Abstract
Gallic acid was microencapsulated in poly-ɛ-caprolactone by the solvent evaporation method and was applied onto biofunctional textile substrates, cotton, and polyamide fabrics using a finishing process. A higher content of microspheres on polyamide was obtained due to the more hydrophobic character of polyamide. Drug release in physiological serum was carried out with treated fabrics submerged into a thermostatized vessel at semi-infinite bath conditions. The kinetic study carried out allowed the determination of the drug-delivery behavior for all systems in the medium. The results showed that the hydrophobicity and affinity of textiles and gallic acid influenced the release mechanism. For cotton, a clear Fickian diffusion was obtained; for polyamide, the diffusion was anomalous. However, no differences were found in the global mass transport. The model could address the need of the medical and health sector for assessing the theoretical amount of drug released from biofunctional textile.
Introduction
Biofunctional textiles are a current strategy to apply an active agent onto the human skin. The functions of biofunctional textiles could be similar to the functions of cosmetic products (hydration, antiaging, antioxidant, etc.) or medical treatments. Microencapsulation is a good technique to deliver different active agents from textile fabrics to the skin [1] and leads to textiles with new properties and added values. Microencapsulation is a procedure to obtain microscopic particles, which enclose solids, liquids, or even gases with thin coatings of the wall material around the substances. Two different structures can be obtained, i.e., microspheres and microcapsules. A microsphere is a solid matrix particle, whereas a microcapsule has an inner core and an outer shell. The active agent in a solid matrix particle is typically dissolved or melted in the same vessel before fabrication processing into microspheres.
When the final application of these microparticles is on the human skin, it is important to use biocompatible materials, such as biopolymers. Poly-ɛ-caprolactone (PCL) is a synthetic aliphatic linear polyester often used in medical applications [2]. PCL has a semicrystalline structure with a glass transition temperature of −60℃ and a melting point ranging between 59℃ and 64℃, depending on the crystalline structure. PCL is soluble in chloroform, dichloromethane, carbon tetrachloride, toluene, benzene, cyclohexanone, and 2-nitrochloride [3,4] at room temperature.
Generally, the active agent is encapsulated, dissolved, or entrapped in polymeric microparticles depending on the methodology used. In this work, the solvent evaporation method, i.e., o/o/w double emulsion technique was used to obtain the microspheres. In this case, the polymer is dissolved in a water immiscible solvent, such as dichloromethane, and the active agent is dissolved in an organic solution. Then, the second solution is added to the first solution (o/o). The resultant solution is then emulsified in an aqueous continuous phase (o/o/w) and then evaporated at the water/air interface [5] to obtain microspheres.
Gallic acid (GA) (3,4,5-trihydroxybenzoic acid) is a phenolic compound that mostly found in tea leaves, berries, citrus fruits, red wine (Figure 1). Apart from its strong antioxidant ability, GA has also been demonstrated to biological activities including antioxidant, antityrosinase, antimicrobial, anti-inflammatory, and anticancer activities [6,7].
Gallic acid molecule.
GA was chosen as the active agent to obtain a biofunctional textile with antioxidant properties. Antioxidants are natural agents that are used to prevent the external aggression of oxidative stress in human beings. The human organism can generate endogenous antioxidants as a defense and can also incorporate them into the diet. The route to apply different compounds is clearly through the skin. When topically applied, these exogenous antioxidants have been demonstrated to diminish the effects of free radicals by using defense mechanisms similar or complementary to those of endogenous antioxidants [8,9]. In this study, GA is used as a tracer to assess its delivery from a biofunctional textile. In previous works, the antioxidant activities of GA applied onto the skin through a textile surface were studied [10,11].
Cotton (CO) and polyamide (PA) are used in this study as biofunctional textiles because of their comfort when in contact with human beings. PA has been used in medical applications due to its excellent wear resistance, strength, toughness, elastic recovery, and other many properties [12]. CO is a natural fiber most commonly used by humans with contact dermatitis and other skin diseases, and has good behavior related to heat conduction and excellent properties related to moisture [13].
A drug-delivery system is commonly used to prolong the therapeutic dose of effectively a single dose and to eliminate or minimize the concentrations exceeding the therapeutic requirements [14]. In issues related to the release of pharmaceutical or cosmetic active agents, it is important to predict their behaviors. In this work, several experiments were done to estimate the drug release mathematical model. The objective of the controlled release is to obtain the right amount of active agent at the right time and right place [15].
