Abstract
The most flame retardants and non-combustible non-woven fabrics are made of oxidized and carbon fibres due to their strong thermal stability. The burning of non-woven fabrics consists of complex combustion mechanisms: their surface, micro and macrostructures together define their combustion features. By microstructure, we mean oxidized polyacrylicnitrile fibres, which finally constitute the base material of the macrostructure. The macrostructure represents the different forms of the product, in which the material results during production. In this paper, the effect of the macrostructure of non-woven fabrics on flammability has been studied. It has experimentally shown that by defining the oxygen index, we can demonstrate the surface and thickness inhomogeneity, which is invisible or cannot be detected by mechanical tests. A feature of non-combustible non-woven fabrics is that their flammability depends on their thickness and area weight; however, the combustion phenomena of felt fabrics depend primarily on their macrostructure. Different oxygen contents have different combustion phenomena, thus an oxygen index can be assigned to each one. Thermoanalytical test results clearly showed the temperature at which the thermal decomposition of the fibres begins, which gives the surface flame when combusted.
Introduction
Flame retardation of flammable natural and chemical fibre fabrics have been extensively addressed for very long time [1–3]. Not only chemical inhibitors are used for flame retardation, but also the exploration of the relationship between structural research and flammability is coming to the fore [4,5]. The other direction of research addresses the thermal stability of substances [6,7]. The flammability of fabrics can be interpreted and measured on two levels: microstructure (fibre as raw material) [8–11] or macrostructure (non-woven products). The flammability of different processing forms made from the same fibre will be different. The limiting oxygen index (LOI) of oxidized fibres as a basic material of non-woven fabrics can be determined by standard method [12].
Felts are made from cut threads of different fibre densities between 1.32 and 1.42 g cm−3. The threads themselves could have different combustion characteristics. As the technology of textile-reinforced composites expanded, a growing demand from the aerospace industry for composite materials with superior properties emerged. In particular, materials with (i) higher specific strength, (ii) higher specific moduli and (iii) low density were required. Other desirable properties are good fatigue resistance and dimensional stability. Non-woven fabric, made from oxidized fibres, is used for fire barrier insulation of aircraft seats. A carbonized version of oxidized felt is used for aircraft brake systems. This felt manufacturing technology differs from the ones described above.
In the field of protective apparel for extreme high temperatures, felts are used as heat-protective liners, where the upper layer is aluminized fabric in general. The felt form and the fabric are also commonly used for welding cover. In the automotive industry, oxidized felt is used to protect the interior of the passenger compartment and to insulate the engine compartment. Its application in the furniture industry is significant. The flame retardancy of the material is utilized as a liner of hotel and hospital furniture, mattresses, prison mattresses, etc.
When investigating felts, we investigated how the secondary processing of cut threads and cables, i.e., the evolved macrostructure, would modify the flammability parameters, and how they would modify the oxygen index of the base fibre. We investigated if the cable retains its original flammability. In the case of woven textiles in one of our earlier works, we have demonstrated the correlation between micro and macrostructures [13].
In our experiments, we investigate how the micro and macrostructure of non-woven fabrics determine the combustibility of the felt products produced from polyacrylicnitrile (PAN)-based oxidized fibres. As with all measurements, reproducibility is the most important requirement here, too, where the evenness of samples plays a significant role. The difficulty of the selecting within the same sample is partly due to the selection of structurally similar parts (sample taking).
Behaviour of oxidized fibres in fire
An oxidized fibre is characterized by the typical microstructural parameters of the structure:
Stabilizing carbon fibres against oxidation is an important requirement, and many methods and processes are used [18–24].
A well-defined oxygen index can be assigned to them. From the features of the macrostructure, area weight (surface density) and thickness are determinant.
Behaviour of nonwowen fabrics in fire
Non-combustible textiles are now being used in many applications. The application is based on good heat resistance and reduced or limited flammability of the oxidized fibre [25–31].
In case of non-woven textiles, the macrostructure should be considered, when burning characteristics are studied.
According to our observations, the combustion characteristics are influenced by: consistency of the sample (the macrostructure examined), conditions of measurement, combined effect of the microstructure and macrostructure during the production process of fibres and products.
