Abstract
Organic coconut oil was investigated as a bio-based phase change material in core, and melamine formaldehyde was used as shell material to fabricate microencapsulated phase change material for thermo-regulation in nonwoven textiles. The microcapsules were synthesized using in situ polymerization method. The produced microcapsules (microencapsulated phase change material) were applied by knife coating in different ratios (1:5 and 1.5:5; MPCM: coating paste by wt.) to 100% polypropylene nonwoven, porous, and hydrophilic layer of a laminated, spunbond, and double-layer fabric. The coated layer was confined within two layers of the fabric to develop a thermo-regulative system on the nonwoven fabric to regulate the body temperature in surgeries. The two layers were composed by applying heat (140°C) and pressure (12 kg/cm2). Organic coconut oil, the fabricated microcapsule, and the composite fabrics were characterized by Fourier transform infrared spectroscopy, differential scanning calorimetry, and scanning electron microscopy. Scanning electron microscopy results revealed that spherical and uniform microcapsules were obtained with an approximate particle size of 2–6 µm. Differential scanning calorimetry results indicated that microencapsulated phase change material and the composite fabrics possessed significant melting enthalpies of 72.9 and 8.4–11.4 J/g, respectively, at peak melting temperatures between 21.6 and 22.8°C within human comfort temperature range. The utilization of coconut oil as a phase change material and the composite integration of this phase change material to a nonwoven fabric bring forward a novelty for future applications.
Keywords
Introduction
Renewable energy sources and energy savings have become highly important in every aspect of our lives. Phase change materials (PCMs) are heat storing materials that are used for functional textiles, buildings, electronics, solar panels, etc. for energy saving. PCMs can store or release heat in the phase changing process. High latent heat capacity, small volume change at phase transition, non-toxicity, non-flammability, and durability to repeated heating and cooling cycles are the primary features for PCMs [1]. There are organic and inorganic PCMs. Organic PCMs have superior properties over inorganic types, considering these features. The most preferred organic PCMs for textiles are paraffin waxes with a straight-chain n-alkanes, polyethylene glycols, and fatty acids, due to their phase change temperatures and thermal storage capacities [2]. They are usually integrated to fiber or fabric in microencapsulated form to not lose the material during solid to liquid phase change. Additionally, microcapsule form increases thermal conductivity [3].
Several research studies on bio-based PCMs are being undertaken [4,5]. Edible oils are one of the most significant bio-based PCMs which have being investigated for thermal energy storage use in several fields. Fatty acids and their mixtures can be produced by the hydrolysis of plant or animal-based fats and oils. Fatty acids, their eutectic mixtures, and derivatives are highly important heat storing materials which are nontoxic, biodegradable, nonflammable, low cost, and a strong renewable PCM alternative to paraffin waxes. They exhibit high heat capacity, appropriate melting temperature range, and good thermal and chemical stability during several melting and freezing cycles [6–8].
Coconut oil is a promising natural source for PCM used in several areas. It has remarkably high latent heat capacity at human comfort temperature [9]. This study investigated the use of coconut oil for thermo-regulation in nonwoven textiles. A new textile application was performed by confining the developed microencapsulated coconut oil within the layers of a nonwoven fabric and making a composite. The novelty of this research is the microencapsulation of coconut oil for thermal energy storage use in textiles and to propose a new treatment technique for integration of this material to nonwoven textiles. According to existing literature, the use of organic coconut oil as PCM in a nonwoven application has not been previously studied.