To quantify the level of retention of active agents due to microencapsulation, the drug-delivery behavior must be studied. The apparent diffusion coefficient is a quantitative parameter that can reflect the whole steps of the delivery using a unique value. Therefore, from the experimental curves obtained, this parameter can be calculated assuming a short-time approach.
The main aim of this study is to evaluate the diffusion system with mathematical kinetic models for the drug release from textiles treated with a microsphere system which contains an active agent such as GA.
Experimental
Materials
The fabrics used were standard spun nylon 6.6 DuPont type 200 woven fabric (PA) (Style 361, ISO 105-F03, 124 g/m2) and plain CO fabric (Bleached and Desized Cotton Print Cloth, Style 400, ISO 105-F02, 100 g/m2). Poly(vinyl alcohol) (PVA, 87–89% hydrolyzed, MW 31,000–50,000 Da) was used as the dispersant in the microsphere preparation; PCL (MW 45,000 Da) was used as the microsphere polymer. Both PVA and PCL were supplied by Sigma-Aldrich, (Madrid, Spain). GA was supplied by Sigma-Aldrich (Madrid, Spain). For high-performance liquid chromatography with UV detector (HPLC-UV) analysis, methanol (HPLC grade), and distilled water were used. Methanol (Carlo Erba, France) was used for extractions of the active agent from textiles. Physiological saline solution Vitutia 0.9% (Sodium chloride) from Laboratorios ERN (Barcelona, Spain) was used as a medium for drug delivery.
Microsphere preparation: Double emulsion
The solvent evaporation method was used to prepare the microspheres; microemulsions (o/o/w double emulsion) were formed.
A 0.26% dispersion of GA in mono-alcohol (2% isopropanol, 98% ethanol) was added to a solution of 0.26% PCL in dichloromethane. A simple emulsion (o/o) was generated by mechanical agitation (ULTRA-TURRAX T25, IKA) for several minutes at 24,000 r/min. Afterward, this simple emulsion was added to a continuous phase consisting of 100 mL of aqueous PVA solution (2%) and was emulsified for several minutes at 24,000 r/min, resulting in a double emulsion (o/o/w). The mixture was maintained under agitation at 400 r/min (20 h) leading to solvent evaporation and consequently microsphere formation. The method used was carried out at 4℃ [16] (see Figure 2).
Schematic diagram of the microsphere preparation.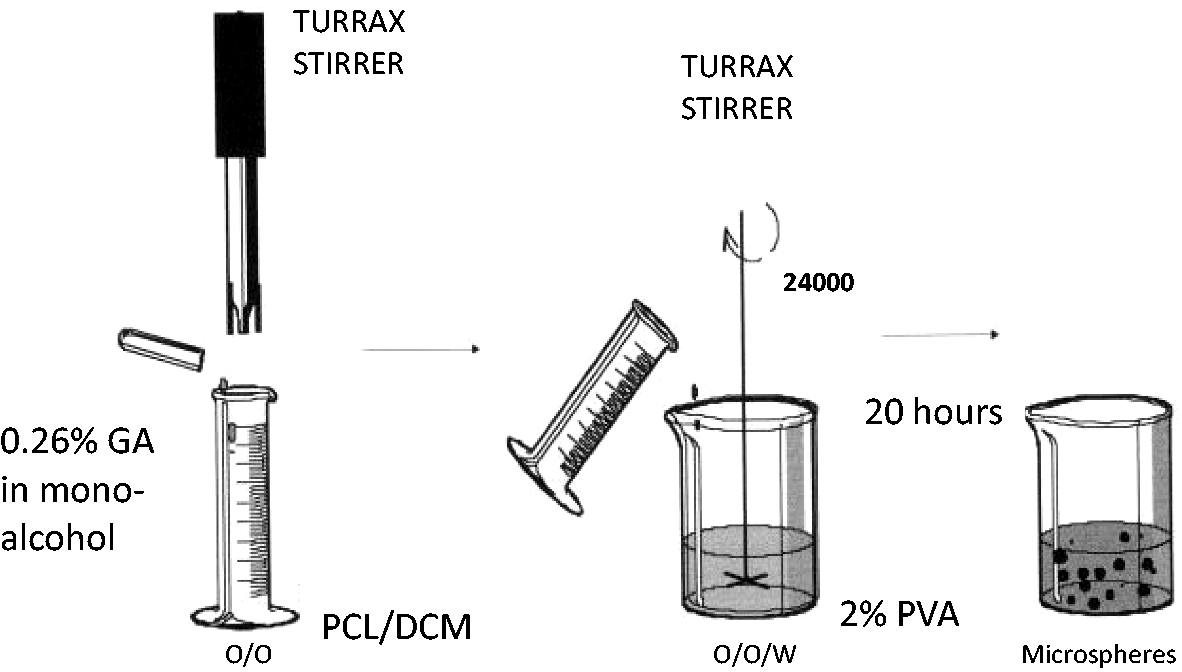
To quantify the encapsulated GA, the PCL–microsphere formulation was precipitated and separated from the supernatant by centrifugation at 14,000 r/min for 15 min using a 5415-Eppendorf centrifuge (Germany). After separation, the supernatant was retained. The initial PCL–microspheres and the supernatant were diluted in methanol and analyzed by HPLC.
