Abstract
To develop waterproof breathable membranes, a Janus lyophobic/lyophilic media composed of a poly(vinylidene fluoride) electrospun membrane and a hydrogel electrospun mat was proposed. Poly(vinylidene fluoride) membranes were electrospun in different electrospinning conditions to evaluate the effect of the fiber diameter on the membrane characteristics. Afterward, a layer of hydrogel was electrospun on the poly(vinylidene fluoride) electrospun mat. The hydrogel was synthesized through the esterification reaction of poly(vinyl alcohol) and poly(acrylic acid). This hybrid membrane can transfer the body-generated moisture to the environment while it prevents the penetration of liquid water from the reverse side. The proposed hybrid membrane represented improved water vapor permeability compared with the neat poly(vinylidene fluoride) membrane (from 12.6 kg/m2/day for the neat poly(vinylidene fluoride) up to 13.6 kg/m2/day for the hybrid) along with good water resistance (up to 70 kPa) and good windproof property (1 ml/cm2/s in pressure drop of 500 Pa). The results indicated that molecular diffusion is the main mechanism of moisture transfer in the poly(vinylidene fluoride) membranes with the fiber diameter ranging from 132 to 1136 nm. It has been found that the experimental effective diffusion coefficient (Dexp) of the neat hydrogel layer (8.7 × 10−6m2/s) is much higher than the neat poly(vinylidene fluoride) membrane (approximately 4 × 10−6m2/s), signifying that apart from molecular and Knudsen diffusions, the surface diffusion has played a major active role contributing to much higher value of water vapor permeability in case of the hydrogel layer and subsequently in hybrid layers.
Introduction
Breathability is an essential feature of wearable membranes. Water vapor permeability (WVP) and, subsequently, breathability is a crucial factor in making a proper heat and moisture exchange between the body and environment, which is a vital issue in clothing comfort [1–4]. Waterproof and breathable membranes have attracted tremendous attention in many fields, which includes separation membranes, medical applications, and especially wearable membranes. They are designed in a way to be impermeable to liquid water and permeable to water vapor [5–8]. Polytetrafluoroethylene (PTFE) membranes have been used extensively for many years as waterproof and breathable membranes. However, these membranes are expensive, and there are difficulties in regulating the porous structure of these membranes [9]. So, it is necessary to develop alternative technologies for producing waterproof and breathable membranes. Electrospinning has been considered as a promising, versatile, and straightforward approach to produce these membranes [10–12].
Electrospun polyurethane membranes have been employed in several types of research for this purpose [13,14]. Ahn et al. [15] have concluded that the WVP of the electrospun poly(urethane)-laminated fabric is significantly more than the conventional waterproof/breathable fabric (laminated poly(tetrafluoroethylene) (PTFE)). However, the waterproofness of the electrospun-laminated fabric was not as good as the PTFE-laminated fabric. Other neat polymers such as poly(acrylonitrile) and poly(propylene) have been electrospun for this purpose [8]. However, these membranes have revealed inadequate hydrostatic pressure and WVP. Therefore, various methods have also been used for modifying and treating the surface of the materials to improve the hydrophobicity. Modifying the surface of electrospun poly(acrylonitrile) with waterborne-fluorinated poly(urethane) and poly(dimethylsiloxane) are some of these attempts, which resulted in improving the hydrostatic pressure up to 83.4 kPa [16,17]. In the mentioned researches, additional process was applied for improving the hydrophobicity of the membrane up to 83.4 kPa. This is while Yang et al. [8] have proposed the poly(vinylidene fluoride) (PVDF) electrospun membrane as a better choice for producing waterproof breathable membranes with waterproofness of 110 kPa which does not need additional modification. PVDF has been widely used in different fields due to waterproofness, good thermal stability, and acceptable mechanical properties [18,19]. Nevertheless, improving the breathability of the waterproof membranes has remained a crucial challenge [20]. So, it is necessary to balance between waterproofness and breathability with contrasting nature. This is also an essential issue in gas drying, humidification/dehumidification applications, and membrane distillation. Janus membranes with the capability of directional and continuous water transfer have attracted researchers’ interest and could be good candidates for using in wearable membranes. Hydrophilic surface modification of hydrophobic membranes such as alkaline hydrolysis [21], incorporating nanoparticles with polymeric membranes [22], producing hydrophilic/hydrophobic hollow fibers, are some of the attempts that have been carried out for this purpose [23,24]. However, research is still ongoing to develop an improved and simple approach for producing waterproof breathable membranes. It seems that utilizing porous hydrogels (electrospun hydrogels) for the hydrophilic part of the Janus membrane is a novel idea for producing membranes with enhanced WVP, which could be considered in different fields such as wearable membranes.
This study aims to develop a new approach for improving WVP of a waterproof membrane. For this purpose, a layer of electrospun hydrogel (a porous hydrophilic layer) was coated on the electrospun PVDF membrane. Hydrogels are networks of polymer chains which have the capability of swelling and retaining a considerable fraction of water within their structures. These materials have also been used increasingly in tissue engineering, drug delivery, contact lenses, agriculture, and waste-water treatment in different forms, such as electrospun mat [25–27]. It was expected that the surface diffusion through the hydrophilic pores would improve the water vapor transfer across the membrane. This hybrid membrane (Janus membrane) has both hydrophobicity and hydrophilicity simultaneously on two different sides. This Janus membrane is able to absorb the body-generated moisture (sweat) and transfer it to the environment while it prevents the penetration of liquid water from the reverse side. PVDF fibers were electrospun in different conditions to produce fibers with different diameters. The influence of PVDF fiber diameter on the water vapor diffusion coefficient of the layers was experimentally and theoretically investigated. The effective diffusion coefficient of the hybrid layer was calculated experimentally and compared with that of PVDF layers.
Experimental details
Materials
PVDF granule (Kynar 720, Arkema, MW: 265 kDa), poly(acrylic acid) (PAA, Sigma, Mv: 450 kDa), and poly(vinyl alcohol) (PVA, Fluka, Mw: 72 kDa) were used in this research. Dimethyl sulfoxide (DMSO, Merck, purity ≥99.9%), acetone (AC, Merck, purity ≥99.8%), N,N-dimethylformamide (DMF, Merck, purity ≥99.8%), ethanol (Merck, purity ≥99.5%), and deionized water were used as solvents in the electrospinning process.
Membrane preparation
The first layer (PVDF)
PVDF solutions were prepared using 21, 23, 24, and 27 wt% of PVDF with different solvent ratios and salt (LiCl) content to produce fibers with different diameters. Solutions were stirred for 3 h at 50°C. The polymer solution was transferred into a glass syringe. A syringe pump was applied to inject the polymer solution through a 22 G blunt-tipped needle, and the feeding rate of the polymer was adjusted to 0.5 ml/h. A high voltage power supply was applied to create an electric field between the nozzle and a rotating collector, which was covered with aluminum foil. The solution properties and the electrospinning parameters for producing samples are presented in Table 1. The hot-press process was applied on electrospun samples at 150°C for 15 min for easier peeling the samples from aluminum foil.
Electrospinning parameters.