The common characteristics of the samples with different structures (cable, felt, cut thread) are that their surface is not smooth but fuzzy. In the case of normal measurements, the sample, vertically fixed in a U-shaped sample-holder, burns downwards when ignited. Especially, the fibres positioned outside make the measurement difficult. These parts of the samples with a fuzzy surface ignite significantly easier than the interior of the material itself. The combustion instantaneously runs down on both sides of the sample, whilst the interior of the sample remains unchanged. The appearance of this so-called surface flame or flashing flame depends on the oxygen concentration. By reaching another pre-defined oxygen content, this surface flame can ignite the sample from below, resulting in an upward spread of burning.
These phenomena are not taking in to account in standards, however, it is uniquely characteristic of the sample. The speed of the upward spread of burning is considerably higher than the downward spread of burning. In the case of such samples, it is especially important to take care of fixing the sample, to properly anchorage it and to ensure that the length of the sample is stretched to the bottom of the frame, serving as a flame stopper.
The majority of carbon fibre-based samples can be characterized by a certain oxygen concentration, including standard LOI. The different flaming phenomena can be observed at different oxygen concentrations, defined by the material and structural characteristics. Surface fuzziness is very problematic in case of felts, but especially in case of threads, which interfere correct measurements.
When selecting samples, it is important to take samples of equal thickness and evenness. In the case of felt samples – even if they are composed of the same fibre – it is required that the selection is based on thickness due to the technological limitations that do not ensure even thickness distribution. Beyond thickness, density and looseness should also be considered. These characteristics can be visually determined, however, differences in LOI (1) and LOI (2) can still occur. By paying attention to them, we can achieve good reproducibility.
Felt samples show the degree of dependency on structure by LOI most sensitively. The danger of re-ignition can be witnessed the most in these samples, because the presence of fuzzy piles is inherent in the material, and it cannot be influenced by mounting. Surface flames can appear in the cases when the top of the sample cannot be ignited and therefore, the usual flame spread cannot be observed. If the material is rather loosely structured (thread), even the surface flame is capable of burning through the whole thickness of the material.
Materials and methods
Samples of non-woven fabrics made of oxidized fibres
In this present study, commercial product felt samples were involved. For characterization of these materials, area weight (gm−2) can be used. Area weight data are obtained from our own measurements. The tested materials have area weight values ranging from 80 up to 1850 gm−2. The thickness of non-woven materials in the production process is typically 5–6 mm. In case of low weight, loose textiles, the variations in thickness and colour may occur. The typical fibre density was provided by the manufacturer. The tested materials and their parameters are given in Table 1.
Micro and macrostructure data for each sample.

Test methods
Scanning electron microscopic observations
To study the morphology of the samples, scanning electron microscopy (SEM) images were obtained by a Phenom XL SEM. For the observing of the samples, the surface (horizontally oriented samples) and cut cross section (vertically oriented samples) were studied. Further preparation was the coating of the sample’s surface, which was made in a vacuum with the evaporation of gold/palladium (Au/Pd). The acceleration voltage was 20 kV, and the samples were imaged under medium vacuum conditions. The actual magnifications have been printed on the SEM pictures.
Thermoanalytical method
Thermal behaviour of the studied materials were followed by thermoanalytical methods (TG/DTG/DTA) using Derivatograph-Q 1500 D. Reference material was alumina (Al2O3), the sample mass was ca. 300 mg, and the samples were heated at 10°C min−1 heating rate up to ∼1000°C in air atmosphere (in static condition). Thermoanalytical test results were evaluated by Winder (Version 4.4.) software.
Thermoanalytical tests have been conducted for several reasons: the temperature of the combustion also depends on the oxygen content. The temperature of the burning in high oxygen concentration exceeds the temperature range of the furnace (1000°C).
Measurement of LOI-index
Certain burning characteristics can only be defined under special circumstances. Burning phenomena and the combustions themselves were performed using standard equipment, at different oxygen concentrations, in counter current and upper edge ignition [31].
The precise descriptions of the measurement are regulated in international standards [12,25–32]. For our tests, oxygen index test apparatus (ISO 4589 Part 2, FIRE Instrumentation and Research Equipment Limited, UK) was used. The most important parts of the instrument are shown in Figure 1.

Typical apparatus for the determination of oxygen index.
The following parameters were used during the measurements: Oxygen analyser: ranged from 0% to 100% O2, repeatability: ± 0.1% O2, linearity: ± 0.1% O2, flow: flow through column adjustable from 0 to 20 ml/min.
