Abstract
The growing interest of green technology concept in the field of biomedical application provides the thrust for the development of eco-friendly materials. Accordingly, the eco-friendly nanocomposites were prepared using in situ-generated silver nanoparticles (AgNPs) in cotton fabrics with various concentrations of aq. AgNO3 solutions (1–5 mM). All these nanocomposites were developed using Cassia alata leaf extract as a reducing agent through simple immersion process. The resulting nanocomposite cotton fabrics (NCFs) were characterized by SEM, EDX, FTIR spectroscopy, XRD and antibacterial activity tests. During and after the generation of AgNPs, the colour of the matrix changed from light brown to dark brown visually indicating the formation of NCFs. The SEM images and EDX spectra revealed the generation of spherical AgNPs in the size range of 20 nm to 119 nm. The X-ray analysis further confirmed the presence of AgNPs in the NCFs under study. These NCFs exhibited significant antibacterial activity against Escherichia coli bacteria. Further, the NCFs under study also inhibited the growth of bacteria in sewerage water. Based on the antimicrobial activity results, these NCFs can be considered for wound dressing and for biomedical applications.
Keywords
Introduction
Increasing demand for degradable materials all over the world urges the attention of scientists to search for innovative materials with custom-made properties [1]. This growing concept of green technology is already initiated almost in all the engineering applications. However, it is implemented effectively in food packaging and medical applications to the large extent [2,3]. In this regard, the development of nano-materials has fulfilled the needs of customers in several occasions by their superiority in properties like strength, physical, thermal stability, transport and antimicrobial properties [4,5]. However, the search for new materials is a dynamic process and needs to be continued until to achieve all the possible benefits. Recently, the doping of metal nanoparticles in low strength biodegradable matrices has attracted the attention of the audience through the green synthesis approach. In this direction, nanoparticles such as silver, copper, gold, TiO2 and ZnO [6–8] have been generated and studied. But still, the utilisation of silver nanoparticles (AgNPs) stands foremost due to their unique antibacterial properties against both Gram-positive and Gram-negative bacteria.
Cotton fabric is one of the cost-effective textile materials used in several applications [9,10]. However, it is extensively used for medical applications especially as an ideal dressing material for the wound healing [11,12]. Sometimes, the cotton fabrics are modified with different techniques to enhance their antibacterial properties [13–15]. In particular, the nanocomposite cotton fabrics (NCFs) coated with AgNPs were developed under different conditions [16,17]. In one of the works, the NCFs with AgNPs were prepared using glucose as a reducing agent and cetyltrimethyl ammonium bromide as a stabiliser. The results emphasised that the NCFs possessed excellent antibacterial property even after 10 washings due to the stable and disaggregated AgNPs [18]. On the other hand, chitosan-modified cotton fabric was used to generate AgNPs for obtaining the uniform antibacterial properties. However, it did exhibit antibacterial activity against Staphylococcus aureus bacteria [19]. The study further reported the effect of time and loading of precursors (i.e., AgNO3 and D-(+)-glucose monohydrate) on the antimicrobial activity through combined synthesising and coating process. From the results, they found that the factors like the concentration of an AgNO3 source solution, type of reducing agent and time of coating influenced the antibacterial performance [20].
Further, the cotton fabrics coated with AgNPs were prepared using flat printing technique. Pluchea dioscoridis leaves collected from Egypt were used as source medium for the formation of AgNPs. It produced a quick colour change in the fabric during process. Results revealed their enhanced stable antimicrobial activity and good UV-protection properties [20]. The rough surface of cotton fabrics after the generation of AgNPs transformed the fibres as hydrophobic which were a suitable characteristic required for rendering the textiles conductive and antibacterial properties [21]. A recent study reported that the nanocomposites with hybrid coating of Ag-single-wall carbon nanotubes (Ag-SWCNTs) exhibited highest antibacterial activity against Escherichia coli bacteria and S. aureus bacteria than the pure AgNPs and SWCNT-coated cotton fabric composites separately even after 20 washings [22]. The AgNPs were identified to firmly fix with cotton fabric's surface due to the curing process, using gamma-irradiation or a thermal method. These textile composites exhibited excellent antibacterial properties against both Gram-positive and Gram-negative bacteria even at a very low concentration of 1mM source solution [23]. However, the condition of the cotton fabric, methods, colourants, addition of chemicals and type of reducing agents can be varied in individual cases. But, still, work needs to be carried out with modified cotton fabrics through various media to improve their antibacterial property. Performing this can alter the parameters like adhesion characteristics of AgNPs, amount of coated particles and, size and shape, as well as better distribution and therefore this may improve the multi-functional properties of the nanocomposites.
