Abstract
In the present study, the electrospinning method was used to develop β–cyclodextrin functionalized polyvinyl alcohol nanofiber webs. The electrospinning parameters were adjusted in order to obtain uniform nanofiber webs by incorporation of varying concentrations of polyvinyl alcohol and β–cyclodextrin in the solutions. Subsequently, β–cyclodextrin/Vitamin C inclusion complexes were added into the polyvinyl alcohol solution, and adjusted parameters were used in the electrospinning process of functionalized polyvinyl alcohol nanofiber webs containing β–cyclodextrin/Vitamin C inclusion complexes. The formation of nanofiber webs was demonstrated by using Scanning Electron Microscopy. The presence of the inclusion complex in the functionalized electrospun polyvinyl alcohol nanofiber webs was proven by using ultraviolet–visible spectroscopy and Fourier Transform-Infrared Spectroscopy. Consequently, it was achieved to functionalize polyvinyl alcohol electrospun nanofiber webs by incorporating β–cyclodextrin/Vitamin C inclusion complex, which might be considered to have useful applications in cosmetics, personal-care products, and topical drug delivery.
Introduction
Materials and surfaces in nanoscale, such as nanofibers and nanofiber webs, have gained prominence in various applications due to their outstanding characteristics like a high ratio of surface area to volume, porous structure in very small sizes, and ability to be modified and functionalized easily [1]. Electrospinning is the most common process to produce nanofiber and nanofiber webs from diversified polymers and polymer blends. By using the electrospinning process, nanofiber webs in diameters from several nanometers to hundreds of nanometers can be obtained [2]. Incorporation of functional materials into the polymer solution enables producing composite nanofiber webs by using the electrospinning process. One of the methods for functionalization of electrospun nanofibers and nanofiber webs is the incorporation of cyclodextrins (CDs) into the nanofiber formation process.
CDs are cyclic oligosaccharides consisting of 6, 7, 8, or more glucopyranose units linked by α–(1,4) bonds. Specifically, CDs with 6, 7, and 8 glucopyranose units are called as α–CD, β–CD, and δ–CD, respectively [3]. Among them, β–CD is the most accessible, the lowest-priced, and generally the most useful one. CDs have a wide range of utilization in textiles, food, pharmaceutical, cosmetic, and agricultural industries [4]. The glucopyranose units, linked in a ring, form a cone shape with a cavity inside, which allows the products, including CDs, to be used for the absorption of unwanted odors or the adsorption of small pollutants in waste water. Besides, this cavity enables CDs to form inclusion complexes with a variety of compounds. For instance, since they are nontoxic materials, they can be used in pharmaceuticals for controlled drug delivery, in beauty products for sustained release of cosmetic additives, and in personal-care products [5–10]. In fact, there are several studies, which showed that CDs can be used effectively for the stabilization and persistent release of drugs, fragrances, or flavors [7,8,11–13].
The incorporation of CDs into electrospinning of nanofiber webs was conducted in several studies up to date. α–, β–, and δ–CDs were incorporated into polystyrene (PS), polymethyl methacrylate (PMMA), polyethylene oxide, and zein fibers by electrospinning method, mainly for the aim of using the obtained nanofiber webs as a molecular filter [10,14–18]. PS and PMMA nanofiber webs were also functionalized by incorporating α–, β–, and δ–CD/menthol inclusion complexes for enhancing the durability and stability of volatile fragrance such as menthol [14,19]. Electrospun polylactic acid nanofiber webs, including α–, β–, and, δ–CD/triclosan inclusion complexes were developed for obtaining antibacterial property [20]. Besides, polyvinyl alcohol (PVA), which is a non-toxic and water-soluble polymer, was functionalized with the addition of α–, β–, and δ–CD/eugenol inclusion complexes and α–, β–, and δ–CD/vanillin inclusion complexes for enhancing the thermal stability and slow release of the guest compound that may be useful in the food industry for prolonged shelf life [21,22]. For the same purposes in the food industry, vitamins (Vitamin A—retinyl acetate and Vitamin E—α-tocopherol) were also used as the guest material in the inclusion complex with β–CD [23,24].
