Abstract
Development of conductive fabrics causes usually deterioration of the some of the comfort attributes of the final textile product. In this work, we propose a simple non-deteriorative method for preparation of electrical conductive fabrics based on wool and polyester as well as the blended fabric thereof. The unmodified fabrics or saponified polyester or oxidized wool were treated in aqueous solution containing heavy metal ion salts, namely, silver nitrate, copper II sulfate using exhaustion technique at 70°C for 1 h followed by reduction step. Nano zinc oxide was also used directly for treatment on the said fabrics. The electrical conductivity of the metal ions-treated fabrics and the influence of sewing on their conductivity was measured. The effect of pretreatment of the fabrics, saponification of polyester or oxidation of wool on their ability to absorb metal ions was monitored. Scanning electron microscopy and energy dispersive X-ray spectroscopy were used to prove the incorporation of metal particles onto the treated fabrics. The change in chemical composition of the treated fabrics was studied using Fourier transform infrared spectroscopy. Atomic absorption was adopted to measure the amount of metal incorporated onto the treated fabrics. The formation of nanometal crystals was proved using X-ray diffraction pattern. Different parameters of comfort, namely, air/water permeability of the treated as well as untreated fabrics were assessed. The treated fabrics exhibit enhanced electrical conductivity as well as some improved comfort properties without any effect on their inherent mechanical properties.
Introduction
In the last few decades, conductive textiles have been spread out very widely. Such fabrics can be used for medical and military applications, protective clothing, and are called smart- or e-textiles [1].
Conductivity in fibers and yarns are being utilized, not only for conducting electricity and sensing but also for delivering antimicrobial [2,3] anti-static [4,5] and electromagnetic shielding [6,7] properties. Currently, researchers and industry have produced conductive yarns out of whole metals known as metallic yarns, hybrid yarns out of a combination of metallic yarns and conventional textile yarns [8], yarns laminated with conductive films, yarn coated with conductive solutions, and yarns made from nanomaterials [9]. The selection of conductive yarns for a smart textile application should be based on the level of conductivity needed, durability of the conductive component, yarn material content, method to be used in fabrication, suitability to wear, and comfort. Some conductive yarns tend to behave less than their corresponding normal textile yarns and their brittle characteristics can damage weaving or knitting machinery during production [10]. Moreover, they may also be uncomfortable to wear due to abrasion [11] and prone that inducing allergic contact dermatitis [12,13]. Recently, clay, metal and metal oxide nanoparticles, and graphene, graphite, and carbon nanotubes (and allotropes) are being studied for conductive textile applications [14].
Recently, nanocoating is an interesting technique in the textile field and became under research due to the materials processed at nanoscale have greater surface areas comparing to conventional coatings [15,16]. Nanoscale materials can be dispersed or converted to conductive polymer composite solutions and coated on yarns to impart conductivity [17,18]. Conductive finishing on yarns is more controllable, durable, and significantly enhances the functionality by incorporating various nanoparticles, i.e. silver nanoparticles, gold [18,19], or creating nanostructured surfaces.
Literatures contain many researches that deal with electric conductive yarns for smart textiles applications. In situ synthesis of silver nanoparticles on nylon fabric has been introduced through a chemical reduction method in order to create a thin layer of silver nanoparticles on nylon fabric producing an electro conductive fabric with considerable antibacterial properties [20]. In 2018, a unique design of a conductive coating on cotton fabric to improve the electrical stability is proposed. The coating has a hierarchical structure composed of an interlayer of silver nanoparticles (Ag NPs), a top layer of conductive hydrogel and a bottom layer of L-cysteine binder [21].
Polyester (PET) as a synthetic fabric and wool as a natural one are prevalently used textile materials for outer garment. Silver and copper nanoparticles were used to achieve antibacterial, coloration, and conductive function for these fabrics [22,23].
This work is devoted to chemical modification of wool, polyester, and wool/polyester fabrics with heavy metal ions or nanometal oxide using exhaustion technique followed by reduction to impart permanent electrical conductivity without any deterioration in their comfort attributes and mechanical properties.
Experimental
Materials and reagents
Heavy weight 100% wool and wool/polyester (70/30) % w/w fabrics of twill structure (228–294 g/m2) with thickness (0.065 mm) were used in this study. In addition, 100% polyester of plain structure of light weight fabric (101–121 g/m2) with thickness (0.027 mm) was also investigated. All the above fabrics were supplied by Misr Co. for Spinning and Weaving, El-Mehalla El-Kubra, Egypt. The yarn count in both warp and weft is shown in Table 1.
Yarn count in wool, polyester, and wool/polyester blended fabrics.