The encapsulation efficacy was calculated using equation (1), it was an index of efficiency of the microspheres preparation process.
Size of PCL–microspheres
PCL–microsphere size distribution and polydispersity index were measured by dynamic light scattering (DLS) (Zetasizer Nano ZS ZEN3600; Malvern Instruments Ltd., Malvern, Worcestershire, UK). The noninvasive backscattering technology was used to minimize multiple scattering effects without the need for sample dilution. The measurement was performed at room temperature using olystyrol/polystyrene cells (Ref 67.754 Sarstedt) to determine the size distribution and polydispersity index. Detection of the light scattered was performed at an angle of 173°. Each sample was measured in triplicate. The data were interpreted by considering the size distribution by intensity. All data were collected and analyzed using the program dispersion technology software (DTS) provided by Malvern Instruments Ltd (IESMAT, Madrid, Spain).
PCL–microsphere application onto textiles
All fabric samples were conditioned under standard atmospheric pressure at 20 ± 2℃ and 65 ± 5% relative humidity (ISO 554-1976) for 24 h prior to further application. The application of PCL–microspheres onto the fabrics was performed by the foulard process twice using a Pad-Dry machine having a measured width of 30 cm (ERNST BENZ AG KLDHT and KTF/m250), with a padding pressure applied to obtain a pickup of 90 ± 5% from the microencapsulation formulations. This was followed by drying at 40℃ in a curing and heat-setting chamber, which was maintained for 10 min. The treated fabric samples were finally conditioned at 20 ± 2℃ and 65 ± 5% relative humidity for 24 h before weighing and performing subsequent experiments.
The thickness of the treated fabrics was measured using a digital micrometer 40 × MAHR (Germany).
Scanning electron microscope of biofunctional textiles
The surfaces of CO and PA fabrics treated with PCL–microspheres were visualized by scanning electron microscope (SEM; model JSM-5610, JEOL, Tokyo, Japan). Samples were coated with a thin layer of sputtered gold prior to examination.
Drug-delivery methodology
To perform the kinetic release study of GA, 4g samples of treated fabrics were submerged into a vessel with 400 mL of physiological saline at semi-infinite bath conditions. The temperature used was 37℃ in a shaking water bath GFL 1083 (Gesellschaff fur Labortechnik mbH, Burgwedel, Germany). At different times, an aliquot of bath was extracted to analyze the GA amount delivered to the bath for three days by HPLC. Each aliquot was taken in triplicate, and the experiment was done twice with each textile fabric.
To detect and quantify the GA, an HPLC equipped with a UV–vis detector was used. The column used was a LiChrocart 125-4/Lichrosorb RP-18 (5 µm) (Darmstadt, Germany). Eighty percent water /80% methanol flowing at a rate of 1 mL/min was applied as the mobile phase. The retention time was approximately 3.3 min. The area below the peak was used to calculate the concentration of GA using external standards that displayed linearity over a concentration range from 0.25 to 100 µg/mL. This experimental methodology prevented any compound from interfering with the analysis of the target substance. This analytical methodology was previously fully validated.
Results and discussion
Size (z-average), polydispersity index, and encapsulation efficiency of microspheres prepared for application onto cotton (CO) and polyamide (PA).

EE: encapsulation efficacy; PCL: poly-ɛ-caprolactone.
PCL–microspheres containing GA were found to be homogenous with a Polydispersity Index lower than 1, size of approximately 1700 nm in diameter, and encapsulation efficiency of 53%. It is important to notice the good encapsulation efficiency of the microspheres.
Finishing process: Pickup and dry product applied on textile fabrics.

CO: cotton; PA: polyamide; PCL: poly-ɛ-caprolactone; o.w.f.: over weight of fiber.
