Abstract
This work describes that bacterial cellulose was successively modified by copper/zinc nanoparticles with the help of direct current and radio frequency magnetron sputter-coating method for electromagnetic interference shielding to enhance its hydrophobic, mechanical, and conductive properties. The surface morphology and properties of bacterial cellulose/copper/zinc nanocomposite were analyzed by various testing methods (X-ray photoelectron spectroscopy, X-ray diffraction) and instruments to analyze smooth deposition of metallic nanoparticles. The conductive and shielding effects were examined by a four-point probe and vector analyzer. The mechanical properties were investigated by a uniaxial testing machine. The combined structure of bacterial cellulose, copper, and zinc nanoparticles (Zn–Np) improves the surface hydrophobicity, mechanical strength, as well as electromagnetic interference shielding effectiveness, while zinc nanoparticles act as a protective layer over the surface of bacterial cellulose/copper nanocomposite. So, the findings revealed that the bacterial cellulose/copper/zinc nanocomposite has excellent hydrophobic (θ; 143.7°), good mechanical (T.F; 39.67 MPa, elongation; 13.1%, Y.M; 5.90 MPa), conductive (50 W; 0.0323 S/m), and electromagnetic interference shielding (28.54 dB) properties.
Keywords
Introduction
In the modern age, communication instruments are being widely used in every sector of life, which include scientific, industrial, and commercial instruments. Although such devices facilitate human life in many ways, however, they also expose risk to the life of individuals in the form of electromagnetic pollution [1].
Recently, many graphene and copper (Cu)-based nanomaterials have been recognized with excellent conductivity and electromagnetic interference shielding [2]. However, graphene-based materials are costly and relatively high thickness materials which limit their commercial applications and mass production [3]. Similarly, Cu nanoparticles are rapidly oxidized in an open environment, which hinders their possible applications. To overcome this problem, more consideration was given to study oxide nanocomposites and metal films [2]. The Environmental Protection Agency of the U.S. confirmed that Cu and several Cu alloys have many potential surface properties [4].
Several coating techniques were investigated for the accumulation of metallic particles of the Cu alloys on insulating substances, for example, vacuum metallization [5], laser sputter coating or vapor deposition, and chemical coatings [6]. Every method has several advantages and disadvantages, for example, toxic reducing agents are known to be employed during chemical coatings [7].
The cellulosic materials are getting high attention in terms of conductive polymers; additionally, more attention toward cellulosic materials due to its usage in several applications of daily life like apparels, medical, and paper industry [8]. The formation of semiconductor by the combination of cellulosic materials and metallic nanoparticles is extensively studied in the modern age due to its high potential characteristics [9]. Preparation of these types of nanocomposite materials has good conductive, insulative, high sensitivity, and absorption properties in response to their usage as compatible electronic gadgets, films, and sensing devices [10]. The formation of nanocomposite materials demonstrates the nanoparticles’ functionalities and cellulosic materials as base to functionalize [11].
It was reported that bacterial cellulose (BC) is a suitable and eco-friendly nanomaterial for the fabrication of high-end nanocomposites. The performance of BC with inorganic or organic materials generates a value-added pathway for the manufacturing of novel multifunctional nanocomposites with compromising presentation. For the preparation of novel functional nanocomposites, BC was used as a template, and good properties of BC in terms of organic or inorganic nanomaterials were obtained including optical and magnetic characteristics [12]. BC is obtained by gluconacetobacter-xylinus, which is probably 50 nm wide and 10 nm thick [13], and has a 3-D structure of bio-synthesized fibers. It gained great attention because of its high purity, a wide range of polymerization, water retaining properties, and well-known high hydration. Moreover, BC has great potential due to its non-toxic, biocompatible, biodegradable, and highly porous material characteristics. Nowadays, BC is being used in a number of applications in terms of electronic gadgets, biosensors, medical field, paper, and packaging industries [2]. It is necessary to get the good edge of the versatile formation and characteristics of BC nanomaterials to generate novel BC-based nanomaterials with advanced functionalities [14]. Several developed procedures have been introduced to expose chances for endowing BC with advanced properties. The application of BC with metal oxides and metals is an approach to make nanocomposites, in which BC performs as a base material. In a way to promote the different characteristics to BC, many scientists have worked on BC in compass to several types of metals and metal oxide nanoparticles, in which Cu, CuO, Ag, and ZnO conclusively exist [15]. Such types of metals and metals oxide are easily available in the market because of their ecofriendly nature and nontoxic behavior [16]. In terms of zinc nanoparticles, which can easily produce over surface of target materials, different techniques like magnetron sputtering, vapor deposition, pulsed laser deposition, screen printing, vapor deposition, sol gel, and thermal evaporation are most widely used methods nowadays [17]. In addition, BC has recently been extensively studied due to its eco-friendly nature [18].
