Abstract
In this study, it was aimed to prepare silver nanoparticles by reduction of silver salt (AgNO3) in situ by means of only synthesized polyacrylonitrile (PAN) and poly(acrylonitrile-co-itaconic acid) (P(AN-co-IA)) polymers, and N,N dimethylformamide (DMF). Thereafter, PAN/Ag and P(AN-co-IA)/Ag nanofibers were prepared via electrospinning. Spectroscopic and morphologic characterizations, electrical and thermal features, and antimicrobial activities of the prepared nanofibers against Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, and Candida albicans were carried out in detail. It was observed that P(AN-co-IA) was much more effective than PAN on the reduction of AgNO3 and formation of silver nanoparticles. Silver nanoparticles and also itaconic acid contributed to decrease the cyclization temperature of PAN by generating sharp exothermic peaks. In addition, electrical conductivity of the nanofibers increased dramatically from E−13 to E−4 related to the presence of silver nanoparticles. Furthermore, incorporation of silver nanoparticles to the nanofiber membranes let bactericidal/fungicidal activities, which started at 6–24 h and continued for up to 168 h, against S. aureus, E. coli, P. aeruginosa, and C. albicans. The prepared silver containing nanofibers can be regarded as good candidates for potential use in the biomedical and pharmaceutical applications.
Introduction
Nanocomposite materials are of great interest due to their ability of gathering unique features of two or more different materials in a single structure [1–3]. The properties of materials are highly dependent on their nanostructure [4]. Polymers, nanometals, and metal nanoparticles are some of the materials used in the production of nanocomposite structures [1–9]. Polymers are regarded as excellent binding materials for the formation of stable colloidal dispersion of metals; therefore, an increasing interest has occurred on the deposition of metal nanoparticles onto polymer latexes [3].
Being regarded as an important engineering polymer with its novel properties such as high strength, high abrasion resistance, and low price, polyacrylonitrile (PAN) can be effectively used in nanocomposites and carbon fiber production. However, it is more beneficial to use PAN in copolymer form for fiber production in order to disorder its strong nitrile–nitrile interactions and obtain fiber easily [10–12].
Itaconic acid (IA) is among the comonomers used for copolymerization of PAN. It reorders the nitrile groups of PAN through its two carboxylic acid groups [13]. Studies of Poly (Acrylonitrile-co-Itaconic Acid) P (AN-co-IA) generally depend on the effect of IA on lowering the initiation temperature of exothermic reactions. In this respect, the effect of IA and methyl acrylic acid on thermo-oxidative properties of PAN was studied by Bajaj et al. [14]. Thermal properties of AN-IA polymer in oxidative and nonoxidative atmospheres were investigated by Yu et al. [15]. Thermal behavior and structural evolution of PAN were analyzed both in the presence and absence of IA by Ouyang et al. [16].
As a noble metal, silver (Ag) has been widely preferred in applications due to its excellent structural, electrical, thermal, and antibacterial properties. It has been effectively used as antibacterial agent because it has the ability to avoid the bacterial replication and bacterial electron transport by generating bonds to microbial DNA [2,3,17–22]. In the applications, it is more effective to use Ag in nanofiber form rather than using as powder.
Nanofibers, a special class of nanomaterials, is mostly prepared by electrospinning method and used in various applications such as filtration, protective clothing, nanosensors, tissue scaffolds, implant coating film, wound dressing, drug nanocarrier, cosmetic skin masks, and micro-/nanoelectronic devices [23,24]. These nanofibers can be prepared from a wide array of polymer types and components to enable desired features and have many advantages such as ultrafine structure, a large surface area to volume ratio, and high porosity with a small pore size. Furthermore, deposition of active agents such as metal nanoparticles onto nanofibers has improved their functions [25,26].
To obtain silver nanoparticles, silver nitrate (AgNO3) was used as a precursor and various reduction methods were tried such as mechanical mixing in the presence of different reductants and photopolymerization with a photoinitiator. Unfortunately, most of the time, production of silver nanoparticles and doping them in a polymer solution caused formation of agglomerates. To solve this problem, Wang et al. tried to prepare silver nanoparticles in situ and homogeneously disperse them in PAN nanofiber web. For this purpose, AgNO3 was added into the solution containing PAN and N,N dimethylformamide (DMF) and stirred for 24 h. Thereafter, PAN/AgNO3 nanofibrous structure was fabricated via electrospinning. For the reduction of Ag+ ions to Ag nanoparticles, the produced nanofiber web was immersed in hydrazinium hydroxide (N2H5OH) solution at room temperature. As a result, it was understood that there was a coordination between nitrogen and silver ions, which made PAN an ideal carrier for in situ preparation of silver nanoparticles [27]. Since reduction of Ag+ ions to silver nanoparticles using N2H5OH required a precipitation process, Sichani et al. used xenon arc lamp for the first time in order to reduce AgNO3 to silver nanoparticles faster. PAN nanofibers containing silver nanoparticles were fabricated via electrospinning and following that reduction of Ag+ ions was achieved by the application of xenon arc light for only 4 h. It was noted that PAN behaved like a stabilizer for silver nanoparticles [28]. Reduction of Ag+ ions to silver nanoparticles was achieved by means of DMF in the existence of PAN by Rujitanaroj et al. Further reduction was promoted by the application of UV irradiation for 1–10 min on PAN/AgNO3 nanofiber mats which were produced via electrospinning. Moreover, it was declared that agglomeration could be prevented due to the interactions between the cyano-nitrogen of PAN molecules and silver nanoparticles [29]. Karbownik et al. produced silver containing nanofibers along with PAN terpolymer which consisted of acrylonitrile (AN), methyl acrylate, and sodium allylsulfonate and utilized as both polymer and stabilizer. Reduction of Ag+ ions to silver nanoparticles was carried out in the presence of DMF and ascorbic acid. It was declared that particle agglomeration was prevented related to the interaction between nitrile groups and polymer micelles [30]. Mahapatra et al. reduced Ag+ ions to silver nanoparticles by three different methods, which were refluxing the solution before electrospinning, using sodium borohydride as reductant and subjecting the produced nanocomposite webs to heat. It was observed that fastest reduction was achieved using reflux, and thus higher percentage of Ag nanoparticles could stay on the nanofiber surface giving satisfactory antibacterial results [31].
Antimicrobial inhibition is very crucial especially in biomedical applications. Infections may cause biomaterial failure in wound dressings, drug release systems, and pharmaceutical applications [32]. Among the antimicrobial agents, metal nanoparticles are one of the most promising agents in terms of physical, chemical, and biological features. When compared to the other metals, silver has a broader spectrum of antimicrobial activity that kills more than 650 disease-causing bacteria, fungi, viruses, and molds [33,34]. Besides, its inhalation, ingestion, or dermal applications are not dangerous [35]. The mechanism of action of silver nanoparticles is cell membrane disruption upon contact with silver surface and formation of reactive oxygen species or inhibition of microbial growth by interacting with DNA after silver ion penetration into the nucleus [35,36]. However, these silver nanoparticles have some stability problems such as aggregation and dissolution causing a challenge in practical application [37]. Recently, there have been few studies examining antimicrobial activities of silver nanoparticles/polymer nanocomposite fibrous membranes. Kharaghani et al. fabricated silver nanoparticle-functionalized PAN nanofibers by a facile wetting process as an antibacterial membrane filter which is exhibiting appreciable antibacterial efficiency against Escherichia coli and Staphylococcus aureus bacteria [34]. In another study, Quirós et al. prepared electrospun cellulose acetate composite nanofibers containing silver nanomaterials supported in sepiolite and mesoporous silica and tested as fungistatic reservoir membranes against Aspergillus niger [38]. In the study of Shi et al., electrospun PAN nanofibers were loaded with silver nanoparticles and these showed excellent antibacterial properties [39]. Shalaby et al. incorporated silver nanoparticles on the surface of PAN nanofiber membranes by electrospinning for use as high disinfection materials [37]. Also, Rujitanaroj et al. prepared PAN nanofiber membranes containing Ag nanoparticles and examined their bactericidal activities against S. aureus and E. coli [29]. In addition, while the cytotoxic effect of silver nanoparticles and the polymer of PAN to human cells is very low [34], IA is nontoxic [40]. Despite these studies, there is still need to develop new nanocomposite carrier systems for silver nanoparticles in order to solve the stability problem and enhance the disinfection ability.
