Abstract
In this study, the antibacterial fatliquor emulsion was prepared from castor oil bearing thymol (1, 2, 4 and 8% w/w) to be used in the leather production. The average particle size and zeta potential of the fatliquors were determined by zeta sizer. The thermogravimetric behaviors of fatliquor emulsions under dry air were analyzed in order to determine the weight losses when they are exposed to constant heating rate. After the emulsification process, fatliquor emulsions were applied into chrome tanned cattle leathers. Antibacterial activities of the fatliquored leather were tested according to standard test method for determining the antimicrobial activity under dynamic contact conditions against Gram-positive and Gram-negative bacteria. Minimum inhibitory concentration (MIC) analysis of emulsion was also determined. The bacterial reduction was measured as 99.9% against S. aureus and 98.6% against E. coli bacteria. MIC values obtained from emulsions against bacteria were determined as 6.25 μL/mL and 12.5 μL/mL respectively. The results of antibacterial tests showed that thymol was very efficient in the fatliquor emulsion against both Gram-positive bacteria and Gram-negative bacteria. Physical characterization of leather was also carried out with optimum fatliquor emulsion (4% thymol w/w) and the results showed satisfactory physical properties on the fatliquored leathers compared to similar leather made with commercially available fatliquors. The results of the study indicated that thymol loaded fatliquor emulsions with small particle size could be a promising solo fatliquor to provide functional properties such as physical, strength, morphological and antibacterial properties to leather as an efficient fatliquor compared to conventional fatliquors.
Introduction
The environmental awareness, competitiveness and increase in customer demands encourage the R&D activities for the development of functional products that are more ecological and cost-effective, shorten the process duration, increase the quality of leather. In the last decade the studies on fatliquoring agents have gained much interest to improve the leather properties. Fatliquoring process is a physicochemical process to ensure the necessary fiber isolation and prevention of fiber damage caused by friction of fibers by penetrating various oil emulsions into the leather three-dimensional woven structure to improve tanning properties and to provide suitable softness and flexibility properties by rotating drums [1]. In addition, the handling and various strength properties of the leather can be improved in this process by using different kind of fatliquors [2]. Vegetable oils are generally used in fatliquoring process owing to their renewable, biodegradable and low cost properties. They provide softness, fullness and physical and mechanical properties to leather [3]. Castor oil with its non-edible performance is one of the finest natural softeners, many different from other oils, depending on the structure of ricinoleic acid [4]. Double bond presents in ricinoleic acid (Figure 1) structure make oil suitable for various chemical reactions and modifications [5]. The hydroxyl group in the structure also provide higher viscosity and polarity that provides reactivity against leather fibers.

Molecular structure of ricinoleic acid.
On the other hand, this double bonds frequently found in other vegetable fatliquors can cause a white stain on the leather under UV light and thus affect the appearance and the use of the leather. However, hydrogenated castor oil is an important derivative of castor oil, which is obtained by catalytic hydrogenation of castor oil and has less unsaturated double bonds in oil molecules [6]. Although castor oil has excellent softening properties, it alone is not sufficient to increase the thermal and strength properties of the leather. Paraffin emulsions with long chain and low chlorine content are used in the leather production as synthetic fatliquors owing to their high emulsifying power, formation of rigid crosslinks and conferring such important characteristics as fullness to the leather and an increase in collagen stability [7,8].
In the leather production, there are several stages for an attack of microorganisms before hides/skins are turned into leather. Because of transport and a long-term storage, it is necessary to preserve the extremely sensitive hides/skins to avoid losses through deterioration caused by bacteria. Hides/skins are also very convenient for the growth of microorganisms due to its structure consists of water, protein, fatty materials, and mineral salts [9]. Microorganism growth on leather can cause protein deterioration, colour changes, strength loss and unpleasant smell [10]. Biocides as antimicrobial agents can be applied during the process to protect the leather fibers against either bacterial or fungal attack. However, the antibacterial agents used in leather production are toxic organics, such as phenolic active ingredients, which would harm the environment [11]. What concerns the leather industry is that chemical biocides are regularly utilized, but mainly to preserve hides during processing rather than to confer antimicrobial properties to the final products [12]. Furthermore, some of these biocides used in leather process have been restricted or prohibited due to human health and ecological problems [8]. Certain biocides like methyl isothiazolinone (MIT) and chlorisothiazolinone (CIT) when used in leather production behave as an irritant [13]. The use of 2-thiocyanomethylthiobenzothiazole (TCMTB), ortho phenyl phenol (OPP) and pentachlorophenol are restricted for leather chemicals due its carcinogenic activity and poor degradability [14]. Organotin compounds like dibutyl tin (DBT) used in leather may contain tributyl tin as an impurity which is highly toxic [10].