Sodium hydroxide and sodium carbonate were purchased from El-Nasr Pharmaceutical Chemicals Company, Egypt. Silver nitrate was supplied by Sisc Research Laboratories Pvt. Ltd., Mumbai, India. Hydrated copper II sulfate was purchased from Loba Chemie Pvt. Ltd., Mumbai, India. Zinc oxide nanopowder (>100 nm) particle size and ascorbic acid (AA) were supplied by Sigma Aldrich. Egyptol® PLM, a non-ionic detergent based on nonylphenol ethoxylate, was kindly supplied by Starch and Detergent Company, Alexandria, Egypt.
Method
Scouring
Wool, polyester, and wool/polyester fabrics were scoured using 2 g/L sodium carbonate and 1 g/L non-ionic detergent at 60°C for 15 min with liquor ratio 1:50, followed by thorough rinsing with cold water, then squeezed and air-dried at ambient temperature.
Fabric pretreatment
Oxidation of wool
Oxidation of wool was made for the rupture of the disulfide bonds of cystine present in wool into cysteic acid that acts as a redox-active site and assisted the reduction of metal ions into metal nanoparticles.
Scoured wool sample was treated in a bath containing diluted solution of H2O2 (3% w/v) at pH 9 (adjusted by sodium silicate solution) at 65°C for 60 min, the material to liquor ratio was 1:25. The sample was then washed thoroughly with distilled water and air-dried.
Saponification of PET
Saponification of PET was carried out to create reactive hydroxyl and carboxylic groups (new functional groups).
Scoured polyester fabric was pretreated with 1 M NaOH solution at 80°C for 60 min, the material-to-liquor ratio was 1:100. The recorded loss in weight of polyester fabric after treatment was about 5%–6%.
Fabric treatment
Untreated wool (UW), untreated polyester (UPET), untreated wool/polyester blend (W/PET), oxidized wool (OW), and saponified polyester (SPET) fabrics were treated with two metal salt solutions, namely, 0.005 M CuSO4·5H2O and 0.005 M AgNO3. In another experiment, the aforementioned fabrics were treated with 0.01 M nano-ZnO at 70°C for 60 min with liquor ratio 1:30.
Reduction step
The reduction of metal ions treated fabrics was carried out by two ways:
1. The metal ions were reduced within the wool fabric vicinity by virtue of its reducing nature producing metal nanoparticles.
2. Fabrics treated with CuSO4·5H2O were reduced using aqueous solutions of 0.11 M AA at 80°C for 60 min with liquor ratio 1:30 to form the copper nanoparticles (Cu NPs). AgNO3-treated fabrics were also reduced using sodium borohydride (SBH) 0.004 M at room temperature for 15 min with liquor ratio 1:30 to form silver nanoparticles (Ag NPs).
Testing and measurements
Scanning electron microscopy and energy dispersive X-ray spectroscopy
Bruker Nano GmbH Scanning Electron Microscope D-12489 Berlin, Germany, was applied to investigate the morphological structure of the treated as well as untreated samples. The samples were mounted on aluminium stubs, and sputter coated with chromium in an S150A with 20 KV scanning voltage. The energy dispersive X-ray spectroscopy (EDX) measurements were reported at 20 KV accelerating voltage and 15 mm working distance. The distribution of particles’ diameter was measured using Image J software program saved on scanning electron microscope (SEM).
Fourier transform infrared spectroscopy
Fourier transform infrared spectroscopy (FTIR) spectra of untreated as well as of treated samples were recorded by using an FTIR spectrophotometer (Nexus 670 Fourier transform infrared spectrometer; Nicolet, USA) in the region of 4000–400 cm−1 with spectra resolution of 4.0 cm−1.
Atomic absorption spectrometry
The metal content per gram of fabric was determined using flame atomic absorption spectrophotometer. Metal ions on the treated fabric was extracted by immersing 0.2 g of the fabric in 20 mL of 15 wt% aqueous nitric acid at 80°C for 2 h. The concentration of silver, copper, and zinc in the extract was measured by Contraa 700 flame atomic absorption spectrophotometer (AAS, Analytik, Jena, Germany) equipped with a silver lamp at 328.1 nm wavelength, copper lamp at 222.6 nm, and a zinc lamp at 213.9 nm
The Ag, Cu, and Zn content in the fabrics was calculated according to equation (1) [23]
X-ray diffraction pattern
The X-ray diffraction analysis was performed at room temperature for copper- and silver-treated fabrics on a Bruker D8 Avance using CuKα as the target with secondary mono-chromator to operate at 40 KV and 40 mA. The scans were performed within the range of 4° < 2θ < 60° with scanning step 0.02° in reflection geometry.
Comfort characteristics
AC electrical conductivity
The electrical conductivity of untreated and treated fabrics before and after sewing was measured using LRC-bridge (Hioki model 3531zHi Tester, Japan).
The relative dielectric permittivity was calculated using the following equation

