Abstract
Batch-adsorption technique was employed to evaluate the adsorption capacity of the nonwovens produced from lignocellulosic milkweed fibers for the removal of heavy metal ions, i.e. Pb2+ and Ni2+, from aqueous solutions. The impact of controllable factors such as solution pH, initial concentration, contact time and operating temperature on the adsorption quantity of Pb2+ and Ni2+ was investigated using Atomic Absorption Spectrophotometer. The kinetic and isotherm of the adsorption process were also modeled. The milkweed fibers demonstrated superior performance for the removal of Pb2+ ions from wastewater over Ni2+ ions. The results demonstrated that the equilibrium concentration of heavy metal ions in solution varies with solution pH and the least equilibrium concentration is observed at pH = 6, regardless of ion type. The results also revealed that the removal percentage of Ni2+ ions reached the value of 65.58% at contact time of 60 min while Pb2+ ions removal percentage was as high as 96.16% at contact time of 90 min. It was concluded that an increase in operating temperature from 26 to 45°C causes to slight decrease of the removal percentage for both Pb2+ and Ni2+ ions by ∼5.7% and ∼9.5%, respectively. The adsorption behavior of milkweed fibers toward Pb2+ and Ni2+ ions followed Freundlich and Langmuir isotherm models, respectively. Moreover, pseudo-second order model was the best to describe the adsorption kinetics of both metal ions.
Introduction
The introduction of heavy metal ions into the environment from agricultural, industrial and natural disposals has become a major health and environmental concern. Removal of heavy metals from industrial wastewaters can be conducted through various treatments such as coagulation, complexation, adsorption by activated carbon, ion exchange, solvent extraction, foam flotation, electro-deposition, cementation, and membrane operations [1–6]. These techniques are not capable to remove the metal completely, require high energy and also generate toxic sludge [7].
In recent decades, various researches have proven the capability of wood-based materials as high-performance filters to remove diverse heavy metals from wastewater. Laszlo and Dintzis [8] argued about ion-exchange capacity of lignocellulosic materials due to their constituent components such as cellulose, hemicellulose, lignin and protein. Du et al. [9] demonstrated that using the lignocellulose biomass to remediate the water contaminated with heavy metal ions is attracting more attentions due to several advantages such as low cost, abundant production, and environmental sustainability.
Many researchers [10–13] have reported that lignocellulosic materials are composed of tannin and lignin, which contain active sites to adsorb the heavy metal ions. Basso et al. [14] demonstrated a correlation between lignin content of various lignocellulosic materials and their removal ability of heavy metals from wastewater. The findings reported by Lee and Rowell [15] indicated the incompatible results. Their investigations on the ability of lignocellulosic fibers to remove copper, nickel, and zinc ions from aqueous solutions showed no correlation between the removal ability of heavy metals and lignin content of fibers. Abbar et al. [1] examined the influence of different processing variables on optimal removal efficiency of heavy metals from aqueous solution using flax fibers. They demonstrated that flax fibers are effective adsorbent to remove Pb2+, Cu2+, and Zn2+ ions from aqueous solutions.
Al-Sayah et al. [16] showed that chemical modification of lignocellulosic fibers could enhance their removal efficiency of heavy metal ions. Du et al. [9] introduced carboxyl modified jute fibers treated under microwave heating and evaluated their ability toward adsorption of Pb2+, Cd2+, and Cu2+ ions from aqueous solutions. They showed that the adsorption capacity of the modified jute fibers is remarkably higher than that of raw jute fibers. Their findings also revealed that the adsorption behavior of the proposed adsorbent follows Langmuir isotherm model.
Milkweed fibers are obtained from a perennial plant, which is grown commonly in southern regions of Asia, the United States and South Africa [17]. Milkweed fibers, which could be extracted from the plant’s seed or stem, possess numerous unique properties such as lightweight and good insulation mainly due to their hollow structure [18]. However, in spite of their unique characteristics, no industrial use of these fibers has been reported and various ideas in this field have remained under research.
Numerous technical applications are considered for eco-friendly textiles produced from milkweed fibers due to their ecological, physical, and chemical characteristics [17–22]. Milkweed fibers, which are categorized as lignocellulosic materials, are composed of different components such as holocellulose, lignin, ash, and wax [17]. Table 1 shows the main chemical compositions of seed-extracted milkweed fibers compared to cotton fibers. As can be observed, milkweed fibers possess significantly higher lignin content than cotton fibers meaning the possibility of heavy metals adsorption by these fibers from aqueous solutions. Although these fibers as an eco-friendly material are widely available around the world; however, to our knowledge, their ability to remove the heavy metals has not been quantitatively examined anywhere.