A promising possibility to overcome these difficulties is the improvement of an idea to eliminate bacteria. An ideal antibacterial treatment for leather should be safe and environmentally friendly. Plant-based antimicrobial compounds as preservative agents offer certain advantages over these harmful biocides [15]. These advantages include their biodegradability, efficacy at low concentrations and eco-friendly natural structure [16]. Thymol (C10H14O), also known as 2-isopropyl-5-methylphenol, is one of the natural preservative product (main monoterpene phenol) found in the essential oil extracted from to the Lamiaceae family (Thymus, Ocimum, Origanum, and Monarda genera), and other plants such as those belonging to the Verbenaceae, Scrophulariaceae, Ranunculaceae, and Apiaceae families [17]. Thymol has bactericidal and fungicidal properties and has also demonstrated antimutagenic effects in combination with other essential oils [18]. The phenolic components are primarily responsible for the bactericidal properties of thymol as shown in Figure 2.

Chemical structure of thymol.
Thymols’ active components, are generally recognized as being safe for human consumption in the United States, have caused researchers to examine its potential to improve its production in different areas such as the cosmetic industry as fragrance ingredient, food industry as a preservative against microorganisms, pharmaceuticals for its preservative properties, and products such as insecticides and other biocidal products [19]. Moreover, antibacterial textiles with improved efficacy and durability were produced by incorporation of nontoxic monoterpene thymol [20].
While the elimination of technical problems caused by a fatliquoring process in leather products has become the most basic expectation, the demands for improving leather properties in the process have increased. These versatile claims have brought a new dimension and technological approach to fatliquoring process. Technological approaches on the process are focused on the production of light, soft, flexible and antimicrobial leather products which do not show quality degradation such as bad smell and fatty spue staining with low environmental and human health risks [21]. Additionally, in general, about 85 weight % fatliquoring agent can penetrate into leather, and that which is unexhausted remains in the wastewater after the fatliquoring process. Therefore, the design of an ecofriendly fatliquoring agent and its application have attracted increasing attention in the leather industry [22].
In the present study, it was aimed to develop a novel and environmentally friendly synthetic and natural blend fatliquor emulsion with functional properties using for leather industry. In the emulsion, thymol was used as natural antimicrobial agents for the first time for determining the antibacterial leather properties without using any toxic chemicals in the production. Hydrogenated castor oil with fewer unsaturated double bonds was chosen as natural oil ingredient for its biodegradable character, superior softening properties and low probability of forming fatty spues. On the other hand, paraffin emulsion was preferred due to high emulsifying power, formation of permanent crosslinks, which will improve the exhaustion rate of emulsion and providing increasement in the thermal, and strength properties of leather. This study will also provide a necessary guidance for the reduction of many chemicals utilized in the fatliquoring/retanning process in order to obtain the required properties of finished leather by one innovative product.
Materials and methods
In the research, thymol (≥98.5%), hydrogenated castor oil, Span 80 and Tween 80 were obtained from Sigma-Aldrich Chemie GmbH. Cloparten 228 (26–28% chlorine content, 18–20 Chain length) were provided by Harke Chemicals. Wet-blue American Cattle hides with 1.0–1.2 mm were used in the leather production trial.
Preparation of fatliquor agents
For preparing O/W emulsion; Cloparten 228, hydrogenated castor oil, Span 80 and Thymol (1, 2, 4 and 8% w/w) as a dispersion phase were heated to about 50°C and mixed in a beaker equipped with a homomixer for 8500 rpm. At the same time, the ingredients of the continuous phase (deionized water and Tween 80) were heated in a separate beaker to about 50°C. After both beakers were mixed until the mixture became uniform. Then, the dispersion phase was placed to continuous phase while stirring. After adding this mixture, the emulsion was stirred for 10 min with high-speed homogenizer at 18.000 rpm. pH of the emulsions have been adjusted to pH 7 during homogenization. The hydrophilic lipophilic balance (HLB) value of the prepared fatliquor emulsion was optimized as 11.2 using Span 80 (HLB 4.3) and Tween 80 (HLB 15.0). The prepared emulsions were cooled to room temperature and this formed emulsion is simply dispersible in water.