Other than the food industry, CDs and their inclusion complexes were proposed to be used as scaffolds and wound odor absorbents while providing an ideal environment for the wound to heal in medical applications. α–CD, β–CD, and δ–CDs were used for enhancing the physical and thermo-mechanical properties of pseudorotaxane nanofibers composed of biomedical poly ε-caprolactone (PCL) polymer with different solvent systems, i.e. chloroform, dimethylformamide, and their mixtures [25–27]. In another study, non-stoichiometric inclusion complexes were added into PCL polymer for the same purposes [28]. The inclusion complexes of vitamins and β–CDs were also observed to be used in medical applications, especially in topical drug delivery materials. As an example, β–CD/α-tocopherol (Vitamin E) inclusion complex was incorporated into PCL nanofibers because of their antioxidant activity and photostability. In this way, they can be thought as an alternative material for the topical delivery of poorly soluble drugs [24]. The complexation between ascorbic acid (Vitamin C) and β–CD and hydroxypropyl-β–CD was studied by using different methods such as high hydrostatic pressure and freeze drying [29,30]. Although these studies proved the formation of inclusion complexes of β–CD/Vitamin C, they did not propose an incorporation of these inclusion complexes into the structure of any material. Considering that ascorbic acid (Vitamin C) has the ability to scavenge free radicals and destroy oxidizing agents, a novel polymeric nanofiber face mask composed of PVA and inclusion complexes of random methylated (RM)-β–CD with ascorbic acid, retinoic acid, gold nanoparticles, and collagen were developed as the only study presenting the application of RM-β–CD/ascorbic acid inclusion complex. In the mentioned study, ethanol was used as the solvent for the preparation of electrospinning solution [31].
As it can be seen from the literature survey, the functionalization of nanofiber webs with CDs is very appealing due to the combination of their unique characteristics that is important for broadening the use of both electrospun nanofiber webs and CDs. However, the formation of inclusion complexes of the vitamins and CDs was noticed in a limited number of studies; although there is a large potential of use in different areas such as cosmetics, food industry, and medical applications as explained by the related literature above.
To expand the limited literature focusing on the formation of CD and vitamin complexes to be used for cosmetic applications by using different polymer/solvent combinations, CD type, and the application of different concentrations of β–CD/Vitamin C inclusion complexes, in this study, functional PVA electrospun nanofiber webs containing β–CD and Vitamin C inclusion complexes at different concentrations were produced. The electrospinning conditions were aimed to be adjusted in order to produce uniform functionalized PVA nanofiber webs. The characterization of these functional nanofiber webs and the presence of additive materials were analyzed by using Scanning Electron Microscopy (SEM), ultraviolet–visible (UV–Vis) spectroscopy, and Fourier Transform-Infrared Spectroscopy (FT-IR) techniques.
Experimental part
Materials
PVA (C2H4O; melting point: 200℃), 99% hydrolyzed with a molecular weight (Mw) of 130,000 g/mol (Sigma Aldrich), was used in this study. β–CD (C42H70O35, Mw: 1134.98 g/mol, melting point: 300℃, and purity ≥ 98.5%) and Vitamin C (ascorbic acid, C6H8O6, Mw: 176 g/mol, and melting point: 189℃) with purity of > 99.7% were purchased from Sigma Aldrich and Scharlau, respectively. Distilled water and acetic acid with purity of 100% were used as the solvents. All materials were used as received without any purification.