The most common methods are situ synthesis, coating, and blending to introduce the conductive materials into the BC matrix [19]. In this study, BC was selected as a base material for nanoparticle electromagnetic interference shielding of nanoparticles. The coating formation by physical vapor accumulation is an eco-friendly method for several materials to functionalize them [20]. Therefore, direct current (DC) megatron coating and RF reactive sputter-coating methods were adopted to investigate the BC/Cu/Zn nanocomposites. Zn was coated over the Cu layer as protective layer and investigated in terms of the electromagnetic interference shielding effect. The results showed that Zn nanoparticle coating on the Cu layer could be an excellent oxidation barrier.
The chemical composition and morphological study of nanocomposite BC were examined by X-ray diffraction (XRD), environmental scanning electron microscopy, and X-ray photoelectron spectroscopy (XPS), respectively. In addition, mechanical characteristics, electrical conductivity, and measurement of contact angle were also investigated.
Experimental procedure
Materials
BC samples were prepared from the textile laboratory of Jiangnan University (Wuxi, P.R.C). Meanwhile, Cu and Zn (purity: 99.99%) were purchased from C.M. Technology Pvt., Ltd. (China). Glucose, bacto-peptone, mannitol, and sodium hydro-oxide were bought from A.B-Chem Technology Pvt., Ltd. Shanghai (China). Similarly, yeast extract and dextrose were purchased from S.C. Reagent Pvt., Ltd., Shanghai (China).
Preparation of BC
The static incubation method was used to synthesize BC [21]. A sterilized nutrient medium (200 ml) comprising of yeast extract (8.0 g/L), disodium-phosphate (10.5 g/L), dextrose (24.5 g/L), and bacto-peptone (10.5 g/L) were made with adjacent pH of 6.0. Gluconacetobacter-xylinus was mixed to the medium and put in an incubator for 24 h at 30°C. After that, 10 mL of bacteria solution was mixed into 200 mL of culture and incubated for six days at 30°C as illustrated in Figure 1. Then, BC pellicles were obtained at day 7 from the upper layer of the culture medium. To get more clear and white form of cellulose, it was rinsed with 0.1 M sodium hydroxide for 8–10 h at 80°C and repeatedly rinsed with distilled water to neutralize [22]. After that, it was frozen in the refrigerator for 24 h and then put in a vacuum chamber to get a completely dry form prior to coating. Finally, an electronic micrometer was employed to find out the thickness of the BC substrate.

Process representation of bacterial cellulose preparation over six days incubation method.
Sputter coating
The deposition of Cu nanoparticles on the BC layer was done by magnetron sputter- (JPG-450) coating technique as described in Figure 2. The cathode end was occupied by a high Cu purity (99.99%) with a diameter of 50 mm. Whereas the substrate BC was placed on the anode which was 80 mm far from the target material (Cu), with a moving speed of 90 r/min to get a smooth accumulation of Cu nanoparticles by DC magnetron sputtering. The high-temperature diffusion movement of sputtered Cu nanoparticles and deformation of the BC substrate was controlled by the supply of H2O [23]. A pressure of 8.0 × 10−4 Pa was provided to the chamber before the supply of Ar gas (purity: 99.99%). The induction of argon gas (Ar) was stabilized on 20 sccm. The sputtering powers (30, 50, and 70 W) were supplied at 0.85 Pa during the sputtering process. Six different timings (10, 20, 30, 40, 50, and 60 min) of coatings were set to control the deposition formation of Cu nanoparticles on BC layer. Similarly, target Zn (purity: 99.99%) with a 50 mm diameter was used. The coating time was 30 min over the surface of BC/Cu to develop a protective layer by the RF sputter-coating method. In the case of Zn, the pressure was provided 1.0 × 10−5 to the chamber for evacuation. After that, gas (argon) was supplied into the vessel at the pressure of 1.05 Pa. Then the process of sputter coating was continued in argon gas with a flow of 50 sccm, and power was set at 100 W. The deposition thickness of Zn nanoparticles on BC/Cu layers was measured the same as way as for Cu nanoparticles.