The size of the sample holder is a 60 × 160 mm, U shaped, double layered vertical metal frame, secured in a glass cylinder with its top end open. The ignition and the removal of the combustion products are made possible through this opening. The source of ignition is a 40 mm long gas-flame as prescribed by standards. The specimen is ignited along the upper edge applying the flame for maximum of 15 s. Burning proceeds downwards against the flow of the pre-adjusted gas mixture. Nitrogen and oxygen content are adjustable as required.
For the purposes of the international standard [12,33,34], the following definition applies. Oxygen index: the minimum concentration of oxygen by percentage volume in a mixture of oxygen and nitrogen introduced at 23 ± 2°C that will just support combustion of a material under specified test conditions. It is recommended that the first trials are performed at low (30%–32%) oxygen concentration.
In our study, initially, the samples did not show any ignition phenomena and were non-flammable. By increasing the oxygen content, at the beginning, only few signs of burn appeared, then as the oxygen concentration increases, the length of the burnt area becomes greater.
Results and discussion
Results of scanning electron microscopy observations
SEM is a useful method to explain material behaviour by the microstructural point of view [35]. SEM tests were performed prior to incineration for similar reasons as thermoanalytical studies. Incinerated samples were not studied with the method because they could not be prepared for further SEM observations (e.g., preparing samples for sputter coating was not possible).
We used SEM observation to find any anomaly or differences from the ideal “smooth” surface and from round cross section of the fibres (Figure 2(a)). In Figure 2(a), freestanding fibre ends are visible. The diameter and the surface roughness of the fibres were the same for all investigated felt samples (Figure 2(a) and (b)). In Figure 2(b), we can see polluting particles on the surface of the fibres. For some fibres, traces of product-making process (i.e., deformations of fibres) were visually observed (Figure 2(c)). Few fibres were found with some morphological deviances, e.g., fibres with surface deposition, twisted or wavy fibres (Figure 2(d)–(i)).
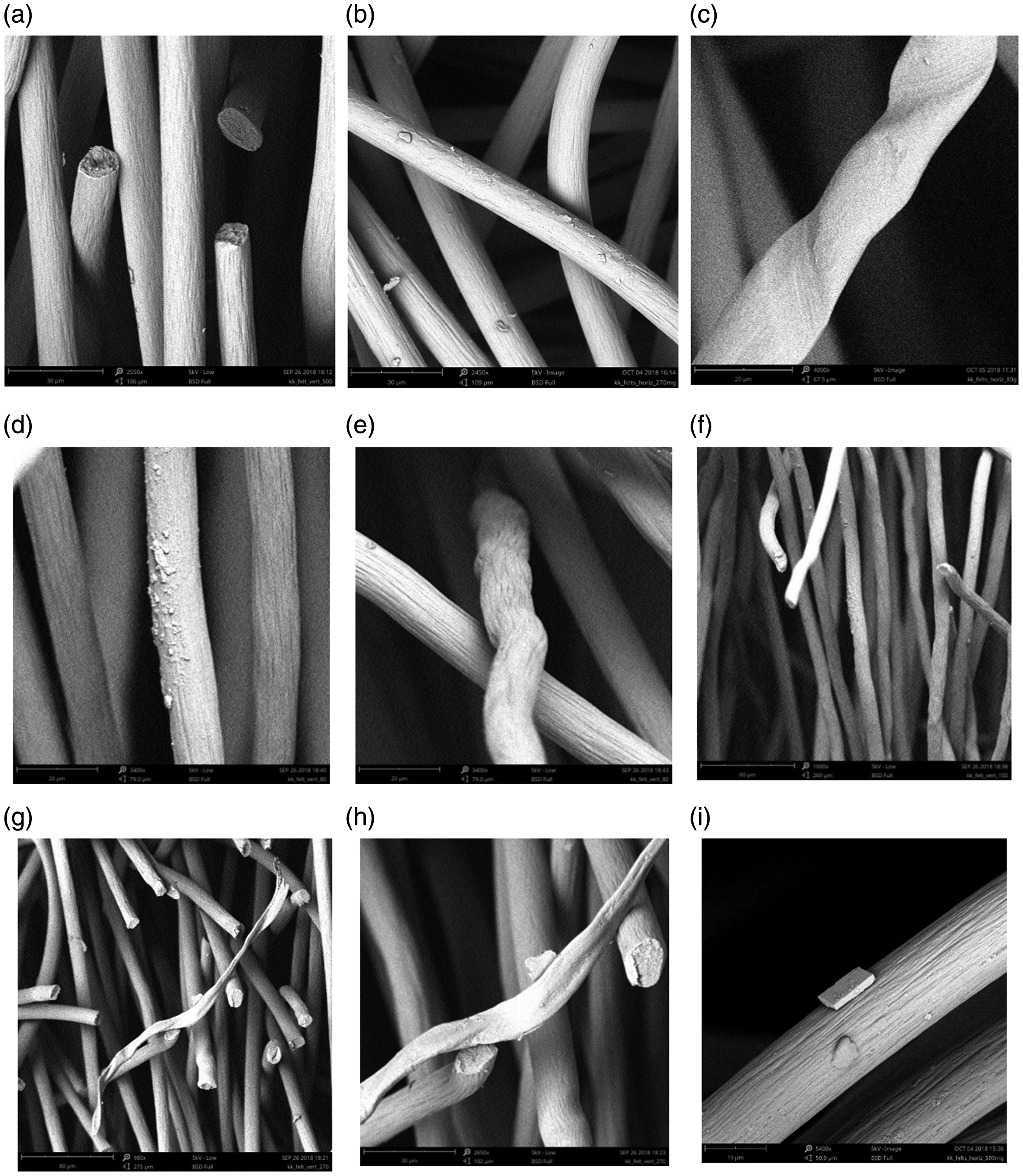