Production of nanofiber webs
In the preliminary trials, PVA was dissolved in distilled water by heating around 75–95℃ around 2 h [32–35]. The PVA content was varied in percentages between 5 and 10% (w/v) as suggested in the literature [32–36]. The dissolution of the polymer was observed visually, and the conductivity of the solutions was measured by using Mettler Toledo SevenMulti conductivity meter (USA). The conductivities (µS/cm) of these solutions were found to be ranging between 92 and 187 that were lower when compared to the earlier studies [33,36]. Besides, dissolution of PVA was incomplete, particularly as the PVA concentration increases. Therefore, in order to dissolve PVA completely and to increase the conductivity of the solutions to the levels specified previously, PVA was dissolved in the 50%–50% (w/w) mixture of distilled water and acetic acid for 2 h at 80℃ by using magnetic stirrer. Since this process led to complete dissolution of PVA and acetic acid was mixed with water in the solution, the conductivity increased to the levels around 490 µs/cm, which is in conformity with the results of the studies in the literature [33,36]; 0.01% (w/v) NaCl was also added to increase the conductivity and charge density of the solutions more [36]. Then, the conductivity of the solutions were measured between 3400 and 5400 (µS/cm) that are compatible with the earlier studies stated before [33,36]. Salt addition to the electrospinning solution, which increases the conductivity of the medium, has a potential to positively affect the morphology of the nanofibers and electrospun mats leading to not only hindering of beads or change in their structure making them more spindle-like but also drop in average fiber diameter, production of smoother nanofibers as already declared in the literature [37–43].
Based on the results obtained in the earlier studies in the literature and the preliminary trials carried out by the authors, PVA nanofiber web samples with β–CD were produced in two different concentrations of PVA as 6% and 8% (w/v). β–CD amount was adjusted to 17%, 25%, and 33% (w/w) with respect to PVA in 6% PVA solutions and 13%, 19%, and 25% (w/w) with respect to PVA in 8% PVA solutions [33,34].
Since the average diameters of the nanofibers produced from 6% PVA solutions were lower, PVA concentration was used as 6% (w/v) with respect to the solvent in the electrospinning of nanofiber webs with the inclusion complexes. The inclusion complexes were prepared separately by mixing equimolar (1:1 molar ratio) amount of Vitamin C with β–CD in water [30,33]. β–CD was dissolved in distilled water at 80℃ for 5 min, cooled down to room temperature, and then Vitamin C was added. After stirring for 30 min, the inclusion complex solution was added to the polymer solution and stirred for additional 4 h at room temperature [33]. In the samples of electrospun PVA nanofiber webs, including β–CD/Vitamin C inclusion complexes, PVA concentration was used as 6% (w/v) with respect to the solvent (distilled water–acetic acid); β–CD concentration was used as 17%, 25%, and 33% (w/w) with respect to PVA, and Vitamin C concentration was used as 3%, 4%, and 5% (w/w) with respect to PVA.
All nanofiber web samples were produced on an electrospinning system with a closed chamber that was equipped with a syringe pump, a high-voltage power supply, and a collector covered by aluminum foil. Solutions were added to a 20 ml syringe with a needle (diameter of 0.8 mm). For adjusting the electrospinning parameters, the feed rate of the solution varied from 1 to 1.5 ml/h, the voltage varied from 15 to 27 kV, and the distance between the nozzle and the rotating cylinder varied from 10 cm to 19 cm in the preliminary trials [33,34]. The best results were obtained with the feed rate of 1 ml/h, voltage of 18kV, and distance of 18 cm. Besides, the distance was enough for water to evaporate to a great amount until the solution leaving the needle tip reaches to the collector covered by aluminum foil [32]. Therefore, these parameters were used in the production of all samples. The electrospinning of nanofiber webs were performed at room temperature, and they were dried at room temperature for 24 h.
Characterization
The nanofiber web sample images were captured by using SEM (Carl Zeiss EVO 40, Germany) in order to study the surface morphologies. Before taking the images, a thin layer of gold was coated by sputtering onto samples to prevent charging of the surface and damaging of the electron beam. Average diameters of the nanofibers from the samples were examined by taking measurements from 50 different regions of each. UV–Vis absorption spectra of the β–CD/Vitamin C inclusion complexes, pure β–CD, and pure Vitamin C in water were obtained by Shimadzu UV-2600 (Japan). The scans were recorded from 200 to 550 nm.