Chemical composition of milkweed fibers compared to cotton fibers [11].

This study aims to investigate the adsorption capacity of the milkweed fibers for the removal of heavy metal ions from aqueous solutions using batch-adsorption techniques. The influence of processing variables such as solution pH, contact time, initial concentration and operating temperature on the adsorption performance is discussed. Modeling the adsorption kinetics and isotherms is another goal of this study.
Experimental and methods
Materials
Milkweed fibers (lmean: 27.9 mm; Dmean: 24.6 µm) were collected manually from the plant’s seeds and then cleaned from any vegetable wastes. In order to remove the wax materials from fiber surface, fibers were washed using an aqueous solution containing 2 g/l detergent at 40°C. The fibers were then rinsed and dried. The dried individual milkweed fibers were converted into a uniform nonwoven layer using a semi-industrial carding line (Figure 1) followed by mechanical needling process.

Semi-industrial carding line.
PbSO4, NiCl2.6H2O, HCl, and NaOH were purchased from Merck Co. (Germany). Aqueous solutions of Pb2+ and Ni2+ with required concentrations were prepared by dissolving weighed amounts of PbSO4 and NiCl2.6H2O in deionized water.
Determination of heavy metal ions concentration was done by Atomic Absorption Spectrophotometer (PerkinElmer Analyst 700, USA). It is a quantitative procedure for determination of heavy metal ions using the absorption of optical radiation (light) by free atoms in the gaseous state.
The impact of controllable factors such as solution pH, contact time, initial concentration and process temperature on the adsorption performance of Pb2+ and Ni2+ ions are investigated. To this end, a specified amount of fibrous adsorbents (0.1 g) was introduced into beakers containing 50 ml of aqueous solution at an initial concentration of 20 mg/l. The initial and final concentrations of solutions were determined using Atomic Absorption Spectrophotometer, and the removal percentage of considered heavy metal ions was then calculated.
FTIR spectroscopy and scanning electron microscopy
A standard pellet with a diameter of 13 mm was prepared by pressing a ground mixture of the milkweed fibers after wax removal and (ca. 1 mg) KBr (100 mg). FTIR spectrum of the prepared pellet was collected on a FTIR MB-series 100 (BOMEM, Canada) at resolution of 4 cm−1, with 20 scans. FE-SEM was utilized to observe the morphology of the milkweed fibers after wax removal. The fibers were sputter-coated with gold for 2 min at 20 mA, and then imaged using Quanta 450 scanning electron microscope (FEG, USA).
Investigating the effect of controllable factors on adsorption of heavy metal ions
Table 2 demonstrates the experimental design for determination of the effect of controllable factors on the adsorption of heavy metal ions.
Experimental design for determining the effect of controllable factors on adsorption of Pb2+ and Ni2+ ions.

The initial pH of the aqueous solution containing metal ions is a determinant factor for the heavy metal adsorption because of its remarkable effect on the chemical speciation of the metal ions as well as the ionization of functional groups located on the adsorbent surface [23]. In order to determine the optimal solution pH for the adsorption of Pb2+ and Ni2+ ions, a specified amount of fibrous adsorbents (0.1 g) was introduced into beakers containing 50 mL of aqueous solution at an initial concentration of 20 mg/l. The initial and final concentrations of solutions were determined using Atomic Absorption Spectrophotometer, and the removal percentage of considered heavy metal ions was then calculated. The solution pH was adjusted using aqueous solutions of HCl (0.1 M) and NaOH (0.1 M).
Effect of initial heavy metal ion concentration on the removal percentage of metal ions was examined by changing the concentration of metal ions (Pb2+ or Ni2+ ions) in the range of 2.5 to 20 mg/l while the pH of aqueous solution was kept constant at the optimal value, i.e. 6, determined in the previous section. Metal ions aqueous solutions of 50 ml with given concentrations were introduced into beakers containing 0.1 g of the fibrous adsorbents. The solutions were gently shaken for a predetermined contact time of 150 min. The amount of heavy metals adsorbed by the fibrous absorbent was then determined.