This work was aimed to prepare the NCFs with in situ-generated AgNPs using Cassia alata leaf extract as a reducing agent. The properties of the NCFs viz., morphology, dispersion and antimicrobial activity under normal and different washing conditions were studied and reported. These NCFs with in situ-generated AgNPs are expected to achieve better antibacterial properties. Based on the results, the NCFs can be proposed as good biomedical materials for the dressing applications.
Materials and methods
Materials
The bleached cotton fabrics were purchased from local textile industry, Tamil Nadu, India. Silver nitrate was purchased from Hi-MediaLaboratories Pvt. Ltd. India. The C. alata leaves collected from the local garden were thoroughly cleaned by repeated washings with distilled water to remove dirt and soil particles before preparing the extract.
Preparation of C. alata leaf extract
The washed and scoured C. alata leaves were chopped evenly. The 10 wt.% of the chopped leaves were immersed in distilled water maintained at 80℃ for about 20 min as followed in our earlier works [9,10,19]. Then the solution was cooled to room temperature and filtered with Whatman no.1 filter paper. The filtered C. alata leaf extract was stored in a freezer until further use.
Process of diffusing C. alata leaf extract into the fabric
Initially, cotton fabrics were washed repeatedly with distilled water for the removal of starch and other sizing materials. Then the cleaned cotton fabrics were immersed in C. alata leaf extract in beakers which were placed on a magnetic stirrer operated at 180 r/min for 24 h. The extract gets absorbed into the fabric with the assistance of free –OH groups in the cellulose units of the cotton fabric. The colour of the cotton fabrics reformed after the diffusion of the leaf extract. Then the leaf extract-diffused cotton fabrics were washed with distilled water for four to five times. The leaf extract-diffused cotton fabrics were used as matrix materials in the preparation of NCFs.
The major constituents present in the C. alata leaf extract are rhein and kaempferol. The structure of these constituents and their amount (0.38% and 0.08%) were discussed by the Singh et al. [24]. It is well known that the cellulose network can act as reducing and stabilizing agent for the AgNPs. Though cellulose initiated the nucleation process, the reduction of Ag + ions is highly facilitated by the carboxylate group (–COO−) present in the flavonoid compounds (rhein and kaempferol). The electrostatic interaction between flavonoid compounds and cellulose leads to the formation of a network structure which could be highly beneficial for the stable accommodation of AgNPs. During the nanocomposite preparation process, the same concentration of leaf extract was used for all the varying concentrations of silver nitrate (1, 2, 3, 4 and 5 mM).
Generation of AgNPs inside the matrix
The leaf extract-diffused cotton fabrics (hereafter termed as matrix) were outstretched at the ends to avoid wrinkling in the fabrics. The fabric pieces were retained separately in 1, 2, 3, 4 and 5mM concentrated aqueous silver nitrate (aq. AgNO3) solutions. These solutions were exposed to sunlight for 12 h to stimulate the dissemination of silver nitrate solution in the form of AgNPs on the cotton fabrics via the in-situ process. The AgNPs-generated cotton fabrics were dried up at the room temperature. The colour of the cotton fabrics impregnated with AgNPs changed from light brown to black indicating the in-situ generation of AgNPs.
Characterization of NCFs with in situ-generated AgNPs
Fourier-transform infrared (FTIR) spectra of the matrix and NCFs were recorded using RXI Perkin Elmer FTIR spectrophotometer in the range of 4000 to 500 cm−1 with 45 scans in each case at a resolution of 4 cm−1. XRD analysis was carried out on a Bruker D8 X-ray diffractometer using a Cu Kα radiation source to investigate the crystalline behaviour of the NCFs. The surface morphology and the presence of AgNPs in NCFs were analysed with the help of Carl Zeiss EVO 18 scanning electron microscope equipped with energy-dispersive X-ray (EDX) attachment. Using the built in SmartTiff programme, the size of the AgNPs generated in the NCFs was measured and the histograms indicating the particle size distribution were plotted in each case.