The FT-IR spectra of nanofiber webs were recorded using PerkinElmer FT-IR C99089 (USA) Spectrum. By FT-IR spectra, the bond structure and the bonding energy of the nanofiber webs were examined, and the functional groups were identified. Thus, by FT-IR technique, the presence and distribution of β–CD and inclusion complexes on the functionalized electrospun PVA samples were investigated within the wavelength range of 500–4000 cm−1.
Results and discussion
Morphology of the resulting electrospun nanofibers and SEM images
The characteristics of the solutions and the morphology of the resulting electrospun nanofibers.
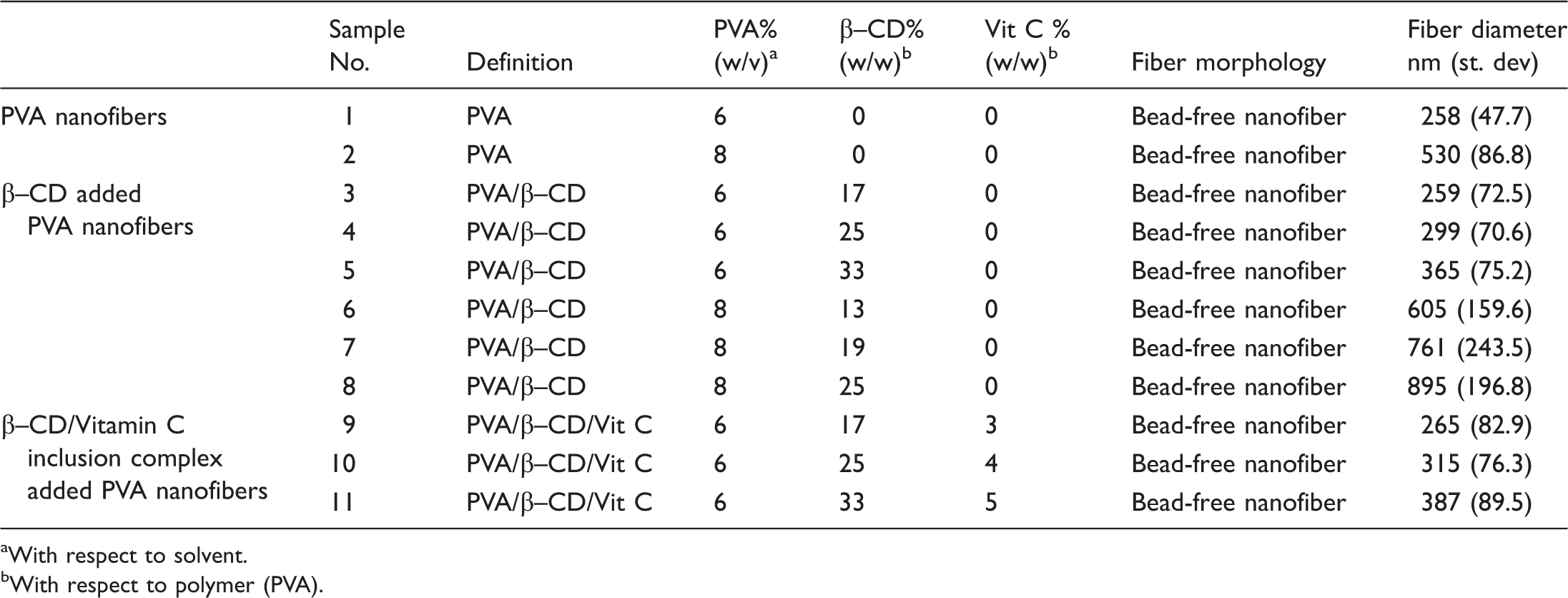
aWith respect to solvent.
bWith respect to polymer (PVA).