The second layer (hydrogel)
The PVA solution (5% w/w) was made by dissolving PVA powder in deionized water, which was heated to 90°C and then stirred for 6 h. The PAA solution (5% w/w) was also prepared by dissolving PAA powder in deionized water and stirred for 6 h at 40°C. PAA and PVA solutions were mixed (PAA: PVA, 5:3) and stirred for an hour. To produce the hybrid membranes, a 3 wt% of PAA-PVA (5:3) was electrospun on the hot-pressed PVDF mat. The schematic of the production process of the hybrid membrane is shown in Figure 1. Electrospinning parameters for producing the hydrogel layer (the second layer (represented by H)) are also written in Table 1. Hybrid samples are represented by HM (HM100, HM200, HM500, and HM1000) in this article, where H shows the hydrogel layer, M shows the PVDF membrane, and the number represents approximately the diameter of the PVDF fibers which were obtained using scanning electron microscopy (SEM) images (Figure 8). Hybrid samples were placed in a vacuum oven at 80°C for 20 h and then heated at 140°C for 20 min under the vacuum. Cross-linking was induced by esterification between the hydroxyl and carboxyl groups of the PVA and PAA during the heating process (heating at 140°C for 20 min).

Schematic of the production process of the hybrid membrane.
Hydrogel characterization
Swelling studies were carried out gravimetrically on the casted hydrogel film. The hydrogel solution (PAA: PVA, 5:3) was casted on to a glass petri dish. In order to maintain a slow solvent evaporation rate and prevent the creation of bubbles in the casted film, the casted solution was placed under a porous cover in room temperature for three days and then placed at 80°C under vacuum for 20 h. Cross-linking was induced by esterification between the hydroxyl and carboxyl groups of the PVA and PAA during the heating process (heating at 140°C for 20 min). Afterward, the hydrogel film was immersed in deionized water and then weighed after removing the excess of water laying on the surface using filter paper. Immersed samples were weighed until equilibrium to plot swelling ratio (S) versus time. Swelling ratio was determined using the following equation
Thermogravimetric analyzer (TGA, SDT Q 600, TA instruments) was used to evaluate the thermal stability and chemical phenomena during heating the PAA–PVA blend. Thermogravimetric analysis was performed under a nitrogen atmosphere, and the heating rate was adjusted to be 20°C/min. The chemical composition of the blend and neat polymers were evaluated using Fourier-transform infrared spectroscopy (FT-IR, NEXUS 670, Nicolet).
Membrane characterization
A SEM (XL 30, Philips) was used to study the morphology of the electrospun membranes. The fiber diameter was also calculated via image processing commercial software “image J.” The thickness of the membranes was obtained using the Mitutoyo micrometer. The water contact angle (WCA) was measured using a contact angle analysis system, including a CCD camera and image processing software. The contact angle of a 5 μl water droplet was measured by processing the picture captured by the camera. The average values of five measurements were reported for each sample. The porous structure was investigated using the gas–liquid displacement method [28]. Samples with a diameter of 28 mm were immersed in isopropyl alcohol as a wetting liquid. Hence, the pores of the membrane were filled automatically due to the favorable capillary force. Then, a non-reacting gas (air) was used to take the wetting liquid away from the pores of the membrane. The pore diameters can be related to the differential pressure using the following equation

