Abstract
The aim of this research was to prepare composite polymer particles containing bismuth vanadate (BiVO4) particles through microsuspension iodine transfer polymerization (ms ITP) for fabric coating as a self-cleaning fabric. To reduce the aggregation of pristine BiVO4 particles during the fabric coating process, composite polymer particles containing the visible-light-driven photocatalyst as BiVO4 particles in self-cleaning fabric applications were investigated in the first time. First, BiVO4 particles were prepared via an aqueous chelating method where the stable precursor solutions of Bi3+ and V5+ with ethylenediaminetetraacetic acid ligand were obtained. After calcination at 500 °C, the BiVO4 particles were obtained. To disperse them well in an oil (monomer) phase in ms ITP, the BiVO4 surface was modified by oleic acid as o-BiVO4 to present a hydrophobic surface. The encapsulation efficiency of the o-BiVO4 (≥60%) in composite poly(methylmethacrylate-divinylbenzene) (P[MMA-DVB]/o-BIVO) particles was significantly higher than that (≈10%) of the pristine BiVO4 particles. Using polyethylene glycol 30 dipolyhydroxystearate (DPHS) as a porogen, porous P(MMA-DVB)/o-BiVO4 particles still maintaining their spherical shape were obtained with an 8% particle of DPHS. Furthermore, increasing the hydrophilic polymer shell by adding 2-hydroxyethyl methacrylate (HEMA) in the oil phase of ms ITP, the P(MMA-DVB-HEMA)/o-BiVO4 particles showed a much higher methylene blue (MB) degradation rate under visible light for 1 h (24 mg MB/g BiVO4 or 96% MB degradation) than that (13 mg MB/g BiVO4 or 52% MB degradation) of the pristine BiVO4. Moreover, the fabric coated with porous P(MMA-DVB-HEMA)/o-BiVO4 particles showed a satisfactory self-cleaning property.
Introduction
Recently, photocatalysis has been one of the most popular techniques for the removal of organic dyes and antibacterial applications [1–8]. For self-cleaning fabrics especially dye degradation, one of the most popular materials coated onto the fabric are photocatalytic materials due to low cost and nontoxicity. Under the light radiation, an electron-hole (h+) containing a positive charge is generated in the valence band (VB) by the electron transition from the VB to the conduction band (CB). The h+ can oxidize the water, providing a hydroxy radical (•OH), whereas the electron from the CB can reduce oxygen, providing a superoxide radical (•O2−), which further reduces water and provides the •OH in the final product as well (Figure 1). The •OH is well known as an oxidizing agent with antibacterial potential [1,2].
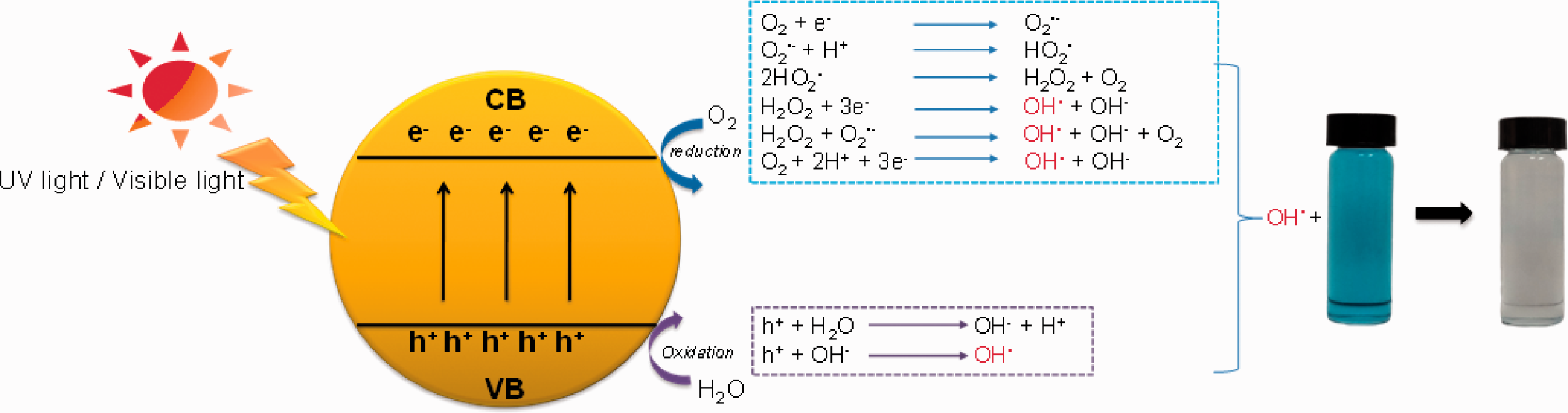
Schematic diagram of the organic dyes degradation mechanism with photocatalytic materials under UV or visible light irradiation.