In this study, P(AN-co-IA)/Ag and PAN/Ag nanocomposite nanofibers were prepared via electrospinning and characterized by scanning electron microscopy (SEM), energy dispersive spectroscopy (EDS), Fourier transform infrared-attenuated total reflectance (FTIR-ATR), ultraviolet (UV)-visible spectroscopy, and atomic force microscopy (AFM). Conductivity of the samples was analyzed by four-point probe. Furthermore, in vitro antimicrobial activities of the prepared nanofiber membranes were evaluated against S. aureus as a gram-positive bacteria, E. coli and Pseudomonas aeruginosa as gram-negative bacteria, and Candida albicans as a yeast using disk diffusion method, susceptibility testing, and time-kill analysis.
Although there are studies on in situ reduction of Ag+ ions to silver nanoparticles in the presence of PAN and DMF, it is regarded as a time-consuming process, and possibility of agglomeration of silver nanoparticles is considerably high [28–31]. In this study, it is desired to obtain silver nanoparticles in situ using P(AN-co-IA), which does not require any post-reduction steps, and shorten the duration of in situ reduction process compared to ones achieved using PAN. P(AN-co-IA) copolymer served as stabilizer as in case of PAN but completed the reduction process two times faster than PAN, which is the novelty. Silver nanoparticles and also IA contributed to decrease the cyclization temperature of PAN by generating sharp exothermic peaks. It was observed that electrical conductivity of the nanofibers increased dramatically from E−13 to E−4 due to the existence of silver in both PAN/Ag and P(AN-co-IA) nanofibers compared to the nanofibers that do not contain silver. Moreover, PAN and P(AN-co-IA) nanofibers did not show any antimicrobial activity against all microorganisms; however, incorporation of silver nanoparticles to the nanofiber membranes let bactericidal/fungicidal activities against all studied microorganisms for both PAN/Ag and P(AN-co-IA)/Ag nanofibers. The bactericidal/fungicidal activities started at 6-24 h and continued for up to 168 h. The prepared silver containing nanofiber membranes are promising candidates for potential use in the biomedical, pharmaceutical, and air and water filtration applications. In addition, the cytotoxicity studies of these newly synthesized PAN and P(AN-co-IA) polymers and PAN, P(AN-co-IA), PAN/Ag, and P(AN-co-IA)/Ag nanofibers to human cells are in progress.
Experimental
Materials
Acrylonitrile (AN (>99.5%)) was provided by Aksa Acrylic Chemistry Company. Itaconic acid (IA (>99.5%)), sodium dodecyl benzene sulfonate (SDBS), and AgNO3 were purchased from Sigma-Aldrich. Ammonium per sulfate (APS), DMF, dimethyl sulfoxide (DMSO), and ethanol were all Merck reagents. S. aureus ATCC 29213, E. coli ATCC 25922, P. aeruginosa ATCC 27853, and C. albicans were provided by American Type Culture Collection (ATCC® CLL-163TM, Manassas, VA, USA). Mueller-Hinton Broth (MHB) and Tyriptic Soy agar (TSA) medium were obtained from Difco, RPMI-1640 medium; levofloxacin and fluconazole were purchased from Sigma-Aldrich. All these reagents were used without further purification.
Synthesis of P(AN-co-IA) copolymer and PAN homopolymer
P(AN-co-IA) copolymer was synthesized by emulsion polymerization utilizing water, SDBS and APS as dispersion medium, surfactant, and initiator, respectively. The polymerization was lasted for 3 h at 70℃ and then stopped using ethanol and then cooled to room temperature as in previous studies [41–43]. The obtained emulsion latex was precipitated and washed with ethanol and water for five times. After that, it was dried in vacuum oven at 60℃. Eventually, P(AN-co-IA) powders could be obtained.
PAN homopolymer was synthesized by emulsion polymerization with the same method used for P(AN-co-IA) copolymer. The only difference was the absence of IA monomer in the polymerization. After precipitation, washing, and drying, PAN powders could be collected.
Preparation of PAN/Ag solutions
PAN (10 wt%) was dissolved in DMF and stirred for 24 h at room temperature for complete dissolution. Then, 10 wt% and 20 wt% AgNO3 (the weight percentage of AgNO3 in the solution was calculated on the basis of PAN weight) were added to the prepared PAN solutions separately and stirred at room temperature for 10 days in order to reduce to Ag+ ions to silver nanoparticles [21,29,31]. During stirring, all samples are kept in the same conditions and protected from sunlight in order to avoid the decomposition of AgNO3.
Preparation of P(AN-co-IA)/Ag solutions
P(AN-co-IA) (5 wt%) was dissolved in DMF and stirred for 24 h at room temperature for complete dissolution. Then, as in the case of PAN/Ag solution, 10 wt% and 20 wt% AgNO3 (the weight percentage of AgNO3 in the solution was calculated on the basis of P(AN-co-IA) weight) were added to the prepared P(AN-co-IA) solutions separately and began stirring at room temperature. During stirring, all samples are kept in the same conditions and protected from sunlight in order to avoid the decomposition of AgNO3. It was observed that five days of stirring were sufficient for the conversion of Ag+ ions to silver nanoparticles. Moreover, it was seen that addition of AgNO3 into P(AN-co-IA) solution immediately caused the solution to become a gel due to the interaction between silver nanoparticles and polymer backbone. Ionization of carboxyl groups and complexation between the COO− groups and Ag+ ions was the main reason for the gel formation [44–47].
Electrospinning of PAN/Ag and P(AN-co-IA)/Ag solutions
To produce PAN/Ag nanofibers, electrospinning was applied to the prepared PAN/Ag solutions. For this reason, prepared solutions were loaded into a syringe having 0.8 mm needle diameter and 15 kV voltage, 1 mL/h feed rate, and 10 cm distance between needle tip and collector were used as optimized electrospinning parameters.
For the production of P(AN-co-IA)/Ag nanofibers, due to the gel form of P(AN-co-IA)/Ag solutions, they were blended with homopolymer of PAN solution in volume ratios of P(AN-co-IA)/Ag/PAN 80:20 (v:v) in order to obtain a spinnable electrospinning solution. The prepared solutions were loaded into a syringe having 0.8 mm needle diameter. Then, the same electrospinning parameters which were 15 kV voltage, 1 mL/h feed rate, and 10 cm distance between needle tip and collector were used as optimized electrospinning parameters.
Figure 1 shows the representative experimental scheme of P(AN-co-IA)/Ag nanofibers beginning from the emulsion latex and ending with nanofiber production process.