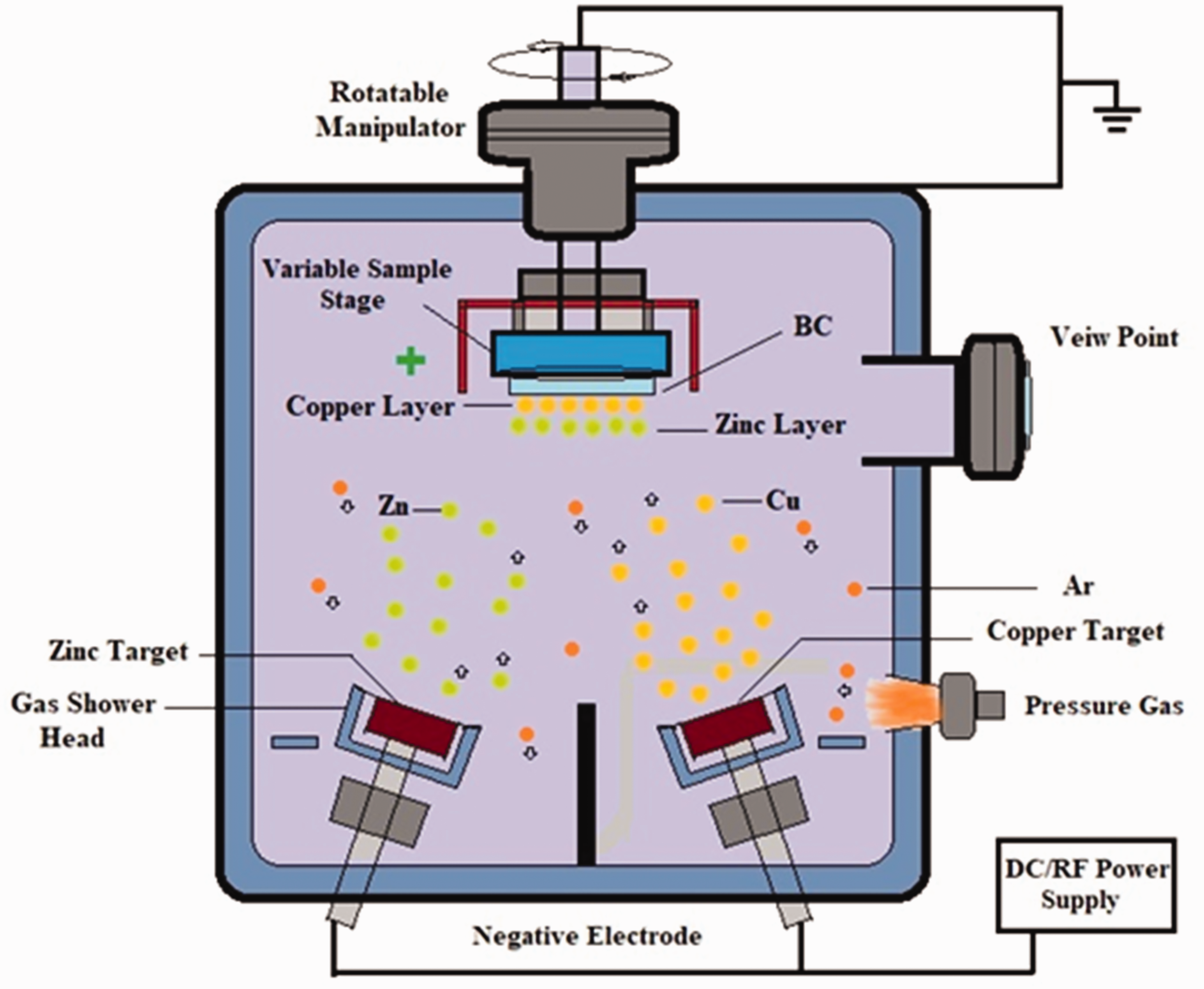
The DC magnetron and RF sputter-coating technique for the composition of BC/Cu/Zn nanocomposite.