Therefore, under presenting water and light radiation, the generated •OH can degrade dye [3–8] and present an antibacterial property [4,5]. The metal oxide with UV light such as Titanium dioxide (TiO2) [3–7] including its composite as RB-21/TiO2 nano-sol [9], and ZnO nanoparticles [10] were then used in textile applications as the self-cleaning fabrics. However, UV radiation is only 5% in solar radiation, which is much lower than visible radiation (46%) [11]. Using photocatalytic materials with visible radiation may have more potential than with UV radiation. Therefore, several studies have been done with various composite materials such as Pt-TiO2 [12], TiO2/TCPP (porphyrin derivative) [13], SnO2−x/GO-5 [14], and graphene oxide (GO) - carbon nitride (CN) hybrid materials [15] to use visible light for photocatalytic applications. However, it was not easy to control morphology. The common visible light photocatalytic material as bismuth vanadate (BiVO4) [16–24] is a good choice to use for self-cleaning fabrics due to simple preparation. Moreover, BiVO4 is a non-toxicity and compatibility for the human cells [25–27]. It has a potential antibacterial agent [28–30] and protein kinase inhibitor considering anticancer therapies [25]. However, when photocatalytic particles are used for fabric coating, the particles are unstable in the dispersed medium and coagulate easily, especially in long-term storage. They are difficult to redisperse in the aqueous medium after drying. Moreover, they do not disperse well during the coating process. Although the alternative way as in situ synthesis on the fabrics were widely reported [31,32], it still needed more study to upscale.
Preparation in the form of a capsule or composite hybrid particle would be a good solution to overcome the above-mentioned drawbacks. One of the most high-performance techniques for microcapsule preparation is microsuspension polymerization via conventional radical polymerization which is a useful technique in an industry, where the capsule shell is produced from the polymer [33–39]. It provides high encapsulation efficiency if the encapsulated materials present more hydrophobicity and hydrophilicity than the polymer shell for oil-in-water (O/W; normal microsuspension polymerization) and water-in-oil (W/O; inverse microsuspension polymerization) emulsion systems, respectively. The polymerization loci in the monomer droplets consist of a monomer, the material core, and an initiator that disperses in the continuous phase containing an emulsifier. During the polymerization, internal phase separation takes place, in which the portion containing lower interfacial tension in the continuous phase moves to the interface and leaves the higher ones in the core. Because the material core hardly dissolves in the continuous phase, the obtained microcapsule is quite large. However, if a hydrophilic monomer is used, a large amount of free particles form in the continuous phase based on the exited radical from the monomer droplet/polymerizing particle [34]. To overcome this drawback, iodoform was added to the polymerization as the hydrophobic chain transfer agent, called microsuspension iodine transfer polymerization (ms ITP) [40,41]. The radical exited significantly depressed, providing a small amount of free particles. Thereafter, this technique was useful for the encapsulation of various material cores with various polymer types [42–45]. To encapsulate the metal oxide into the capsule in the O/W emulsion system, the surface of the metal oxide was modified to present hydrophobicity before envelopment with the polymer shell [46–48]. The encapsulation efficiency significantly improved [48].
In this work, a polymer particle containing BiVO4 was prepared by ms ITP using a hydrophilic polymer for the first time. It is well-known that the photocatalytic activity of the encapsulated BiVO4 has been done with visible light and water. To improve the diffusion of water into the composite particle, both hydrophilicity and porosity of the composite polymer particle were designed. The more hydrophilic polymer shell as 2-hydroxyethyl methacrylate (HEMA) was implemented to be the copolymer shell of the composite polymer particle for the first time. Moreover, the low HLB value as polyethylene glycol 30 dipolyhydroxystearate (DPHS) was incorporated in the monomer droplet during ms ITP to form the porous in the composite particle. Furthermore, the obtained composite polymer particle was used for dye treatment. The self-cleaning performance of fabric coated by composite polymer particles was also studied.
Experiment
Materials
MMA (Aldrich, 99% purity) was purified by passing through a column packed with basic aluminum oxide. DVB (Aldrich, 80% purity) was washed with 1 M sodium hydroxide (NaOH; BDH, Prolabo, 97% purity) solution to remove the polymerization inhibitors before use. Reagent-grade benzoyl peroxide (BPO; Merck, 75% purity) was purified by recrystallization in methanol. Poly(vinyl alcohol) (PVA; Aldrich, degree of saponification 87–90% and molecular weight 3–7 × 104 g mol−1) was used as received. Toluene (RCI Labscan, 99.5% purity), oleic acid (OA) (Aldrich, 90% purity), hydroquinone (Sigma-Aldrich, reagent plus, 99% purity), ethyl alcohol (Duksan, AR grade, 99.9% purity), iodoform (CHI3; Aldrich, analytical reagent, 99% purity), hexane (RCI Labscan, analytical reagent, 99% purity), PEG 30 dipolyhydroxystearate (DPHS; Croda, technical grade), ethylenediaminetetraacetic acid (EDTA; Vetec, reagent grade, 98% purity), ammonium metavanadate (NH4VO3; LOBA Chemie, analytical reagent, 98% purity), bismuth (III) nitrate (Bi[NO3]3·5H2O; Aldrich, reagent grade, 98% purity), ammonium hydroxide (AppliChem Panreac, analytical reagent, 30% purity), and methylene blue (MB; Unilab, laboratory reagent, 100% purity) were used as received.
Preparation of BiVO4 nanoparticles
Bismuth vanadate (BiVO4) nanoparticles were prepared via an aqueous chelating method based on previous works [49]. Briefly, the precursor solutions of Bi3+ and V5+ were separately prepared by dissolving 0.05 mol of Bi(NO3)3·5H2O and NH4VO3 in 50 ml of EDTA solution (1.0 M; 0.10 mol), respectively. After mixing both solutions, where the mole ratio of Bi3+: V5+: EDTA was 1:1:4, the pH solution (final volume of 100 ml) was adjusted to 8 by addition of NH4OH. Thereafter, the water was evaporated at 100 °C before the dried powder was calcined at 500 °C for 6 h to obtain BiVO4 particles.