Air permeability was measured using Shirley Air Permeability Tester in different pressure drops (25, 50, 75, 100, 150, 200, 300, 400, and 500 Pa). Hydrostatic pressure was investigated using a home-made setup shown in Figure 2. Samples were placed inside the module cell with an effective diameter of 40 mm. The deionized water was poured in the module cell and the flexible pipe connected to it. The hydrophobic side of the membrane (electrospun PVDF) was in contact with water. The pressure was applied from an air compressor through a pressure gauge to the surface of water poured in the pipe. The applied pressure was increased gradually with a rate of 6 kPa/min. The experiment was kept on until the observation of three droplets of water on the outer side of the membrane in the mirror, and the required pressure was reported. The reported value of the air permeability and the hydrostatic pressure were the average values of five specimens. Tensile properties of the electrospun samples were investigated using Instron 5566 Universal Testing Machine with a 50 N load cell. Samples were cut into strip shape (5 × 20 mm2) and tested with the extension rate of 5 mm/min. The average value of five specimens was reported. The adhesion of two layers was measured through 180° peeling test. For this purpose, samples were cut in strips (1 cm × 6 cm). The hydrogel mat was clamped by the movable clamp of the instrument (Instron 5566 Universal Testing Machine with a 50 N load cell), and the PVDF mat was held by the stationary clamp and peeled apart at a constant displacement rate of 50 mm/min. The average of five highest peaks was calculated for each sample.

Schematic of the hydrostatic pressure test.
Moisture transfer through the membrane
The water vapor permeation of the mats was in situ monitored using a TGA instrument. In order to evaluate the moisture permeability of the membranes, a piece of a sample (7 × 7 mm2) was fixed on an open of a platinum TGA pan containing deionized water (40 mg) using double-sided copper tape, and it was fully sealed, as illustrated in Figure 3. The weight loss of samples (due to water evaporation through the sample) was measured using very sensitive mass sensing of the TGA instrument. This test was carried out at 25°C, and airflow (100 ml/min, dry air) was also being passed over the sample during the test. The WVP during 24 h was determined using this method.

Schematic of TGA pan and the sample placed on it to perform WVP test.
Moisture is transmitted through the membrane due to water vapor concentration difference of the two sides of the sample. The mass transfer resistances of the membrane in this test method are shown in Figure 3. The steady-state flux of water vapor (i) through the three resistances is as follows (30)

Here, 1/Ki can be determined by dividing the height of the air gap (L) with the water vapor diffusion coefficient in the air (Dva) at the experimental temperature (Dva ≈ 2.5 × 10−5 m2/s at T = 25°C) [30]. In order to obtain the value of Ku, the weight reduction of a full of water TGA pan (without the membrane), which was exposed to the dry airflow, was in situ monitored [31–33]. Hence, Ku can be obtained using the following equations

Theoretical effective diffusion coefficient of the membrane (DTheo) was also obtained using the Bosanquet equation, which is a limiting case of the dusty gas model (DGM). According to this model, the effective diffusion coefficient of the membranes (the PVDF membrane) can be obtained by reciprocal additivity law of the molecular diffusion (DM) and Knudsen diffusion (DK) as follows [35–37]

The effective diffusivities of PVDF membranes were calculated using experimental data (equation (5)), and also, it was calculated through the theoretical equation (equation (8)). The effective diffusion coefficient of the hybrid membranes was also calculated experimentally and compared with the PVDF membranes.
Results and discussion
Hydrogel characterization
The hydrogel was synthesized by blending the PAA and PVA with the ratio of 5:3 corresponds to a 4 COOH/OH mole ratio. This PAA to PVA ratio is the proper figure as it seems that the greater extents of the cross-links have been formed in this ratio, which is consistent with the findings of Jin et al. [40]. The swelling ratio of the selected hydrogel is shown in Figure 4. As seen, the sample was swelled about 2.2 times and then reached equilibrium in 30 min.

The swelling ratio of PAA–PVA (5:3) hydrogel.
The results of thermogravimetric measurements of neat polymers and the blends are shown in Figure 5. As can be seen, the thermal degradation of PVA has a two-step mechanism, which is consistent with the literature. The first peak is related to the dehydration of PVA, which leads to the formation of polyene structures, and it will be degraded at higher temperatures [41]. The rate of weight loss curve (DTGA) also has two separate maxima at 360 and 456°C. Two maxima can also be observed for neat PAA at 305 and 410°C. PAA also has a two-step thermal degradation curve. The first step involves the formation of anhydride and then its degradation. A small step can be seen in the TGA curve of the blend, which is started at approximately 130°C. This thermal event can be attributed to esterification between COOH groups of PAA and OH groups of PVA, which is associated with the release of water; in comparison with literature, Arndt et al. [42] were also observed such thermal event in their research. The second step at 225°C attributed to the formation of anhydride, which takes place between two carboxyl groups of PAA. At higher temperature, thermal degradation of the blend takes place.