Surface characterization
The surface morphology and chemical composition of pure BC, BC/Cu, and BC/Cu/Zn were investigated by XPS (Escalab 250Xi, England), whereas XRD was evaluated by Bruker–AXS. All samples were scanned over at room temperature in open environment. The field emission scanning electron microscopy (FE-SEM) was taken into account by the FE-SEM 4800 type by which at 5 μm resolution, FE-SEM images were obtained. For the hydrophilic property of pure BC, BC/Cu, and BC/Cu/Zn nanocomposite, the DCAT-21 (Germany) type was used to find out the contact angle by static method in which a drop of water was put over the surface of pure BC and both nanocomposites. Whereas, contact angle was measured by looking on drop of water with the help of goniometer.
A four-point probe (Jandel) was used to find out the conductivity of the samples. This four-point method is called four-terminal sensing method in which electrical impedance was measured. Instead of two-point prob, this four-prob method was more accurate and capable to obtain the values; because, in this method, current caring and voltage sensing electrode were used separately. All samples were tested 10 times in a similar way and aggregated. EMI SE of the samples was measured by vector network analyzer (VNA) 8573ES (Donghua University, China) with the help of flange coaxial method by which the shielding effect of both nanocomposites was measured. The range and accuracy of the measuring outline were 26–40 GHz in terms of curve values.
Mechanical testing
Mechanical characteristics of the samples were determined by the uniaxial testing machine (Instron 1185, I.C, US) with a tensile value of 10 mm/min by 2 cm distance between clamps. An aggregated value was determined by 10 replicate trials of each sample of 5 cm long and 1 cm wide.
Results and discussion
XPS analysis
To determine the organic composition of Cu, Zn, nitrogen, and carbon elements, the surface variation of organic bonding was determined by XPS [24]. Figure 3(a) describes the distinctive peaks of Cu elements. In addition, peaks at 931.22 and 951.45 eV subsequent toward Cu2p1 and Cu2p3 peaks of Cu+, respectively, clearly shows the existence of Cu [25,26]. Similarly, it can be seen in Figure 3(b) that peaks at 1021.2 and 1044.25 eV, ascribing to the characteristics peak of Zn2p3 and Zn2p1 of Zn individually, ensured the existence of Zn in the sample. Additionally, the peaks at 390.46 and 284.75 eV corresponding to N1s and C1s revealed the presence of nitrogen and carbon [27]. As indicated in Figure 3(c), showing the existence of those elements, the formation of nanocomposites was examined through X-ray photoelectron spectroscopy.

XPS survey spectra of BC/Cu/Zn nanocomposite: (a) copper elements; (b) zinc element; (c) nanocomposite.
The atomic content percentages of various elements in BC/Cu/Zn nanocomposite were observed as given: N (2.57%), C (42.75%), Cu (1.12%), and Zn (53.56%) individually, by which Cu may be oxidized during the sputter-coating process. Moreover, it is signifying that Cu nanoparticles sheets were fully coated by protective layer Zn nanoparticles.