Preparation of polymer composite particles containing BiVO4
Before encapsulation of BiVO4 particles, their surface was modified to present hydrophobicity by coating with OA (o-BiVO4). The 5.0 g BiVO4 particles were dispersed into a mixture solution of toluene (20 g) and OA (10 g). Thereafter, the dispersion was transferred to a round-bottom flask, sealed with a silicone rubber septum and purged with a vacuum/N2 cycle five times (finally in N2) and stirred at 200 rpm at 70 °C overnight before toluene evaporation. Polymer microcapsules containing BiVO4 nanoparticles were prepared by ms ITP (the recipes are shown in Table 1). First, accurate amounts of MMA and DVB (with and without HEMA) as the monomers, CHI3 as the chain transfer agent, and BPO as the initiator (with and without DPHS as porogen) were homogeneously mixed as an oil phase before dispersing the pristine and modified BiVO4 nanoparticles. Thereafter, the dispersion oil phase was poured into a PVA aqueous solution (1 wt%). The monomer droplets containing BiVO4 nanoparticles dispersed in an aqueous phase as an O/W suspension were produced by applying a high shear rate with homogenization of 5,000 rpm for 5 min. The obtained suspension was subsequently transferred to a round-bottom flask, sealed with a silicone rubber septum, and purged with a vacuum/N2 cycle five times. The suspension was finally polymerized at 80 °C for 3 h followed by 90 °C for 5 h. The schematic diagram for the preparation of porous composite polymer particles was shown in Figure 2.
Reagent amount for the preparation of polymer particles containing BiVO4 nanoparticles by ms ITP.

M1: MMA, M2: DVB.
aDPHS.
bPristine BiVO4.
co-BiVO4.
d0.25 g DVB and 0.25 g HEMA.

Schematic diagram of the preparation of porous composite polymer particles using DPHS.
Dye treatment
The MB solution of 5 mg/l (CMB,0) with 50 ml (VMB) was used as an artificial dye. Accurate weights of various kinds of polymer particles and BiVO4 such as P(MMA-DVB), pristine BiVO4 nanoparticles, P(MMA-DVB)/o-BiVO4, P(MMA-DVB)/o-BiVO4/DPHS, and P(MMA-DVB-HEMA)/o-BiVO4/DPHS were separately added to the MB solution with a mild stirring rate under visible light (EVE X-FIRE 25 watt). The MB degradation efficiency in terms of mg MB/g BiVO4 of the various kinds of materials was investigated by measuring the remaining MB in the solution at various times. Before MB measurement, each MB solution containing polymer composite particles was centrifuged, and the appropriate amount of supernatant MB solution was withdrawn. The remaining MB concentration (CMB,i) was measured by UV-visible spectrophotometry (UV/Vis, Lambda 35, PerkinElmer Inc., America) at the wavelength of 663 nm in which the absorbance (Abs) of MB in each sample was extrapolated in the calibration curve of the standard solution of MB (1–5 mg/l). The degradation amount of MB (WMB) in terms of mg MB was obtained through equation (1), whereas the MB degradation efficiency (DE) was presented in terms of mg MB/g BiVO4 calculated by equation (2).

Self-cleaning fabrics
The 100% cotton knit fabric with a structure of interlock having a fabric weight of 237 g/m2 including wale per inch (WPI) and course per inch (CPI) of 36 and 35, respectively, was coated with polymer composite particles using the Dip-Pad-Cure process as follows. The suspension of the polymer composite particles was mixed with a special self-crosslinking polyether polyurethane binder (Nano-PU), where the concentrations of the polymer composite particles and the binders were 2.5 and 0.3 wt%, respectively, in the final volume of 100 ml. Thereafter, fabric with a size of 20 cm × 20 cm was immersed in the prepared suspension (dip) and left for 2 min before compressing (pad), curing at 120 °C for 3 min. The schematic preparation was shown in Figure 3. The add-on efficiency (%) of the polymer composite particles was measured by gravimetry and calculated by equation (3).

The schematic diagram of the coating of polymer composite particle on the cotton fabric for self-cleaning fabric preparation using dip-pad-cure.
Besides, the durability test of the coated fabric was studied based on AATCC 135–2004 method. The triplicate accurate weight of the coated fabric was separately included with other clothes to obtain the final cloth mass of about 2 kg. Thereafter, the detergent amount of about 66 g was added. The clothes were washed at various times by the washing machine using a normal mode. The performance of the coating of polymer composite particle on the fabric was investigated in the term of percent weight loss calculated from equation (4).
When %Add-onb (g) and %Add-ona (g) were the %Add-on of polymer composite particles before and after washing, respectively.
