phase. This paper hopes to provide a crucial laboratory link between the mask developers and the certification agencies in the times of urgency. Needless to mention that commercialization of the same is subject to certification from authorized agencies, following standard procedures.
Introduction
The world is facing an unprecedented crisis in the form of COVID-19 (coronavirus, SARS-CoV-2 disease) pandemic, a highly infectious disease. As of 15th November, 2020, more than 54.4 million cases and 1.3 million deaths [1] indicate the severity of the current situation. The modes of transmission for SARS-CoV-2 in populations include droplets, aerosols and physical contact, very similar to some recent pandemics (Severe acute respiratory syndrome (SARS)-1, Middle East respiratory syndrome (MERS), Influenza) [2–4]. Wearing of face mask/respirator is one of the key public recommendations made by the scientists [5], designated authorities and governments. This is posed as a crucial defense to control the transmission from the infected to healthy individuals, implying better public health. Although the research studies on masks have been consistent, resurgence is seen during the pandemic period [6], periodically bringing the focus on aspects related to their effectiveness in controlling the airborne transmissions.
A face mask/respirator is designed to reduce the concentration of inhaled particulates to which the wearer is exposed. Apart from filtration efficiency, main factors crucial to its design are pressure drop, moisture absorption, reusability and comfort fit [7,8]. In general, face masks refer to loosely fitting masks while respirators are face fitted via sealings. Non-powered air purifying particulate respirators are segregated as N-series (for workplace free of oil aerosols) and R- & P- series (exposure includes oil-based liquid particulates) respirators [9]. N-series respirators are classified as per their capture efficiency for the most penetrating particle size (MPPS) of 0.3 µm. Thus N-95 class refers to 95% capture efficiency for the MPPS at the testing conditions specified as per the documented protocols given by certifying agencies. Face masks/respirators remove particles from the inhalable stream via aerosol filtration processes i.e. interception, diffusion, impaction and electrostatic deposition. The efficiency of removal depends on the aerosol characteristics (concentration, particle size and charge), filter characteristics (solid fraction, fiber diameter etc.), environmental conditions (RH and temperature) and flow rate of the air stream.
For particle capture considerations, decrease in fiber diameter and increase in respirator thickness is of importance in mask designs [10]. Breathability is improved in surgical (medical) masks, albeit compromising the filtration characteristics. Surgical masks are commonly used in healthcare settings where the properties such as splash resistance and blood penetration resistance become equally important [11]. A balance between the quality factor [12] and aspects such as cost and availability of the raw material, other fiber characteristics (e.g. splash resistance, static charge etc.) and risk/exposure profile finally determines the choice amongst the options of respirators, surgical masks and nanofiber masks [13]. Another issue which could be important is the decontamination enabling the re-usage of masks, provided the performance does not degrade. Several scientific bodies recommend guidelines to be followed for the use and certification of the respirators and surgical masks. For example, in United States of America, Occupational Safety and Health Administration (OSHA) recommends a compliance program where the use of National Institute for Occupational Safety and Health (NIOSH)-approved respirator is mandated. Similarly, standard testing procedures in terms of documents and protocols are in place worldwide (American Society for Testing and Materials standards, Bureau of Indian standards, European Union protocols, Korean Food and Drug Administration protocols etc.). The various filtration test methods differ in terms of aerosol type, flow rate/face velocity and sample type and extensive studies have been carried out on optimizing effectiveness of different types of masks under these guidelines. The effectiveness of N-95 respirators and surgical masks in health care setting has been extensively reviewed [14,15]. As per the convenience and due to the concept of MPPS (generally in the range of 0.2 to 0.5 µm) for aerosol filtration, optical based detection (> 0.2 µm particle size) has been preferred in these test methods. As infected individuals release considerable numbers of submicron particles in the exhaled breath [16,17], detailed measurements in this size range have also been reported [18,19]. These mostly include electrical mobility measurements for interpretation and methodology development [20–22].
