Abstract
In this paper, polyacrylonitrile (PAN) nanofibrous membranes (NFMs) were prepared by electrospinning technology. Copper (Cu) or/and zinc (Zn) nanoparticles (NPs) were deposited on the surface of PAN NFMs by spark ablation technology to fabricate a surface structure with antibacterial and hydrophobic properties. The deposition of NPs was controlled using current, voltage, carrier gas flow rate, and load time. The fabricated NFMs were characterized by FE-SEM, EDX, XRD, and water contact angle. Moreover, the antibacterial properties of the fabricated NFMs were studied using the agar-diffusion test method. The results showed that Cu, or/and Zn NPs successfully deposited on the surface of PAN NFMs, which improved the hydrophobicity by increasing the water contact angle from 46° to 128°, 130°, and 136°, respectively. Besides, the Zn@PAN NFMs sample was showed good antibacterial property against both Gram-negative and Gram-positive bacterial strains. Overall, the NPs@PAN NFMs based spark ablation technology might afford an easy-to-operated design pattern for constructing potential materials for water treatment applications.
Keywords
Introduction
Developed countries are still suffering from a lack of safe, fresh, and clean water due to contamination of water [1,2]. The existence of bacteria in water (e.g., E. coli, S. aureus, and B. subtlilis) is one of the major causes of water contamination, which is considered as a concern to the global economy because of their threat to human health [3,4]. Contaminated water can transmit diseases such as diarrhea, cholera, dysentery, typhoid, and polio; accordingly, 485 thousand people are estimated to die each year due to diarrhoeal only [5,6]. Therefore, with the aim of treating the water from such bacteria, it is very urgent to provide an inexpensive, durable, reliable, simple and robust disinfection approach by advancing an effective membrane for water treatment.
Polymeric membrane-based separation approach is the most useful disinfection strategy due to its self-standing, easy to package, and cost-effectiveness [7–9]. However, membranes for the disinfection of large volumes of water with high efficiency are still in the process of exploration [10,11]. Based on a polymeric membrane, various traditional disinfection approaches have been applied in water treatment such as filtration, chlorination, and ozonation [12,13]. Notwithstanding the fact that these approaches have high efficiency and multifunctional to treat the water, there are significant challenges faced when applying these methods due to the toxic disinfection byproducts, managing the spreading of illnesses, high operating costs and time-consuming. Recently, to overcome these problems, the progress in nanotechnology has provided reasonable clarification for meeting the current challenges.
Electrospun nanofibrous membranes (NFMs) have various typical structures, such as a high specific surface area, small pore size, tortuous porous structure, high porosity, unique mechanical properties, good flexibility, cost-effective production and easy to functionalization according to different application [14–18]. Electrospun polyacrylonitrile (PAN) NFMs offers promising properties, such as sufficient hydrophilicity, good mechanical strength, and low swelling rate [9,19–21]. Nevertheless, aforementioned advantages of PAN NFMs also bring the unfavorable factor-membrane fouling [22]. Researches indicate that bacteria and proteins contaminate PAN NFMs immediately during the filtration procedure, which led to resistant the chlorination and eventually provides severe membrane fouling that influences negatively on the permeability and the throughput of the membrane [23,24]. Additionally, likely secondary contamination, energy-consuming, and lower productivity, which does not only increase the cost of the filtration process but also decreases the life of the membrane [25,26]. Consequently, the functionalization of PAN NFMs to fabricate a simple, efficient and inexpensive membrane for water filtration remain challenging.
Metallic nanoparticles (M NPs) have been widely applied in medical, energy, environmental areas owing to their small particle size and high surface area. Among M NPs, the antibacterial properties of silver nanoparticle, known as the most common nanoparticle, are well recognized and numerous mechanisms for their bactericidal effects have been anticipated [27,28]. Although only a few studies have described the antibacterial properties of zinc and copper nanoparticles (Zn and Cu NPs), they have also shown Zn and Cu NPs to have a substantial role as bactericidal agents [29]. Besides, Zn, and Cu NPs are considered as an inexpensive antibacterial disinfectant [30–32]. M NPs have been combined with different NFMs by the suspension of M NPs directly into the polymer solutions to enhance their stability and disinfection ability [33,34]. At the same time, the NPs aggregation and dissolution diminish antimicrobial efficiency and limit their potential practical applications [35,36]. How to combine M NPs with NFMs together via environmentally sustainable methods remains challenge [37–39]. The electric nanoparticles generator currently has been used to deposition of NPs on the surface of polymeric materials due to its good environmental sustainability, low cost, low energy consumption, and high efficiency [40,41]. Nevertheless, little attention has been paid to applying the electric nanoparticles generator method to deposition the NPs on the surface of NFMs for water treatment.