XRD analysis
To elaborate on the advance study of pure BC, BC/Cu, and BC/Cu/Zn nanocomposites, XRD was employed, and outcomes are shown in Figure 4. Pure BC displayed major peaks at 2θ = 14.9°, 18.7°, and 23.2° (Figure 4(a)), which typically corresponds to the crystallographic planes (100), (010), and (110) of cellulose Iα [28]. However, it is seen from Figure 4(b) that BC/Cu sample demonstrated similar major peaks as the BC but with a newly identifiable sharp peak at 2θ = 43.7°. This new peak corresponds with (220) plane which was due to the presence of Cu [29]. Figure 4(c) illustrates that the addition of Zn to BC/Cu did not yield any new distinct peak but rather intensified the peak attributed to the Cu plane (220). This shows an obvious enhancement of the degree of crystallinity of BC, with Cu’s average size determined toward 14.2 nm according to the Debye–Scherrer equation, whereas Zn is 40 nm [30]. In summary of the XRD, the description recommended that the combined structure of Cu and Zn sheets instantaneously developed over the surface of BC.

XRD representation of experiments: (a) pure bacterial cellulose; (b) BC/Cu nanocomposite; (c) BC/Cu/Zn nanocomposite.
Mechanical strength analysis
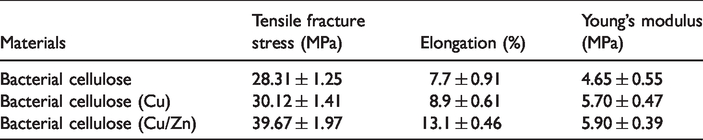
Mechanical strength characteristics were determined by tensile strength and elongation at break point. Tensile strength represents the strength of nanofibers, while elongation at break shows the stretchability of nanofibers prior to breakage. Under uniaxial tensile loading for BC layer, BC/Cu and BC/Cu/Zn nanocomposites’ typical tensile stress–strain were measured. It was observed that BC, BC/Cu, and BC/Cu/Zn have a linear elastic behavior up to the yield point and a non-linear elastic behavior up to breakage point from yield point. The rupture caused was particularly for fibers and nanocomposite layers slippage. The value of the pure BC for tensile fracture stress was about 28.31 MPa, with elongation break 7.7% corresponding to the result mentioned in the literature [31].
The addition of Cu and Zn nanoparticles on the BC surface plays a vital role to increase the rupture stress of nanocomposite, that is why the maximum coating time (Cu: 60 min, Zn: 30 min) samples were selected to find out the mechanical strength characteristics of the samples. Table 1 illustrating that, it increases from 28.31 to 30.12 MPa for BC/Cu and 28.31 to 39.67 MPa for BC/Cu/Zn. More elongation percentage was also enhanced from 7.7 to 8.9% and 7.7 to 13.1%, respectively. The average value of Young’s modulus of pure BC, BC/Cu, and BC/Cu/Zn was 4.65, 5.70, and 5.90 MPa, respectively. Compared with the BC film, breaking strength, elongation, and Young’s modulus are enhanced by a coating of Cu and Zn nanoparticles.
Representation of fracture stress, elongation percentage, and Young’s modulus of pure bacterial cellulose, bacterial cellulose/Cu, and bacterial cellulose/Cu/Zn nanocomposite.
Contact angle analysis
Figure 5(a) and (b) illustrates the BC/Cu nanocomposites’ results after DC magnetron sputtering. Additionally, it can be observed that in Figure 5(b) and (c), Cu nanoparticles on BC and Zn nanoparticles layer were accumulated on BC/Cu nanocomposite. The accumulation can be controlled by the flow of Ar gas during the sputtering of Cu and Zn nanoparticles [32]. Figure 5(d) shows BC, that is a structure of ultra-fine nanofibers with interlaced mesh [18,33]. In addition, Figure 5(e) describes a uniform Cu nanoparticles layer which was deposited on the substrate of the BC surface, whereas Figure 5(f) illustrates that a protective layer of Zn nanoparticles was obtained on BC/Cu nanocomposite. To measure and elaborate the wetting characteristics of composite materials, contact angle has great importance. As shown in Figure 5(g) to (i), the water contact angles explained the layers’ characteristics. Furthermore, the effect of time on the moistening actions of BC substrate was deposited through Cu and Zn nanoparticles. The contact angle between water and BC nanofiber was determined at 17.8° as shown in Figure 5(g), illustrating the high absorbency and hydrogen bonding from themselves [34]. It can be seen that, in Figure 5(h), by increasing the time of sputter coating, the water contact angle of BC/Cu enhanced gradually. When Cu nanoparticles’ sputter-coating time reached 60 min, it occurs that Cu layers on the BC surface show hydrophobic behavior with water contact angle 123.8°. It reveals that the nucleation power obstacle was high on the surface with greater contact angles [35]. Similarly, Figure 5(i) describes the change in wetting behavior of the Zn protective layer over BC/Cu nanocomposite by time. Similarly, the water contact angle 143.7° corresponding to BC/Cu/Zn nanocomposite in ascending order with time, attributing to lower surface energy that can be caused by surface oxidation. Conclusively, pure BC was modified and factionalized through Cu nanoparticles and the protective layer of Zn nanoparticles which provides superior hydrophobic characteristics. Hence, the modified surface of pure BC with Cu and Zn nanoparticles turned to it in many applications such as anti-static, anti-glazing, anti-oxidization, self-cleansing, and so on.