TGA and DTGA curves of PVA, PAA, and PAA: PVA (5:3).
The FT-IR spectra of the hydrogel sample was collected and shown in Figure 6 to verify the presence of esters, which make bridges between hydrophilic polymer chains. In this figure, neat PVA, neat PAA, as well as its blends cured at two distinct temperatures 140 and 300°C for 20 min, and also uncured blend has been presented. Low-temperature curing (140°C) is related to hydrogel preparation, and the high-temperature curing (300°C) is evidence for anhydride formation, which was claimed in the TGA signal. All the spectra are normalized with respect to the C–H stretching band (at 2800–3000 cm−1). The spectrum of neat PVA reveals O–H stretching at 3387 cm−1 and C–H asymmetric stretching at 2921 cm−1. The spectrum of neat PAA has characteristics peaks at 3167 cm−1 (O–H stretching), 2921 cm−1 (C–H asymmetric stretching), and 1709 cm−1 (C=O stretching). The O–H stretching vibration at 3387 cm−1 from neat PVA was gradually shifted to higher wavenumber in blend samples (PAA–PVA-140 and 300) and also weakened and broadened. The intensity of the O–H stretching band (at 3000–3800 cm−1) decreased by applying the heating process in cured samples (PAA–PVA-140 and 300) in relation to the unheated blend (PAA–PVA-0) which indicates the consumption of the hydroxyl groups during the esterification reaction. The C=O stretching peak was slightly shifted from 1709 cm−1 in neat PAA toward higher wavenumber in blends. These changes indicate the creation of new hydrogen-bonding interactions between polymers in blends. Since an ester has characteristic peaks at similar wavelengths to carboxylic acid and alcohol groups, it is difficult to distinguish the formation of an ester bond in the spectrum. As can be seen, a shoulder has appeared in PVA–PAA-300 at 1807 cm−1. This shoulder can be attributed to the formation of anhydride in higher temperatures, which is consistent with TGA results, and it is also consistent with literature [43,44].

The FT-IR spectra of PAA, PVA, and their blends (PAA: PVA, 5:3).
Electrospun membrane characterization
As it was mentioned, the purpose of this study is to propose a new approach for improving the WVP of the electrospun PVDF membrane by coating a hydrogel layer on it. Both hybrid and neat PVDF membranes were evaluated in this study. The production process of the hybrid layer is shown in Figure 1. As it was mentioned, the hydrogel solution (PAA: PVA, 5:3) was electrospun on a PVDF mat and then heated at 140°C for 20 min by means of creation of the cross-links through an esterification reaction. The results which have been reported in the previous part (swelling, FT-IR, and TGA curves) confirmed the creation of cross-links between the polymers chain. The SEM image of the hydrogel layer after the heating process is shown in Figure 7. The diameter distribution of the fibers is shown in this figure as well. The average diameter of the hydrogel fibers is 175 nm.

SEM image and fiber diameter distribution of the hydrogel layer.
SEM images and diameter distributions of electrospun PVDF are shown in Figure 8. The average diameter of sample M100, M200, M500, and M1000 is 132, 193, 576, and 1136 nm, respectively. Fibers with the desired diameter have been produced by tuning the solvent ratio, polymer concentration, LiCl content in the solution, and applied voltage. As can be seen, an increase in the acetone content in the solution resulted in the production of coarser fibers, which would be because of quick evaporation of acetone (because of high vapor pressure) and faster solidification of polymer jet. Hence, the polymer jet would not be stretched effectively. Coarser fibers have been produced by increasing the acetone content, the concentration of polymer, and decreasing in voltage (M500 and M1000). LiCl has been used to reduce the diameter of the fibers and the number of beads (M100 and M200) in this research. Adding salt to the polymer solution, decreasing the concentration of the polymer in the solution, and increasing the voltage up to 22 kV were the factors that affected the reduction of the fiber diameter and the number of beads. The optimal electrospinning parameters for producing desired electrospun fibers were reported in Table 1. The thickness of the PVDF membranes was adjusted to be 55 ± 5 μm by adjusting the electrospinning duration. The thickness of the hybrid membranes was also 55 ± 5 μm (which composed of a 15 μm hydrogel layer on a PVDF layer).