The AC resistivity (ς′) of the prepared samples has been estimated from the dielectric parameters. As long as the pure charge transport mechanism is the major contributor to the loss mechanism, ς′ can be calculated using the following relation

Air permeability
Air permeability of the examined fabrics was measured on FX 3300 air permeability tester (TEXTEST AG, Switzerland) at a pressure of 100 Pa according to ASTM D737 standard method.
Water permeability
The water permeability of the treated as well as the untreated fabrics was assessed according to ASTM–D 1913 (American Test Method for Water Repellency; Water Spray Test new edition 2010).
Physico-mechanical properties
Tensile strength
The tensile properties of untreated and treated fabrics were evaluated using an Instron Tensile Tester (USA) according to ASTM D 76 Standard Specification for Textile Testing Machines. The average dimensions for each sample is (5 × 20 cm).
Roughness
Surface roughness of treated and untreated fabrics was measured according to ASTM D7127-17 standard test method using surface roughness tester Model SE 1700∞ (Kosaka Laboratory Ltd., Japan).
Bending stiffness
The bending stiffness of untreated as well as treated PET fabrics was determined according to the standard test method ASTM D1388-2018. The bending stiffness was taken as a measure of the fabric drapability.
Results and discussion
Intelligent and smart textiles are now being produced with various electric conductive properties. Electrically conductive fabrics have attracted interest due to their assembly facility with wearable devices. Herein, we propose a simple method for fabricating conductive fabrics with comfort properties.
Comfort characteristics
Electrical conductivity
Electrical conductivity is the movement of electrically charged particles through a transmission medium. Figure 1 shows the conductivity of different fabrics treated with nanometals as well as untreated ones. It is clearly observed that the conductivity of the untreated wool and wool/polyester fabrics is higher than that of untreated polyester fabric. This is due to the higher moisture absorption of wool in which more dissociative water molecules can be ionized [24]; moreover, this hygroscopicity nature of wool facilitate the motion of ions or impurities resulting in the higher electrical conductivity than that of polyester. Generally, all metal-treated fabrics exhibit remarkable higher electrical conductivity than the untreated ones. This could be explained by the fact that the conductive metals are adequately adsorbed on the fabric surface until it reaches the percolation threshold, which is known as the critical point at which electrical conductivity increased sharply [25]. The conductivity percentage of the metal-treated fabrics increased to different extents relative to the respective untreated fabrics, i.e. 52.2% for Cu NPs–treated wool, 46.6% for ZnO NPs–treated oxidized wool, 35.7% for Zn NPs polyester, and 49% for Ag NPs wool polyester. This difference in conductivity for the metal-treated fabrics may be related to the homogenous distribution of metals nanoparticles in fabrics. It was found that, the pores between adjacent metal nanoparticles must be minimized, and accordingly the conductivity of current through these particles is continuous [22]. It can be noticed from Figure 1 that the electrical conductivity of oxidized wool is higher than that of the native wool. Oxidation of wool with H2O2 decreases the electrostatic charges on the material, presumably due to formation cysteic acid residues along keratin macromolecules which results in an increase in the hydrophilicity of the cuticle which permits conduction of the charges to the cortex [26].

Electrical conductivity of different fabrics treated with different nanometals.
In comparison between the conductivity of metal-treated wool, oxidized wool and wool polyester using external reducing agent (SBH and AA) and/or self-reduction, the results of this investigation are shown in Figures 2 and 3. These figures emphasize also that reduction of metal ions either with wool macromolecule side chain (self-reduction) or by reducing agents resulted in similar improvement in its electrical conductivity. So from economic and ecological point of view, it can be concluded that reduction of metal ions by wool is more preferable than using external hazard reducing agent.