Characterizations
The particle size and morphology of BiVO4 nanoparticles were determined by dynamic light scattering (DLS, Delsa Nano C, Beckman Coulter, Germany) and transmission electron microscopy (TEM, Tecnai 20, Philips, the Netherlands), respectively. The BiVO4 particles (ca 10 wt%) dispersed in the dodecyl sulfate (1 wt%) solution were measured by DLS with the concentration mode at a light scattering angle of 165° at room temperature. Before TEM observation, each BiVO4 particle suspension was diluted to approximately 50 ppm. Several drops of the suspension were placed on a carbon-coated copper grid, which was then dried at room temperature in a desiccator before measurement. The crystallinity of the BiVO4 nanoparticle powder was measured by X-ray diffraction. The chemical structure of the o-BiVO4 was investigated by Fourier transform infrared spectroscopy (FTIR, Nicolet™ iS™ 5, Thermo Fisher Scientific Inc., America) using a frequency range of 400–4000 cm−1. The dried o-BiVO4 particles were washed 3 times with ethanol to remove the free OA from the BiVO4 surface before FITR measurement. The polymer composite particles were observed with an optical microscope (OM, SK-100EB & SK-100 ET, Seek Inter Co. Ltd., Thailand) and a scanning electron microscope (SEM, JSM6510, JEOL, JEOL Ltd., Japan) to investigate the inner structure of the microcapsules and the morphology of the surface, respectively. For SEM observation, the dried polymer composite particle was placed on a nickel SEM stub and coated with Au. Percent conversion and total BiVO4 contents or loading experiments (LE) in polymer composite particles were measured by gravimetry. The polymer composite particle suspensions (ca 1.5 g) were transferred directly to a crucible and weighed. Before drying at 80 °C, several drops of hydroquinone solution (1 wt%) as the inhibitor were added. After obtaining constant weight (Wd), the dried polymer composite particles were subsequently calcined at 500 °C for 10 min and weighed (Wc). The weight difference (Wd–Wc) between dried polymer composite particles and particles after calcination was the polymer weight including a residual surfactant, CHI3, and initiator. The monomer conversion was obtained by comparing the weight of dried polymer excluding residual substances with that of the original monomer. Total BiVO4 contents (Wc or Wbivo4) or percent loading experiments (LE) in polymer composite particles were determined by comparing the weight of the residual BiVO4 after burning with the weight of the dried microcapsule [(Wc/Wd) x 100]. Based on the calculation of theoretical BiVO4 loading in the polymer composite particles as shown in equation (5), encapsulation efficiency in terms of percent encapsulation (%EE) was obtained from equation (6).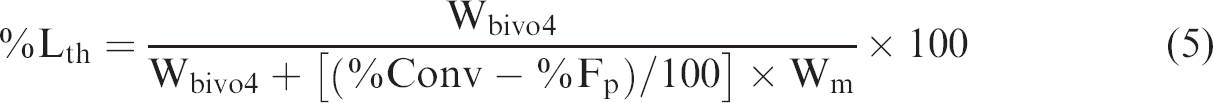

Results and discussion
Preparation of BiVO4 nanoparticles
The BiVO4 nanoparticles were prepared via an aqueous chelating method [49]. In the alkaline solution, four carboxyl groups of an EDTA molecule were ionized, giving four negative charges of carboxylate ion. They could capture the Bi3+ and V5+ cations in the confined space of EDTA molecules where the yellow transparent solution without any precipitation was obtained, as shown in Figure 4(a). After water evaporation and calcination, the yellow powder of BiVO4 (Figure 4(b)) was obtained. The prepared BiVO4 particles were measured by DLS by dispersing in SDS (1 wt%) solution to provide good dispersion during the measurement. However, it would be difficult to prevent the particle agglomeration in which the measuring size was in sub-micrometers (≈500 nm) because the BiVO4 particles prepared by this method should be nanometer-sized. However, the particle size distribution curve was not very broad, with a PDI of 0.32 (Figure 5(a)). Moreover, regarding the XRD patterns of the prepared BiVO4 (Figure 5(b)), all of the diffraction peaks agreed with the XRD patterns of standard BiVO4 (JCPDS Card number 01-083-1699) [50]. The major characteristic peaks observed at both 28.8° and 28.9°, respectively, evidenced the monoclinic BiVO4 structure possessing a good photocatalytic property [51,52].

Photos of precursor BiVO4 solution (a) and BiVO4 powder (b) after calcination.