Coconut oil is comprised of high amount of low molecular weight saturated fatty acids that majorly come from lauric oil. Marina et al. [10] studied Malaysian and Indonesian commercial virgin coconut oil and found out that the main part of the oil was lauric acid (46–48%), and the rest consisted of myristic acid, stearic acid, caprylic acid, capric acid, linoleic acid, and caproic acid. The study concluded that the percent of the contents might differ according to geographical origins. Wi et al. [11] produced shape-stabilized PCMs using coconut oil and palm oil that were placed into exfoliated graphite nanoplatelets by vacuum impregnation method for use in buildings or similar areas to save energy. Udangawa et al. [12] fabricated biomass microfibers using coconut oil in core and cellulose in shell by coaxial electrospinning. Nemeth et al. [13] microencapsulated coconut oil in calcium alginate shell using interfacial coacervation-crosslinking method to obtain thermal energy storing material. Khorasani et al. [14] fabricated microcapsules using coconut oil-based alkyd resin in core and poly (melamine-urea-formaldehyde) in shell, aiming to develop a self-healing material against corrosion on metallic structures. Hee et al. [15] studied different wall materials in microencapsulation of virgin coconut oil by spray-drying method. Hee et al. [16] developed virgin coconut oil microcapsules using spray-drying method with supercritical carbon dioxide to obtain a healthy food product. Bakry et al. [17] prepared a broad review on microencapsulation techniques of oils and their benefits for various industries such as food, pharmaceutical, agricultural, and textile.
This study consists of microencapsulation of organic coconut oil as a bio-based PCM and the integration of these fabricated microcapsules (MPCM) to a nonwoven double-layer fabric used for surgeries to obtain a thermo-regulative property. The microencapsulation was processed by in situ polymerization, which is one of the mostly used encapsulation methods [2]. Melamine formaldehyde was used as the shell material of the microcapsule. The polymer is one of the widely studied wall material in literature for microencapsulation of PCMs for thermal energy storage [1,2,8,18–21]. It was reported to be a highly stable material to chemicals and mechanical actions such as pressure, abrasion, or shear [1,6]. The produced MPCM was incorporated at the hydrophilic surface of the nonwoven fabric by coating, and then the coated layer was confined within two layers of the fabric as a sandwich composite under heat and pressure. It is usually not possible to apply high amounts of microencapsulated PCMs on textile fabrics to maintain the physical appearance or features. On the other hand, this technique provided an opportunity to include a high amount of MPCM in the coating paste by confining it within the layers of the fabric. Thus, this study took forward a novel process for future nonwoven PCM applications as well as revealed the thermal storage enthalpies of microencapsulated organic coconut oil and the treated fabrics.
Experimental
Materials
Sri Lanka origin 100% organic coconut oil (LifeCo Company, Turkey) was used as a phase change core material. Pure melamine (99%; Aldrich) and formaldehyde (Merck, 37% aqueous solution by weight) were utilized as shell material of the microcapsules. As an emulsifier, sodium dodecyl sulphate (SDS) (Fluka Chemika) and as a crosslinking agent for the melamine formaldehyde polymer shell Melateks 700 (Melamin Kemicna), containing methylol were used. Extra pure ammonium chloride (Riedel-de Haen) was also used as a nucleating agent for the encapsulation [4,8], and triethanolamine (99% pure) from Merck was used for adjusting the pH in reactions. The fabricated microcapsules were coated on a double layer, laminated, and spunbond nonwoven fabric which was supplied from Felix Plastik Laminasyon Ambalaj Malzemeleri San. ve Tic. Ltd Sti., Turkey. The structure of 100% polypropylene nonwoven layer was porous and hydrophilic. The weight of fabric was 56 g/m2. In coating, Orgal HC 77 was used as an acrylic polymer binder. The emulsifier and the thickener were Orgaprint EMU and Orgal M 420, respectively. Organik Kimya San. ve Tic. A.S. (Turkey) kindly supplied these coating materials.