Representative experimental scheme of P(AN-co-IA)/Ag nanofibers. DMF: dimethylformamide; P(AN-co-IA): poly(acrylonitrile-co-itaconic acid); AgNO3: silver nitrate.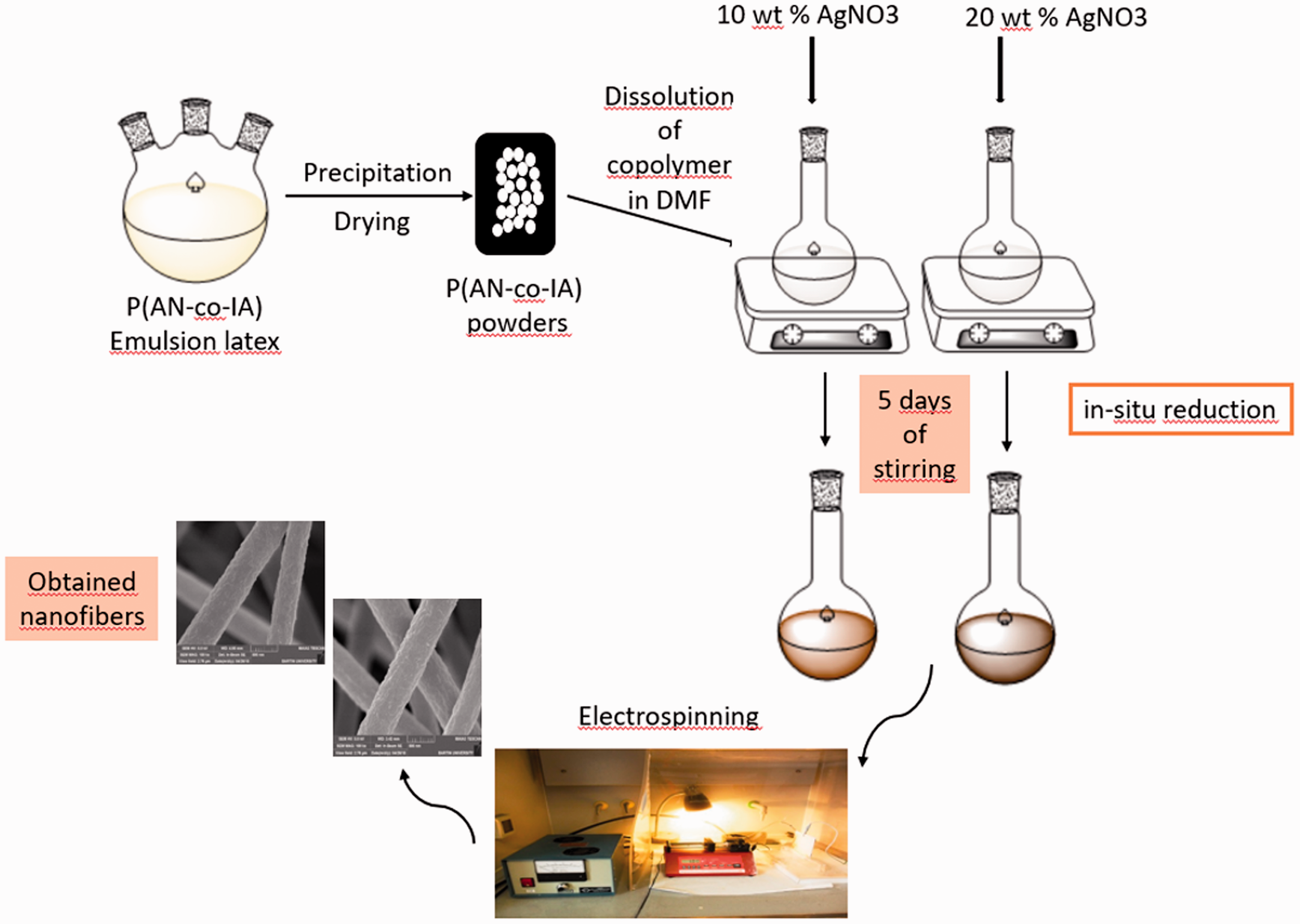
Antimicrobial activity studies of produced nanofibers
Disk diffusion method
To determine in vitro antimicrobial activities of the produced nanofibers of PAN, P(AN-co-IA), PAN/Ag (prepared from 20 wt% AgNO3), and P(AN-co-IA)/Ag (prepared from 20 wt% AgNO3) and the powders of PAN and P(AN-co-IA) polymers and AgNO3, disk diffusion method based on the Clinical and Laboratory Standards Institute (CLSI) criteria was used as a prescreening test against various microorganisms [48]. For this purpose, S. aureus ATCC 29213 as a gram-positive bacteria, E. coli ATCC 25922 and P. aeruginosa ATCC 27853 as gram-negative bacteria, and C. albicans ATCC 10231 as a yeast fungus were used.
Briefly, the cell suspensions (equivalent to 0.5 McFarland) were inoculated over plates containing solidified agar medium onto sterile Petri plates by spread plate method. The nanofiber samples with and without silver nanoparticles were cut in the area of approximately 1 cm2, and the powder solutions in DMSO were immersed on empty sterile disks (in diameters of 6 mm). Then, the samples were placed over the solidified agar gel under aseptic conditions. The plates were placed into an incubator at 37℃ for bacteria and at 25℃ for yeasts. After 24 h, the zone inhibitions were observed.
Susceptibility testing
In vitro antimicrobial activities of the nanofiber samples with and without silver nanoparticles against S. aureus ATCC 29213, E. coli ATCC 25922, P. aeruginosa ATCC 27853, and C. albicans ATCC 10231 strains were investigated. Minimum inhibitory concentrations (MICs) of the nanofibers were determined by microbroth dilution method based on the CLSI criteria [49]. For comparison, the antimicrobial activities of the powders of PAN and P(AN-co-IA) polymers and AgNO3 were also assessed.
The samples used in the study were dissolved in DMSO and then diluted in MHB medium (Difco, Detroid, MI, USA). While the inoculums of the bacteria were prepared using 4–6 h cultures in MHB medium at the final concentration of 5 × 105 cfu/mL (colony forming unit), the inoculum of the yeast strain was prepared using 24-h culture in RPMI-1640 medium (Sigma, St. Louis, MO, USA) at the concentration of 0.5–2.5 × 103 cfu/mL in the test tray. The trays were covered and placed in plastic bags to prevent evaporation and incubated at 37℃ during 18–24 h for the bacteria and at 25℃ during 48 h for the yeasts. The MICs were defined as the lowest concentration of compound giving no visible growth. The activity of DMSO, which is used as solvent in the study, was also tested as negative control, and the results were taken into account when calculating the results. Levofloxacin was used as reference antibacterial for bacteria, and fluconazole was used as reference antifungal for yeasts. The MIC values of levofloxacin and fluconazole were within the accuracy range in CLSI [50] throughout the study.
Time-kill analysis
Time kill analysis were performed to investigate the antimicrobial activities of the nanofiber samples with and without silver nanoparticles against various microorganisms over time according to the time killing curve method which was reported by National Committee for Clinical Laboratory Standards (NCCLS) [51]. S. aureus ATCC 29213, E. coli ATCC 25922, P. aeruginosa ATCC 27853, and C. albicans ATCC 10231 strains were used in the study. The rate of killing of the microorganisms was determined by incubating the test organisms with different concentrations of nanofiber samples or powders in MHB or RPMI-1640 medium for bacteria and yeast, respectively.
PAN/Ag (prepared from 20 wt% AgNO3) and P(AN-co-IA)/Ag (prepared from 20 wt% AgNO3) nanofibers and AgNO3 powder were prepared at the final concentrations equivalent to 1 × MIC and 10 × MIC. The polymers of PAN and P(AN-co-IA) were also weighted in the same amounts, and all of the samples were added into test tubes containing 5 mL of MHB or RPMI-1640 medium. Then, bacterial or fungal suspensions were added to each tube to give a final concentration of 1–5 × 105 cfu/mL. One tube containing only bacterial or fungal inoculum of 1–5 × 105 cfu/mL was prepared as control. The tubes were incubated on a shaker water bath at 37℃ for bacteria and at 25℃ for yeast at an agitation speed of 70 r/min. At predetermined time points (0, 2, 4, 6, 24, 48, 120, and 168 h), samples were removed and diluted in saline, if necessary. Moreover, 100 µL aliquots were spreaded onto TSA agar plates and incubated overnight after drying. Then, surviving colonies were counted (cfu/mL) after incubation. The time-kill curves were constructed by plotting the log10 colony counts (cfu/mL) against time in different concentrations.
Characterization
The obtained nanofiber samples were investigated morphologically by SEM (MAIA3 TESCAN), EDS (MAIA3 TESCAN), and AFM (Nanosurf Easyscan2); spectroscopically by FTIR-ATR spectroscopy (Perkin Elmer) and UV-visible spectroscopy (Perkin Elmer). Conductivity of the produced nanofibers was measured by four-probe instrument (ENTEK, FPP-460), and thermal properties of the samples were analyzed using differential scanning calorimetry (DSC; Perkin Elmer, DSC 4000).
Results and discussion
UV-visible spectroscopy
UV-visible spectroscopy characterization was performed using PAN and P(AN-co-IA) nanofibers prepared with and without AgNO3. Reduction of AgNO3 to silver nanoparticles could be observed from UV-visible characterization as well as visual observation of color change of the solutions [28].
Reduction of Ag+ ions to silver nanoparticles occur in several steps such as primary nucleation, secondary nucleation, crystal growth, and agglomeration of some adjacent particles into aggregated clusters [29]. Reduction of Ag+ ions to silver nanoparticles in the presence of DMF starts with the oxidation reaction of DMF at room temperature according to following mechanism:
As the reaction continues, the color of the solutions changes from transparent to light yellow, dark yellow, brown, and finally black. Therefore, without any spectroscopic characterization, color change of the solution can be regarded as visual confirmation of Ag nanoparticles formation [28–30].
In this study, color change of the solutions for both AgNO3 containing PAN and P(AN-co-IA) solutions was clearly observed. The color of the AgNO3 containing PAN solutions changed from transparent to light yellow after five days of stirring and became dark yellow after 10 days of stirring. Figure 2 depicts the color of PAN/Ag solutions after 10 days of stirring. However, the color of the AgNO3 containing P(AN-co-IA) solutions became light brown, brown, and dark brown just after half an hour, 2 h, and five days of stirring, respectively, as shown in Figure 3.
PAN/Ag polymer solutions prepared from 10 wt% AgNO3 (left) and 20 wt% AgNO3 (right) after stirring of 10 days. P(AN-co-IA)/Ag polymer solutions prepared from a 10 wt% AgNO3 and b 20 wt% AgNO3.

UV-visible spectrum of PAN and PAN/Ag nanofibers was investigated in the range of 200–1100 nm and was shown in Figure 4.
UV-visible spectrums of PAN/Ag nanofibers. PAN: polyacrylonitrile; AgNO3: silver nitrate.