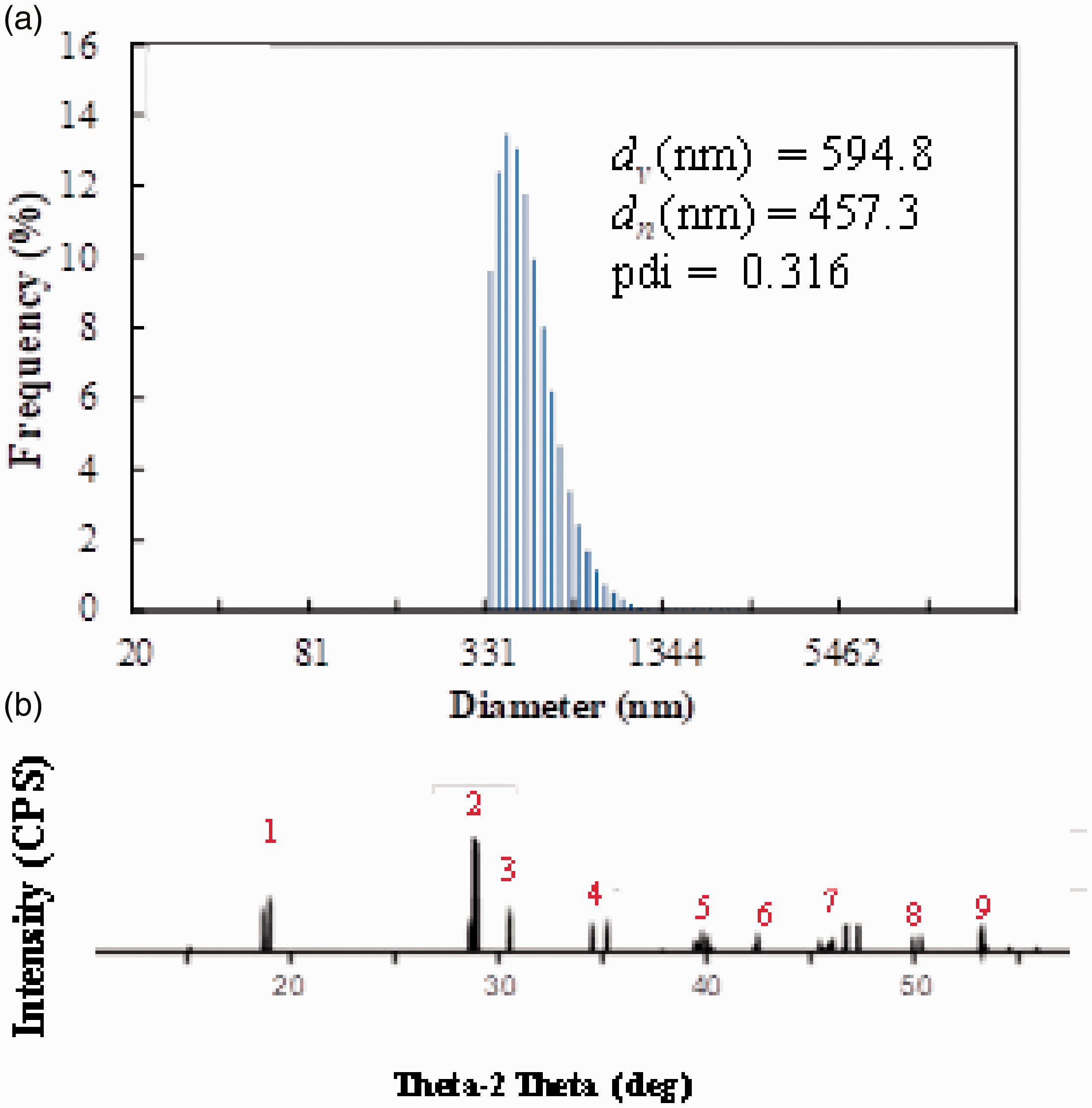
DLS histogram (a) and XRD pattern (b) of BiVO4 nanoparticles.
Preparation of polymer composite particles containing BiVO4
Before ms ITP of polymer composite particles containing BiVO4 particles, the surface of BiVO4 particles should be modified to provide enough hydrophobicity to disperse in the monomer phase. It is well-known that the polymerization loci of suspension polymerization including ms ITP are in the monomer droplet. In this work, the polymer composite particles were prepared in the O/W emulsion system. Therefore, to incorporate BiVO4 particles into the polymer particles, the BiVO4 particles must disperse well in the oil phase or monomer droplet during the polymerization. OA with an HLB value of about 1 is normally used as an emulsifier in an inverse (W/O) emulsion system [53] or surface coating of metal oxides [48,54–56] to improve their hydrophobicity. Therefore, in this work, BiVO4 particles were coated with OA. The FTIR technique was then used to confirm the coating performance of OA, as shown in Figure 6. The FTIR spectra of BiVO4 particles, OA, and o-BiVO4 particles are shown in Figure 3(a) to (c), respectively. The two main peaks of OA were observed at 2854 and 2923 cm−1, corresponding to the symmetric and asymmetric -CH2 stretching, respectively, whereas the carbonyl peak of the carboxylic group was observed at 1,710 cm−1 [57,58]. All of them were observed in the spectrum of o-BiVO4. This indicated that the BiVO4 particles were successfully coated with OA.