Preparation of the microcapsules (MPCM)
The method has already been partially described in our recently published work [9]. The microencapsulation of coconut oil in melamine formaldehyde shell was carried out by in situ polymerization [18]. The process of microencapsulation was completed in three steps: preparation of pre-polymer solution of melamine and formaldehyde, preparation of oil in water emulsion, and the formation of the microcapsules. The polymer reaction mechanism was given in Figure 1. The pre-polymer solution was prepared by dissolving of 4 g of melamine and 20 mL of formaldehyde in 20 ml of distilled water by magnetic stirring at 70°C. During the pre-polymer preparation, the pH of the solution was maintained at 8.5–9 [18–20,22] by dropwise addition of triethanolamine. As the solution became transparent, 1 g of melamine and 10 mL of distilled water were added, and the solution was stirred till it turned transparent. Twenty grams of coconut oil, 5 g of the emulsifier (SDS), and 1 g of the crosslinking agent (Melateks 700) in 150 mL distilled water were stirred mechanically at 1700 r/min for 2 h at 50°C in order to prepare the emulsion. The addition of the crosslinking agent assisted the capsule formation. The pH of the emulsion was maintained at 4.0–4.5 by dropwise addition of acetic acid. The pre-polymer solution was added to the emulsion solution dropwise while stirring at 600 r/min. As the reaction started, 1 g of a nucleating agent (ammonium chloride) was added to the emulsion [4,18]. The encapsulation reaction was carried out for 90 min at 60°C by stirring at 600 r/min. The reaction was eventually terminated by maintaining the pH of the solution at 8.5–9. The microcapsules were filtered, washed with 30% ethanol solution several times to eliminate any residue on the microcapsules, and then dried at 50°C for 24 h.

The formation reaction of melamine formaldehyde prepolymer (a) and melamine formaldehyde polymer (b) [23].
Coating of MPCM on nonwoven fabric and forming a composite
Coating pastes were prepared with the recipes indicated in Table 1 using mechanical stirring to obtain a homogenized solution. Before adding the MPCM, the pH of the coating paste was adjusted to 8–9 by adding 25% ammonium hydroxide aqueous solution dropwise. Two recipes were applied on the hydrophilic layer of the nonwoven fabric, with ratios of 1:5 and 1.5:5 by weight (mass of MPCM: mass of coating paste without MPCM) for Recipe 1 and 2, respectively, using a fabric-coating machine from Turkey ATAC GK 40 RKL. The coated nonwoven fabric samples were then dried at 110°C for 15 min. The coated hydrophilic layer was placed within two layers of the fabric, and the three layers were composed under 140°C with a 12 kg/cm2 of pressure for 4 min in a press machine for composites, HURSAN Pres from Turkey, to form a thermo-regulative nonwoven composite. Coating add-on % was calculated as 46 and 55% for Recipe 1 and 2, respectively, by measuring the dry weight of coating pastes in the composite fabrics. The add-on % of MPCM in the composite fabrics for Recipe 1 and 2 was then calculated as 7.7 and 12.7%, respectively.
Coating recipes for each treatment.

Results and discussion
The surface morphology of the MPCM-treated nonwoven fabric layers was analyzed by scanning electron microscopy (Zeiss EVO-MA 10) (Figure 2). SEM images indicated that the spherical microcapsules were obtained in an approximate particle size of 2–6 µm, and the microcapsules were coated and fixed on the fabric surface uniformly. The particles on the surface of the microcapsules were considered to be aggregates of melamine formaldeyde which might be formed depending on the reaction rate and pH [23].

SEM images of (a) MPCM, (b) untreated fabric surface, and (c, d) coated fabric surface.
Fourier transform infrared spectroscopy (Perkin Elmer Spectrum 100 FT-IR) in the frequency range of 380 and 4000 cm−1 was utilized to examine the chemical compositions of organic coconut oil and the fabricated microcapsule (Figure 3). The absorption peak at 3300 cm−1 was attributed to O_H and N–H stretching vibrations of melamine formaldehyde microcapsule which was crosslinked with melamine resin that contains methylol. The absorption peaks placed at 2921 and 2853 cm−1 were assigned to the C–H bond stretching vibrations. The peaks at 1545 and 1490 cm−1 were assigned to the C=N and C–N stretching vibrations of melamine formaldehyde microcapsule. The peak at 813 cm−1 corresponded to stretching vibration of the triazine rings. The strong peak at approximately 1750 cm−1 was attributed to C=O bending vibration of fatty acid, which was only seen in organic coconut oil spectrum. FT-IR results confirmed [18,20] that the microencapsulation was processed successfully.