The analysis of the nanofiber diameter determined from the SEM images revealed that the nanofiber web samples with 6% PVA had average diameters no more than 300 nm; whereas nanofiber web samples with 8% PVA had average diameters no more than 550 nm. In addition, β–CD added PVA nanofiber web samples had the fiber diameters no more than 400 nm for 6% PVA and 900 nm for 8% PVA. When the diameters of the fibers and the uniformity of the samples were taken into consideration, these results were both reasonable and satisfactory comparing to the literature [38–41], which may be attributed to NaCl utilization in the electrospinning process [37–43]. However, as expected, average fiber diameters were found higher with the increase in the polymer concentration.
Since the average fiber diameter of the nanofiber web samples increased when the polymer (PVA) ratio was increased from 6% to 8%, 6% PVA concentration was used to produce the nanofiber web samples, including β–CD/Vitamin C inclusion complexes. Figure 1 shows the SEM images of electrospun β–CD/Vitamin C incorporated PVA nanofiber webs obtained from visually homogeneous solutions of 6% PVA with the addition of 17%, 25%, and 33% β–CD and equimolar amount of Vitamin C. It is apparent that the nanofibers were successfully produced in a uniform structure without any beads and in a continuous form. The diameters of the fibers within the nanofiber web samples with the inclusion complexes had the average diameters between 265 and 387 nm, which were produced with the parameters employed in the electrospinning process of 6% PVA. The addition of additives increased the average fiber diameter of the nanofiber web samples; which was confirmed statistically by applying t-test at the significance level of 0.05.
SEM images of nanofiber web samples. (a) 6% PVA; (b) 6% PVA/17% β–CD/3% Vit C; (c) 6% PVA/25% β–CD/4% Vit C; and (d) 6% PVA/33% β–CD/5% Vit C.
Absorption spectrum of β–CD/Vitamin C inclusion complex
The UV–Vis spectroscopy measurements of Vitamin C and β–CD/Vitamin C inclusion complex solutions in distilled water were displayed in Figure 2. As it can be seen from the spectra, the absorption of β–CD/Vitamin C inclusion complex solution was similar to Vitamin C in absorption spectrum shape (the peaks were observed at around 295 nm), but the absorbance of β–CD/Vitamin C inclusion complex was higher than that of Vitamin C alone. Therefore, these facts were the indicators of inclusion complex formation [44–47].
UV spectra of Vitamin C and β–CD/Vitamin C inclusion complex.
FT-IR analysis
FT-IR spectrum was used to generate a plot of absorbance (au) against wavenumber (cm−1) for the electrospun nanofiber web samples within the wavelength range of 450–4000 cm−1. In Figure 3, the FT-IR spectra of the pure PVA, β–CD, Vitamin C, 6%PVA FT-IR spectra of 6% PVA, β–CD, Vitamin C, 6% PVA/33% β–CD, and 6% PVA/33% β–CD/5% Vit C nanofiber web.
Corresponding stretches on pure PVA spectrum were observed as C–O stretching vibration in the region of 1000–1250 cm−1 and CH2 bonding at around 1400 cm−1. In addition, the peak at around 2900 cm−1 on PVA spectrum was observed due to CH/CH2 bonding [48]. When the FT-IR spectrum of β–CD was observed in Figures 3, the broad peak around the wavelength of 3300 cm−1 (OH stretching) and the characteristic peaks of β–CD were observed at around 1000–1200 cm−1 assigned to the coupled C−C/C−O stretching vibrations and the asymmetric stretching vibration of the C−O−C glycosidic bridge [49,50].