Observation with SEM: (a) Figure SEM1: vertically oriented felt sample of 500 gm−2, with cut cross-sections; Magn: 2550x; (b) Figure SEM2: horizontally oriented felt sample of 270 gm−2, Magn: 2450x; (c) Figure SEM3: horizontally oriented felt sample of 80 gm−2, deformations of fibres; Magn: 4000x; (d) Figure SEM4: felt sample of 80 gm−2, surface deposition; Magn: 3420x; (e) Figure SEM5: felt sample of 80 gm−2, deformed shape – wavy fibre; Magn: 3420x; (f) Figure SEM6: felt sample of 150 gm−2, twisted fibres; Magn: 1000x; (g) Figure SEM7: felt sample of 270 gm−2, polluting fibre; Magn: 980x; (h) Figure SEM8: felt sample of 270 gm−2, polluting fibre; Magn: 2650x; (i) Figure SEM9: felt sample of 500 gm−2, contamination on the surface; Magn: 5400x.
Figure 2(d) gives an example of deposition found on the surface of an individual fibre. Deposition can be either a microcrystallic or amorphous solid material formation without any well-defined shape. Figure 2(g) and (h) shows another type of fibre without having the round cross section; with ribbon-like shape. We called this “polluting fibre”, because we can clearly distinguish between ideal (a), wavy (e) and twisted (f) fibres with round cross section from ribbon-like fibre without round cross section. Figure 2(i) shows some contamination with a perfect crystal shape of a most probably inorganic material, with orthorhombic shape. This material is different from the material of the fibres, and so it is called contamination.
These microstructural differences are influencing also the burning characteristics of fabrics.
Results of thermal analysis
The temperature range of the thermoanalytical test was up to 1000°C, which is less than the temperature of the combustion of oxidized fibres. This means that the thermal analyzes do not reflect to the total pyrolysis of the carbon fibres. The explanation of the appearance of the surface flame is clear: the surface flame of the macrostructure – the fibrous material – can be linked to functional groups that easily detach from the surface of the carbon fibres. These can be pollutions, surface treatments or presence of temporary compounds during production, which are only available on the surface. Material of the fibres in the studied felts with different area weights is identical (Table 1), so they behave similarly by thermoanalytical approach.
The TG curves show that leaving groups from the surface of the fibres give ca. 50% of the total mass loss. According to the DTA curves, this starts at about 380°C, where the second exothermic reaction begins, i.e., the initial oxidation (Figure 3). The process lasts approximately up to 600°C, and then the carbon fibre becomes stable and does not give any more surface flame.