The effect of contact time on the adsorption of Pb2+ and Ni2+ ion by the fibrous adsorbent was investigated. The specified amount of nonwoven (0.1 g) was introduced into beakers containing 50 ml of metal ion aqueous solution at an initial concentration of 20 mg/l. The prepared solutions were gently shaken for varying time periods ranging from 30 to 150 min at pH value of 6. The removal percentage of both heavy metal ions was then calculated by measuring the final concentration of each solution.
Effect of temperature on the removal percentage of metal ions was investigated by varying processing temperature in the range of 26–45°C. The same mass of adsorbents (0.1 g), as well as initial concentration of the solution (20 mg/l), were considered for this test. The solutions were gently shaken for a contact time of 150 min and an optimal pH of 6.0. Finally, using Atomic Absorption Spectrophotometer, the percentage removal of adsorbates was calculated. Each measurement was carried out three times and the mean value was reported.
Adsorption capacity
The amount of adsorbate (Pb2+ and Ni2+) removed at equilibrium condition by the fibrous adsorbent was calculated by equation (1)
The amount of solutes removed at a given time by the fibrous adsorbent was calculated using equation (2). The removal percentage of Pb2+ and Ni2+ ions from the solution, R (%), was calculated using equation (3)
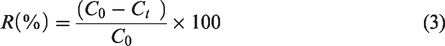
Results and discussion
Morphology and chemical composition of milkweed fibers
Scanning electron microscopy (SEM) images captured from the surface of milkweed fibers used in this work are depicted in Figure 2. According to the SEM images, milkweed fibers have a nonporous surface with hollow cross-section. The hollow structure provides a higher surface for adsorption purposes.

SEM images captured from the surface of milkweed fibers.
FTIR spectrum of milkweed fibers is shown in Figure 3. From Figure 3, it can be observed that the milkweed fibers exhibit absorption peaks around wavenumbers of 3437, 2920, 1456, 1378, 1161, and 1051 cm−1, which are due to chemical compositions of lignocellulosic fibers [24]. The absorption peaks at 1738 cm−1 and 1249 cm−1 are attributed to the stretching vibrations of C = O and C–O groups, which are prevalent in lignin and hemi-cellulosic structures [24].

FTIR spectrum of milkweed fibers.
Removal of Pb2+ and Ni2+ ions from solutions using milkweed fibers under different operating conditions
In this section, the removal percentage of Pb2+ and Ni2+ ions under different operating conditions including solution pH, initial concentration of Pb2+ and Ni2+ ions in solution, contact time and temperature is investigated.
Effect of solution pH
In order to investigate the impact of pH on the removal percentage of heavy metal ions, adsorption capacity tests for both Pb2+ and Ni2+ ions were conducted at different pHs ranging from 3 to 8. The equilibrium concentrations of heavy metal ions in solution at different pHs were measured while the initial concentration of heavy metal ions, adsorbent mass and solution volume are the same. Additionally, the removal percentage of Pb2+ and Ni2+ ions at various pHs is illustrated in Figure 4. From Figure 4, it can be concluded that the equilibrium concentration of heavy metal ions in solution varies with solution pH and the least equilibrium concentration is observed at pH = 6, regardless of ion type. Concurrently, the maximum removal percentage of ions occurs at pH = 6 as shown in Figure 4. Increasing the solution pH from 3 to 6 significantly enhances the adsorption capacity of milkweed fibrous adsorbent. Further increasing the solution pH to values of 7 and 8 leads to inferior removal of Pb2+ and Ni2+ ions. Therefore, pH = 6 was chosen as the optimum pH for both Pb2+ and Ni2+ ions during subsequent investigations.

Effect of solution pH on heavy metal ion (Pb+2 and Ni2+) removal percentage. The operating conditions are listed in Table 2.
The same optimum pH has been also reported in previous researches in which the adsorption capacity of lignocellulosic fibers has been evaluated [1,14,15,25]. According to Chang et al. [26], the concentration of hydrogen ions (H+) is high at low pH, i.e. acidic medium. This causes a competition between H+ ions and heavy metal ions to occupy the active sites on the surface of the adsorbent. An increase in the pH to values 4.0 to 6.0 reduces the concentration of H+ ions. This allows for better adsorption of heavy metal ions [27,28]. Thus, in order to evaluate the maximum adsorption capacity of the milkweed fibers, the subsequent adsorption tests were conducted at the optimum pH value, i.e. pH = 6. Additionally, the findings revealed the superior performance of milkweed fibers to adsorb Pb2+ ions compared to Ni2+ ions. The maximum adsorption of Pb2+ ions by milkweed fibers is about 95%, which is significantly higher than that of Ni 2+ ions, i.e. 65%.