In the present work, the electric nanoparticles generator method was used to deposit Zn, Cu, and Zn/Cu NPs on the surface of PAN NFMs. The surface morphologies and antimicrobial properties of the resultant Zn@PAN, Cu@PAN, and Zn/Cu@PAN NFMs were systematically examined. To the best of our knowledge, there is no previous report for applying the electric nanoparticles generator for deposition NPs on the PAN NFMs as a platform in any antimicrobial membrane for water treatment. Owing to outstanding properties of NFMs, the NPs were dexterously deposited successfully on the surface of PAN NFMs, which were achieved good antimicrobial properties. Due to the unique advantages, the fabricated NFMs may be used to solve the water treatment and disinfection of bacteria issues in practical engineering applications.
Experimental
Materials
PAN (Mw = 75,000) and N, N-Dimethylfomamide (DMF) were obtained from Shanghai Lingfeng Chemical Reagent Co. Ltd. Escherichia Coli (E. Coli) and Staphylococcus Aureus (S. Aureus) were purchased from Songon Biotech Shanghai Co., Ltd.
Preparation of the PAN NFM
Firstly, PAN NFM was fabricated by applying the Electrospinning machine (SS-2534) Yongkang Technology Development Co., Ltd., a homogeneous solution of 10 wt % PAN was prepared by dissolving PAN powder in the DMF by stirring for 8 h at ambient temperature (Scheme 1(a)). The electrospinning process was carried out under a constant temperature of 28 °C and a relative humidity of 30% using a syringe and a 21 G needle. The electrospinning parameters were set as follows: the voltage was 10 kV, the feeding rate was 1 mL/h, and the distance between the needle and the collector was fixed at 15 cm. The NFM was collected using polypropylene nonwoven fabric, and then the collected NFMs were dried in a vacuum oven at 70 °C for 24 h to remove the solvent.
Deposition of nanoparticles on PAN NFM
Nanoparticles generator instrument (VSP-G1, FuNa Scientific Instrument Co., Ltd) was used to synthesize multi-scale metal nanoparticles on the surface of PAN NFM. A circular sample of PAN NFM with a diameter of 47 mm was placed on the stage of the nanoparticle generator instrument (Scheme 1(b)). Two Cu and Zn electrode rods were connected to the positive and negative electrodes, respectively. A supply of electricity will generate sparks to form metal nanoparticle aerosols. The Cu and/or Zn NPs were deposited on the surface of the PAN NFM by diffusion and electrostatic deposition. The nanoparticles were deposited on PAN NFMs by adjusting the voltage, current and carrier gas flow rate to control the size to be 10 nm and 30 nm for Cu NPs and Zn NPs, respectively. After 15 min, the power and gas-flow were turned off. Then the carrier gas was cleaned to remove the remaining NPs from the instrument. In the case of loading double-layer nanoparticles, firstly, the Zn nanoparticles with a size of 30 nm were loaded on the surface of the PAN NFM. After 15 min, the power supply, and the gas-flow were turned off, and the Zn electrode rod was replaced to continue to load the Cu with a size of 10 nm on the surface of the PAN NFM. As listed in Table 1, three kinds of structures including Cu@PAN, Zn@PAN, and Cu/Zn@PAN NFMs were prepared.
Sample preparation.

Instrumentation
The morphology of NFM was analyzed by field emission-scanning electron microscopy (FE-SEM, S-4800, Hitachi, Japan). The FE-SEM images were analyzed using ImageJ software to get the diameter. The results were averaged from hundred replicates and presented as the average ± the standard deviation. The elemental compositions were analyzed by using energy-dispersive x-ray spectroscopy (EDX, JEM-2100F, JEOL Ltd, Japan), which was connected with the FE SEM. Wide-angle x-ray diffraction analysis was recorded with 2θ between 5° and 60° by an x-ray diffractometer (XRD; D/max-2550 PC, Rigaku, Japan), operating at a 1°/min scanning rate and along with Cu Kα radiation at 40 kV and 200 mA.