Thermoanalytical test results: (a) Thermoanalytical test results of felt sample 80 gm−2; (b) Thermoanalytical test results of felt sample 150 gm−2; (c) Thermoanalytical test results of felt sample 500 gm−2; (d) Thermoanalytical test results of felt sample 1850 gm−2.
This temperature range is sufficient to cause surface flames. They are easily available at low temperatures and spread rapidly.
Since the raw material, carbon fibre itself does not burn in the air, conventional flammability and other test methods, such as thermoanalytics, where the oxygen concentration is 21%, do not give full scale information about the characteristics of combustion. That is why we need to increase the oxygen content of the atmosphere, which is already capable of initiating pyrolysis. This is the purpose of LOI measurement.
Examination and classification of the burning phenomena of non-woven fabrics
Typical burning phenomena
In general, burning with a flame is considered a burning, also glowing for its external appearance. A flame phenomenon is always in connection with the presence of a gas phase. Glowing, in most cases, simply results in the “evaporation” of the substance. The literature also calls this “erosion” [13]. Burning off results in mass loss, called primary burning, which belongs to the LOI (1).
Samples as per their external appearance show two types of combustion phenomena (Figure 4): solid-phase burning (glowing, without flame) can lead to complete carbonization too, which may result in shrinking. Glowing can also be self-sustaining and spreading (Figure 4(c)) after the igniting flame is removed, burning with a strong flame that may appear on the surface of the sample or on the full thickness of the sample; it corresponds to the measurement standard (Figure 4(b)). In this case, we witnessed, that after combustion, the structure of material disintegrated.

Burning of non-woven fabrics: (a) felts before the start of the test; (b)burning with a flame with standard oxygen index test; (c) downward solid-phase burning (glowing).
It is recommended that the first trials are performed at low (30%–32%) oxygen concentration [12]. Initially, our samples do not show any ignition phenomena and is non-flammable. By increasing the oxygen content, initially only burning marks appear, then as the oxygen concentration increases, the length of the burnt area becomes greater. It is characteristic of felts that after burning, a carbonized disintegrated material: a charred dusty skeleton remains (Figure 4(b)). The appearance of the surface flame may have several impacts on the complete burning of the sample. The typically three types of burning phenomena occur at different oxygen contents, to which individual oxygen indices can be assigned. Measurements have confirmed that different LOI values could be assigned to the different burning phenomena, to which new flammability parameters can be defined: LOI (1), LOI (2), LOI (3).
The parameters are defined as follows: LOI (1): the oxygen content, at which a surface flame appears at the moment of ignition, and it swiftly spreads downward on the surface, while the sample inside remains unchanged; LOI (2): the oxygen content, at which a surface flame appears at the moment of ignition, and it spreads downward on the surface, and the sample would be ignited from its bottom (standard LOI); LOI (3): the oxygen content, at which a surface flame appears at the moment of ignition, and it swiftly spreads down on the surface, while the sample inside remains unchanged. After that sample can be ignited again at a higher oxygen content.
LOI (2) and LOI (3) values are 6–8 times higher than LOI (1).
The appearance of the surface flame is bound to a specific oxygen concentration: LOI (1). The surface flame, at a definable oxygen concentration, is able to ignite the sample from below, which will result in an upward burning. This cannot be regarded as standard, but the oxygen concentration can be measured, at which this phenomenon occurs, i.e., a LOI value can be assigned to it. At this point, we can interpret LOI (3), which means tertiary combustion (Figure 4(c)). The rate of the upward burning is far greater than the standard downward burning, so, before we could measure a significant burning, the surface of the material would have already burned. Re-ignition can be dangerous during the use, because the presence of fluffs is the property of the material, and thus, proper burning cannot be observed. There may also be a surface flame, when the top of the sample cannot be ignited, so, it is not possible to observe proper burning. If the material is of quite a loose structure, even a surface flame is able burn through the entire thickness of the material. Surface flame can also be caused by the burning of the additional finishing or the residual oxygen-containing functional groups or contaminants, found on the surface of the threads, but not belonging to the structure itself. This phenomenon is disturbing because it repeatedly ignites the lower edge of the sample. It burns violently, the rate of which is higher than of the standard downward burning, making it sometimes impossible to measure the LOI (2) [9,18,28,29]. Generally, three different LOI values can be assigned to the three combustions and the combustion range, which is the lowest oxygen content, when it shows the first flame, and which is the one that shows a stable self-sustaining combustion.