(a–c) development of nanoparticles on bacterial cellulose; (d–f) SEM pictures of pure bacterial cellulose, BC/Cu and BC/Cu/Zn nanocomposite; (g–i) representation of contact angles related to samples.
Protective mechanism of Zn–Np layer
Figure 6 describes the protection/resistance process of Zn nanoparticles layers accumulated on Cu layers. Due to the greater surface energy of Cu nanoparticles, Cu layers were certainly oxidized as compared to Cu foil. In the ambient atmosphere, there were two oxidation mechanisms that can occur, as shown in Figure 6(a). In the first stage, the ambient atmosphere converts it in Cu2O. More with an accumulation of Cu2O layers, this oxidation would be limited, which may decrease the movement of the election and Cu+ [36]. So, it further converts into CuO from Cu2O in the second oxidation process. Sometimes, in the sputter-coating process, it was investigated that the Cu nanoparticle layer becomes oxidized by the residue of O2 which originated from a pre-pumped vacuum along with the impurity of working gas Ar. The first oxidation of Cu goes to Cu2O and further oxidizes by the ambient atmosphere which leads it to CuO [37]. As shown in Figure 6(b), Zn nanoparticle coating plays a role in the protective layer by forming a barrier to prevent further oxidation of CuO. Probably, when Zn layers is exposed to the open atmosphere with the formation of the Zn oxide layer, it further reacts with vapor form of H2O in the air to produce zinc hydroxide. This zinc hydroxide may respond to CO2 in the air to produce a thin, resistant, stubborn, and moderately insoluble dull gray layer of zinc carbonate which holds tremendously to the fundamental Zn layer, which provide the protection from additional oxidation; hence, Zn can be good protective layer for Cu and provide protection to substrate films with Cu, when film played the role of conductance [38].

(a) representation of Cu oxidation in the ambient atmosphere; (b) protection mechanism Zn nanoparticles from oxidation.
Shielding mechanism
In EMI shielding effect, probably, three mechanisms can be involved (Figure 7): the absorption of electromagnetic energy (SEA), multiple reflection residual loss (SEML) of electromagnetic radiation, and multiple reflection loss (SEMR) of electromagnetic radiation; all of them individually reflect on the layer of the substrate (SER). Reflection performs a significant function in shielding mechanisms for homogeneous conductive substrate materials, in which mobile charge caries or e– (present) inside the material may affect the incident electromagnetic waves. Material thickness has its own significant role regarding absorption. So, absorption is a secondary mechanism. Interior reflection which occurs inside of shielding materials generates multiple reflections called the third shielding mechanism. The following equation can deliberate the total EMI SE of materials

Diagrammatic representation of the shielding method in which SER corresponding to powers reflection on the substrate; SEML multiple reflection residual loss of EM radiation; SEA absorption of electromagnetic energy; and SEMR multiple reflection loss.