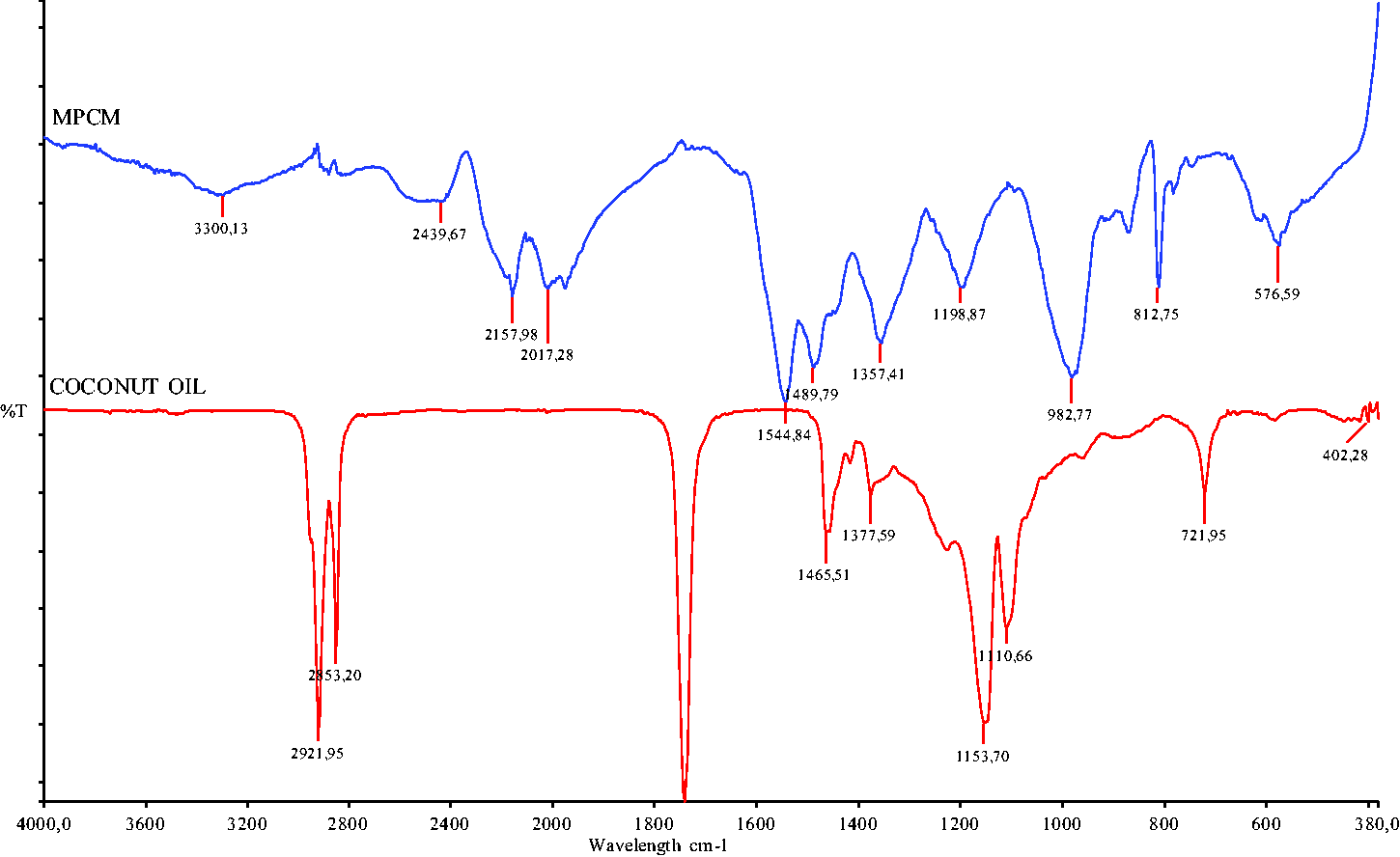
FT-IR spectra of organic coconut oil and MPCM.