In the past epidemics, as well as in the present COVID-19 situation, there has been a dearth of supply of N-95 respirators for health care workers [23] and for other essential service workers. Hence, urgent efforts are undertaken to develop new designs or to explore alternative arrangements encompassing decontamination strategies. This is also true for surgical masks where any new design/fabric needs a quality check for the desired features. Although cloth masks are widely used by general public, generally effectiveness evaluations are not performed for the same. Different materials for developing the masks have been explored w.r.t. their performance [24–30]. In addition to capture efficiency and breathing resistance, prevention of leakage in respirators [31,32] and ascertaining the fit of the mask without or after any decontamination treatment [33,34] are important issues. New designs need testing in a set-up which either is accessible at ease or can be made in a laboratory or industrial setting. A simple methodology to determine whether the risk of the use of designed mask is acceptable is the utmost desirable deliverable of such a set-up. Some studies published in recent past made effort in this direction by discussing the set-up/methodology for similar contexts [35–37].
The objective of this work is to evolve a quick methodology, which can be adopted very easily by a standard aerosol laboratory, for testing the quality of a face mask/respirator in terms of its particle (intrinsic and full mask) capture efficiency, pressure drop and the fit factor. The setup adopted for this purpose comprises of an intrinsic sampler and a full mask sampler. A sequential procedure to investigate the design of the mask leading to the set specifications and desired characteristics has been evolved. The test conditions may not conform to a particular standard, but are more tuned towards getting first level assessment. Nevertheless, the setup can be tuned for stricter test conditions depending on the requirement. The study discusses experiments with 3 different types of masks and enumerates the results with a view to evolve an appropriate methodology for the present context.
Material and methods
For the assessment of the ‘material particle capture efficiency’, a laboratory set up was made with a glass pipe having provisions to hold the filter material, connect optical particle counter and a manometer, upstream and downstream across the test specimen. This was also capable of measuring the pressure drop across the specimen. For the full mask testing, a glass jig acting as a mannequin was connected to a cylindrical aerosol sampling station at one end while the other end was used for air suction without and through the test mask. After validating the set-up and the methodology, 3 types of masks were employed as ‘test specimens’ in the current work. The details are presented as under:
Aerosol instrumentation
We used optical particle counter (OPC), condensation particle counter (CPC) and scanning mobility particle sizer (SMPS) for the measurement of aerosol characteristics. A laser particle counter (Grimm OPC model 1.108) was used for measuring the size distribution of test particles in the size range of 0.3–20 µm (particle optical diameter). It works on the principle of light scattering by the particles and follows Mie scattering patterns for the deconvolution of size distribution from the light intensity profile. Results from OPC measurements have been used for studying and interpreting the variation of filtration (capture) efficiency as a function of particle size. In addition, SMPS was used in these experiments to cover size range down to nucleation mode particles. SMPS measures mobility size distribution of test particles by size classifying particles on the basis of their electrical mobility. Grimm SMPS (model no. 5403) was used in this work covering the size distribution measurements in the size range of 0.01–0.9 µm (equivalent mobility diameter). OPC and SMPS sampled the particle-laden air with the flow rates of 1.2 Lmin−1 and 0.3 Lmin−1 from the aerosol sampling station, respectively. We also used our setup for the estimation of ‘fit factor’ utilizing the measurements of total number of particles (of all sizes) carried out with a CPC. Grimm CPC (model no. 5416), sampling at the flow rate of 0.3 Lmin−1 and measuring total number concentration of particles (>4 nm particle diameter) was employed for this purpose. CPC was preferred for the estimation of fit factor (FF) as it can be used as a ‘total number concentration counter’ (similar to other commercial instruments used for FF estimations) operating at a high measurement frequency. Other relevant details about these instruments and sampling protocols have been provided elsewhere [38,39]. TSI Portacount respirator fit tester (model no. 8038) was used for validating the results for FF obtained from our set-up. It is a modified condensation nuclei counter designed to measure respirator fits. It samples aerosol particles at the flow rate of 0.35 Lmin−1 and measures the FF by comparing number concentration of test particles sampled from ambient environment and a mask. Aerosol atomizer (TOPAS ATM226) was used for the generation of ‘NaCl test particles’ during these measurements. These particles were chosen due to their availability in any working laboratory and convenience in terms of aerosolization. NaCl particles have also been used as test particles in standard testing procedure code [40] which was followed for matching operating conditions in this work. These test particles were passed through an ‘aerosol neutralizer’ and ‘nafion dryer’ before passing through the mixing chamber and the test section. We also conducted few tests under ambient atmospheric conditions and the interpretations are discussed in a later section.