Felts react sensitively to macrostructural changes and show different burning phenomena. There is a significant difference between the burning of unfixed, loose, almost cotton wool-like felts and the more compact, quite dense felts. According to our observations, the oxygen indices of felts are the most sensitive to the macrostructure of the product amongst other macrostructural forms.
The effect of compactness on the flammability of felts
Our observations show that solid-phase combustions (such as glow propagation) are typical in the case of dense thread in close contact with each other. In such a case, the specific heat transfer surface plays a role, which can be expressed in the LOI value. The deviation of oxygen index of even up to 10 units can be observed at a material of the same mass and quality. After the surface flame has run through, the sample can no longer be re-ignited, while its interior will remain intact.
Loose felts only exhibit surface flame at 25%–28% oxygen content. After removing the external burnt surface, they become re-ignitable again. The measurements were taken in two steps. First, we measured the minimum oxygen index (LOI (1)), where the surface flame appears. Later, when the surface has burnt, by removing the burnt surface, we tried to ignite the same sample again. Re-ignition was again followed by the appearance of a surface flame. This flame occurred at an increased oxygen concentration. Thus, we defined a new oxygen index (LOI (2)), where the sample can be ignited and become flammable. There was no surface flame during re-ignition. The spread of the propagation may be a rapid flare or a prolonged local glowing. In the case of two characteristic felt samples, the measurement data of the above-described phenomena are summarized in Table 2.
The LOI values in function of fibre density.

LOI: limiting oxygen index.
It is clearly visible that felts respond to even a relatively low-density difference from the aspect of the oxygen index. Oxygen index differences within the same sample indicate composition and thickness inhomogeneity. Observations indicate that light colour areas burn faster and there is a charred, sparse, loose structure left behind them.
The effect of area weight on the flammability of felts
Two of the characteristics of compactness are thickness and area weight (gm−2), i.e., how loose or compact a non-woven fabric is. Their thickness is typically 5–6 mm during production. However, one can find, in some samples, thinner or thicker areas as well. The burning of low area weight (below 300 gm−2) fabrics is characterized by surface flames, often at 25 to 28 oxygen index (Figure 5).

Area weight parameters influencing the oxygen index of non-woven fabrics.
Solid, high-density felts burn evenly, without surface flames (Figure 2(c)). After burning, the structure remains, the structure does not disintegrate (Figure 2(a)).
Above a certain compactness (∼300–400 gm−2), there was no visible surface flame, only a standard downward burning spread without flame. The disappearance of the surface flame occurs at the combustion in the air of 50% oxygen content (Table 2). Thus felts are also able to burn with flames, surface flames or just glow. The oxygen index of denser felts without surface flames is clearly defined by the area weight as shown in Table 3.
Area weight and LOI values of PN based felt samples (fibre density: 1.37 g cm−3).

The oxygen index value is in a linear relation to area weight. As far as combustion phenomena, at an oxygen index above 50%, only glow appears without surface flames; it may happen above 500 gm−2 area weight.
The inhomogeneity of samples
Inhomogeneity is one of the characteristics of felts during production. Its value is also defined by oxygen index. In the case of felt samples, a maximum and a minimum oxygen index can be measured, which can be specified as a range. The magnitude of the range indicates the unevenness of the flammability of a sample. The oxygen index values here correspond to the standard LOI values. Further inhomogeneity may also occur from thickness unevenness. Macrostructural changes also appear in the combustion characteristics. For example, thin parts burn through completely (with a low oxygen index), while thicker areas cannot be ignited at all (with a high oxygen index).
By selecting an inhomogeneous sample, the following oxygen indices were defined: high density (1.379 or 1.387 g cm−3) fibre sample LOI: 47–55 Δ LOI: 8 mixture of fibres with 1.379 and 1.387 g cm−3 (50%–50%) LOI: 36–38.6 Δ LOI: 2.6 low density (1.341 g cm−3) fibre-based sample LOI: 31–32.5 Δ LOI: 1.
Based on oxygen index measurements, the deviations in inhomogeneity are high in case of felts made from high fibre density oxidized threads (1.38–1.39 g cm−3), i.e., the difference between the minimum and maximum measurable LOI is high: 8. Low fibre density (under 1.37 g cm−3) show a lower fluctuation in oxygen index.