Particle size and zeta potential analysis
Particle size and zeta potential of the prepared fatliquors were measured a Malvern Zeta Sizer Nano ZS. The measurement size range of the instrument was 0.1–10000 nm. Emulsion samples were prepared as 0.1 mg/mL concentration by diluting with the deionized water.
Thermogravimetric analysis
The thermal degradation behavior of the fatliquor sample was investigated by using Perkin Elmer TGA 8000 instrument. The samples were placed in ceramic crucible and the flow rate of dry air in the system was set at 100 mL min−1. The analysis of the samples was performed between 30–800°C with a heating rate of 10°C. The main degradation process of the samples was observed with the thermogravimetric (TG) curves.
Application of fatliquor agents
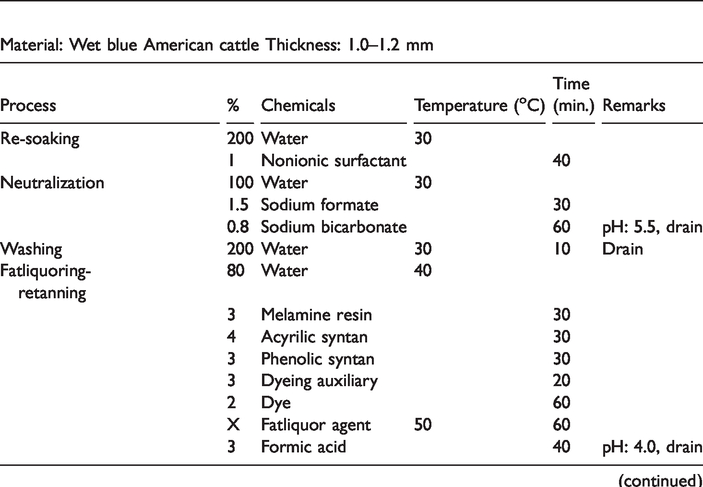
After preparation of thymol loaded fatliquoring emulsions were used on the leather production recipe with the percentage of 10, 15, 20. The recipe used in the production was given in Table 1. In the recipe, 30% more was added on the wet-blue weight, and the chemicals were calculated on the hides’ weight. After application of fatliquors, leathers were left to dry for two days. In order to compare the effects of emulsions on leathers, no fatliquoring emulsion was given to the control group leathers.
Leather production recipe.
Antibacterial activity tests
The antibacterial tests were carried out on fatliquored leather according to the ASTM Standard E 2149-01 against E. coli ATCC 23716 and S. aureus ATCC 6538. The inoculum was diluted to 0,5 McFarland turbidity standard (corresponding to a concentration of 1,5–3,0 × 108 cfu/ml) using sterilized Ringer solution. The concentration of bacterial dilution was also controlled by spectrophotometry by measuring the absorbance at 625 nm. This solution was used to prepare the working bacterial dilution employed in the assays by properly dilute it in a sterile buffer (0,3 mM KH2PO4, pH=7, 2±0, 1) to reach a final concentration of 1,5–3,0 × 105 cfu/ml. For antimicrobial activity evaluation purposes, fatliquored leather samples (2 cm×2 cm) were introduced in a sterile 250 mL flask containing 50 mL of the working bacterial dilution. The flask was then placed in a bath at 37 °C with orbital stirring. After 1 hour under stirring, 1 mL of the solution was aseptically collected to determine the bacterial concentration by standard plate count techniques with nutrient agar. The results of colony counting were converted to colony forming units per millilitre (cfu/ml) and used to calculate the bacteria reduction in percentage (%).
The percentage of colony number reduction was determined according to the following equation (1).
B-A= death rate constant
A = cfu per milliliter for the flask containing the treated substrate after the specified contact time
B = “0” contact time cfu per milliliter for the flask used to determine “A” before the addition of the treated substrate.