Effect of initial concentration of heavy metal ions in solution
The removal percentage of Pb2+ and Ni2+ ions at different initial concentrations was measured while adsorbent mass, solution volume and pH were the same. In addition, the variation of removal percentage of Pb2+ and Ni2+ ions with initial concentration of these ions in solution is shown in Figure 5. It is clearly observed that increasing the initial concentration of Pb2+ and Ni2+ ions in solution from 3 to 51 mg/l decreases the removal percentage of Pb2+ and Ni2+ ions by milkweed fibers. Specifically, the removal percentage of Ni2+ decreases from 65% to ∼40% when Ni2+ initial concentration increases from 3.7 to 50.5 mg/l. For Pb2+, increasing the initial concentration from 3.7 to 48.6 mg/l leads to a decrease in removal percentage from 95% to ∼79%. This concurs well with the findings reported by Elaigwu et al. [28] who concluded that the initial concentration is a profound factor, which affects the adsorption of metal ions. At a given pH and low initial concentrations of heavy metal ions, the ratio of adsorption sites to the initial number of cations including H+ and heavy metal ions (Pb2+ or Ni2+) is high. This means that more active sites on the surface of milkweed fibers are available for heavy metal ions. At the same pH and high initial concentrations of heavy metal ions, the mentioned ratio becomes smaller. Under these conditions, the active sites are limited and heavy metal ions have lower chance for occupying the active sites.

Effect of initial concentration of heavy metal ions (Pb+2 and Ni2+) on their removal percentage. The operating conditions are listed in Table 2.
Effects of contact time
The equilibrium concentration of heavy metal ions in solution at different contact times was measured while initial concentration of heavy metal ions in solution, adsorbent mass and solution volume were the same. Additionally, increase of the removal percentage of Pb2+ and Ni2+ ions with contact time is depicted in Figure 6. Considering Figure 6, it can be observed that the adsorption process occurs in two stages. At the beginning of this process, removal of Pb+2 and Ni2+ ions increase rapidly and then it continues slowly until the equilibrium state is reached. The adsorption rate at the first stage for Ni2+ is significantly higher than that of Pb+2. It can be said that an increase in contact time from 20 to 120 min remarkably enhances the removal percentage of both metal ions. The findings also revealed that the highest removal percentages of Ni2+ and Pb2+ ions are 65.58% and 96.16%, respectively, which are reached at contact times of 60 and 90 min, respectively. After reaching the optimum time for both heavy metals ions, the removal percentage decreases slightly. This result agrees well with observations reported by Elaigwu et al. [28] and Ho et al. [29]. The adsorbent structure and its available active sites determine the time needed to reach the equilibrium state. This can be explained based on the existence of numerous unoccupied active sites on the adsorbent surface, which are available for heavy metal ions at the beginning of the adsorption process. Continuing the adsorption process, the available sites gradually decrease which in turn makes the process slower [30]. Note that during equilibrium process between adsorption and desorption, the rate of adsorption is faster than the rate of desorption. At short times, adsorption is more pronounced but as time progresses, desorption becomes noticeable and some metal goes back to the solution.