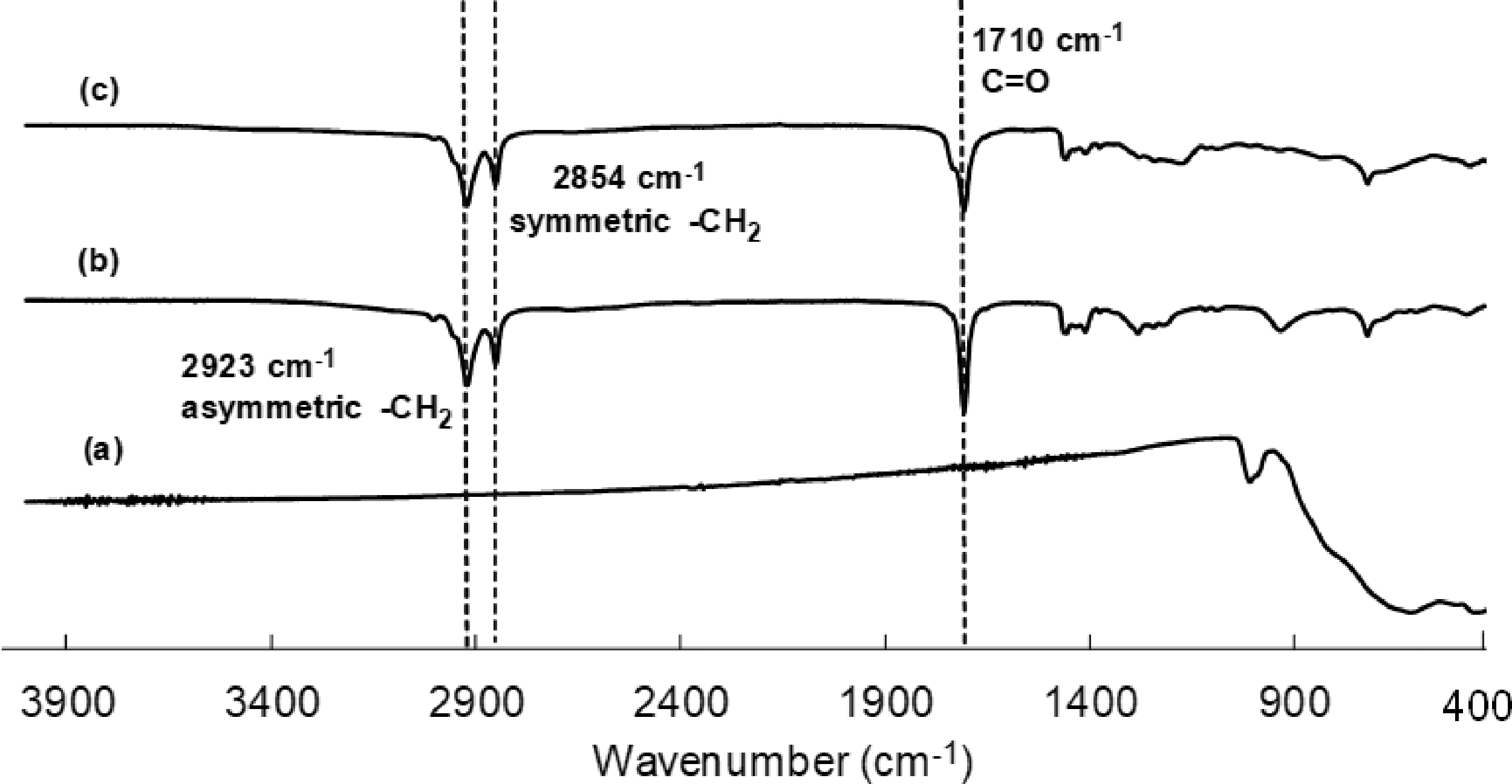
FTIR spectra of BiVO4 (a), oleic acid (b), and o-BiVO4 particles (c).
In previous works [34,40,41], a large amount of free particle in microsuspension polymerization formed competition with microcapsules or particles when hydrophilic monomers were used. This phenomenon is based on the radical/oligomeric radical exited from the polymerizing particles in the early stage of the polymerization and formed new particles in the aqueous phase. Although CHI3 of ms ITP (in this work) could depress this phenomenon it seems to work well only the moderate hydrophilic monomer as MMA. The performance of CHI3 was reduced with the more hydrophilic monomers [41]. Therefore, based on the previous work [42], low temperature at 80 °C was used for 3 h (early stage of the polymerization) to reduce the generation of the radicals. After the internal viscosity of polymerizing particle increases (with the conversion) the oligomeric radical would difficult to exit from the polymerizing particle. Thereafter, the temperature was then increased to 90 °C (5 h) to shrink the polymerization time. The influence of the OA coating on the encapsulation of BiVO4 particles in ms ITP was studied by comparison between the o-BiVO4 and the pristine BiVO4 particles using P(MMA-DVB) as the polymer shell. The blank sample of the P(MMA-DVB) particles (Run 1) was also synthesized. After the polymerization, suspensions without any coagulation were obtained for all conditions. White (Figure 7(a)) and yellow (Figure 7(b) to (c)) milky suspensions were obtained from P(MMA-DVB) and polymer composite as pristine BiVO4 (Run 2) and o-BiVO4 (Run 3) particles, respectively. After centrifugation at 3,000 rpm, all micrometer-sized particles were precipitated. Transparent supernatant was obtained for P(MMA-DVB) (Figure 4(a’)), whereas a white, cloudy solution containing a few free particles (1 wt%) with a size of about 80 nm was observed for P(MMA-DVB)/o-BiVO4. This indicated that the iodine chain transfer agent showed a good performance to trap the oligomeric radicals, preventing the radical from exiting the polymerizing particles [40,41,44]. Moreover, most of the o-BiVO4 was incorporated inside the precipitate microparticles. In contrast, some of the pristine BiVO4 particles (Figure 7(b)) still dispersed in the supernatant as a yellow solution was observed. The more hydrophobic surface of BiVO4 provides more potential to incorporate BiVO4 particles into the composite particles. The results agreed with OM observation, as shown in Figure 8. In all cases, monomer droplets (Figure 8(a) to (c)) and polymer composite particles (Figure 8(a’) to (c’)) were spherical, with a size range of about 5–20 µm. It is well-known that the polymer particles/capsules prepared by suspension or microsuspension polymerization where the monomer droplet generated by mechanical high shear rate are broad particle size distribution. However, the monomer droplets and polymer particles were not different in both shape and size (Figure 8(a) and (a’)) with the absence of BiVO4 particles. When the pristine BiVO4 and o-BiVO4 particles were added in the ms ITP, most of the pristine BiVO4 particles as the dark part either escaped from the monomer droplet or coagulated outside the monomer droplet (Figure 8(b)). Therefore, only a few pristine BiVO4 particles (%EE of 10) incorporated inside the polymer composite particles (Figure 8(b’)). Using o-BiVO4 improved the hydrophobicity of the particle surface, most of them as the dark part existed from the monomer droplet generation (Figure 8(c)) to the polymerization finish (Figure 8(c’)), as high %EE (60%) was obtained. To confirm this assumption, the partitioning study [59–61] of both the pristine BiVO4 and o-BiVO4 was then investigated using the same ms ITP recipe without initiator and PVA. As seen in Figure 9(a), most of the pristine BiVO4 particles dispersed in the water (bottom) phase. In contrast, the monomer (top) phase contained all of the o-BiVO4 particles (Figure 9(b)). This indicated that the OA coating on the BiVO4 surface provided a good performance to retain o-BiVO4 in the oil (monomer) phase, resulting in o-BiVO4 located in the composite polymer particles as well. Therefore, o-BiVO4 was used for further study.