It is known that existence of silver nanoparticles in the samples could be verified by the examination of absorbance band between 300 and 600 nm [27,28,30,52]. However, the exact position of the absorption maxima depends on the used concentrations of AgNO3, the amount of deposited metal, the form of the sample, and the size of the nanoparticles [28,52]. If silver nanoparticles have diameters smaller than 5 nm, they show a high absorbance band with a peak nearly at 400 nm. If they have diameters of ∼10 nm, they exhibit absorption band between 410 and 450 nm. Moreover, when the absorption band shifts to higher wavelengths, it means that the produced silver nanoparticles have diameters bigger than 10 nm [27]. In Figure 4, it can be clearly seen that Ag containing PAN nanofibers showed an absorbance band between 400 and 500 nm without a sharp peak, while no absorbance was seen for pure PAN nanofibers in that range. With the light of these information, it can be interpreted that the produced PAN/Ag nanofibers contain Ag nanoparticles with coarser diameters than 10 nm.
UV-visible spectrum of P(AN-co-IA) and P(AN-co-IA)/Ag nanofibers was also examined between 200 and 1100 nm and was demonstrated in Figure 5.
UV-visible spectrums of P(AN-co-IA)/Ag nanofibers. P(AN-co-IA): poly(acrylonitrile-co-itaconic acid); AgNO3: silver nitrate.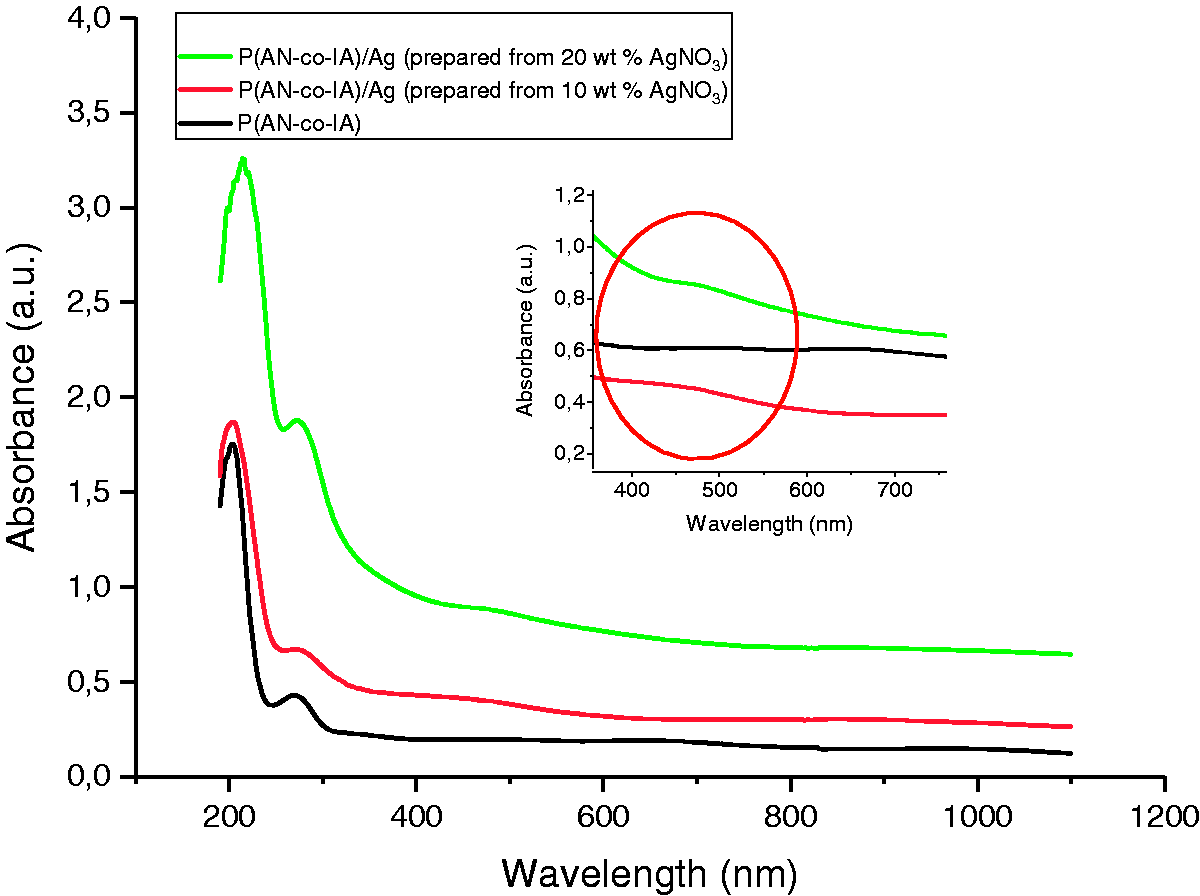
For the verification of Ag nanoparticle formation, 300–600 nm wavelength was examined deeply. It was seen from Figure 5, while pure P(AN-co-IA) nanofibers did not show any peak in that range, Ag containing P(AN-co-IA) nanofibers reflected absorbance between 400 and 500 nm with maximas approximately at 450 and 470 nm for the samples prepared from 10 wt% and 20 wt% AgNO3, respectively. Sichani et al. also mentioned that both dielectric constant and range of the nanoparticles affect the absorption maximum of UV-visible spectra [28]. Since UV-visible spectrum give preliminary information about the size and distribution of the produced silver particles, according to literature [27,28], it can be said that the produced nanoparticles in this study both have higher diameters than 10 nm and size distribution. In addition, increase in the intensity was related to the increase in the amount of reduced silver [28]. From Figure 5, it can be monitored that P(AN-co-IA)/Ag nanofibers prepared from 20 wt% AgNO3 had higher intensity than the P(AN-co-IA)/Ag nanofibers prepared from 10 wt% AgNO3, thus P(AN-co-IA)/Ag nanofibers prepared from 20 wt% AgNO3 have higher amounts of reduced silver than the other sample. Moreover, the intensities of P(AN-co-IA)/Ag nanofibers are higher than the intensities of PAN/Ag nanofibers (Figure 4) which is also a proof that P(AN-co-IA) reduced more amount of AgNO3 to Ag nanoparticles than PAN.
FTIR-ATR spectroscopy
To examine the effect of existence of Ag nanoparticles in both PAN and P(AN-co-IA) nanofibers, FTIR-ATR spectroscopy was used and demonstrated in Figures 6 and 7, respectively.