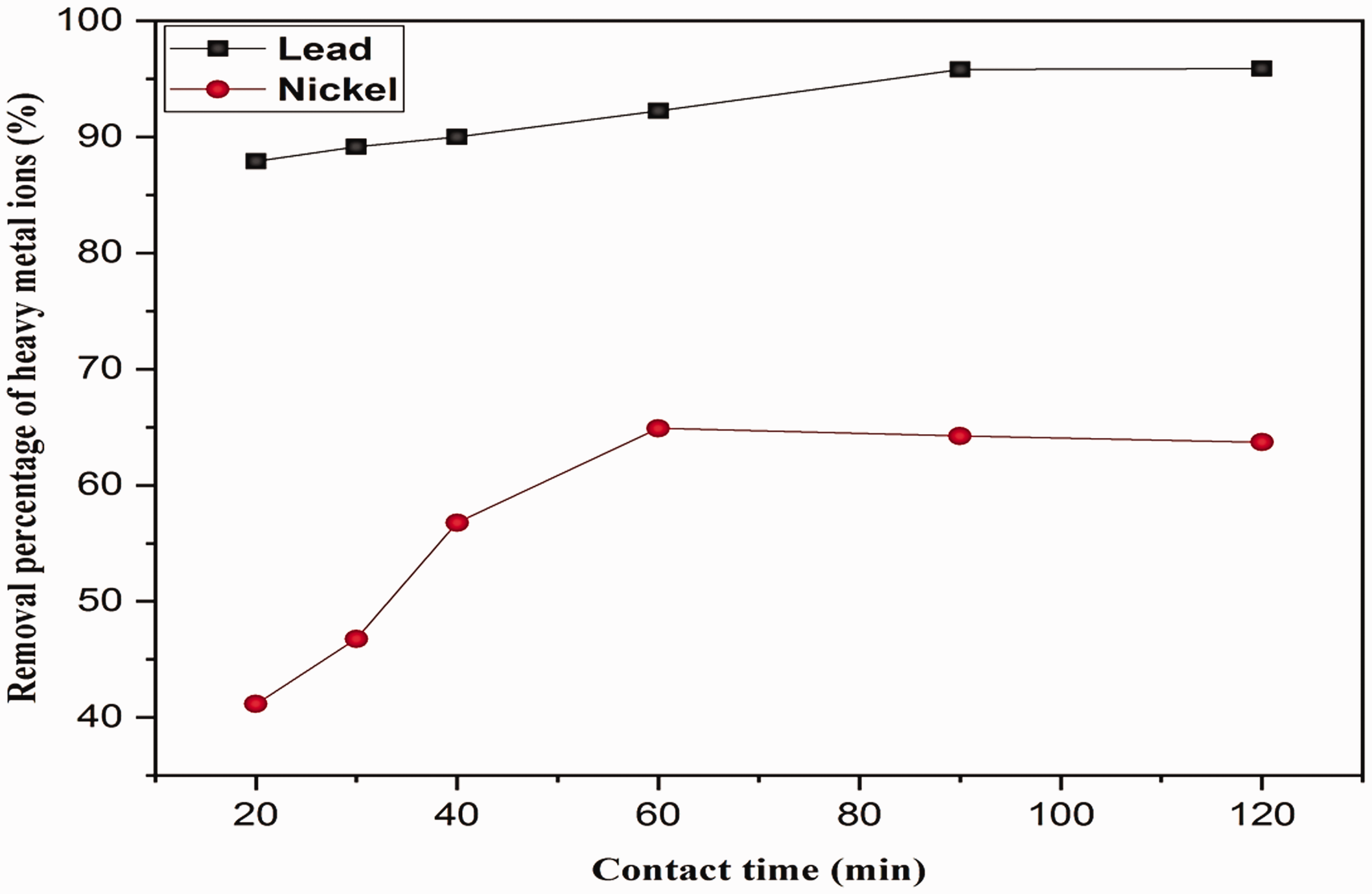
Effect of contact time on the removal percentage of heavy metal ions. The operating conditions are listed in Table 2.
Effects of temperature
The nature of adsorption process depends on operating temperature [31]. The variation of equilibrium concentration of heavy metal ions in solution with operating temperature is shown in Figure 7 according to which one can witness that operating temperature adversely affects adsorption of heavy metal ions by milkweed fibers. Additionally, Figure 7 reveals that an increase in the operating temperature from 26 to 45°C causes to a slight decrease of the removal percentage for both Pb2+ and Ni2+ ions by ∼5.7% and ∼9.5%, respectively. It is observed that the highest removal percentage occurs at the temperature of 26°C, irrespective of ion type. This may be due to this fact that with increasing temperature, the attractive forces between fiber surface and heavy metal ions are weakened and the ion adsorption decreases. This is in line with the results reported by Horsfall and Spiff [32] who studied the effect of temperature on removal efficiency of Pb2+ from wastewater using Caladium bicolor biomass.

Effect of operating temperature on the removal percentage of heavy metal ions. The operating conditions are listed in Table 2.
Adsorption kinetic studies
Adsorption kinetic studies of Pb2+ and Ni2+ ions by milkweed fibers were conducted using two kinetic models including simple Elovich kinetic and pseudo-second order models. The models were fitted to the experimental data using regression techniques, and accordingly, the model’s coefficients were determined. Kinetic studies are advantageous due to their pivotal role in providing data on the mechanisms of adsorption as well as rate controlling steps. Pseudo-second order model is described using equation (4) [33].

A plot of qt values versus ln t demonstrates a linear behavior for the applicability of the simple Elovich kinetic model.