Electrical conductivity of fabrics treated with Ag NPs.

Electrical conductivity of fabrics treated with Cu NPs.
Electric conductivity after sewing
The effect of sewing of the metal-treated fabrics on their electrical conductivity was studied and the results were presented in Table 2.
The effect of in sewing (sewing stitch type SSa–1) on the electrical conductivity metalized fabrics (self reduction in case of wool or using reducing agent in case of polyester).
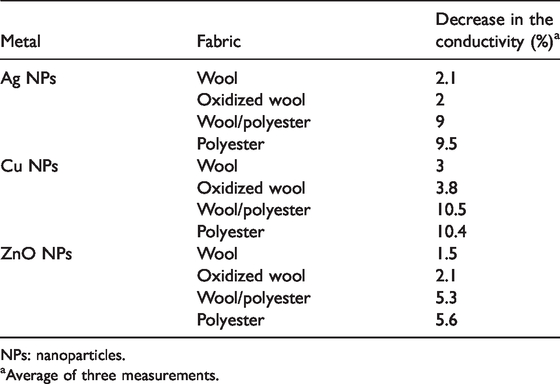
NPs: nanoparticles.
aAverage of three measurements.
The data of Table 2 shows that there is a limited decrease (1.5%–10.5%) in the electrical conductivity of the said fabrics after being sewn. The limited decrease in the electrical conductivity of the metalized fabrics may be attributed to the increase if the air gaps due to sewing process as well as the bad electrical conductivity of the polyester threads used in the sewing process.
Physico-mechanical properties
Table 3 summarizes the physical and mechanical properties that would have an impact on the comfort characteristics of metal treated as well as untreated fabrics.
Physico-mechanical properties of the nanometal-treated fabrics (average of three measurements each).
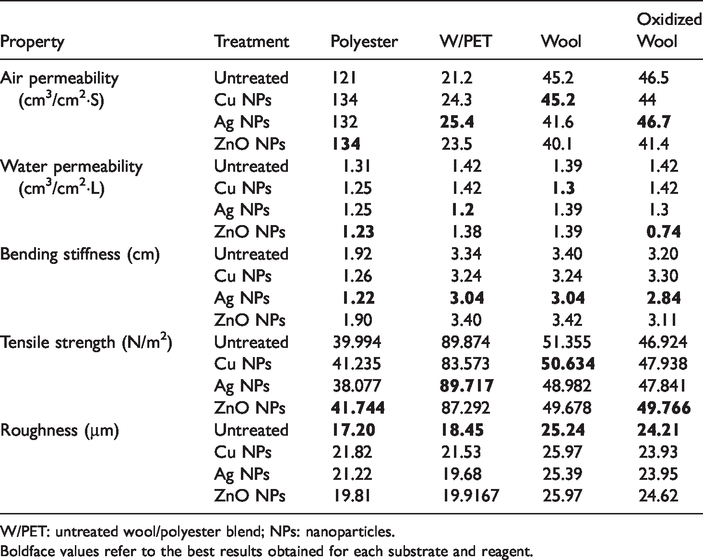
W/PET: untreated wool/polyester blend; NPs: nanoparticles.Boldface values refer to the best results obtained for each substrate and reagent.
Permeability is a part of apparel comfort and defined as the rate of air/water flow passing perpendicular through a known area. It is a measure of how well a fabric allows the passage of air/water through it [27]. Data of Table 3 imply that most of the treated fabrics have higher, or nearly equal, air permeability than the corresponding untreated fabrics. For each substrate, the rate of air that flows through the fabric depends on the nature of nanometal loaded onto the fabric, which affects the fabric porosity. The boldfaced results in Table 3 refer to the samples that exhibited the best results regarding air permeability for each fabric material. The situation is different in case of water permeability. The decrease in water permeability in case of PET, W/PET, W, and OW relative to the corresponding untreated fabrics reaches 6.1%, 15.5%, 6.5%, and 47.9%, respectively. This might be due to engagement of most of the hydrophilic groups along keratin macromolecules of wool fabrics as well as the limited number of such groups along PET chains with metal ions via covalent of coordinate bonds. This will decrease the water combining capacity of the fabrics and hence enhances their water permeability.
Results in this table indicate also that the drapability of the fabrics loaded with silver nanoparticles is higher than the analogous untreated copper ions or zinc oxide–treated ones.
The surface roughness of polyester and wool/polyester fabrics increases by metallization. However, the increase in surface roughness of wool and oxidized wool was not so high after being treated with copper and silver, respectively. Additionally, there is no observable change in the tensile strength of all fabrics after different metal treatments.
Atomic absorption spectrometry
The amount of metal particles taken by the different treated fabrics was measured using atomic absorption spectrometry and the results are tabulated in Table 4.
Metal contents of the different treated fabrics.