SEM images and fiber diameter distribution of PVDF electrospun fibers: (a) M100, (b) M200, (c) M500, and (d) M1000.
The mechanical performance of a membrane is a vital issue in practical applications. Therefore, the tensile properties of the membranes were assessed by tensile testing. The stress–strain curves of PVDF membranes are shown in Figure 9(a). High resistance to deformation can be observed in the first part of the curves which followed by a yield point and after that a gradual decrease in modulus due to fiber slippage.
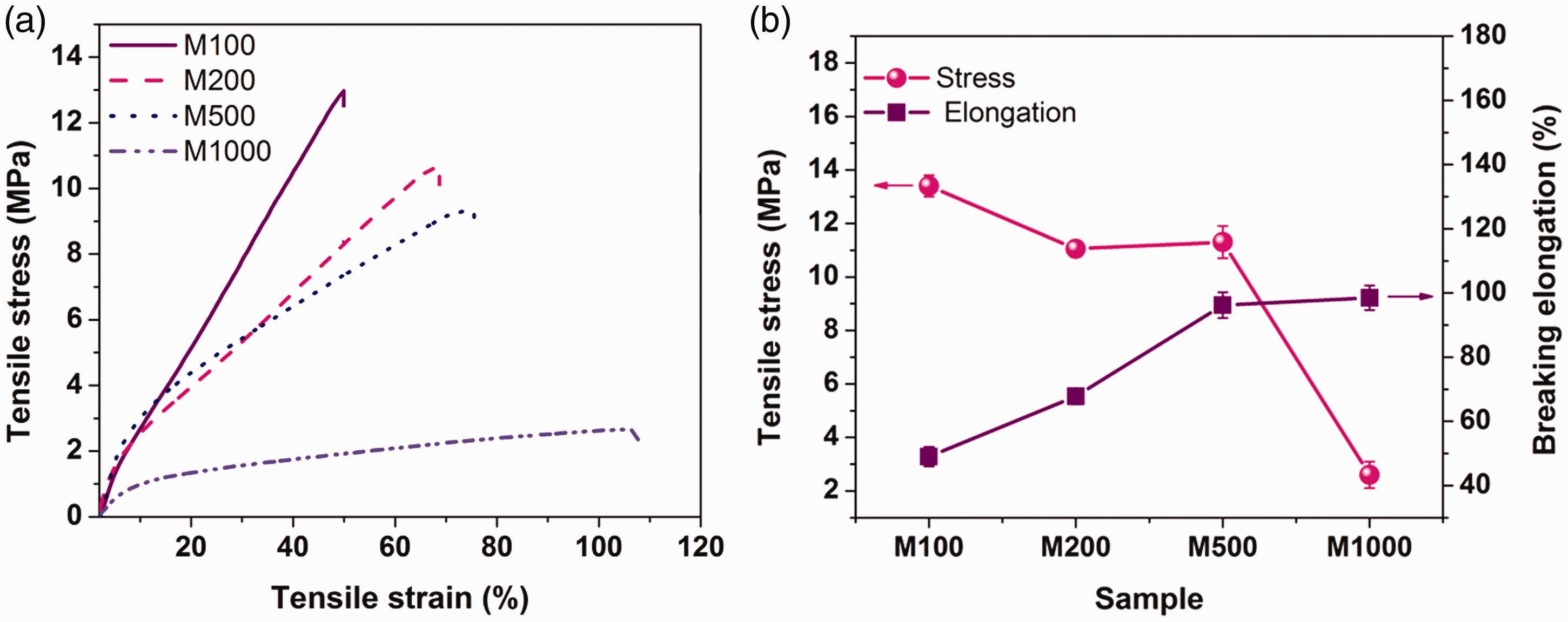
(a) Stress–strain curves of the PVDF mats; (b) tensile stress and strain of the PVDF membranes.
As shown in Figure 9(b), the tensile strength increased from 2.6 to 13.4 MPa with the decreasing of the fiber diameter of the samples (from M1000 to M100). The tensile strain decreased from 98.5 to 49%, with the decreasing of the average of the fiber diameter of the samples (from M1000 to M100). The mechanical properties of fiber mats are affected by individual fiber properties, the geometrical arrangement of the fibers, and interaction among fibers [45–47]. Coarser fibers form a membrane which is more porous and subsequently has less fiber-to-fiber contacts at the same area density. Hence, these porous structures have lower tensile strength and more tensile strain as a result of fewer bonding points among fibers. As shown in Figure 9(a), a larger strain after the yield point is observable in sample M500 and M1000, which is the result of less bonding points among fibers and more fiber slippage. Besides, thinner fibers have stretched more during the electrospinning process, which resulted in higher molecular orientation, and more contribution to enhancing the tensile stress of a single fiber [47,48]. Based on the results, the sample composed of thinner fibers (M100) represented better mechanical performance. As it was mentioned, 180° peeling test was performed to assess the adhesion of the layers. The average of five highest was calculated for each sample, and the values are 2.4, 3, 4.1, and 5.8 cN/cm for M100, M200, M500, and