Figure 8 shows the plots of simple Elovich and pseudo-second order kinetic models for the adsorption of Pb2+ and Ni2+ ions by the milkweed fibers. Table 3 represents the coefficients of kinetic models describing the adsorption of Pb2+ and Ni2+ ions by milkweed fibers.
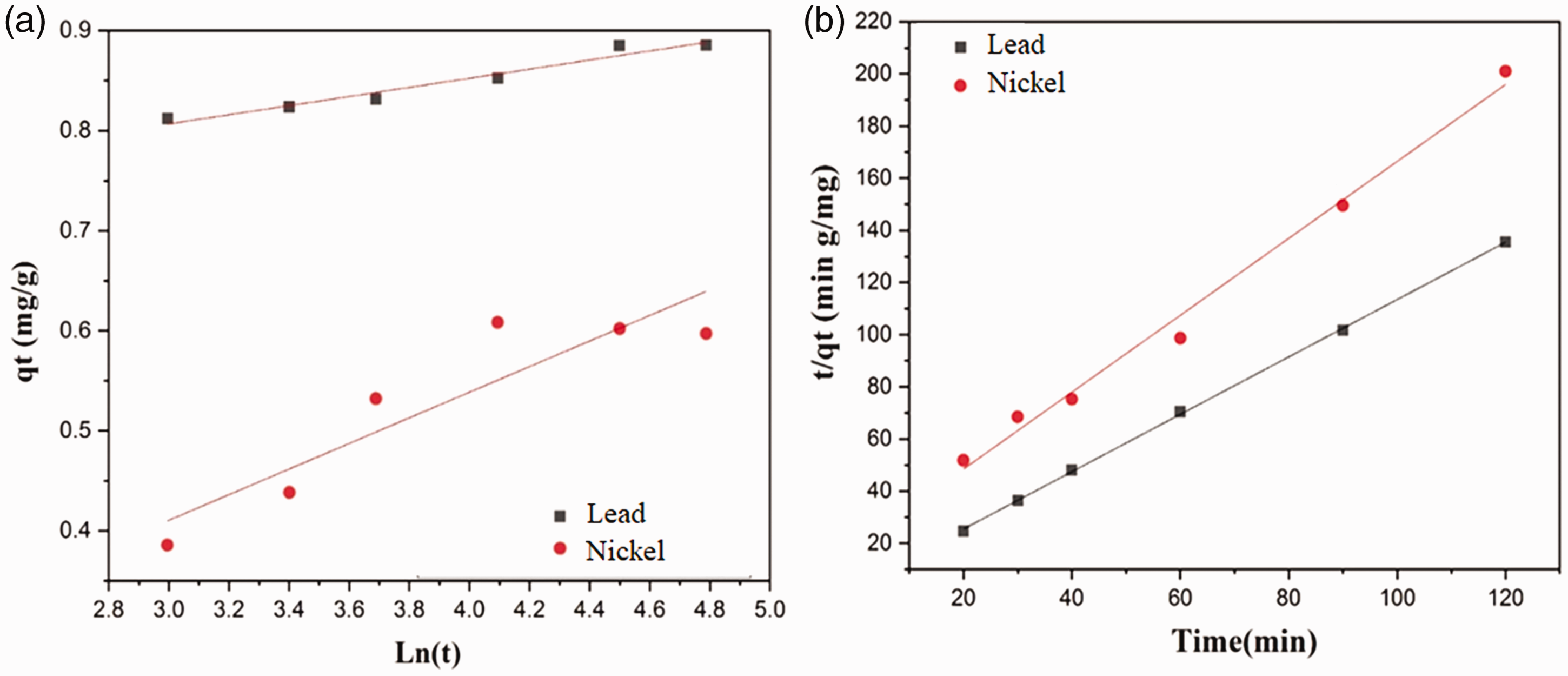
(a) Simple Elovich and (b) pseudo-second order plots for the adsorption of heavy metal ions by the milkweed fibers.
Coefficients of both simple Elovich and pseudo-second order kinetic models describing the adsorption of Pb2+ and Ni2+ ions by the milkweed fibers.
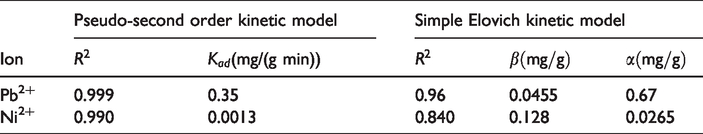
From Table 3, it can be concluded that for both Pb2+ and Ni2+ heavy metal ions, R2 values calculated for pseudo-second order kinetic model are more than 0.99 while this value for simple Elovich kinetic model is not satisfactory. This means that the adsorption kinetic of Pb2+ and Ni2+ ions by the milkweed fibers can be described very well by the pseudo-second order model. On the other hand, simple Elovich model provides no reliable data for describing the kinetic of adsorption of Pb2+ and Ni2+ ions by the milkweed fibers.