Antibacterial activity assessment
Evaluation of antibacterial properties of textiles-Part 1: Agar diffusion method “GB/T 20944.1-2007” was applied to qualitatively evaluate of antibacterial properties of the PAN and NPs@PAN NFMs. A bacterial suspension (E. Coli ATCC25922 and S. Aureus ATCC 25923, Shanghai, China) of 400 µL (1 × 108 CFU/mL) was evenly spread on agar plates using sterilized swabs. All the NFMs samples were cut into a circle with a diameter of 1 cm and then placed on agar plates containing a suspension of bacteria at 37 °C for 36 h. After 36 h, check whether there is an inhibition zone around the sample. The inhibition zone was measured and calculated according to the formula: H = (D-d)/2. Where H = inhibition zone (mm), D = inhibition zone measured after incubation showing antibacterial activity (mm), and d = inhibition zone before incubation (mm).
Results and discussion
The microstructure of PAN and M NPs decorated NFMs
The FE-SEM was used to observe the surface morphologies of the PAN NFMs before and after deposition the NPs, as shown in Figure 1. As shown in Figure 1(a) and (a’), the FE-SEM image shows randomly oriented, three-dimensional nonwoven PAN NFMs, with an average diameter of 147 ± 26 nm (Figure 1(e)). After the Zn, Cu, and Zn/Cu NPs were deposited on the surface of the PAN NFMs, the monodisperse NPs have can be clearly observed on both at the nanofibers and in between the nanofibers of PAN NFMs, and the fiber surface becomes obviously uneven. Here the results show that the NPs prepared by the nanoparticles generator instrument were successfully constructed on the surface of PAN NFMs. From Figure 1(b) and (b’), it can be observed that the Cu NPs have a spherical and non-uniform distribution on the surface of the PAN NFMs, with small particle size. From Figure 1(c) and (c’), it can be clearly observed that the Zn NPs have a spherical and uniform distribution on the PAN NFMs. From Figure 1(d) and (d’), it can be found that the surface of the Cu/Zn@PAN NFMs has a large number of deposited NPs, since the Cu/Zn@PAN NFMs has two layers of the NPs. The Cu/Zn NPs on the PAN NFMs are obvious with uneven distribution and wide particle size. Comparing the surface morphology of the three NPs deposited samples, the average nanofiber diameters of PAN NFMs after deposition of Cu, Zn, and Cu/Zn NPs were increased to 171 ± 29, 169 ± 25, and 170 ± 25 nm, respectively (Figure 1(f) and (h)). It was found that the uniformity of Zn NPs distribution is the best, which may be related to the PAN NFMs [42]. Some literature shows that PAN NFMs can prevent Zn NPs aggregation to a certain extent. Cu and Cu/Zn NPs aggregate on the PAN NFMs surface, which may be related to the NPs size and the process parameters [43,44].

Representative FE-SEM images of (a) PAN, (b) Cu@PAN, (c) Zn@PAN, and (d) Cu/Zn@PAN NFMs (a), (b), (c), and (d) are the corresponding high magnification FE-SEM images (e), (f), (g), and (h) are the corresponding diameter distribution.
Moreover, EDX analysis was used to determine the amount of Cu and Zn elements in the NFMs, and also the distribution of the Cu, Zn, and Cu/Zn NPs in the NFMs was monitored by EDX mapping and represented in Figure 2. EDX analysis of all samples exhibited that the presence of significant amounts of C, N, and O which proved that the obtained NFMs are composed of PAN. In addition, the weight ratio and EDX mapping of the corresponding elements were also inserted in Figure 2. The EDX spectrum of the PAN NFMs before deposition of NPs do not show the signals of Cu or/and Zn (Figure 2). In contrast, as can be seen from Figure 2, the existence of distinctive energy peaks for Cu and Zn elements demonstrates that the Cu, Zn, and Cu/Zn NPs have been formed successfully on the PAN NFMs. Besides, EDX mapping of the corresponding elements showed that the Zn NPs have been well distributed on the surface of the PAN NFMs; while the Cu and Cu/Zn NPs have been shown some aggregation.