SBH: sodium borohydride; NPs: nanoparticles.
The data of Table 4 indicated the following:
The adsorption power of textile materials for different metal ions used in this investigation is in the order: oxidized wool > wool > wool/polyester blend. This adsorption power of textiles substrate for metal ions is a function of the number of electron pair donor groups along the respective macromolecules which is responsible for capture of the metal ions.
In the presence or absence of reducing agent, the amount of adsorbed silver ions on wool, oxidized wool, as well as wool/polyester blended fabrics is higher than copper ions. This is may be due to the higher ability of Ag+ ions to make electrostatic interaction force with the negative charge of carboxylate groups or lone pair of nitrogen and sulfur of the amino acids (–COOH, –NH2, and –SH) along the keratin macromolecules of wool than Cu+2 ions [23].
Oxidation of wool fabric resulted in the formation of oxidized cystine moieties (cysteic acid, for instance) which would enhance the efficiency of wool to bind with metal ions.
The adsorption of ZnO NPs by polyester fabrics is much higher than CuSO4 or AgNO3. This may be attributed to the fact that ZnO is used in its nano form which facilitates its adsorption by the fabric surface more than Cu+2 or Ag+.
It is worth to mention that, the high metal uptake by any textile substrate does not assure the best electrical conductivity. Other factors affect the conductivity of textile substrate, such as the morphology of the metal particles and their homogenous distribution onto fabrics, fabric structures, and fabric gaps [28].
Fourier transform infrared spectroscopy
FTIR analysis was used to monitor any alteration in the chemical composition of the used fabrics after being modified using metal ions.
Figure 4(a) to (c) shows the FTIR spectra of the untreated wool, oxidized wool, wool/polyester blend fabrics, as well as the corresponding fabrics treated with Ag NPs, Cu NPs, and ZnO NPs. The characteristic peaks of wool appear at 3273 cm−1 (N–H), 2930 cm−1 (C–H), the protein amide I at 1630 cm−1 corresponds to with (C = O) stretching vibration, the protein amide II which absorbed at 1519 cm−1 was related to (N–H) bending and (C–N) stretching vibrations [29,30], and protein amide III at 1237 cm−1. Compared to untreated fabrics, the peaks of treated fabrics found to be red shifted with stronger intensity.

FTIR spectra of different fabrics treated with different metals (a) UW and W fabric treated with Ag+ and Cu+2 ions and ZnO nanoparticle, (b) OW fabric treated with Ag, Cu and Zn nanoparticle, (c) W/PET fabric and W/PET treated with Ag+ and Cu+2 ions and ZnO nanoparticle, (d) UPET fabric and PET fabrics treated with Ag+ and Cu+2 ions and ZnO nanoparticle, and (e) saponified polyester fabric treated with Ag+ and Cu+2 ions and ZnO nanoparticle.UW: untreated wool; UPET: untreated polyester; SPET: saponified polyester.
Figure 4(d) and (e) shows FTIR spectra of the untreated polyester, saponified polyester fabrics, and Ag NPs–, Cu NPs–, and ZnO NPs–treated fabrics. The characteristic peaks of untreated polyester appear at 2963 cm−1 (–CH3), 1710 cm−1(C = O), 1243 cm−1 (–CH2), and 1093 cm−1 for C–O bond. The peaks show enhancement in their intensity after treatment with the nanometal.
The results obtained from the FTIR confirm the in situ synthesis of Ag NPs and Cu NPs on the fabrics successfully. Additionally, the spectra of the metalized fabrics have no new peaks different from those of their respective untreated samples, which mean that no alteration in the chemical composition of the fabrics take place after being treated with metal NPs. This would assure the hypothesis which said that loading of metal NPs on the fabric surface takes place via physical deposition process which leads to shifting in the bands and its intensity only [31].
X-ray diffraction pattern
The X-ray diffraction pattern was used to ensure the reduction of Ag+ and Cu+2 ions by reducing agents or by wool (Figures 5 to 7).
Figures 5 and 6 show prominent 2θ peaks in the diffractogram at 20.8° and 9.7° which corresponds to wool [32,33]. Figure 7 shows characteristic 2θ peaks in the diffractogram at 17.5° and 25.6° which corresponds to polyester fabrics [34].