Modeling the adsorption isotherm
Adsorption isotherms can mathematically express the isotherm analysis of the equilibrium data for the adsorption of heavy metal ions by fibrous absorbent. In this research, Langmuir and Freundlich isotherm models were applied to describe the adsorption equilibrium data. For this purpose, batch adsorption experiments were conducted and the amount of heavy metal ions adsorbed by the milkweed fibrous absorbent at equilibrium (qe) and the equilibrium concentration of heavy metal ions in solution (Ce) were measured.
The Langmuir isotherm model indicates the monolayer adsorption on a uniform surface containing a finite number of adsorption sites [1]. This model can be expressed by equation (6)

The value of RL refers to the shape of Langmuir isotherm plot, which could be linear (RL = 1), unfavorable (RL > 1), favorable (0 < RL<1), and also irreversible adsorption (RL= 0) [34].
Freundlich isotherm model characterizes the relation between the non-ideal and reversible adsorptions. It is also applied to adsorption on heterogeneous surfaces along with the interaction between adsorbed molecules. The application of the Freundlich equation also implies that adsorption energy decreases exponentially corresponding to the completion of the adsorption centers of the adsorbent [35]. Freundlich isotherm is described by an empirical equation as equation (8)

The linearized Langmuir and Freundlich isotherms associated to Pb2+ and Ni2+ ions are plotted in Figures 9 and 10, respectively. Additionally, the calculated constant values are tabulated in Table 4. The results show that the Freundlich isotherm model can describe Pb2+ adsorption remarkably better than Langmuir isotherm model. The value of n for Pb2+ is greater than 1 indicating that this heavy metal ion is favorably adsorbed by milkweed fibers (see Table 4).
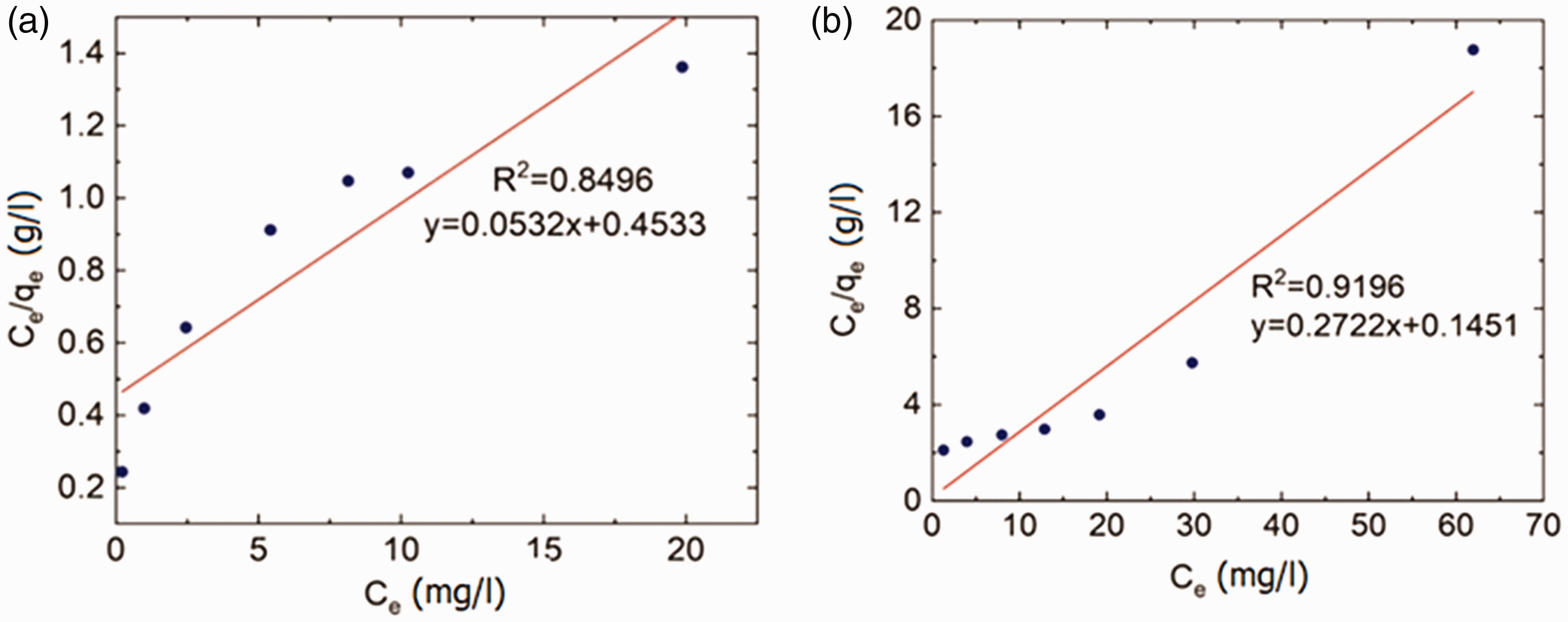
Langmuir plots related to isotherm adsorption of (a) Pb2+ and (b) Ni2+ by milkweed fibers at 26°C.