The difference in area weight due to the deviations in thickness can be up to 50% (between 190 and 300 gm−2) (Table 1).
A thin (with low area weight) sample easily ignites, and the flame can ignite the sample even from below. In the case of a thicker sample, the surface flame is not able to do so (Table 4). On a larger sample, selecting the thickest and thinnest parts, one may witness high deviations in oxygen index. This means that, within a sample, there will be both combustible and non-combustible areas.
The ignition behaviour of felts with different thicknesses at different oxygen concentrations.

Different oxygen indices can be determined within one sample, depending on the part of the sample we measure: the thick or the thin one. A maximum and a minimum oxygen index can be defined for one sample. In Table 5, we summarized the inhomogeneity and the different LOI values of our samples.
The characteristic and defined oxygen indexes of certain samples.

LOI: limiting oxygen index.
The thin (low area weight) sample (Samples 1 and 2) is easily ignited, and the flame can ignite the material even from below. In the case of a thicker sample, the surface flame is unable to do so (Samples 5 and 6). Contrary to the burning phenomena present in fibrous materials so far, the sample tested, after being ignited, only glows, does not burn with flame in the oxygen range (51%–60%) examined.
It is an important phenomenon that cables (base fibres) in a macrostructure format reduce the oxygen index of the product (fabrics), with the exception of high compactness felts. In this case, an increase of 15–20 LOI can be reached. This fact deserves a special attention, because it is not enough to know the oxygen index of fibres and cables (base fibres), it will not be by far the same as the oxygen index (Table 6) of the product (fabrics). In fact, the biggest changes are made by the macrostructural forms themselves.
The influence of different area weight of non-woven fabrics on oxygen index.

LOI: limiting oxygen index.
Summary
The aim of this study was to determine the effect of the macrostructure of non-woven fabrics on its flammability.
Since the raw material, the oxidized fibres do not burn in the air, conventional flammability and other tests, which occurs in the air (or in nitrogen) (e.g., UL94) do not give enough information about the combustion or provide only incomplete information. That is why we need to increase the oxygen content of the atmosphere, which is already capable of initiating pyrolysis.
The oxidized fibres of our felt samples show similar thermoanalytical behaviour. The thermal analysis showed the beginning of the thermal decomposition of fibres, which gives the surface flame at the combustion. DTA curves show that oxidative pyrolysis runs from 380–400°C up to about 680–700°C.
In this temperature range, the burning of functional groups of the surface of the fibres is performed, which can be seen as a rapid flame spread during the LOI test.
In the case of felts, the flammability (oxygen index) of the product is more sensitively influenced by both microstructural and macrostructural parameters than the density of the fibres used. This also means that the flammability of felt can be defined by several different oxygen indexes. To classify the flammability of the product, it is not enough only specify one oxygen index or the oxygen index of the oxidized (base) fibres. The area weight and surface consistency of felts also influence how the sample burns during the test, i.e., if it flames or not. To classify a product, one can define as many oxygen indexes as the number of burning phenomena.
Non-woven fabrics are considered to be safety flame-retardant that has LOI values higher than 50; it can be reached by felts with an area weight over 500 gm−2. When the oxygen index of the oxidized fibre itself is higher than 50, it can be regarded as a thermodynamically stable structure against burning and flames.
The decisive role of the macrostructure is proved by the fact that different production forms (non-woven fabrics) made from similar microstructural oxidized or carbon fibres will have different combustibility parameters. Interestingly, not even manufacturers have their products classified for flammability; they only provide the oxygen index value for the base material (cable formats, base fibres). In this paper, we have clearly shown that the evolution of the oxygen index is influenced by further processing.
We recommend a new method for classifying felts, which does not only provide one oxygen index but as many oxygen indices can be assigned to a sample as many the burning phenomena are. Additionally, with an oxygen index range, one can provide data for the inhomogeneity of a product that is not recognized by visual observation.
It was demonstrated that the flammability of a fabric is defined by both the characteristics of the fibre (here oxidized fibre – microstructure) and by the different woven structures of the fabrics (macrostructure).
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Authors acknowledge the support for the purchase and use of scanning electron microscope by the Hungarian Research Grant NVKP_16-1-2016-0019 “Development of concrete products with improved resistance to chemical corrosion, fire or freeze-thaw”.