FTIR-ATR spectrums of PAN and PAN/Ag nanofibers. PAN: polyacrylonitrile; AgNO3: silver nitrate. FTIR-ATR spectrums of P(AN-co-IA)/Ag nanofibers. P(AN-co-IA): poly(acrylonitrile-co-itaconic acid); AgNO3: silver nitrate.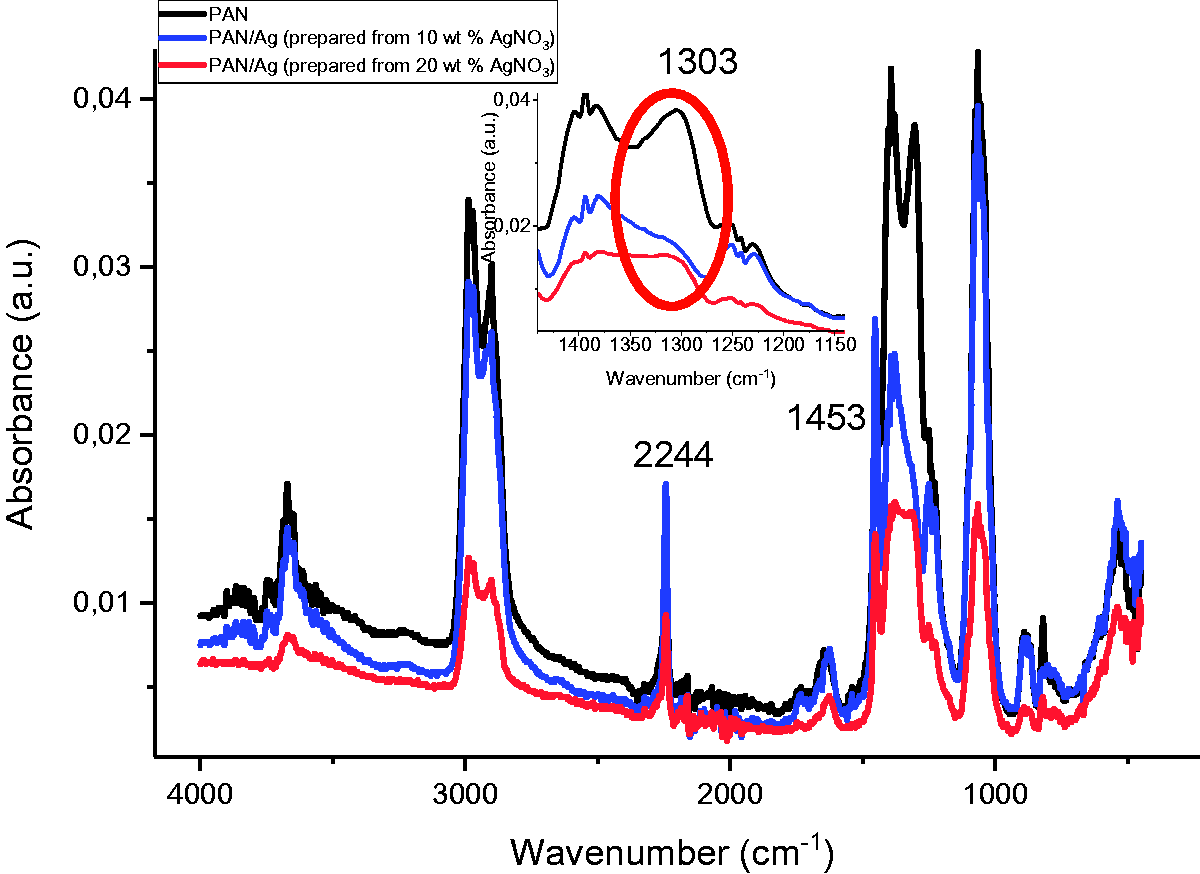

The positions of all peaks of for PAN and PAN/Ag nanofibers were almost the same as seen in Figure 6. The presence of Ag nanoparticles in the samples did not make any considerable shift in the C≡N triple bond (at 2244 cm−1) [28,45]. However, the intensity of the peaks of PAN/Ag nanofibers decreased compared to PAN nanofibers and even according to the increase in AgNO3 amount from 10 wt% to 20 wt%. Thus, it can be said that nitrile groups in PAN chains polymerized by the existence of Ag+ ions [28]. The only shift was seen at 1303 cm−1 that the sharp peak disappeared and just turn to be a shoulder with the presence of Ag nanoparticles. In addition, the peak at 1453 cm−1 corresponded to bending vibration of –CH [43].
FTIR-ATR spectrums of P(AN-co-IA) and P(AN-co-IA)/Ag nanofibers (shown in Figure 7) also mention that the positions of the peaks were nearly the same for P(AN-co-IA)/Ag nanofibers. The peak at 2244 cm−1 was related to the C≡N triple bond, and there was no noticeable shift on the position of it with the presence of silver nanoparticles. However, the intensity of this peak increased due to presence of silver nanoparticles but decreased with the increase in as the amount of used silver salt (from 10 wt% to 20 wt%) as in case of PAN and PAN/Ag nanofibers [28]. New peaks occurred at 3657 cm−1 and 1663 cm−1, and a shift from 1731 cm−1 to 1721 cm−1 was seen for silver containing P(AN-co-IA) nanofibers which could be taken as a proof for the ionization of carboxyl groups and interaction of Ag+ ions between P(AN-co-IA) copolymer [45,53,54]. The interaction of Ag+ ion with P(AN-co-IA) is also shown in Figure 8. Related to the interaction of Ag+ ion with P(AN-co-IA), a new shoulder at 1314 cm−1 occurred, and the peak at 1235 cm−1 shifted to 1237 cm−1 by giving three broad peaks.
Interaction of silver ion with P(AN-co-IA).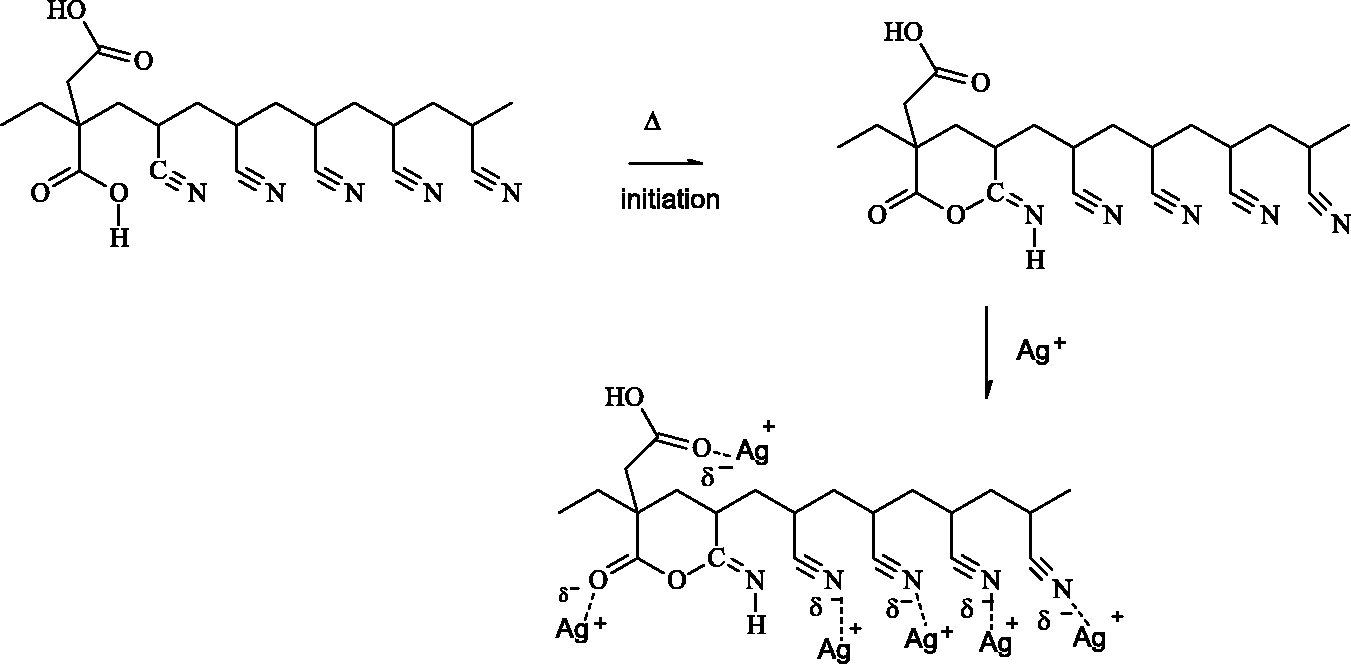
SEM and EDS
To observe the surface morphology of the produced nanofibers, SEM analysis was performed. Average diameter of the nanofibers was also determined using a special program, Image J. Figure 9 [43] shows SEM images of PAN and P(AN-co-IA) nanofibers along with their diameter distribution graphics. Figure 9 [43] shows that PAN and P(AN-co-IA) nanofibers are continuous, smooth, and fine with the average diameters of 120 and 132 nm, respectively.
SEM images of PAN (left) and P(AN-co-IA) (right) nanofibers [43].
Figure 10 is related to the SEM images of PAN/Ag and P(AN-co-IA)/Ag nanofibers, prepared using 10 wt% AgNO3, with different magnification ratios, and Figure 11 shows SEM images of PAN/Ag and P(AN-co-IA)/Ag nanofibers, prepared using 20 wt% AgNO3, with different magnification ratios. Also, the nanofiber diameter distribution graphics were added onto the SEM images.
SEM images of (a) and (b) PAN/Ag nanofibers (prepared from 10 wt% AgNO3) with 50 kx and 100 kx magnification ratios, respectively. (c) and (d) P(AN-co-IA)/Ag nanofibers (prepared from 10 wt% AgNO3) with 50 kx and 100 kx magnification ratios, respectively. SEM images of (a) and (b) PAN/Ag nanofibers (prepared from 20 wt% AgNO3) with 50 kx and 100 kx magnification ratios, respectively. (c) and (d) P(AN-co-IA)/Ag nanofibers (prepared from 20 wt% AgNO3) with 50 kx and 100 kx magnification ratios, respectively.

It can be accurately observed from Figures 10 and 11 that silver nanoparticle containing nanofibers are continuous and have rougher surface compared to pure PAN and P(AN-co-IA) nanofibers. No beads were seen related to the electrospinning parameters and especially due to the strong elongation forces applied on the jet during electrospinning [28]. Rough surface morphology of nanofibers which include silver nanoparticles was also observed in the studies conducted by Chaudhary et al. [55] and Ucar et al. [56].
Nanofiber samples and their average nanofiber diameters (nm).

PAN: polyacrylonitrile; P(AN-co-IA): poly(acrylonitrile-co-itaconic acid); AgNO3: silver nitrate.