Test set-up
Figure 1(a) shows the set-up used for the evaluation of intrinsic efficiency and pressure drop of the filter media. It comprises of a cylindrical glass pipe (to be named as ‘intrinsic sampler’ hereafter) of volume 2.2 liters (length: 140 cm, diameter: 4.5 cm) having provisions to connect aerosol particle stream, aerosol instrumentation (OPC, in this case) and a manometer. The ‘test filter media’ (cut piece of diameter 5.5 cm corresponding to face area of 23.75 cm2) was placed in the line of air stream using a clamping arrangement. The sealing was ensured by performing ‘zero aerosol test’ i.e., passing clean air through the inlet, closing all other ports and ensuring near zero aerosol number concentration through the outlet port. A ‘full mask sampler’ has been utilized for the evaluation of full masks. The block diagram of this sampler and the associated setup has been shown in Figure 2(a). The core part of this set-up is a leak proof face mannequin (a modified 2 liters round glass flask) which had 3 circular openings on its round bottom; 2 openings of diameter 1.5 cm and one larger opening of diameter 2.5 cm, representing the facial nostrils and mouth. The mask to be tested can be attached over these openings in ‘face fix position’ (i.e., attached by means of elastic bands symbolic of as on a human face) or ‘sealed position’ (wherein the mask edges were completely sealed onto the openings on the mannequin to give the most conservative results). The face area in this configuration is the tested area of the fabric which was ≈250 cm2, 180 cm2 and 230 cm2 for N-95 respirator, surgical mask and cloth mask, respectively. The sampling arrangement has been designed so as to sample the aerosol particles from the ‘exposed environment’ (NaCl or ambient particles, outside the mask) and ‘inhaled stream’ (inside the mask) alternatively and continuously by a controlled switching arrangement. This sampler can be connected to an aerosol sampling station or any other measurement network depending on the requirements of the test protocols. A closed mixing chamber was used for sampling atomizer generated test particles through the mask attached on the mannequin. For the case of direct atmospheric sampling, the mannequin was kept open to the atmosphere. The volume of the chamber and of the mannequin also affects the number concentration at the steady state conditions and hence should not be too restrictive against the working range thresholds of the measuring instruments. Figures 1 and 2 also shows the aerosol instrumentation used in the present work. The availability of the aerosol instrumentation and the affordability in terms of the fabrication of simple set-up shown here indicates that any common aerosol laboratory can perform the test experiments similar to this work depending on the context and the need. This can assist the mask developers and they can approach such laboratories for testing the masks against the set specifications as first level assessments. This can also be utilized to attain desired parameters by varying the configuration of the components such as layers of filtration media during the development phase itself. Set-ups similar to Figures 1 and 2 have been used elsewhere for the evaluation of surgical face masks [40,41] and other masks [21], respectively.

Block diagram and representative picture of the setup used in experiments for intrinsic particle capture efficiency and pressure drop measurements.

Block diagram and representative picture of the setup used in experiments with full mask sampler (inset of Figure 2(b) shows the mannequin).