M1000, respectively. As seen, with increasing the PVDF fiber diameter in the mat, the adhesion force of the layers increased, which can be interpreted by the mechanical interlocking theory. Hence, it seems that the larger pore size between the fibers can lead to more mechanical interlocking and subsequently, more adhesion. The values of the adhesion force were compared with the other researches, and it seems that they are comparable with them. These values are also acceptable compared with the adhesion force that Rombaldoni et al. [49] could create between the substrate fabric and electrospun layer by treating the substrate layer using plasma.
The maximum pore size and the porosity of the PVDF mats are presented in Figure 10. The maximum pore size of the membrane increased from 761 to 3860 nm with increasing the fiber diameter from 132 to 1136 nm. However, the porosity of the membranes has slightly increased by increasing the fiber diameter. The porosity and the maximum pore size of the hydrogel layer were also measured, and the values are 80.86% and 1.02 μm, respectively.
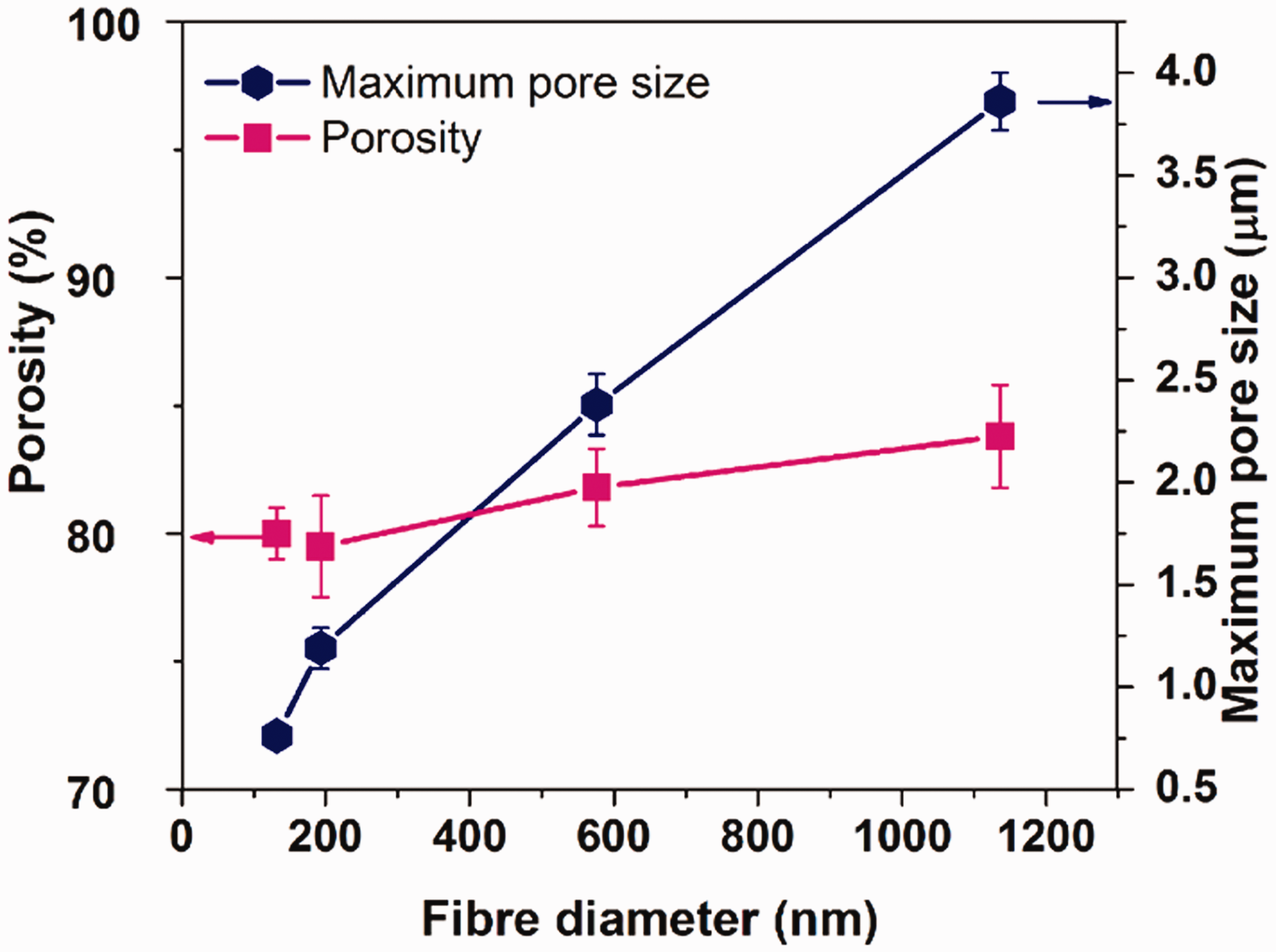
The maximum pore size and the porosity of the PVDF samples.
Performance of hybrid membrane
Waterproof property
The hydrostatic pressure of the neat PVDF and hybrid membranes are shown in Figure 11(a). As seen, the hydrostatic pressure of the PVDF membrane has increased from 10 to 100 kPa with decreasing the fiber diameter from 1136 to 132 nm. However, the hydrostatic pressure of hybrid samples has increased from 10 to 70 kPa by decreasing the fiber diameter of the PVDF substrate. It seems that the presence of a hydrophilic layer on the other side of the sample results in a reduction of hydrostatic pressure. Although the hydrostatic pressure has decreased compared with the neat PVDF membrane, but it is still good for waterproof applications (especially sample HM100). Therefore, it is better to select the hybrid samples composed of PVDF membrane with thinner fibers (HM100) to reach an appropriate water-resistance (70 kPa). Meanwhile, this result can be described by the Young–Laplace equation,