Freundlich plots related to isotherm adsorption of (a) Pb2+ and (b) Ni2+ by milkweed fibers at 26°C.
Results calculated for both Langmuir and Freundlich isotherm models.

The results of adsorption isotherms indicate that Langmuir was the best to model the Ni2+ ion adsorption process (R2 = 0.92), indicating that milkweed fibers have a monolayer adsorption capacity for Ni2+ ions. The RL values in the present study were found to be 0.83 and 0.80 for Pb2+ and Ni2+ ions, respectively, indicating that the adsorption of these heavy metal ions by the milkweed fiber is favorable.
Regarding the adsorption of Pb2+ ions by flax fibers investigated by Abbar et al. [1], the maximum adsorption capacity (qmax) value has been reported as 10.74 mg/g, whereas in this study the value of qmax associated to milkweed fibers for adsorption of Pb2+ ions was obtained as 18.79 mg/g. This indicates the outstanding adsorption capacity of milkweed fibers in comparison to flax fibers. Also, the findings reported by Shukla and Pai [37] shows that the qmax value of jute fibers to remove Ni2+ ions (3.37 mg/g) is nearly the same as the qmax value (3.67 mg/g) obtained for the milkweed fibers in this work.
In Freundlich equation, a variation of the slope (1/n) between 0 and 1 is associated with a chemisorption process [38]. This means that adsorption of heavy metal ions by milkweed fibers is the results of the chemical adsorption mechanism. This result is in agreement with findings reported by Chung et al. [39] who studied the adsorption capacity of Kapok fibers as a lignocellulosic fiber.
Conclusion
This study was undertaken to examine the adsorption performance of milkweed fibers to remove the heavy metal ions (Pb2+ and Ni2+) from wastewater. The experimental results revealed the following conclusions: The equilibrium concentration of heavy metal ions in solution varies with solution pH, and the least equilibrium concentration is observed at pH=6, regardless of ion type. Increasing the solution pH from 3 to 6 significantly enhances the adsorption capacity of milkweed fibrous adsorbent. Further increasing the solution pH to values of 7 and 8 leads to inferior removal of Pb2+ and Ni2+ ions. The milkweed fibers demonstrated superior performance for the removal of Pb2+ ions from wastewater over Ni2+ ions. The equilibrium adsorption was reached within 90 min for Pb2+ ions and 60 min for Ni2+ ions. The findings also revealed that the removal percentage of Ni2+ ions reaches the highest value of 65.58% at contact time of 60 min while Pb2+ ions removal was the highest, i.e. 96.16%, at the contact time of 90 min. The removal percentage of Ni2+ decreases from 65% to ∼40% when Ni2+ initial concentration increases from 3.7 to 50.5 mg/l. For Pb2+, increasing the initial concentration from 3.7 to 48.6 mg/l leads to a decrease in removal percentage from 95% to ∼79%. The adsorption capacity for Pb2+ and Ni2+ ions decreased with increasing the temperature. An increase in operating temperature from 26 to 45°C causes to a slight decrease of the removal percentage for both Pb2+ and Ni2+ ions by ∼5.7% and ∼9.5%, respectively. Moreover, pseudo-second order model was the better model to describe the adsorption kinetic of both heavy metal ions investigated in this study. The results of adsorption isotherms indicated that Langmuir was the best to modeling Ni2+ ions adsorption by the milkweed fibers. Also, the Freundlich model can describe the adsorption of Pb2+ ions by the milkweed fibers better than the Langmuir isotherm model.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