Test specimen
Three types of masks were selected for the present study. The first one was a commercial ‘N-95 respirator’ which served as a reference specimen. It was a convex shape design with polypropylene as the filter media. The headband on the specimen was sewn on the outer surface and designed to provide a leak-proof fit. It was certified by NIOSH for its performance criterion (capture efficiency >95% for 0.3 µm non-oil based particles) complying with standard testing procedures [9]. A commercially available ‘Surgical mask’ was the second candidate for these tests. This specimen was a 3-ply disposable mask having loose-fitting face seal fit. This was picked in order to demarcate the expected differences in terms of aerosol capture efficiency and pressure drop in comparison to a standard N-95 respirator. For the third specimen, we preferred a ‘Cloth face mask’ representing the public choice in the current circumstances. The samples used in these experiments were home-made single layered quilter’s cotton fabric (Threads per inch or TPI ≅ 85–100) with stitched cloth head band. The choice of these specimens was made so as to validate our setup covering the wide difference in their characteristics. While N-95 respirator was expected to give collection efficiency close to or more than 95%, efficiencies are expected to be lesser for the other two variants. Although certifications are provided for commercial N-95 respirators and surgical masks, cloth masks are generally used without any testing or certification. Set-up and methodology similar to the present work can be used for performance evaluation of cloth masks in terms of their particle filtration characteristics.
Experimental methodology and statistical analyses
Standard testing procedures mostly focus on testing the masks or the material at the worst case scenario or the most conservative test conditions. Attaining and maintaining these conditions in a simultaneous aerosol sampling manifold is not simple and requires a considerable amount of efforts in terms of engineering design, flow matching, isokinetic sampling and/or instrumentation. For a quick preliminary estimate, test conditions can be tuned in accordance with the capabilities of the set-up. For our case, we used number concentration of test particles as the quantitative parameter for efficiency estimates. The statistical parameters of the size distribution of steady state atomized NaCl particles varied slightly between different experiments (Count median diameter: 70–100 nm, Geometric standard deviation: 1.8–2.2, Total number concentration: 50,000-60,000 cm−3). We also carried out few representative measurements with atmospheric aerosol particles. Flow rate for aerosol capture efficiency (intrinsic as well as full mask) and pressure drop was fixed at 28.3 Lmin−1. This is above the normal breathing rate of 7.5 Lmin−1 and light activity breathing rate of 13–25 Lmin−1 for an average adult [42]. The corresponding face velocities work out to be 19.86 cm/sec for intrinsic measurements and 1.88–2.36 cm/sec for full mask, respectively. Flow rate and face velocities in this range have been used by several researchers for different contexts in the past [26,43]. These velocities are also in the range given in standard testing procedures for evaluating medical face masks [44]. Pressure drop measurements for the filter material have been carried out at 8 Lmin−1 in accordance with available codes [40,41]. Sampling flow rate for the ‘fit test’ on the mask sampler was taken as 0.3 Lmin−1, corresponding to the sampling flow rate of the CPC as also used in a commercial fit tester. Each specimen was tested 5 times and the mean values and standard deviation (1 σ) were estimated in accordance with error propagation rules. As can be seen, the experimental test conditions were matched with those for standard testing procedures wherever possible. However, in view of the primary purpose of this work i.e. obtaining first level assessments, set-up as well as experimental procedure has been made flexible and convenient. The main differences of the set-up in comparison to the established standard testing procedures [45] are the use of polydisperse test aerosol system, number concentration as the testing parameter, flexibility in terms of face velocities and options to evolve in-house methodology as per the immediate need. These test conditions are easy to attain and can be adopted in a quick time while performing the evaluation tests at the design and development phase itself.
Results and discussions
Aerosol capture efficiency for the filter media/material can be defined as the ratio of captured (filtered) to the inlet aerosol concentration. It can be estimated using the upstream (inlet) and downstream (outlet) aerosol concentration for the case of ‘intrinsic sampler’. For the case of full mask, it can be estimated from exposed (outside the mask) and inhaled (inside the mask) concentration. The formulation used in the present work for the intrinsic particle capture efficiency (ηi) and the full mask particle capture efficiency (ηFM) in % are as below.