XRD pattern spectra of oxidized wool treated with Cu NPs.

XRD pattern spectra of oxidized wool treated with Ag NPs.

XRD pattern spectra of polyester treated with Ag NPs and Cu NPs.
In Figures 5(a) and (b) and 7(b), we observe 2θ low intense peaks at 30°, 41°, 50°, and 61° which might correspond to Cu and Cu2O nanoparticles. These peaks emphasize the formation of a mixture of Cu and Cu2O nanoparticles on the fabric [35]. Figures 6(a) and (b) and 7(a) have 2θ low intense peaks at 39°,43°, 45°, and 46°, which correspond to Ag nanoparticles. It has been reported that the XRD pattern of Ag NPs are found to be at 2θ peaks at 38°, 45°, and 65° [36].
Scanning electron microscopy and energy dispersive X-ray spectroscopy
SEM and EDX of untreated and metal-treated fabrics were used to assign the change in the morphological structure of the fabrics (Figures 8 to 10).
In general, SEM images for the untreated fabrics (wool, polyester, and wool/polyester) show clear and smooth surface (Figures 8(a), 9(a), and 10(a), respectively).

SEM and EDX analysis for: (a) untreated wool fabric, (b) wool treated by CuSO4.5H2O, and (c) oxidized wool treated by ZnO NPs.

SEM and EDX analysis for: (a) untreated polyester fabric and (b) polyester treated by ZnO NPs.

SEM and EDX analysis for: (a) untreated wool/polyester fabric and (b) wool/polyester treated by AgNO3.
On the other hand, the micrographs for the different treated fabrics show uneven distribution of metal that covered the fabrics surface.
The incorporation of the metal particles onto the treated fabrics was also confirmed by the EDX. There are elemental peaks at 1.00, 8.00, and 9.00 keV for copper-treated wool fabric [37] (see Figure 8(b)). Moreover, there are different peaks of zinc and oxygen signals of zinc oxide nanoparticle appear at around 1, 8.601, and 9.5 keV for Zn beside peak at around 0.5 keV for O [38] (see Figures 8(c) and 9(b)). The signals at 3 keV for elemental silver in the EDX spectra of wool/polyester-treated fabric proved the immobilization of Ag NPs into fabric [39], see Figure 10(b).
EDX was used to map Cu, Ag, and Zn NPs on wool, polyester, and wool/polyester fabrics. As shown in Figure 11, Cu, Ag, and Zn NPs are homogenously distributed all over the tested samples.

EDX mapping of (a) Cu NPs on wool fabric, (b) Ag NPs on wool/polyester fabric, and (c) ZnO NPs on polyester fabric.
Conclusion
Conductive wool, polyester, and wool/polyester blended fabrics were prepared by simple and easily applicable method using Cu+2 and Ag+ ions as well as ZnO nanoparticles. Unlike saponification of polyester, pre-oxidation of wool was found to be effective in enhancing the subsequent metallization step. Metallized oxidized wool was found to be superior regarding its electrical conductivity, compared with untreated wool. Sewing of the treated fabrics has limited effect on its electrical conductivity. The incorporation of metal nanoparticles on the treated fabrics was proved by SEM and EDX. Moreover, XRD and FTIR charts emphasized the formation of nanometals in case of Cu+2 and Ag+ treatment. Comfort results and physic-mechanical properties of the metal-treated fabrics show that the proposed method of treatment has no negative effect on the comfort attributes of the treated fabrics.
The treated fabrics would be a probable candidate for the production of conductive goods used for medical applications, such as suits for physical therapy.
Footnotes
Acknowledgement
The authors are indebted to Eng. Basma A. Ashmawi for her technical assistance during preparation of this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financed by the National Research Centre, Cairo, Egypt (grant number AR 110302).