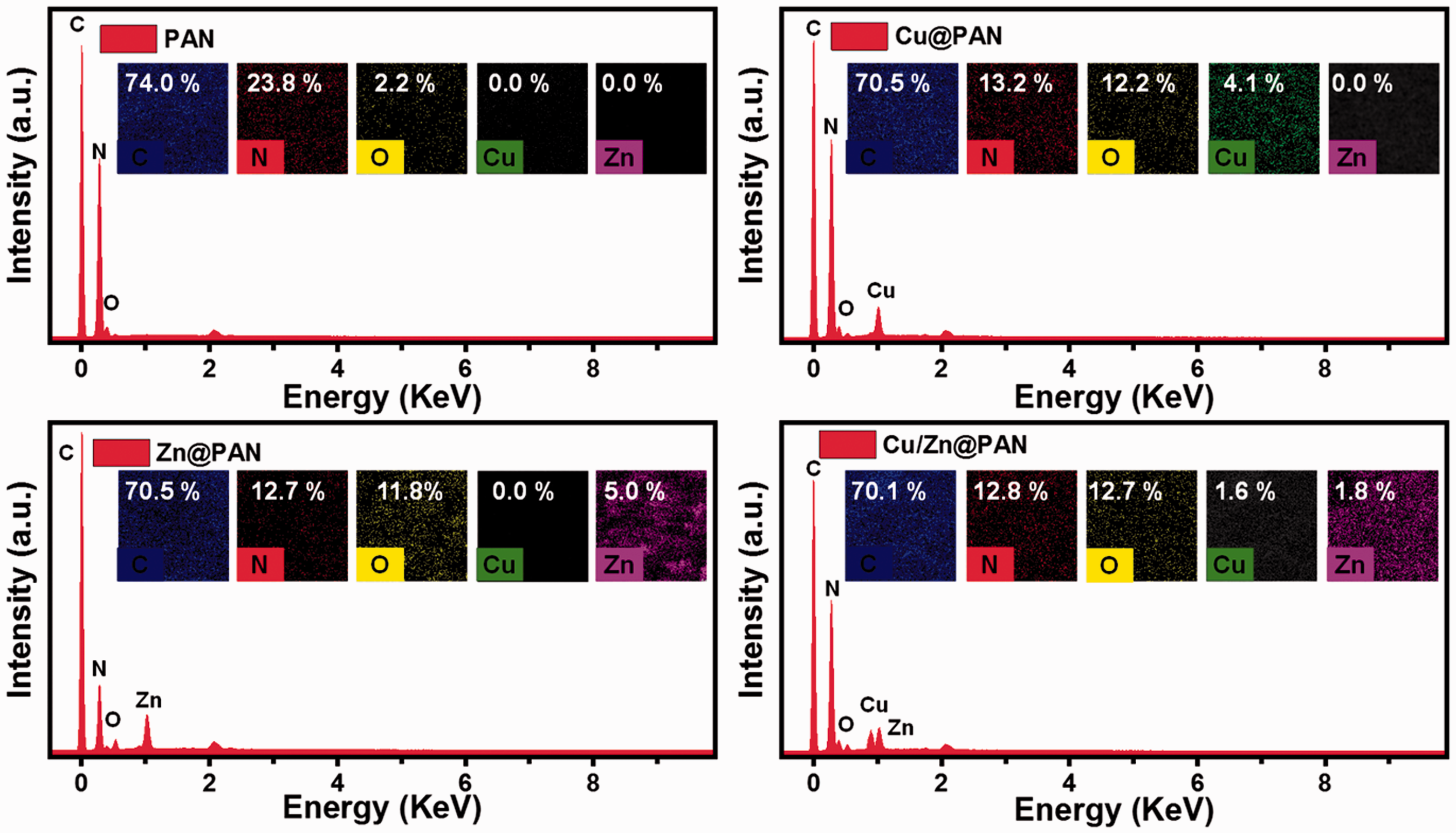
EDX spectrum of PAN, Cu@PAN, Zn@PAN, and Cu/Zn@PAN NFMs, insets show the corresponding elemental mapping and weight ratio.