The efficiencies measured are for polydisperse aerosol spectrum and are reported at stated size selected as the geometric mean size of the particle size range corresponding to the size channel of the instrument. For example, stated particle size of 0.35, 0.55 and 1.25 µm corresponds to the number concentration measured in 0.3–0.4, 0.5–0.65 and 1.0–1.6 µm, respectively. The lowest mean size is close to the generally selected MPPS of 0.3 µm when using latex monodisperse particle suspensions. The difference can however be reduced by selecting the optical particle counter having lowest size channel, covering particle sizes lesser than 0.3 µm.
Intrinsic efficiency and pressure drop
The first step in our measurements pertains to the testing of the filter media material for the aerosol capture efficiency and pressure drop. The material, intrinsically, should qualify against the set specifications during the initial development phase. It is also applicable for the context of sterilization methods, where the process should not degrade the material in terms of the initial specifications. These tests were performed in intrinsic sampler with atomized NaCl aerosols as the test particles and OPC as the measurement system. The average capture efficiency for 0.35 µm particles was measured as 96.19, 40.08 and 14.22% for N-95, surgical mask and cloth mask, respectively. This is listed in Table 1 along with the capture efficiencies for few other sizes as well. As seen, all three test specimens gave expected capture efficiency in this test.
Aerosol capture efficiency and pressure drop (intrinsic) measurements for the three test specimen.

The pressure drop, ΔP (in mm H2O) at 28.3 Lmin−1 (face velocity of 19.86 cm/sec) was measured as 21, 14, and 18 mm H2O for filter material of N-95, surgical mask and cloth mask, respectively. The differences in pressure drop values are as expected for the tested specimens. Airflow resistance of sealed surgical mask has been shown to be approaching that of the N-95 respirator in a comparative study performed with N-95 respirator, nanofiber respirator and surgical mask [46]. The set-up used for these measurements is similar to the setup shown for testing surgical/medical face mask. With similar face area (≈5 cm2) and sampling flow rate (8 Lmin−1), pressure drop for surgical masks can be compared with the specified values [41]. We measured ΔP as 3 mm H2O for tested surgical mask in this configuration corresponding to breathability resistance of 0.13 mm H2O/cm2 which is well below the prescribed upper limit (4-6 mm H2O/cm2) for surgical masks [41]. The material to be used as filtering media for surgical masks can be tested in such an ‘intrinsic sampler’ for both the filtration efficiency and the breathing resistance. Measurements with cloth mask and N-95 respirator in this kind of set-up can also be used for assessing the performance of different materials/configurations in terms of pressure drop characteristics. These pressure drop data can be obtained at any sampling flow rate provided the conditions for isokinetic sampling are maintained.
Full mask sampler-Measurements with optical particle counter
As the capture efficiency tests are specified at MPPS (0.2–0.5 µm) range or sometimes at other stated higher particle sizes such as 1 µm, measurements with OPC covering these size ranges are sufficient for first level assessments. In an aerosol laboratory as well as filtration related industrial set-ups, optical sizers are generally available. In recent times, low cost sensors [47] are also being used for aerosol measurements but issues are also reported for high accuracy applications [48]. It should be noted that proper aerosol measurements require an appropriate understanding of the instruments, set-up, sampling and data interpretation. Any new set-up requires a validation check in terms of leakages, representative and true sampling and engineering aspects e.g. flow and pressure matching. After validation, the developed set-up and a calibrated OPC is sufficient to infer the parametric estimations required for filter/full mask testing. Table 1 summarizes measurements performed in full mask sampler with OPC for all three specimens.
Table 1 gives the comparison of the capture efficiencies with the mask fitted in full mask sampler in both face fix and sealed position. The average capture efficiencies are supplemented with the uncertainties in the above table. These uncertainties are the combination of experimental fluctuations (including variations in the generator output) and the uncertainties associated with the response curve of the measuring instrument. These numbers are also compared with the intrinsic efficiencies obtained for the same specimens when measured in intrinsic sampler. As is observed, sealing slightly improved the efficiency for N-95 respirator moving it closer to the intrinsic efficiency of the filter material. Any significant difference in the efficiency values between the intrinsic efficiency and the sealed mask indicate the leakage through stitches, joints etc. For the case of surgical mask, difference observed between face fix and sealed position is indicative of an inappropriate fit on the mannequin jig. This difference is expected to arise due to leakages through mask sealing, owing to the flat fit as compared to the cup-shaped fit of the N-95 respirator. For cloth mask, higher standard deviation was observed, possibly due to slight variation in texture quality (also indicated by TPI range of 85–100) within the selected 5 specimens. For all the masks, efficiencies for the full masks in sealed fit were close to the intrinsic efficiencies within the error estimates. It should be noted that only N-95 respirator, among the selected specimens, is designed to fit perfectly on the face. However, the observed results for the other two loosely fitting masks can be used for improvisations so as to achieve better performance.