MIC of the prepared emulsion (4% Thymol) was determined against Gram-negative E. coli ATCC 23716 and Gram-positive S. aureus ATCC 6538 as representative microorganisms according to the broth dilution method with some modifications. Stock solutions of samples were prepared as 2 mg/mL using 0.5 McFarland turbidity standard (1 × 108 cfu/ml) of cultivated bacterial solutions were prepared and diluted in 5 × 105 cfu/ml. 10 µL cultivated bacterial solution in 100 µL of LB broth (Miller) was loaded into each well of 96-well plate followed by 110 µL of fatliquor emulsion. A negative control was prepared by adding 110 µL of LB broth without culture, The well cell culture plate was gently vortexed, and incubated for 24h at 37°C. MIC was determined as the lowest concentration showing no visible signs of microorganism growth.
Physical properties of fatliquored leathers
The manufactured leathers were subjected to the tests of shrinkage temperature (IUP 16), tensile strength (TS 4119 EN ISO 3376:2011), filling coefficiency (EN ISO 2589:2002), double edge tear load (TS 4118-2 EN ISO 3377-2), lastometer test for grain crack and grain burst (TS 4131). Prior to the tests, all leathers were conditioned according to the standard of EN ISO 2419:2012 and the sampling was done in accordance with the standard of EN ISO 2418:2002. Scanning Electron Microscopy (SEM) analysis was performed to control the changes in the leather fibers produced with fatliquor emulsions. For SEM analysis, leathers were first coated with gold by Cressington Sputter Coater and then SEM images were obtained by using TESCAN VEGA3 SEM device.
Results and discussion
Particle size and zeta potential of the fatliquors
Average particle size and zeta potential are important parameters in assessing the stability of emulsions [23]. In addition, the particle sizes of the fatliquors used in leather production determine the lubrication efficiency. As the particle size of the emulsions used in the fatliquoring of leathers decreases, the oil emulsions have more surface area per unit volume. Increasing the surface area promotes chemical reactions and increases the binding of the oil emulsions to the leather fibers [2]. The average particle size and zeta potential of the prepared fatliquor emulsions were given in Table 2.
Particle size and zeta potential of the fatliquors.

The results of the zeta potential analysis showed that all emulsions had high stability (ζ= –40.7 mV to –47.6 mV). The average particle size of the emulsions varied between 1.59 - 1.89 µm. The fatliquor emulsions used in leather production must have a particle size that can pass through collagen fiber pores in order to exhibit the desired performance properties. During collagen production, a few microfibrils form a fibril with a diameter of 0.2–0.5 μm. Together with the fibrils, a 3 μm diameter collagen strand is formed [24]. The deep penetration of the fibrous collagen structure of the fatliquor emulsions used in the fatliquoring process significantly increases the tensile and tear strength properties of the skin [25]. When the particle sizes of the emulsions were examined, it was thought that all emulsions would penetrate deep into the collagen matrix structure. They are also found electrostatically stable.
Thermogravimetric behavior of the fatliquors
The thermogravimetric behavior of the fatliquor emulsions was shown in Figures 3 and 4.

TGA curve of control emulsion (without thymol).

TGA curve of fatliquor emulsion containing 4% thymol.
TGA is one of the techniques for characterizing thermal and oxidative stability of fatliquor emulsion samples by measuring changes in the amount and rate of weight change in a material with gradual increase in temperature as a function of time in a controlled atmosphere. As shown in Figure 3, TGA curves show that oxidative degradation occurred in a three continuous step in the temperature range 120.44–728.38°C. In these temperature ranges, the mass losses occur at levels of 53.5% for control emulsion sample. From the results shown in Figure 4, oxidative degradation occurred in the temperature range 122.31–638.6°C with 33.66% mass loss. TGA combustion mechanisms of oils are still under investigation as vegetable oils ingredients are varying according to their origins and reaction severities and conversion amounts of triglycerides to monoglycerides result in different combustion behaviours. Oil samples generally exhibit two combustion reaction regions such as low temperature oxidation and high temperature oxidation steps [26]. During low temperature oxidation step the mass loss of free moisture, water and volatile substances [27]. When we examined the Figures 3 and 4 in detail, mass loss of control emulsion in low temperature oxidation started at 83.22°C and finished at 120.44°C with 41.447% and mass loss of thymol loaded emulsion occurred between 64.20–122.31°C with 61.649% weight loss. The primary reason for the differences in the initial temperature and percentage loss of mass of the first degradation step in the combustion behavior between these two emulsions is the presence of thymol’s volatile component in the emulsion.