The different tendencies found for the two textiles in Table 2 (higher pickup on CO than on PA but lower dry product applied on CO than on PA) are due to the different properties of the textiles. On the one hand, pickup depends on the experimental conditions: foulard pressure and the fabric thickness, and on the other hand, the amount of dry product detected by the weight after application depends also on the affinity of PCL–microspheres toward the textiles and their moisture retention. In this case, PCL–microspheres have more affinity toward PA than toward CO.
CO is more hydrophilic than PA; thus, CO absorbed more water than PA during the finishing process, and therefore, higher pickup was obtained on CO than on PA at the same foulard pressure. The PCL polymer has a crystalline structure and is highly hydrophobic; these two characteristics could be responsible for the strong interaction between PA and PCL–microspheres, so the amount of dry product retained on PA after the drying process is higher than on CO. This behavior is similar to that found in previous studies [10,11].
The treated fabrics with PCL—microspheres were analyzed by SEM and the pictures are shown in Figure 3.
SEM micrographs of PCL–microspheres with GA. (a) Cotton fabric (×1000). (b) Polyamide fabric (×1000).
It can be seen that CO fibers allow the microspheres to be placed in corners and spaces which creates a proper fiber structure and PA accepts the microspheres between fibers. Visually, PA retains more microspheres than CO. This is in accordance with the higher amount of dry product present in the PA fabric.
The release of an active agent in a non-erodible core–shell system can show different profiles of delivery. In Figure 4, four possible theoretical curves (A, B, C, and D) show the global behaviors of the release phenomena in different situations. Curve A shows a perfect release profile. It shows a system where the rate of delivery is controlled by the diffusion of the active agent molecules through the external membrane. The rate of release depends strongly on the internal-external concentration gradient.
Theoretical release curves expected for different types of non-erodible delivery systems. (a) Membrane reservoir-type free of lag time and burst effects; (b) same as (a), with burst effects; (c) matrix or monolithic sphere with square root time release; (d) system with first-order release [17].
If there exist some molecules that are retained in the shell, then a lag-time on the release will be obtained. Then, there will be two controlling steps and diffusion will undergo a transitional intermediate state. Curve A in Figure 4 displays a system with no lag-time. When the encapsulated material migrates to the external membrane of the microcapsule, there will be a “burst-effect,” as shown by line B in Figure 4.
If the microcapsule acts as a microsphere (the entire amount of active agent is distributed in the polymer matrix), the Higuchi equation is useful up to 60% release. In this case, a plot of percentage released versus square root of time is linear, as shown by line C. First-order release is represented by curve D. The curve will be linear when the log of the percentage of core material remaining in the capsule is plotted versus time [17].
To quantify the whole phenomenon, some kinetic approaches can be done. The Korsenmeyer–Peppas equation (2) can be used to account for the coupled effects of Fickian diffusion and viscoelastic relaxation in polymer systems by including both processes

Drug delivery models based on the parameter n.

The transport in swelling systems can be described by Fick's second Law, so the diffusion of the active agent can be assumed as a plane surface for short times of liberation using the Higuchi equation for the apparent diffusion coefficient approximation (equation (3)), where D is the apparent diffusion coefficient of drug release, and δ is the width of the planar matrix [23,24].

The most widely used model to describe drug release from matrices is derived from Higuchi for a plane geometry (equation (4)), which is applicable for systems of different shapes as well.

These models have been used in pharmaceutics for the delivery from pills and from hydrogels. The only condition of substrates to be applied, are the “intervention” of water molecules in the system studied, as “initiators” of the process through the wetting and swelling effect on the substrates [21–24].
To perform the analysis of the mechanism of the drug-delivery system, the treated fabric samples were submerged into a semi-infinite bath of physiological saline.
In Figure 5, it can be seen that PA releases GA more quickly than CO, and PA reaches equilibrium before CO.
Kinetic release of GA (M) applied on textile fabrics in a bath of serum at 37 ℃.
The drug-delivery study was done by applying two types of analyses using data obtained from the drug-delivery analysis (Figure 5). First, a theoretical assessment was done to explain the mechanism of drug release, and afterward, a quantitative assessment with semiempirical equations was carried on using the values of fabric thickness and others:
– Korsenmeyer–Peppas equation was applied to determine the mechanism of drug release (n): from the plot of log Mt/M∞ versus log t, the value of n was determined from the slope of the obtained line (equation (2)). – The apparent diffusivity, D, was calculated using the approach of Fick's second law (equation (3)). – The mass transport coefficient was obtained using the Higuchi equation. By plotting Mt/M∞ versus √t, KH is obtained from the slope of the obtained line (equation (4)).