Fit factor estimations
The Fit tests for face masks are reported in terms of fit factor (FF) which is the ratio of ambient to the inhaled concentration. In terms of the full mask particle capture efficiency (ηFM in %), the minimum value of FF can be represented as
For comparison of FF obtained from ‘Portacount’ and our setup (‘full mask sampler’), we used the CPC as the measurement system in out setup. This ensured that both instruments gave the results based on concentration of particles in almost similar size ranges. The estimations of FF were made for both ‘face fix position’ and ‘sealed position’ for first test specimen i.e. N-95 respirator. As can be seen in Table 2, FF estimated from both instruments used with the set-up matched closely.
Fit factor on the mask sampler estimated by CPC and Portacount.
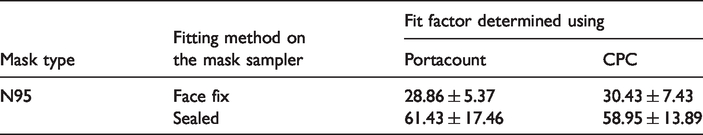
The average FFs shown above can also be compared in terms of particle capture efficiency. These values came to be 96.54% and 98.37% when measured from Portacount and 96.71% and 98.30% when measured from ‘full mask sampler’ for face fix and sealed fit, respectively. The setup can also be used for determining the FFs on human subjects similar to the other systems. FFs for different nominal breathing rates (following OSHA protocols or any other user defined protocols) can be obtained by modifying the mask sampling arrangement. In such a case, combination of CPC measuring the total number concentration as an integral counter and a specially fabricated sampling probe (taking sample from air/chamber and CPC, alternatively) can provide FF values for different activity routines such as talking, bending etc. The details of such an extension are beyond the scope of this work. In the present context, measurement of FFs from our setup and standard commercial instrument used for this purpose were performed for validating the set-up only. Close matching of the FFs obtained from our system and Portacount ensured that no leakages exist in the aerosol sampling manifold.
Efficiency evaluations in sub-micron and nano size ranges
As the aerosol filtration characteristics are size dependent, measurements in the particle sizes other than those covered by OPC size range become relevant as well. In addition to OPC measurements, we generated data from SMPS and CPC when the test specimens were investigated in full mask sampler. This enables to extend interpretations to sub-micron (< 1 µm particle size) size range covering ultrafine (< 0.1 µm particle size) and nano size ranges (<0.02 µm particle size). This kind of added information may be useful for some specific contexts and applications. For example, a user may be more interested in filtering out nanoparticles in a specialized place e.g. microelectronics fabrication area. Also, some standards notify the performance in terms of particle capture efficiency at 0.1 µm [40]. This is relevant in the present pandemic situation too because although the transmission of vectors of COVID-19 infection takes place in particle size ranges well above ultrafine size range, the virus itself (unattached) is in the range of 0.06–0.14 µm [49]. For this context, biological effectiveness evaluations (similar to bacterial efficiency test) could be quite important. However if the filter media/mask fails in terms of particulate filtration efficiency for equivalent size test particles, it is unlikely that it will pass such evaluations performed at limiting test conditions. Figure 3 compares the capture efficiencies obtained for all test specimens at 0.02 µm, 0.05 µm, average of 0.06–0.14 µm, 0.35 µm, 0.55 µm and 1.25 µm particle sizes. These values are taken for sealed position of the masks on the jig of full mask sampler. The first three values are obtained from SMPS measurements while the last 3 are the same which were given in Table 1.