Antibacterial test results
In the present day, where environmental and health problems are prioritized, natural products like active agent from essential oils are a promising and important alternative with their antimicrobial properties for the textile and leather industry. Thymol, are very effective ingredients to yield materials with antibacterial activity against both Gram-positive and Gram-negative bacteria [28]. The antibacterial effects of the leather treated with thymol loaded fatliquor emulsion with different percentage were shown in Table 3.
Reduction of microorganism growth on leather with fatliquors.
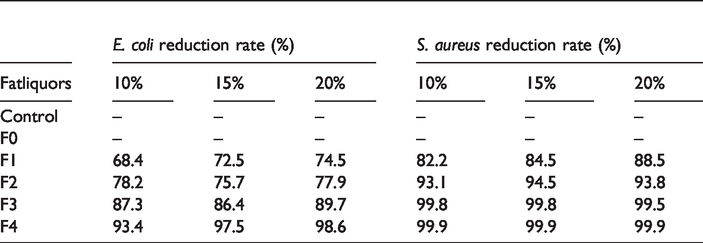
The effectiveness of the antibacterial emulsions regarding the dynamic shaking test in accordance with the standard ASTM E 2149-01 exists when the reduction of microorganisms is higher than 75%. From the results shown in Table 3, it can be noted that leathers fatliquored with thymol loaded emulsions (F1, F2, F3, F4) showed significant antimicrobial activity against two tested bacteria while control group leathers and leathers fatliquored with F0 did not show antibacterial activity. When weight percent of thymol increased in the emulsions, antimicrobial action of the thymol loaded emulsion was increased. However, it has been observed that when the amount of the emulsion used in the fatliquoring process increased, the reduction rate of microorganisms does not change much. Emulsion with 4% Thymol (F3) was chosen optimum fatliquor for providing antibacterial properties to leather against both bacteria. Emulsion with 4% Thymol (F3) showed satisfactory inhibitory effects regarding to the growth of both tested microorganisms, with MIC of 6.25 μL/mL. against S. aureus and with MIC of 12.5 μL/mL against E. coli. Rukmani and Sundrarajan [20] also declared that thymol was provided antibacterial properties to ungrafted and grafted fabrics against both E. coli and S. aureus by agar diffusion method. Inclusion of the antibacterial agent into grafted fabric showed an enhanced inhibition effect with durability after 10 cycles of washing. When we make a comparison between Gram-negative and Gram-positive bacteria, research has demonstrated that Gram-negative organisms are slightly less sensitive to thymol than Gram-positive bacteria as literatures revealed [29,30]. Thymol’s antibacterial activity is explained by the disruption of the bacterial cell membrane integrity, which leads to increased permeability to ATPases proteins [19]. Cyclic hydrocarbons are lipophilic and therefore are prone to accumulate in lipids. One of the possible mechanism where cyclic hydrocarbons act is accumulation of lipophilic hydrocarbon molecules in the lipid bilayer and distortion of the lipid-protein interaction and the second possibility is direct interaction of the lipophilic parts of the protein [31].
Determining the physical properties of leathers
Fatliquoring is one of the most important factors in changing the fibre molecular structure and so the state of physical, chemical and morphological properties of leather during the leather production. It is found that fatliquoring process lubricates and opens up the collagen fibre structure and thereby makes the three-dimensional collagen matrix more extensible. The amount and the type of fatliquor agent affect physical properties such as fullness, tensile and tear strength, elongation, water, air and air vapor permeability, absorption of water and electrical conductivity [32]. Shrinkage temperature, filling efficiency, tensile strength, elongation at break, double edge tear load, grain crack and grain burst analyses results of fatliquored leathers were shown in Table 4.
Physical properties of leathers fatliquored with thymol loaded emulsion.

From the results given in Table 4, it can be seen that the physical and mechanical properties of leather treated by thymol loaded emulsion were higher than control groups. Fatliquor emulsion improved the thermal stability of leather compared to the control group leathers. An increase in the fatliquoring proportion enhanced the thermal stability of leather. The formation of rigid crosslinks, making large cooperative units in the helix structure, enables high collagen stability in the leathers [33]. It has been reported that the paraffins used in the leather industry form covalent cross-links with the peptide groups of collagen, and that long hydrocarbon chains wrap around the fibers. In addition, they form intermolecular hydrogen bonds with the hydroxyl groups of hydroxyproline amino acids, which have a great effect on thermal stability [34]. Onem et al. [33] stated that the shrinkage temperature of collagen varied according to the number of intermolecular and intramolecular cross-links in its three-dimensional helix structure. Considering this statement, the greater hydrothermal stability of treated leather in comparison with control leather observed in the results of this study is to be expected.