According to the reduction method, stabilizer and initial concentration of AgNO3, nanofiber diameters can increase in the presence of silver nanoparticles [56]. From Table 1, it can be evaluated that increasing the amount of AgNO3 caused to produce coarser nanofibers. This could be due to silver salt, changing in conductivity cause to fiber diameter increase. When the salt is added to solution, the diameter decreases, but if the additive amount increased, the fiber diameter tends to increase too. In addition, either the amount of deposited silver nanoparticles on the surface [39] or agglomeration of silver nanoparticles occurred in the syringe tip during electrospinning might be another reason for that increase [55]. Similarly, Chaudhary et al. observed that in case of silver nanoparticles existence in PAN nanofibers, surface morphology of the nanofibers changed from smooth to rough, and nanofibers had diameters between 600 and 900 nm [55]. In addition, Huang et al. produced PAN/Ag nanofibers in the range of 500–800 nm [57]. Table 1 also informs that in the presence of silver nanoparticles, P(AN-co-IA) matrix allowed to obtain smaller nanofiber diameters than PAN matrix, which might depend on the increased charge density during electrospinning process [58].
EDS analysis results of atomic% of Ag nanoparticles present in the produced samples.
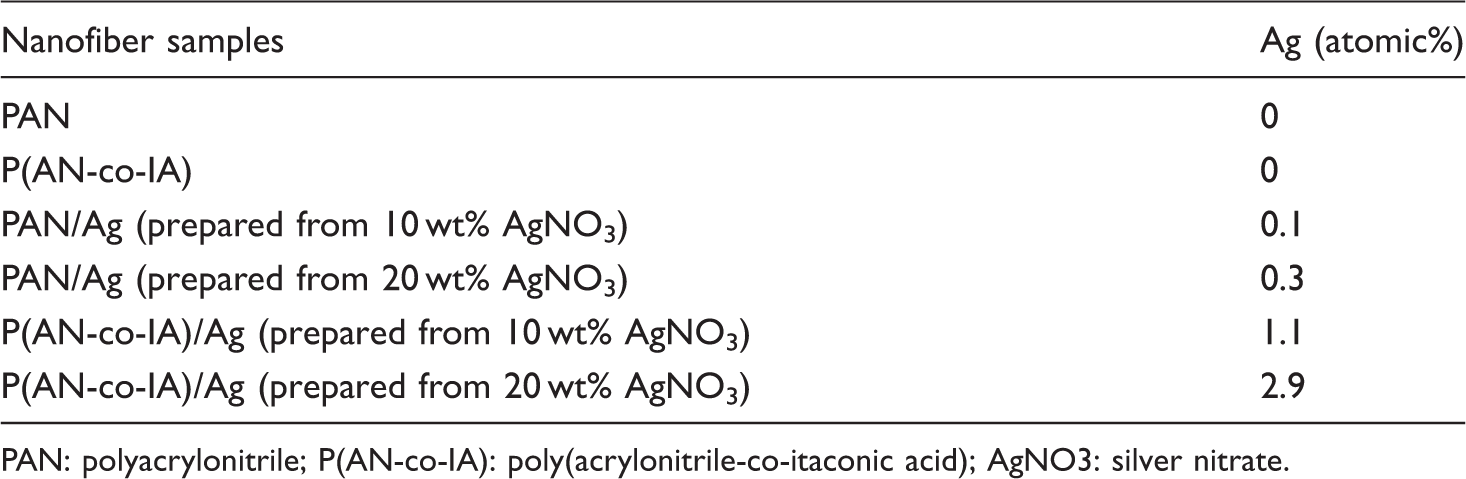
PAN: polyacrylonitrile; P(AN-co-IA): poly(acrylonitrile-co-itaconic acid); AgNO3: silver nitrate.
As expected, neat PAN and P(AN-co-IA) nanofibers did not contain any silver nanoparticles, but the other samples contained silver nanoparticles. Final atomic% of silver nanoparticles in the samples had a direct relation with the amount of AgNO3. Furthermore, for the same amount of AgNO3, it was observed that P(AN-co-IA) nanofibers had much more silver nanoparticles than PAN, which can be taken as an evidence for the enhanced reduction of AgNO3 to silver nanoparticles by P(AN-co-IA) polymer.
Atomic force microscopy
Surface morphology of both produced nanoparticles and nanofibers of PAN, P(AN-co-IA), PAN/Ag, and P(AN-co-IA)/Ag was also analyzed using AFM which uses a tip to evaluate the surface features by scanning the sample.
For nanoparticle observation, thin films were prepared via spin-coating process using prepared electrospinning solutions of PAN, P(AN-co-IA), PAN/Ag and P(AN-co-IA)/Ag compositions. Figure 12 displays AFM images of PAN nanoparticles with different magnification ratios.
AFM images of PAN nanoparticles with magnification ratios of (a) 10 µm, (b) 5 µm, and (c) 2.5 µm.
Although some agglomerates were seen in the images, it can be said that produced PAN nanoparticles were almost distributed homogeneously. Moreover, surface roughness values of PAN nanoparticles varied as 72 nm, 61 nm, and 55 nm related to the magnification ratios of 10 µm, 5 µm, and 2.5 µm, respectively.
Figure 13 shows AFM images of P(AN-co-IA) [32] and P(AN-co-IA)/Ag nanoparticles with different magnification ratios.
AFM images of (a) P(AN-co-IA) nanoparticles (Baskan et al 2017), (b) P(AN-co-IA)/Ag nanoparticles with 10 µm magnification ratio, and (c) P(AN-co-IA)/Ag nanoparticles with 5 µm magnification ratio.
Similar to AFM images of PAN nanoparticles, it can be said that nanoparticle distribution of P(AN-co-IA) and P(AN-co-IA)/Ag samples was homogeneous even though presence of some agglomerates. Surface roughness values of the nanoparticles were calculated as 16 nm, 111 nm, and 86 nm for P(AN-co-IA), P(AN-co-IA)/Ag (magnification ratio of 10 µm), and P(AN-co-IA)/Ag (magnification ratio of 5 µm), respectively. It can be accurately declared that silver containing P(AN-co-IA) samples had rougher surfaces than the pure P(AN-co-IA) sample.
Furthermore, AFM image of P(AN-co-IA)/Ag nanofibers was demonstrated in Figure 14.
AFM image of P(AN-co-IA)/Ag nanofibers.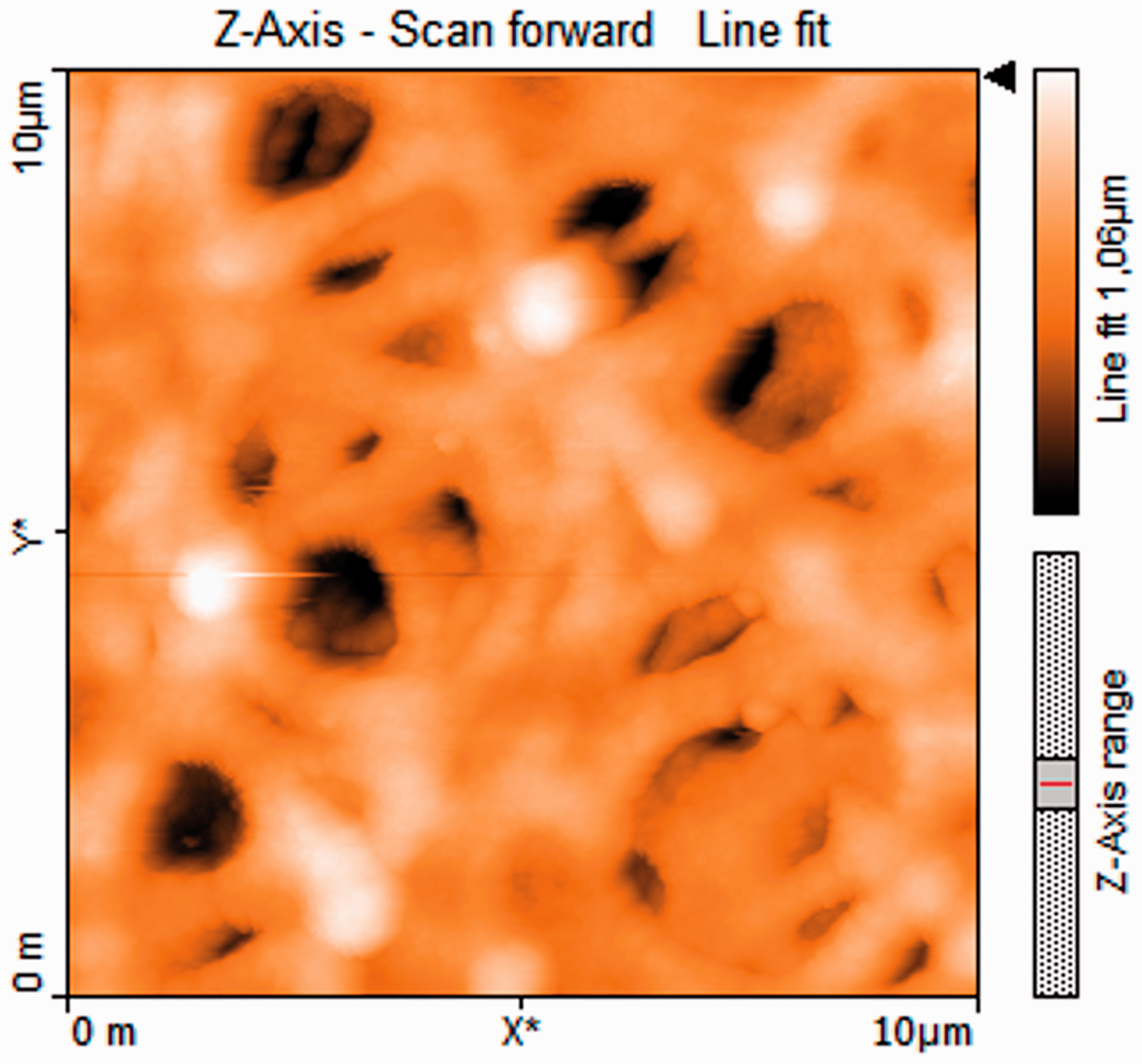
Deposition of Ag nanoparticles over the surface of nanofibers can be observed from Figure 14, and it is clear that brighter regions existing in nanofiber mats are related to the silver nanoparticles. Surface roughness value of P(AN-co-IA)/Ag nanofibers was measured as 129 nm which was higher than the surface roughness values of both PAN and P(AN-co-IA)/Ag nanoparticles. This might be mainly because of the either deep inclusion of Ag nanoparticles in the polymer chain during the reduction process or deposition of the produced nanoparticles over the nanofiber surface [55].