Aerosol capture efficiencies of the test specimen for nano and sub-micron sizes estimated under sealed fit condition on the full mask sampler.
The capture efficiency pattern for N-95 respirator and surgical mask followed the typical U-shaped filtration efficiency pattern (minimum around 0.3 µm) as expected. However for the cloth mask, the efficiency minimum was found to be in the particle size range 0.06–0.14 µm. The particle size corresponding to efficiency minimum was also found to be different for different fabrics and in the same size range for some commonly used fabrics [21]. It can also be inferred from the above figure that capture efficiency for this size range was measured at 96.94%, 52.44% and 8.27% for N-95 respirator, surgical mask and cloth mask, respectively. The measured efficiency in the size-range 0.06–0.14 µm (measured as mobility equivalent diameter) and is expected to be similar to the viral capture efficiency considering the shape and size characteristics of corona virus. We also compared the capture efficiency for all particles (cumulative of the particle size range of the instrument) measured by the instruments (OPC, SMPS and CPC) for all three specimens. Figure 4 shows these values measured for sealed fit position in full mask sampler.

Aerosol capture efficiency for all particle sizes (cumulative of all particles sizes in the measurement range) as measured by OPC, SMPS and CPC.
As seen from Figure 4, the efficiency of N-95 respirator and cloth mask showed marginal differences by including particle sizes below 0.3 µm when compared to the efficiency obtained for OPC covered size range. But these differences were seen to be significant for the case of surgical mask. This was due to the notable improvement of filtration efficiency for the surgical mask for the particles lesser than 0.3 µm. For the case of N-95 respirators, the efficiencies were more than 95% for all particle sizes, leaving no scope for an observable change by including lower size particles. For the cloth mask, the efficiency minimum shifted to the size range 0.06 -0.14 µm, thereby inducing a small decrease in efficiency for SMPS and CPC covered particle sizes. In contrast to N-95 respirator and cloth mask, the efficiency increased from ≈ 40% to ≈ 90% for the mean particle size of 0.35 µm and 0.02 µm, respectively for the surgical mask (Figure 3). This influenced the filtration response by covering more particle sizes compared to OPC size range and resulted in an observable change by including smaller sizes.
Efficiency evaluations using atmospheric aerosols
Filtration efficiencies cannot be performed using ambient atmospheric aerosols for OPC size (0.3–20 µm) ranges in case of very stringent applications. The reason being, the number concentration of ambient aerosol particles in these size ranges is low (compared to the concentration in sizes <0.3 µm) and may increase statistical errors. Further the dynamic behavior of the atmospheric aerosol spectrum affects the consistency of the results. However, if the measurements are targeted in ultrafine size ranges, atmospheric measurements may be used for a first-estimate or guiding purposes. Such an exercise can be adopted during initial stages of the development phase of an in-house filter media material/mask. However the dynamic nature of the atmospheric aerosol spectrum should be kept in mind and the test/exercise should be completed in a short fraction of time. We demonstrate one example where we measured the effect of sealing on an in-house mask. For this, indoor ambient atmospheric particles (number concentration: 11000–12000 #cm−3, geometric mean: 60–80 nm, geometric standard deviation: 2.3–2.6) were chosen as the test particles. Figure 5 shows the efficiency evaluated by the combined SMPS-OPC measured particle size spectrum using full mask sampler.

Demonstration of the effect of mask sealing on the jig on the capture efficiency for an in-house developed N-95 mask in the entire particle size range (0.01–3 µm).
The above figure shows the conventional U-shaped capture efficiency curve for an aerosol filter. While the minimum efficiency was measured at 89–90% for 0.2–0.4 µm particle sizes for face fix position, it improved to 94–95% for sealed position. This significant difference could be arising due to imperfections in the face fit and could be rectified by slight modifications in the design of the mask. This demonstrated the capability of such a laboratory set up during the R&D stages.