Water contact angle and XRD analysis
The wettability of the PAN NFMs surface before and after the deposition of the NPs were evaluated by using the water contact angle. As can be seen from Figure 3(a) the PAN NFMs before the deposition of NPs shows a good hydrophilic property with a water contact angle of 46°. After NPs have been deposited on the surface of PAN NFMs, the wettability of the NFMs has changed significantly, due to the existence of the NPs on the surface of the NFMs. As shown in Figure 3(a), the water contact angle of the Cu, Zn, Cu/Zn NPs@PAN NFMs are 128.5°, 130°, and 136°, respectively. These results showed the successful using the nanoparticle generator to construct a hydrophobic structure of the PAN NFMs. In addition, the structure of Cu, Zn, Cu/Zn NPs@PAN NFMs and PAN NFMs was characterized by using XRD and is shown in Figure 3(b). The spectrum of the PAN shows the presence of a broad peak at 2θ value 26° indicates PAN NFMs [45]. On the contrary, the spectrum of the Cu@PAN, due to very small size and low content of Cu NPs as compared to PAN NFMs, very low intense peaks at 41° and 50° are observed that indicates the formation of Cu NPs on the surface of PAN NFMs. Besides, the spectrum of the Zn@PAN shows the presence of a broad peak at 2θ value 28°, 35°, and 50° indicate the formation of Zn NPs on PAN NFMs. Moreover, the spectrum of the Cu/Zn@PAN shows the presence of a broad peak at 2θ value 28°, 35°, and 40° indicates the formation of Cu and Zn NPs on the PAN NFMs. Here the XRD results show that the Cu, Zn, and Cu/Zn NPs have been deposited successfully on the surface of PAN NFMs.

(a) The water contact angle and (b) XRD spectrum of PAN, Cu@PAN, Zn@PAN, and Cu/Zn@PAN NFMs.
Antibacterial activity
The antibacterial performance of PAN and NPs@PAN NFMs was evaluated using an agar-diffusion test method as a qualitative analysis, which shown in Figure 4. As shown in Figure 4(a), the PAN NFMs did not show any antibacterial activity for S. aureus. Whilst, Zn@PAN and Cu/Zn@PAN NFMs displayed inhibition-zone diameters for S. aureus. The Zn@PAN showed the largest average inhibition-zone with a diameter of 1.30 mm for S. aureus. While Cu/Zn@PAN NFMs displayed inhibition-zone with a diameter of 0.25 mm. Although no inhibition zone appeared in the Cu@PAN NFMs, the bacteria reproduction under the NFMs was smaller than that of the PAN NFMs. It can be seen that the Zn@PAN NFMs has good antibacterial activity on S. aureus, while the Cu/Zn@PAN NFMs and the Cu@PAN NFMs have a limited antibacterial activity for S. aureus.

Antibacterial activity of Cu@PAN, Zn@PAN, and Cu/Zn@PAN NFMs towards (a) S. aureus and (b) E. coli, respectively.

Diagram illustration of (a) fabrication of the PAN NFMs and (b) deposition of the nanoparticles on the surface of the PAN NFMs.
Moreover, Figure 4(b) shows the antibacterial activity of PAN, Cu@PAN, Zn@PAN, and Cu/Zn@PAN NFMs towards E. coli. no inhibition zone appeared in all the samples. Besides, a large number of bacteria multiply under the PAN NFMs, which is indicated the PAN NFMs did not show any antibacterial activity. However, no bacteria reproduction under the Zn@PAN and Cu/Zn@PAN NFMs appeared, and a relatively small number of bacteria reproductions under the Cu@PAN NFMs have been observed. The results here indicated that the Cu@PAN, Zn@PAN and Cu/Zn@PAN NFMs have limited antibacterial activity for E. coli.
Conclusion
In summary, Cu, Zn, and Cu/Zn NPs deposited PAN NFMs were successfully fabricated and characterized as an effective substance in the reduction of bacteria using the nanoparticles generator instrument. Depositing of NPs improved the hydrophobicity by increasing the water contact angle from 46° to 136°. The antibacterial properties of the prepared NFMs were studied using biofilm-forming Gram-positive (S. aureus) and Gram-negative (E. coli) bacteria. The Zn@PAN exhibited good antibacterial potential against S. aureus and limited antibacterial activity against E. coli. On the other hand, Cu@PAN and Cu/Zn@PAN NFMs exhibited limited antibacterial potential against both Gram-negative and Gram-positive bacterial strains. Although there are still many potential challenges, such as individuals, evenness of the NFMs, potential application and so on, our findings may open up ways to simply water treatment in real life as a preventive approach.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the Fundamental Research Funds for the Central Universities (2232020G-01); the seed project of Key Laboratory of Textile Science and Technology of Ministry of Education (KLTST201622); and 111 Project “Biomedical Textile Materials Science and Technology” (BP0719035).