Using equation (2) on the values of the first steps (Figure 6(a)), the exponent n is obtained, which is indicative of the drug delivery mechanism (Table 4 and Figure 6(b)).
First steps of the GA delivery experiment. (a) GA delivery percentage versus time. (b) GA delivery using log for application of equation (2). (c) GA delivery percentage versus square root of time for application of Fick's second law (equations (3) and (4)). n values obtained from fitting drug release experimental data by equation (2). CO: cotton; PA: polyamide.

The CO fabric shows a very clear Fickian diffusion behavior (n ≤ 0.5); in this system, no other influence besides the absorption of aqueous media where the microspheres are dispersed in exists. When dried, the mechanism for the delivery of GA is the capability of the fiber to absorb the bath; once the fiber absorbs water, the microspheres are given the environment needed to liberate GA.
In contrast, the PA fabric has an anomalous diffusion behavior (0.5 < n < 1). In addition to the Fickian diffusion mechanism described for CO, there is an effect due to the chemical affinity of PCL toward PA and the lower absorption of water due to PA hydrophobicity which is conducive to the retention of microspheres on the surface resulting in two overlapping release mechanisms.
Even though the anomalous diffusion mechanism is detected on PA; since the n value is very close to 0.5, Fick's second Law (Figure 6(c)) could be applied to both fabrics using values from the first steps of the drug-delivery experiment (Figure 6(a)). The apparent diffusion coefficients are estimated for plane surfaces.
Estimated parameters K and D/δ 2 , measured thickness, and calculated diffusion coefficient.

CO: cotton; PA: polyamide.
To clearly describe how the system is working, it is necessary to distinguish between the affinity of microspheres (due to the external polymeric matrix) and the affinity of GA (due to its chemical structure and the hydrophilic character).
From the data obtained of dried product applied in the foulard treatment (Table 3), there is a clear affinity between PCL polymer and PA substrate. When microspheres are fixed inside the fabric, GA is liberated to its surroundings. In the case of PA, the GA liberated has no interactions with the substrate and therefore is directly liberated to the media. In contrast, the GA liberated in CO finds affinity for the substrate, and therefore, they are absorbed on the fabric. In Figure 6, it is possible to see the delay time caused by this effect during the first minutes. The molecules of GA then will be delivered to the media. This behavior can be observed also in Figure 5 at long times of delivery. Some molecules of GA tend to be reabsorbed and liberated several times in CO before reaching equilibrium. This phenomenon cannot be observed in the case of PA due to the lack of chemical affinity toward GA.
There is, in the case of CO, a transitional step that decreases slightly the rate of global delivery. This behavior is quantified by the apparent diffusion coefficients. The higher value obtained for PA confirms the lack of delay on the delivery; meanwhile, the lower value of the coefficient for CO confirms the existence of some kind of delay due to this transitional effect.
Conclusions
Microspheres with a diameter of approximately 1700 nm, good polydispersity index (<1), and more than 50% of GA encapsulated were prepared using the double emulsion method. Different amounts of dry products on textiles were obtained after foulard application due to different affinities of the microspheres toward CO and PA. Spherical microspheres were visualized by SEM analysis, and the images confirmed their size and their amount on the two fabrics. A higher content of microspheres on PA was obtained due to its more hydrophobic character.
Drug-delivery modeling showed that CO fabric had a clear Fickian diffusion because this system had no other influences other than the absorption of aqueous media in which the microcapsules were dispersed. On the other hand, the PA fabric showed an anomalous diffusion behavior; this behavior could be due to the high affinity of PCL and PA. At the same time, PA had a more hydrophobic character than GA, and both properties would lead to an overlap of two release mechanisms.
No differences were observed between the two fabrics in terms of global mass transport (KH were similar). However, the diffusion coefficient was found to be higher for PA than for CO. When the microspheres were fixed inside the fabric, GA was liberated to its surroundings. In the case of PA, the GA liberated had no interactions with the substrate, and therefore was directly liberated to the media. In contrast, the GA liberated in CO found affinity for the substrate, and therefore, it was absorbed on the fabric.
The comparison of two textiles with a drug-delivery system (GA-PCL) improved the understanding of the release mechanisms, which may provide useful insights when designing medical and cosmetic textiles.
Footnotes
Acknowledgements
The authors are indebted to the Service of DermoCosmetic Assessment (DC-IQAC) for allowing the use of its HPLC equipment.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