From Table 4, it was observed that the increase in the tensile strength of the leather with the increase of the emulsion in the fatliquoring process. The tensile strength value of the leathers was observed in control group leathers as 13.5 N/mm2, while the highest strength value was obtained in leathers treated with 25% emulsion with 22.54 N/mm2. When the double edge tear load results were examined, there was a significant increase by 83.36% with the addition of emulsion into the leather. The penetration of the emulsions used in the fatliquoring process into the inner fibers of leather significantly increases the tensile and tear strength properties of the leather [35]. Covington and Alexandar [36] claimed that the influence of fatliquors on strength properties is considerable. Fatliquoring reduces the strength of the individual fibres but results in an increase in the bulk leather strength. This is attributed to the improved lubrication between the fibres permitting a more uniform distribution of the applied load. With the implication of fatliquors as a lubricant into the leather, the fibres and fibre bundles more easily slide internally and relatively to each other and so produce a lower stress under the tensile and tear force. Moreover, thymol loaded fatliquoring emulsion application improved the thickness and plumpness of skin matrix up to 13.46% when compared to control samples. Grain crack and grain burst properties of emulsion treated leathers also exhibited sufficient improvement.
From the Figure 5, it can be seen that the collagen fibre bundles of the fatliquored leather (Figure 5(b)) were evidently separated when compared to the control group sample (Figure 5(a)). The collagen fibres of fatliquored leather were effectively lubricated after thymol loaded fatliquoring emulsion treatment, and thus they were more likely to be oriented under external forces, which resulted in the increase of leatherʼs strength properties as shown in Figure 5.

SEM images of cross sections of (a) control group leather and (b) fatliquored leather.
Conclusion
In the leather production, three or more different commercial fatliquors are mixed and applied to the leather during the fatliquoring process to obtain the required leather properties such as softness, fullness and strength. Each of these different fatliquors provide different characteristics to the leather. This situation leads to increase the waste load of tannery effluent. Recent years, leather industry faces certain threat because of the upcoming stringent laws and regulations of waste disposal of toxic chemicals like some commercial antimicrobial agents. To overcome these difficulties, leather industry has to change its traditional production techniques. Use of natural products instead of harmful chemicals, reduction of different many chemicals in the processes are some of the alternative solutions in the production.
In this study, thymol loaded vegetable-based antibacterial fatliquor emulsions were prepared and applied in the fatliquoring process as solo fatliquor to wet-blue cattle hides and following results have been drawn: Thymol shows good inhibitory effect in the emulsion on bacterial growth and provides a good antibacterial effect on leathers against both organisms instead of dangerous synthetic biocides and the numbers of both Gram-positive and Gram-negative bacterial cells on leather treated with thymol loaded emulsions decreased with increased active thymol content. 4% thymol content in emulsion was chosen as an optimum percent for determining the satisfactory antibacterial effect. Prepared emulsions are found thermally and electrostatically stable and have a small particle size to penetrate efficiently into the collagen matrix. A single type of fatliquor emulsion, which is highly degradable and has been ecologically improved, was successfully applied to the leather with high exhaustion rate without any stability problem. Leathers produced with novel fatliquor emulsions showed satisfactory physical parameters such as tensile strength, elongation at break, double edge tear load, which generally a standard fatliquor parameter requires. Filling efficiency and shrinkage temperature values of treated leathers were found considerable compared to some retanning agents features.
The above findings revealed that fatliquoring with this novel fatliquor will provide an environmentally friendly process which overcomes the risk of use of many different fatliquors and commercial synthetic antimicrobials as well as their natural character. To the best of my knowledge, this is the first study in which the antibacterial activity of thymol as natural antimicrobial agents in fatliquoring emulsion has been demonstrated for leather industry.
Footnotes
Acknowledgements
The author would like to thank for bursary given by “Turkish Council of Higher Education”, “Fraunhofer Institute UMSICHT” for their facilities and support. The author also acknowledge the project of “Industrial Doctorate Program of Textile and Leather–2007 DPT 001” supported by T.R. Ministry of Development.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