Aqueous suspension photos before (a-d) and after (a’-d’) centrifugation at a speed of 3,000 rpm of various polymer particles prepared by ms ITP: P(MMA-DVB) (a and a’), P(MMA-DVB)/BiVO4 composite particles (b and b’), and P(MMA-DVB)/o-BiVO4 composite particles (c and c’).

Optical micrographs of monomer droplets (a-c) and various polymer particles (a’-c’) prepared by ms ITP: P(MMA:DVB) (a and a’), P(MMA:DVB)/BiVO4 (b and b’), and P(MMA:DVB)/o-BiVO4 (c-c’).

Binary phase photos of partitioning study of o-BiVO4 (a) and m-BiVO4 nanoparticles.
As mentioned in the introduction, BiVO4 could generate •OH under radiation with the existing H2O. To enhance the catalytic property of BiVO4 encapsulated inside P(MMA-DVB) particles, a porous polymer composite would be a good way to increase the total surface area. It is well known that low molecular weight nonionic emulsifiers can incorporate inside polymer particles [59–62]. Because they contain the hydrophilic part in their molecules, the incorporated emulsifiers could absorb water during polymerization at a high temperature, which finally provides a hollow, multi-hollow, and porous structure particle depending on the emulsifier content. In previous work [44], DPHS as a polyhydroxystearate (PHS)-polyethylene oxide (PEG)-polyhydroxystearate (PHS) block copolymer containing two lipophilic segments end-capping both sides of a large hydrophilic segment showed potential to produce a porous structure of the microcapsule containing fragrance. The microcapsule shape changed with the amount of DPHS. At a high concentration of DPHS in the microcapsule, DPHS molecules diffused to absorb at the capsule particle/water interface in competition with the polymer chains, providing a DPHS domain at the interface, which resulted in porous particles. Therefore, to produce P(MMA-DVB)/o-BiVO4 composite porous particles, various amounts of DPHS were added in the oil phase before the polymerization. Figure 7 shows the particle morphology of P(MMA-DVB)/o-BiVO4 composite particles with various concentrations of DPHS as 0 (Run 3), 5 (Run 4), 8 (Run 5), and 10 (Run 6) wt% composite particles, observed by SEM. At all DPHS concentrations, spherical particles were obtained. The porosity increased with the content of DPHS, where a smooth surface of P(MMA-DVB)/o-BiVO4 without DPHS (Figure 10(a)) was observed. Moreover, not only porosity but also the size of the composite particles seemed to decrease with DPHS from 5 to 10% (Figure 10(b) to (d)). The PEO segment absorbed on the particle interface might supplement PVA chains for particle stabilization. However, at 10 wt% of DPHS (Figure 10(c)), much more porosity was formed, in which the composite particles could not maintain their shape, obtaining largely broken particles. Therefore, 8 wt% of DPHS was selected for further study. Because the hydrophobic P(MMA-DVB) was the polymer shell, a low diffusion rate of H2O molecules toward the incorporated BiVO4 would occur. To improve such a drawback, a more hydrophilic monomer such as HEMA was used as a comonomer (Run 7) of MMA and DVB with an optimal amount of DPHS as the P(MMA-DVB-HEMA)/o-BiVO4 composite particles.

SEM micrographs of P(MMA-DVB)/o-BiVO4 prepared by ms ITP using DPHS as a porogen at various amounts (wt% composite particle): 0 (a), 5 (b), 8 (c), and 10 (d).