Steps for face mask testing to obtain first level assessments
Based on the studies carried out, we evolved a set of guidelines for carrying out the performance evaluations of the face masks/respirators in the laboratory, before approaching a certifying agency. These are presented below. Check the specified characteristics of the filter media/mask material at an appropriate test condition. A set-up similar to ‘intrinsic sampler’ can be used for this purpose. We measured the intrinsic capture efficiency and the pressure drop of the filter material in our setup as first step. Any such set-up should be tested for leakage, presence of background particles and isokinetic sampling before the measurements. The material should pass the ‘set specifications test’ or the ‘standard reference test’ during this step. Perform the leakage test of set-up intended to test full masks at the test conditions. We developed ‘full mask sampler’ for this purpose and tested the employability by following standard aerosol sampling protocols and utilizing a comparison of fit factor with a commercial instrument. In absence of the latter, full mask sampler should be thoroughly tested for any leakage paths (zero testing), representative sampling etc. For design of mask, perform a ‘jig fit test’ using ambient atmospheric aerosols as test particles. This should give the first hint of efficacy of the mask design. Perform test experiments with the set-up made for the full mask testing. In this step, test conditions should be taken as per any standard testing reference document or an in-house testing methodology. We used 3 test specimens viz. N-95 respirator, surgical mask and cloth mask for conducting experiments in full mask sampler. Send the tested masks to authorized certification centers after attaining the set specifications.
Conclusions
We have presented a quick laboratory methodology for testing the effectiveness of face masks/respirators in terms of their particle capture efficiency. The set ups developed for the same are simple and can be put together quickly in a standard aerosol laboratory. An ‘intrinsic sampler’ was developed and employed for testing the material of the masks. A glass mannequin was fabricated and used as a ‘full mask sampler’ for testing of full masks. A provision to place the mask on the mannequin in ‘face fix’ and ‘sealed’ position was made with the full mask sampler. Three test specimens viz. commercial N-95 respirator, surgical mask and a cloth mask was used for demonstration and validation experiments. The primary parameter of investigation in these experiments was aerosol capture efficiency of filter media/mask. Experiments were performed at a carrier flow of 28.3 Lmin−1 while adopting an atomizer for the generation of test particles. Optical particle counter was used to measure the number concentration of test particles in the size range >0.3 µm. Scanning mobility particle sizer and condensation particle counter were also used during the experiments for specific purposes. In the first level, testing experiments with intrinsic sampler, average capture efficiency for 0.3 µm particles was measured as 96.19, 40.08 and 14.22% for N-95, surgical mask and cloth mask, respectively. Fit factor estimated with full mask sampler was validated with a commercial instrument in the second step.
The set-up is intended to optimize the time needed to develop the filtration media/mask with desired specifications and to the performance in the design and development phase itself. As an example, any significant difference in filtration characteristics measured for the media (intrinsic sampler) and the full mask in sealed fixture (full mask sampler) indicates a leakage through the joints, seams etc. The set-up can be tuned for stricter compliances as per the context and the need. Such a set up will help the mask developers to improvise their product, before finally approaching the certification agencies. This will be helpful to reduce the time gap between the development and the finally certified product ready for commercialization. This is utmost important in times of urgency as is due to COVID-19 pandemic. The test rigs and the methodology as used in this work are simple and quick make-shift arrangements and can be adopted for first level assessment of filtration effectiveness of masks. Local laboratories with the know-how of checking the performance of non-certified masks and assisting the mask developers before the certification stage could be an important step for the near future. Additional inferences on the textile characteristics can be utilized in order to improve the performance. This can also be a helpful strategy in the general context of severe air pollution management.
Footnotes
Acknowledgements
The authors acknowledge the motivation and support given by Shri R. M. Suresh Babu, Director, Health, Safety and Environment Group, BARC for carrying out this work in the laboratory and for giving suggestions to improve the manuscript. We greatly appreciate the gesture of Mr. Nishant Mittal, TSI Instruments India Pvt. Ltd. for making the Portacount instrument readily available to us for a short duration during the course of these experiments.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