Electrical conductivity
Electrical conductivity values (S/cm) of the obtained nanofiber samples.

PAN: polyacrylonitrile; P(AN-co-IA): poly(acrylonitrile-co-itaconic acid); AgNO3: silver nitrate.
It is well known from the literature that neat PAN has 10−12 S/cm electrical conductivity and when the value reaches to 10−7 S/cm, it becomes a semiconductive material [59]. It is also possible to increase the electrical conductivity of PAN by incorporation of metal nanoparticles like silver into the structure [56]. Table 3 shows that while neat PAN nanofibers were really insulators with 3.24 E−13 S/cm electrical conductivity value, the presence of Ag nanoparticles made them to become conductive materials and gain ∼2.70 E−5 S/cm electrical conductivity. P(AN-co-IA) nanofibers also showed nearly the same behavior. Existence of Ag nanoparticles in P(AN-co-IA) nanofibers considerably contributed to the electrical conductivity values. They were increased from 2.64 E−12 to ∼ 2.80 E−4. In addition, a slight increase was seen in the conductivity values related to the used AgNO3 amount. It was mentioned that using even a small silver salt amount [28] and changing the initial concentration of AgNO3 [29] can influence the electrical conductivity dramatically.
The relation between electrical conductivities and atomic% Ag values of the obtained nanofibers was revealed in Figure 15.
Relation between electrical conductivities and atomic% Ag values of the obtained nanofibers.
Figure 15 informs that electrical conductivity of the produced nanofibers was directly affected by atomic% of silver nanoparticles existing in the samples. Thus, in order to increase the electrical conductivity, the amount of the reduced silver nanoparticles should be increased and initial concentration of silver salt should be optimized.
Differential scanning calorimetry
DSC was applied in order to examine the thermal behavior of the produced nanofiber samples.
DSC thermograms of PAN, PAN/Ag (prepared from 20 wt% AgNO3), and P(AN-co-IA)/Ag (prepared from 20 wt% AgNO3) nanofibers were shown in Figure 16.
DSC thermograms of PAN, PAN/Ag (prepared from 20 wt% AgNO3), and P(AN-co-IA)/Ag (prepared from 20 wt% AgNO3) nanofibers. PAN: polyacrylonitrile; P(AN-co-IA): poly(acrylonitrile-co-itaconic acid); AgNO3: silver nitrate.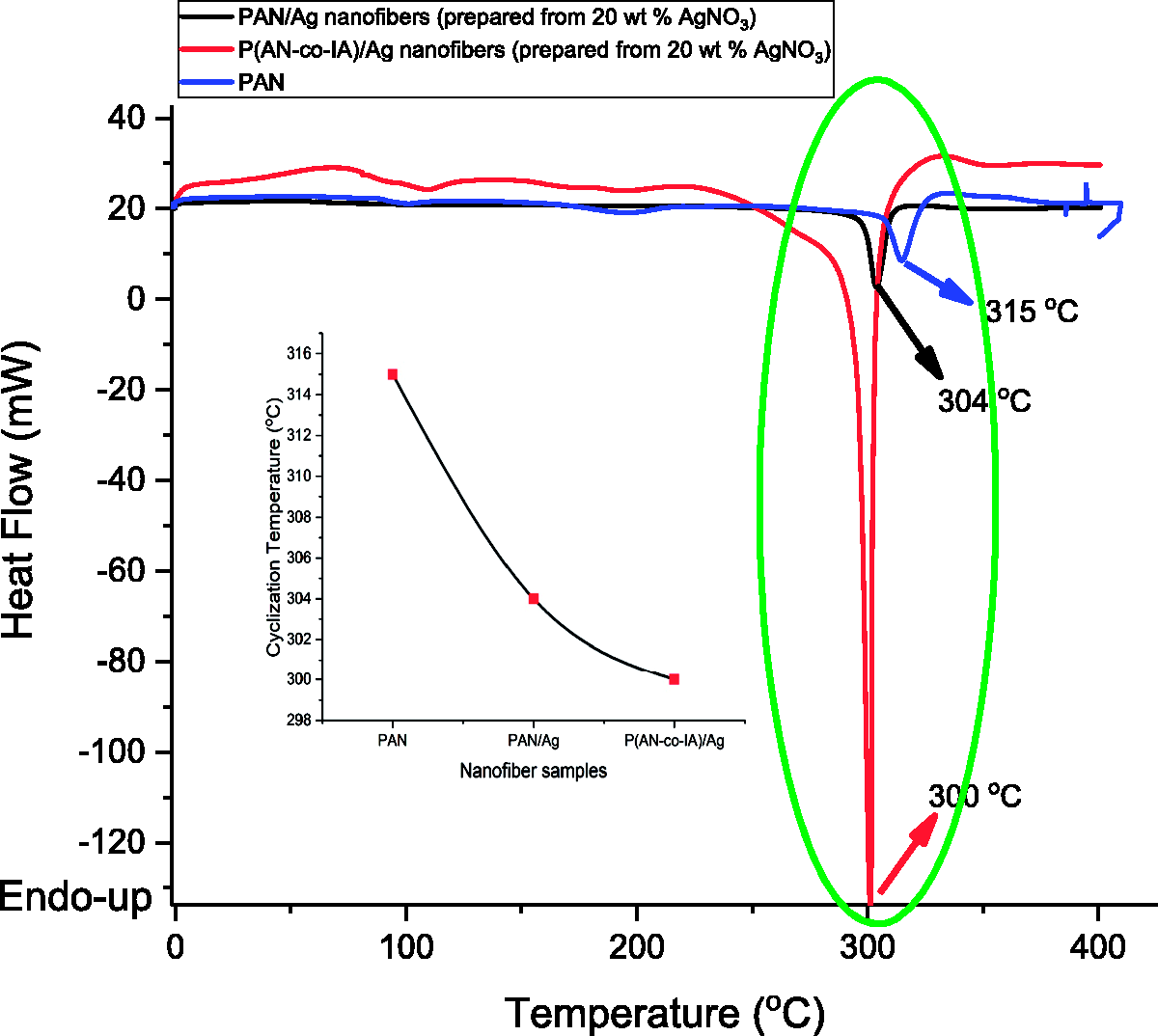
During the transformation of C≡N groups into C=C and C=N groups, some chemical reactions such as cyclization, dehydrogenation, and oxidation occur. These chemical reactions allow PAN fibers to have ladder-like molecular structure and become heat resistant [56].