Differential scanning calorimeter (Perkin Elmer Diamond DSC) was used to measure the melting and crystallization temperatures, latent heat enthalpies of organic coconut oil, MPCM, and the MPCM-treated fabric samples. The heating and the cooling rates were 10°C/min, in the temperature range of –10 and 50°C, under nitrogen atmosphere with a flow rate of 25 mL/min (Figure 3). The encapsulation ratio (R) which shows the PCM core content of the microcapsules [24,25] and the encapsulation yield (EY%) that indicates the ratio between the mass of dry microcapsules (mMPCM) and the total mass of the polymer shell components and the core (mt) [26] were calculated using equations (1) and (2), respectively

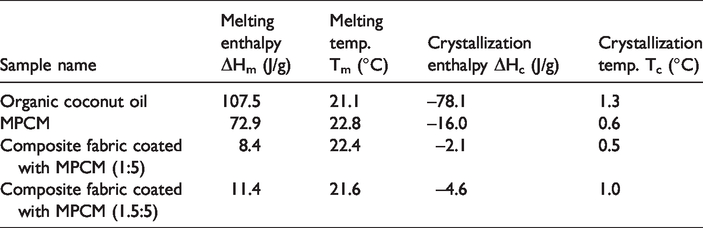
DSC graphs of the fabricated microcapsule and the core PCM organic coconut oil were shown in Figure 4. The DSC analysis indicated that the melting enthalpy of MPCM and core PCM organic coconut oil was 72.9 and 107.5 J/g at peak melting temperatures of 22.8 and 21.1°C, respectively (Table 2).

DSC curves of organic coconut oil and MPCM.
DSC measurements of organic coconut oil, MPCM, and fabric samples.
The encapsulation ratio was determined as 67.8% using equation (1), which shows the core content of the microcapsule. The encapsulation yield of the microencapsulation was calculated as 54.5%, using equation (2).
DSC measurements of the MPCM incorporated fabrics were shown in Table 2. The results demonstrated the latent heat enthalpies of the developed systems. The latent heat capacities of the fabrics during melting and crystallization provide thermo-regulative effect [6]. The most common method to assess the thermal storage and release properties of textiles is DSC measurement [27]. Thermal storage and regulation features of PCM integrated fabrics in literature were generally evaluated by DSC measurement [21,28–31]. The DSC results of this experimental work indicated that the composite fabrics possessed 8.4 and 11.4 J/g latent heat for Recipe 1 and 2, respectively (Table 2).
Some physical properties of the fabrics were tested and assessed by comparing before and after treatment. Thickness of the untreated fabric, the treated composite fabric, and the coating film were evaluated by Cloth Thickness Tester (R&B, James H. Heal & Co. Ltd.). The average results were found to be 0.36, 0.64, and 0.29 mm, respectively. Tear strength of the untreated fabric and the treated composite fabric were tested according to ASTM 1424-96. The results were determined as 1088 and 896 g (CD, cross machine direction) before and after treatment, respectively, which showed 17.6% loss after treatment.
Conclusion
PCMs are thermal energy storing materials which are used for smart textiles usually by incorporation to fiber or to fabric in finishing. In this study, a sandwich composite technique including coating was implemented to integrate a microencapsulated bio-based PCM to a nonwoven textile for thermal regulation functionality. MPCM-containing organic coconut oil in core was produced successfully by in situ polymerization and then applied by coating to a double-layer nonwoven fabric used for surgeries. Following the coating, a composite fabric was produced by confining MPCM-coated surface within two layers of nonwoven fabric under heat and pressure to build up a thermo-regulative system.
DSC measurements of the developed composite fabrics revealed that the fabrics possessed 8.4 and 11.4 J/g latent heat of melting with coating ratios of 1:5 and 1.5:5 (MPCM: coating paste by wt.), respectively. The enthalpy figures were found as remarkably high considering the PCM treatments on fabric in literature [21,28–31]. The most effective PCMs to be utilized in textiles are the ones with melting points in between 15 and 35°C [3]. This experimental work advances the potential use of organic coconut oil as a bio-based heat storing material in textiles due to its high latent heat capacity in human comfort temperature. Furthermore, this study proposes a new method to implement a high amount of microencapsulated PCM by making a composite for nonwoven textiles.
Footnotes
Acknowledgements
We thank Organik Kimya San (Turkey) for the chemicals used in the coatings and Felix Plastik Laminasyon Ambalaj Malzemeleri San (Turkey) for the nonwoven fabric.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Marmara University Scientific Research Project Unit (BAPKO) for the grant (Project No: FEN-C-DRP-200318-0117).