Conductivity analysis
The electric conductivity for EMI shielding effect materials has a significant role due to its interference for electromagnetic waves [38]. In addition, to investigate the impact of timing at electric conductivity of BC/Cu nanocomposite corresponding to various powers, a four-point probe method was used to obtain the measurements. In Figure 8, the increase in the timing of Cu nanoparticle accumulation on the substrate BC leads it to more conductive [39]. Due to an increase of time, Cu nanoparticles were affixed with BC substrate and developed a smooth surface layer by which electric conductivity was maintained in a good way. The electric conductivity (0.0323 S/m) at 60 min with 50 W was relatively higher as compared to 30 and 70 W, because low power may lose the Cu coating, and conductivity of BC/Cu nanoparticles can be decreased by surface brutality influenced at comparatively high power.

The impact of timing on electrical conductivity under the various powers: (a) 30 W, (b) 50 W, and (c) 70 W over BC/Cu nanocomposite.
Electromagnetic interference shielding analysis
The electromagnetic interference of shielding characteristics is highly relevant to its materials’ variation reflection coefficient. Figure 9(a) illustrates that the initial stage of EMI shielding of BC was consecutively low as compared to BC/Cu and BC/Cu/Zn in the frequency range of 26–40 GHz. The value of EMI of BC in this frequency range describes that it is purely a fibrous material, while the addition of Cu nanoparticles on the BC surface enhances its EMI shielding effectiveness. Cu nanoparticles make a tight structure on BC substrate, while the addition of Zn nanoparticles as a resultant of a protective layer over BC/Cu enhances its more EMI SE value up to 28.54 dB on the frequency range of 40 GHz. The addition of Cu and Zn nanoparticles on the BC substrate leads it to more conductive and in terms of Zn protective layer which decreases its air permeability. The disposition thickness over time may also be a reason to enhance its electromagnetic interference shielding. The EMI shielding effect with the passage of time decreases rapidly in terms of BC/Cu nanocomposite as compared to BC/Cu/Zn nanocomposite as illustrated in Figure 9(b). On day 1, the shielding effectiveness of BC/Cu was 100% but gradually decreased when it was measured after several days (5–30). The possible consequences may be due to the compact nanosheet formation of CuO2 from Cu as a P–B ratio on the BC substrate which breaks the composition of Cu nanosheet. The second reason may be the formation of CuO from Cu2O in an ambient atmosphere, which may be the cause of gradual crystalline structure of Cu nanosheet. In addition, it was due to the formation of CuO over the Cu layer with overlapping to each other in a compact structure, and the underlayer surface was gone in descending order [36].

(a) Electromagnetic interference shielding effectiveness findings of BC, BC/Cu, and BC/Cu/Zn nanocomposite on day 1 after coating; (b) effectiveness % of EMI shielding with the passage of time (0–30 days).
In Table 2, the resultant comparison of this study and previous works have been given with different substrates which describe that electromagnetic shielding effectiveness of BC/Cu and BC/Cu/Zn has reasonable results which can be attributed to control and regulation of disposition thickness of nanoparticles.
The assessment of various substrates encompasses their electrical behavior.

Conclusion
We have successively fabricated Cu/Zn nanoparticles on the surface BC by DC megatron and RF sputter-coating technique for electromagnetic interference shielding. The surface of BC material was modified by the addition of Cu and Zn nanoparticles, in which Zn layers provide a good protective mechanism to the underlying surface of Cu particles from oxidization. To get the improved characteristics of the final nanocomposite, Cu and Zn nanoparticles were deposited evenly on the BC surface with different coating times, while the best results were obtained at 50 W and 60 min. The EMI shielding effectiveness (28.54 dB), good mechanical (T.F; 39.67 MPa, elongation; 13.1%, Y.M; 5.90 MPa), excellent hydrophobic (θ; 143.7°), and good conductive (50 W; 0.0323 S/m) characteristics results show a satisfactory demonstration for prepared BC/Cu/Zn nanocomposite material. In addition, the choice of time and power for sputter coating plays a major role to get good EMI shielding effect. So, according to our study, the BC/Cu/Zn nanoscale structure can be a good material for electromagnetic inference shielding-related applications in daily life such as in apparels, medical, paper industry, compatible electronic gadgets, decorative films, energy storage, and sensing devices.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by National first-class discipline program of Light Industry Technology and Engineering: LITE2018-21 and the 111 Project (B17021).