Cyclization reactions of PAN takes place through a free radical mechanism and can only be initiated at high temperatures. PAN displays an exothermic peak with a maxima nearly at 300℃, and this maxima is defined as cyclization temperature (Tc). However, it is possible to decrease this cyclization temperature up to 250℃ by the incorporation of IA monomer into PAN structure [16,60]. Figure 16 shows that while cyclization temperature of PAN nanofibers was 315℃, it was 304℃ and 300℃ for PAN/Ag and P(AN-co-IA)/Ag nanofibers which were prepared from 20 wt% AgNO3, respectively. It was understood that both silver nanoparticles and IA contributed to decrease the cyclization temperature of PAN by generating sharp exothermic peaks. In case of IA existence in PAN structure, cyclization reactions initiates with an ionic mechanism rather than a radical mechanism and shows doublet exothermic peak with maximum points approximately at 180℃ and 310℃, respectively [16]. However, only a single and sharp exothermic peak of P(AN-co-IA)/Ag nanofibers was seen in Figure 16. It can be said that incorporation of silver nanoparticles into P(AN-co-IA) copolymer resulted in a new material which exhibits different thermal features than neat P(AN-co-IA).
Cyclization temperatures and enthalpy values of produced PAN, PAN/Ag, and P(AN-co-IA)/Ag nanofibers.

PAN: polyacrylonitrile; P(AN-co-IA): poly(acrylonitrile-co-itaconic acid); AgNO3: silver nitrate.
It was seen that silver nanoparticles increased ΔH values of the nanofibers when compared to pure PAN nanofibers. Ucar et al. also studied on the thermal behavior of Ag nanoparticle containing PAN nanofibers and observed that the enthalpy of the nanofibers increased due to the silver nanoparticles. Moreover, it was mentioned that reduction method of AgNO3 to silver nanoparticles had effect on not only the enthalpy values but also cyclization temperature [56]. Considering this, it can be concluded that reduction of AgNO3 to silver nanoparticles in the presence of P(AN-co-IA) rather than PAN had a great influence on ΔH values.
Antimicrobial activity studies
Disk diffusion method
Ag nanoparticles are broad spectrum antimicrobials exhibiting very strong bactericidal activity against both gram-positive and gram-negative bacteria and antifungal activity against Candida species [33,61]. As a result of disk diffusion method for prescreening the antimicrobial activity, it was observed that the powder of AgNO3 and the nanofibers of PAN and P(AN-co-IA) containing Ag nanoparticles produced inhibition zones against the studied microorganisms. Obtained inhibition zones against E. coli ATCC 25922, P. aeruginosa ATCC 27853, S. aureus ATCC 29213, and C. albicans ATCC 10231 cultures in plates were shown in Figure 17.
Observed inhibition zones of (a) PAN nanofiber, (b) PAN/Ag nanofiber (prepared from 20 wt% AgNO3), (c) P(AN-co-IA) nanofiber, (d) P(AN-co-IA)/Ag nanofiber (prepared from 20 wt% AgNO3), (e) PAN powder, (f) P(AN-co-IA) powder, and (g) AgNO3 powder against four different microbial cultures in plates.
Microdilution susceptibility testing
MIC values determined for the nanofiber samples with and without silver nanoparticles and powders.

PAN: polyacrylonitrile; MIC: minimum inhibitory concentration; P(AN-co-IA): poly(acrylonitrile-co-itaconic acid); AgNO3: silver nitrate.
Time-kill analysis
Antimicrobial activities of the nanofibers of PAN, P(AN-co-IA), PAN/Ag (prepared from 20 wt% AgNO3), and P(AN-co-IA)/Ag (prepared from 20 wt% AgNO3) and AgNO3 powder at 1 × MIC and 10 × MIC concentrations were evaluated by counting the viable cells of S. aureus, E. coli, P. aeruginosa, and C. albicans suspensions at different time intervals after contact with the samples. For comparison, the growth control of the microorganisms was also assessed. Bactericidal or fungicidal activity was defined as a 99.9% (≥3 log10) reduction of initial cfu/mL, according to the NCCLS (1999) guidelines. The lower detection limit for the time-kill assay was 1 log 10 cfu/mL.
According to the results, which were shown in Figure 18, nanofibers without silver nanoparticles (PAN and P(AN-co-IA)) did not show any antimicrobial activity against any microorganism during the studied 168-h time period as expected. However, bacteriostatic/fungistatic activity was observed with all nanofiber samples containing silver nanoparticles (PAN/Ag and P(AN-co-IA/Ag)) against S. aureus, P. aeruginosa, and C. albicans at almost all time points, bactericidal activity was started from 24 h to 48 h until 120 h at 10 × MIC. Especially against E. coli, it starts from 6 h at 10 × MIC and 24 h at 1 × MIC. In addition, fungicidal activity was observed in 120 and 168 h at 10 × MIC.
Time-kill curves of nanofibers of PAN, P(AN-co-IA), PAN/Ag (prepared from 20 wt% AgNO3), and P(AN-co-IA)/Ag (prepared from 20 wt% AgNO3) and AgNO3 powder at 1 × MIC and 10 × MIC concentrations against: (a) E. coli, (b) P. aeruginosa, (c) S. aureus, and (d) C. albicans strains after 0, 2, 4, 6, 24, 48, 120, and 18 h. The x-axis represents the killing time (hours), and the y-axis represents the logarithmic counts of survival microorganisms. PAN: polyacrylonitrile; MIC: minimum inhibitory concentration; P(AN-co-IA): poly(acrylonitrile-co-itaconic acid); AgNO3: silver nitrate.
Nanofibers with silver nanoparticles and AgNO3 powder samples showed their most effective bactericidal activities against gram-negative bacteria E. coli and P. euruginosa started from 6 h and 24 h at 10 × MIC and 1 × MIC, respectively. Additionally, they showed bactericidal or fungicidal activities at their 10 × MIC started after 24–48 h against S. aureus and C. albicans.
In conclusion, bactericidal/fungicidal activities were obtained with nanofibers incorporating silver nanoparticles, especially at 10 × MIC concentration and AgNO3 powder at 1 × MIC and 10 × MIC concentrations. Moreover, these activities started at 6–24 h and continued for up to 168 h.
Conclusions
In this study, PAN homopolymer and P(AN-co-IA) copolymer were synthesized by emulsion polymerization. PAN and P(AN-co-IA) polymers were dissolved in DMF separately, and two different amounts, 10 wt% and 20 wt% AgNO3 (the weight percentage of AgNO3 in the solution was calculated on the basis of PAN and P(AN-co-IA)weight) were added to the prepared PAN and P(AN-co-IA) solutions. Reduction of AgNO3 to silver nanoparticles was achieved by means of PAN, P(AN-co-IA), and DMF without using any additional reductant or process. Color change of the solutions confirmed the formation of silver nanoparticles as well as UV-visible spectroscopy, FTIR-ATR spectroscopy, and EDS analyses. It was also seen that P(AN-co-IA) was much more effective on reduction process than PAN. Surface morphology of the produced nanofibers was investigated by SEM and AFM. SEM images of the nanofibers showed that silver containing nanofibers had rougher surfaces and existence of silver nanoparticles in the nanofibers increased the nanofiber diameters from ∼ 120 nm to ∼ 500 nm. When the diameters of PAN/Ag and P(AN-co-IA)/Ag nanofibers were compared, it was observed that finer diameters could be obtained in case of using P(AN-co-IA). Electrical conductivity of the nanofibers increased dramatically from E−13 to E−4 related to the presence of silver nanoparticles. In addition, DSC characterization displayed that both silver nanoparticles and IA contributed to decrease the cyclization temperature of PAN by generating sharp exothermic peaks. Also, reduction of AgNO3 to silver nanoparticles in the presence of P(AN-co-IA) rather than PAN had a great increment on ΔH values, which is attributed to the cyclization of the nitrile (C≡N) groups present in the structure of the polymers. The last but not the least, incorporation of silver nanoparticles to the nanofiber membranes let bactericidal/fungicidal activities against S. aureus, E. coli, P. aeruginosa, and C. albicans for both PAN/Ag and P(AN-co-IA)/Ag nanofibers. The bactericidal/fungicidal activities started at 6–24 h and continued for up to 168 h. As a conclusion, it can be interpreted that silver containing nanofibers showed satisfying antimicrobial results and can be regarded as good candidates for potential use in the biomedical and pharmaceutical applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors want to thank ITU Scientific Research Center project coded as MDK-2018-41127 for their financial contribution to the study. In addition, Havva Baskan specifically wants to thank The Scientific and Technological Research Council of Turkey (TUBITAK) 2211-A (application number: 1649B031500306) and 2214-A (application number: 1059B141800375) programs for their financial support to this study.