When the FT-IR spectrum Vitamin C was observed, C=C and C=O stretching of the five-membered heterocyclic lactone ring in Vitamin C was indicated at around 1600 cm−1 and 1700 cm−1, respectively. Besides, C-H stretching at around 3000–3500 cm−1, and various vibrational bands; which are connected with the CH2 scissoring, twisting and wagging, and the C-H deformation modes were observed in the region 1200–1500 cm-1. In particular, the weak bands at around 1300 cm−1 are attributed to CH2 scissoring. The band at around 1200 cm−1 is originated by C–O–C stretching. In addition, very strong C–O–C stretches are seen at around 1100 cm−1. In addition, the bands in the region 1000 cm−1 are probably originated by C–O–H bend. The peaks present below 900 cm−1 are linked with the C–C ring stretching and lactone ring deformation [51].
In the spectra of 6% PVA/33% β–CD and 33% β–CD/5% Vit C inclusion complex added PVA nanofiber web samples in Figure 3, C–O stretching vibration in the region of 1000–1250 cm−1 and CH2 bonding at around 1400 cm−1 were observed, as expected. Due to the existence of the characteristic peaks of β–CD at around 1000–1200 cm−1 assigned to the coupled C−C/C−O stretching vibrations and the asymmetric stretching vibration of the C−O−C glycosidic bridge, this region became sharper in the spectra of the β–CD included 6% PVA nanofiber web sample when compared to the pure PVA spectrum. Therefore, this can be stated as the indication of β–CD in this nanofiber web.
When the spectrum of the 6% PVA nanofiber web samples, including β-CD/ Vitamin C inclusion complex in Figure 3 was analyzed, the characteristic peaks of Vitamin C were not observed, since they were suppressed due to the formation of inclusion complex. In addition, any other new peak was not observed, indicating that any chemical bond was not created while the inclusion complex was formed.
Conclusions
In the present study, β–CD functionalized PVA nanofiber webs were produced by using electrospinning method. Besides, Vitamin C was added into the PVA nanofiber webs with the successful formation of β–CD/Vitamin C inclusion complexes.
As a parameter of polymer concentration, two different concentrations of PVA as 6% and 8% (w/v) were used to produce the nanofiber web samples. Since the average fiber diameter of the nanofiber web samples increased when the polymer (PVA) ratio was increased from 6% to 8%, 6% PVA concentration was determined as the appropriate polymer concentration for β–CD/Vitamin C inclusion complex included PVA nanofiber webs. The use of 50% distilled water–50% acetic acid combination as the solvent enabled complete dissolution of the polymer and to reach the conductivity levels required for the electrospinning process mentioned in the literature.
The results from the study revealed that homogenous and bead-free electrospun PVA nanofiber webs functionalized by 17%, 25%, and 33% (w/w) β–CD concentrations mixed in an equimolar amount of Vitamin C with regard to the polymer concentration can be produced. The morphology of the nanofiber webs produced in the study was observed by SEM images, and the presence of the inclusion complex in the functionalized electrospun PVA nanofiber webs was proven by using UV–Vis spectroscopy and FT-IR.
The findings from the study contributed to the literature by using different polymer/solvent combinations, CD type, and the application of different concentrations of β–CD/Vitamin C inclusion complexes. Considering the properties of Vitamin C to be a natural compound having the inflammatory and antioxidant ability and to stimulate collagen growth and reduce skin discoloration, the findings can be considered as the first step for the usage of β–CD/Vitamin C inclusion complexes for the cosmetic, personal-care products, and topical drug delivery applications.
The fundamental aims of this study were to succeed in obtaining a β–CD/Vitamin C inclusion complex, to obtain uniform nanofibers without any beads, and, finally, to functionalize PVA electrospun nanofiber webs with the incorporation of the inclusion complex to demonstrate the potential of these webs to be employed in cosmetics, personal-care products, and topical drug delivery and thus, improving the performance of the product and investigating the related effects of the properties of the solution and the parameters of electrospinning process, which will be the aim of the further studies was not been focused on. Moreover, within this respect, the future works can be established for evaluating the controlled release of the Vitamin C in-vitro and in-vivo skin distribution studies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the ITU Scientific Research Fund (ITU-BAP, Project no: 37163) for the financial support of this study.