Dye treatment with polymer composite particles
In this work, the MB solution was used as the dye model for the treatment performance of the obtained polymer composite particles. The MB treatment-time plots of various kinds of materials such as P(MMA-DVB), pristine BiVO4, P(MMA-DVB)/o-BiVO4, and P(MMA-DVB-HEMA)/o-BiVO4 particles are shown in Figure 11. The blank materials of the P(MMA-DVB) particles (Figure 11(a)) showed that they could not absorb MB (ca. ≈1 mg MB/g particle or ≈5% total MB) throughout the study. In the first 30 min, all materials dispersed in the MB solution without providing radiation, and particles containing more porous surfaces, such as P(MMA-DVB)/o-BiVO4 (Figure 11(c)) and P(MMA-DVB-HEMA)/o-BiVO4 (Figure 11(d)) particles, showed more potential to absorb MB (≈8 mg MB/g BiVO4) than (≈4 mg MB/g BiVO4) the pristine BiVO4 particles (Figure 11(b)). After visible radiation was applied for 30 min, the degradation rate of MB using both composite particles was significantly higher than that of the pristine BiVO4 particles. This may be due to the lower total surface area of the pristine BiVO4 based on the agglomeration. The MB degradation gradually increased and reached only 13 mg MB/g BiVO4 or 52% MB degradation. In the case of both composite particles with and without HEMA, the porosity seemed no different because the MB absorptions were almost the same before applying visible radiation. However, the P(MMA-DVB-HEMA)/o-BiVO4 containing more hydrophilic polymers provided more potential to degrade MB; MB degradation reached over 95% after applying visible radiation for 60 min. It was only 90% for the composite particles without HEMA. This result indicated that the HEMA segment was the main driving force to generate more •OH, where the H2O diffusion rate from the outside to the inside composite particles improved.

The MB treatment-time plots of various kinds of materials: P(MMA-DVB) (a), pristine BiVO4 (b), P(MMA-DVB)/o-BiVO4 (c), and P(MMA-DVB-HEMA)/o-BiVO4 (d) particles.
Self-cleaning cloth
The P(MMA-DVB-HEMA)/o-BiVO4 particles were coated on the cotton fabric using the Dip-Pad-Cure method. The composite particles better distributed on the fabric (Figure 12(a)) than the pristine BiVO4 particles (Figure 12(b)). This indicated that P(MMA-DVB-HEMA)/o-BiVO4 particles showed higher colloidal stability and exhibited good dispersion in the medium during the coating process. Moreover, the durability test of the P(MMA-DVB-HEMA)/o-BiVO4 particle coating on the fabric was studied in terms of percent weight loss using equation 4. It was found that the weight losses (%) were gradually decreased with the wash cycle which were 1, 2, 6, and 10% for 5, 10, 15, and 20 wash cycles, respectively. This indicated that the self-cleaning fabric was quite a high durability. For a self-cleaning study, the coated fabric (Figure 13(a)) with an 8% add on was quite yellow due to the composite particles containing yellow BiVO4 particles, whereas a white uncoated fabric (Figure 13(b)) was observed. The fabric after coating represented the yellowish seemed the drawback of the white fabric. However, if the yellow fabric was needed, it would be the new candidate of the colorants as the same as silver [63], gold [63,64], copper oxide [65], and iron oxide [66] nanoparticles. Importantly, it can degrade dye and also contains antibacterial property.

SEM micrographs of the cotton fabric coated by P(MMA-DVB-HEMA)/o-BiVO4 composite particles (a) and the pristine BiVO4 particles (b).

Photocatalytic MB degradation on fabrics coated with P(MMA-DVB-HEMA)/o-BiVO4 particles (a, a’, and a’’) compared with the uncoated fabric (b, b’, and b’’): before (a and b) and after (a’ and b’) MB staining, and after visible radiation for 4 h (a’’ and b’’).
After several drops (200 µl) of the MB solution (5 mg/l) were applied to the coated (Figure 13(a’)) and uncoated fabrics (Figure 13(b’)), wide areas of blue color were seen. After applying visible light for 4 h, the blue color on the coated fabric (Figure 13(a’’)) was slightly discolored, whereas the blue on the uncoated fabric (Figure 13(b’’)) changed a bit. This result showed that the fabric coated with P(MMA-DVB-HEME)/o-BiVO4 particles exhibited a self-cleaning property. Moreover, comparing with the other materials in the previous reports shown in Table 2, the self-cleaning fabric developed in this study represented quite high dye degradation performance and self-cleaning property.
Comparison of dye treatment performance of various photocatalytic materials.

GO: graphene oxide; CN: carbon nitride; MB: Methylene blue; MO: Methyl orange; RhB: Rhodamine B; GY: Gardenia yellow; NPs: nanoparticles.
Conclusions
Composite polymer particles containing BiVO4 particles were successfully prepared by ms ITP without any secondary free particles in the continuous phase. To improve the encapsulation efficiency, BiVO4 particles were coated with OA to increase the hydrophobicity on their surface. To improve the photocatalytic property of the incorporated BiVO4 particles, producing porosity in the composite particles and increasing the hydrophilicity of polymer shells were achieved using DPHS as a porogen and P(MMA-DVB-HEMA) as a polymer shell, respectively. The obtained composite porous P(MMA-DVB-HEMA)/o-BIVO4 particles showed good potential to degrade MB, with about 24 mg MB/g BiVO4 or over 96% MB treatment within 1 h under visible radiation. Moreover, after the cotton fabric was coated with the obtained composite particles, it worked well as a self-cleaning fabric. Based on representing the yellow including dye degradation and antibacterial properties, it would be the new candidate for the colorants of the textile applications.
Footnotes
Acknowledgements
Special thanks are due to Dr.Nanjaporn Roungpaisan and Asst.Prof. Sunee Hathaiwaseewong at Textile engineering department, Faculty of Engineering, Rajamangala University of Technology Thanyaburi for the useful suggestions of fabric properties and coating.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by research and researcher for industry (RRi) of Thailand Research Fund (TRF) (MSD60I0101) and STP Chem Solution, Co., Ltd. (given to C.K.).