(a) hydrostatic pressure of PVDF and hybrid membranes and (b) static WCA of PVDF and hybrid layers.
Figure 12 shows the water droplet on the surface of the hydrogel mat right after the insertion and 10, 20, and 30 s after the insertion of the droplet. As can be seen, the droplet spread on the surface and was adsorbed quickly by the hydrogel.

Water droplet on the surface of the hydrogel mat right after the insertion and 10, 20, and 30 s after the insertion of the droplet.
Windproof property
The air permeability test was employed to assess the windproof performance of the membranes. Decreasing the amount of air permeability is a necessity in windproof applications. As can be seen in Figure 13, the air permeability of the membranes is affected by the fiber diameter. As seen, samples which made up of finer fibers (M100, M200, HM100, and HM200) exhibit better windproof performance compared with the samples composed of coarser fibers. By increasing the fiber diameter, the amount of air could pass through the neat PVDF membranes increases. However, electrospinning a hydrogel layer on the PVDF electrospun membrane led to a reduction in the air permeability of hybrid samples (HM500 and HM1000). It seems that the presence of an electrospun layer, which composed of finer fibers, and subsequently smaller pore size resulted in the reduction of the air permeability in these samples. Nevertheless, there is no significant difference between the air permeability of samples HM100 and HM200 with samples M100 and M200. It should be mentioned that, since the values of the air permeability of the samples M100, M200, HM100, and HM200 were pretty close together (below 1 ml/cm2/s), the points were placed almost on each other in this figure. It seems that since the diameter and the morphology of the hydrophilic and hydrophobic fibers in these hybrid samples (HM100 and HM200) are approximately the same, there is no difference in the air permeation of samples M100, M200, HM100, and HM200. Ryu et al. [52] have also examined the gas permeability of electrospun Nylon 6 mats with different fiber diameters (from 90 to 500 nm). They also have concluded that the gas permeability of the electrospun membrane is affected by fiber diameter and pore size.

Air permeability of the membranes.
Breathability
The WVPs of the samples are shown in Figure 14. Achieving a higher amount of WVP is a necessity for producing a better breathable membrane. To gain this goal, the electrospinning of a hydrophilic layer on the PVDF membrane is proposed in this article. The results also indicate that the WVP of the hybrid membranes has been increased up to 13.6 kg/m2/day, which is 1 kg/m2 more than the neat PVDF membrane in a day. The Dexp of the membranes was also calculated using the WVP data through equation (5), and the results are shown in Figure 15(a). Actually, the Dexp of the membranes was calculated by eliminating the influence of the other mass transfer resistance (1/Ki and 1/Ku) on the flux, as described in “Moisture transfer through the membrane” section. As seen, the Dexp of the hybrid membranes is much more than that of neat PVDF membranes. The Dexp of the neat hydrogel membrane was calculated by using experimental data through equation (5), and its value is 8.7 × 10−6 m2/s, while Dexp of the neat PVDF membranes is approximately 4 × 10−6 m2/s. As it is mentioned in detail in Appendix 1, moisture transfer occurs via a combination of Knudsen diffusion and molecular diffusion in the neat PVDF membranes. However, the Dexp of the neat hydrogel layer is approximately two times more than that of neat PVDF membranes. Hence, it seems that another mechanism of mass transfer is accompanied by the molecular diffusion and Knudsen diffusion in hydrogel layers, which is surface diffusion. Surface diffusion can play an important role in strongly adsorbing species. According to this, water vapor molecules may adsorb on the pore walls of the porous membrane and migrate along the pores [53,54]. Surface diffusion occurs in parallel with the other mechanism. Hence, the effective diffusion coefficient can be written as follows

WVP of PVDF and hybrid membranes.
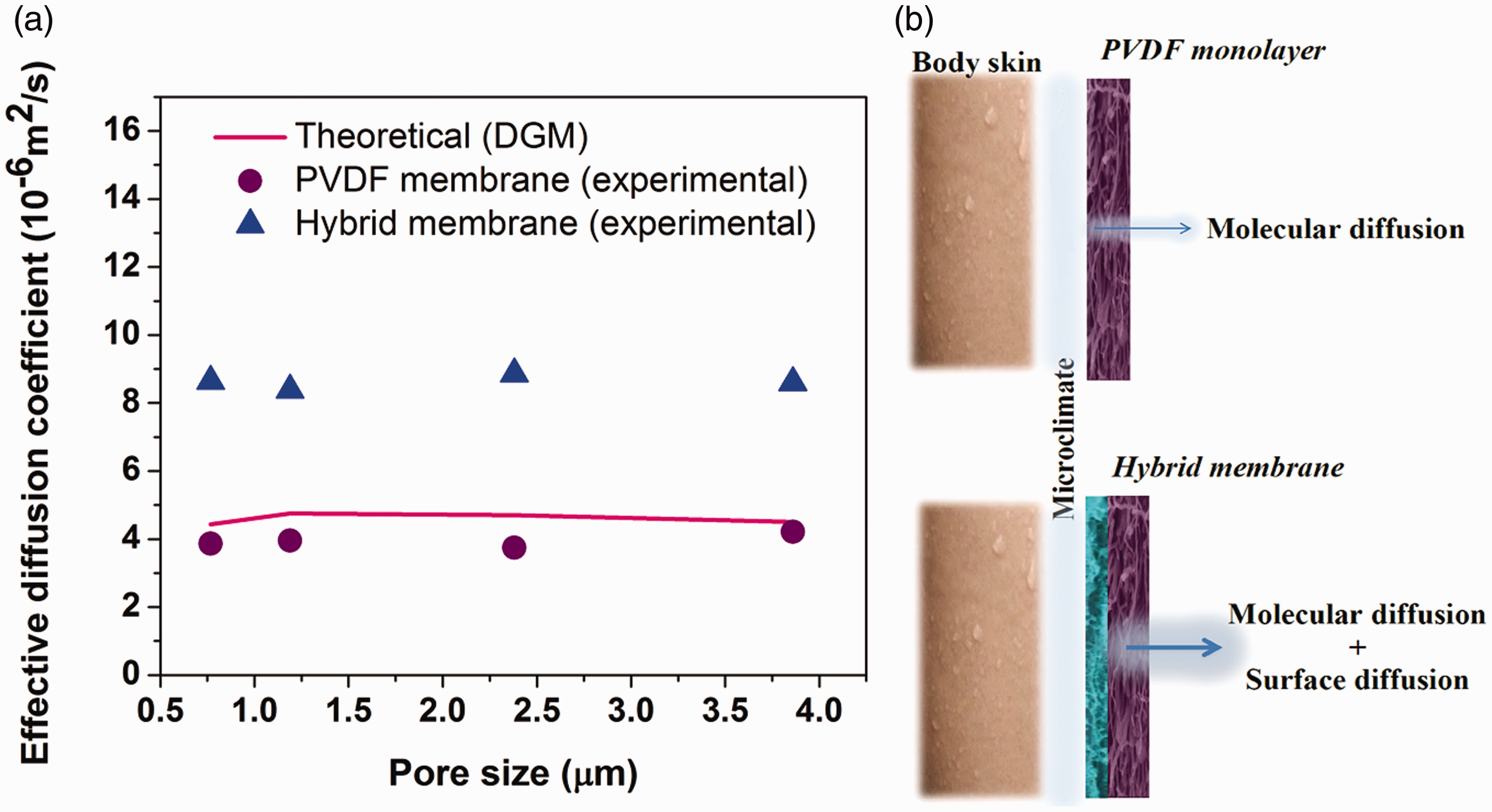
(a) Dexp of PVDF and hybrid membranes and Dtheo of PVDF membranes versus pore diameter and (b) the scheme of the moisture transfer mechanism through the membranes.
The Dtheo of the PVDF membranes was calculated using equation (8), and the results are shown in Figure 15(a). The values of the effective diffusion coefficient calculated using the theoretical model, and those calculated experimentally are in good agreement, and they are in the same order of magnitude (10−6 m2/s). So, it seems that equation (8) is a good model for hydrophobic membranes. Nevertheless, the contribution of the surface diffusion should not be neglected for strongly adsorbing diffusant such as water vapor in the hydrogel.
The other result that can be concluded from Figure 15(a) is that the values of the effective diffusion coefficient of the membranes have not changed significantly by increasing the pore size. The Knudsen numbers were calculated for the membranes with different pore size, and the values are between 0.026 and 0.13. Two possible mechanisms for describing the mass transfer through the produced membrane during the WVP test (for 0.01 < Kn < 1) are Knudsen and molecular diffusion (see Appendix 1). Since, mean pore radius affects the Knudsen diffusion and has no influence on ordinary molecular diffusion (according to equations (11) and (13) in Appendix 1), it seems that the molecular diffusion is the dominant mechanism of moisture transfer in PVDF membranes. Besides, the calculated Knudsen numbers for the membranes are near the lower limit (0.01), so the dominance of molecular diffusion is not unexpected. Therefore, it can be concluded that the membrane composed of finer fibers exhibited not only enhanced strength, waterproof, and windproof properties but also good breathability. So, hybrid membranes composed of finer PVDF fibers are appropriate candidates for waterproof, windproof, and breathable membranes.
Conclusion
In summary, a hydrophobic/hydrophilic Janus membrane composed of PVDF and hydrogel electrospun mat was produced via electrospinning in order to be used as a waterproof and water vapor permeable membrane. Hydrogel solution composed of PVA and PAA (3:5) was electrospun on a layer of electrospun PVDF, and then it was heated at 140°C for 20 min by means of creation of cross-links between the polymer chains of PVA and PAA and producing a limited swelling hydrogel. TGA and FT-IR analysis were also performed to trace the esterification between the hydroxyl groups and the carboxyl groups of these polymers and validate the esterification reaction condition. The optimized hybrid membrane (HM100) represented an improved WVP (from 12.6 kg/m2/day for the neat PVDF up to 13.6 kg/m/day for the hybrid) beside good windproof property (1 ml/cm2/s in pressure drop of 500 Pa). Although the hydrostatic pressure of the hybrid membranes has decreased compared with the neat PVDF membrane, but it is still good for waterproof membrane (70 kPa for sample HM100). The influence of the PVDF fibers diameter on the performance of the membrane was also evaluated. The results indicated that the membranes composed of thinner fibers represented not only better mechanical performance, waterproofness, and windproof property but also a good WVP compared to membranes those composed of coarser fibre. The theoretical deduction indicated that molecular diffusion is the main mechanism of water vapor permeation across the PVDF membranes, and that is why there is no significant difference between the amounts of WVP of the membrane composed of different fiber diameter. It has been found that the experimental effective diffusion coefficient (Dexp) of the neat hydrogel layer (8.7 × 10−6 m2/s) is much higher than the neat PVDF membrane (approximately 4 × 10−6 m2/s), which indicates that the surface diffusion has played a major role in moisture permeation through the membrane apart from molecular and Knudsen diffusions. So, incorporating a porous hydrogel layer sped up the water vapor transfer and enhanced the breathability of the membrane.
Footnotes
Acknowledgements
The authors would like to thank Ms Leili Mahmoudi, Mr Hamid Fattahi Juybari, and Dr Bahareh Yousefi for experimental help.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