Antibacterial activity
The antibacterial test was conducted against both control (matrix) and NCFs with in situ-generated AgNPs using the standard procedure indicated in the literature [20–22]. All the samples of NCFs cut in circular discs were tested against E. coli bacteria. The diameters of zone of clearance of all the NCFs were measured after 5, 10 and 15 washings to determine the consistency of antibacterial activity. In addition to that, the antibacterial study was also performed under sewage water condition. The antibacterial test procedure under stable sewage water condition is as follows: the sewage water was filtered through 250 µm for uniform particle size. The samples were cut into the circular shape of 2 cm diameter. Twenty millilitres of filtered sewage water were poured in petridishes. The NCFs were placed at the centre of the petridishes. The petridishes were incubated from 24 to 96 h. After 96 h the petridishes were analysed and the zones of clearance in each case were photographed.
Results and discussion
Appearance of the NCFs
The images of the pure cotton fabric and matrix are shown in Figure 1(a) and (b), respectively. From Figure 1(b) it is evident that the colour of the matrix was light brown due to the diffusion of leaf extract into it. The photographs of NCFs made using different concentrated aq. AgNO3 source solutions are shown in Figure 1 (c) to (g). It can be observed that for NCFs, the colour changed to dark brown which deepened with concentration of the source solutions. The change in colour preliminarily indicates the in situ-generated AgNPs in the NCFs. Further, the colour of the fabrics was uniform which visually confirms the uniform distribution of the AgNPs in the NCFs. To confirm the in situ generation of AgNPs, their size and distribution, the scanning electron microscope (SEM) analysis was carried out.
Digital images of (a) cotton fabric; (b) matrix and NCFs with in situ-generated AgNPs using 1 (c), 2 (d), 3 (e), 4 (f) and 5 mM (g) aq. AgNO3 source solutions, respectively.
Nanoparticles size and their distribution in NCFs using SEM analysis
The SEM images were recorded to visualise the AgNPs formed in the NCFs. As an example, the SEM images for the NCFs prepared with 1 and 5 mM aq. AgNO3 source solutions are presented in Figure 2(a) and (b), respectively. The corresponding EDX spectra are presented in Figure 2(c) and (d), respectively. Using the SEM images, the particle diameter (in nm) was measured using SmartTiff programme and the size distribution of AgNPs generated in NCFs prepared with 1 and 5 mM aq. AgNO3 source solutions are presented as histograms in Figure 2(e) and (f), respectively.
SEM images (a and b); EDX spectra (c and d) and particle size distribution (e and f) of the in situ-generated AgNPs in the NCFs using 1 and 5 mM aq. AgNO3 source solutions.
From Figure 2(a), it is evident that the AgNPs generated in the NCFs were roughly spherical. The EDX spectra had the peaks corresponding to the Ag confirming the formation of AgNPs. From Figure 2(e) and (f) it is evident that when 1 mM source solution was used, the particle size varied between 20 and 60 nm with an average of 45 nm. On the other hand, in the case of NCFs made using 5 mM source solution, the particles were in the higher range of 40–90 nm with an average of 64 nm. Further in the case of the NCFs made using 1 and 3 mM source solutions, most of the particles were in the range of 40 to 49 nm and 70 to 79 nm, respectively.
From these observations, it is evident that with increasing concentration of the source solution, the particle size increased. This is understandable as the concentration of the source solutions increases, more number of Ag + ions diffuse into the matrix leading to the agglomeration of some of the generated AgNPs. This argument is in good agreement with a similar observation made by Sivaranjana et al. [25]. In the case of the NCFs made using other concentrated source solutions, the average size of the AgNPs generated was in between that of nanocomposites made using source solutions of 1 and 5 mM concentrations.
FTIR analysis
The chemical interactions between the AgNPs generated and the matrix molecules were examined with FTIR spectra. The FTIR spectra of white cloth, matrix and the NCFs with in situ generated AgNPs using 1 mM to 5 mM source solutions are presented in Figure 3. The chemical compounds present in the C. alata leaf extract were already identified by Liu et al. [26]. The extract has mainly 12 compounds and among these, six flavonoids were found to be biologically active and these may be responsible for reducing AgNO3 to AgNPs. Further, the extract has oxidised compounds in keto and enol forms and the keto form is known to give dark colouration [27]. Hence, it may be the reason for the change in the colour of the fabric when the leaf extract was diffused into the white fabric. In addition to these, they also identified other compounds such as 17-hydrotetratriacontane, n-dotriacontanol, n-triacontanol, palmitic acid cerylester, stearic acid and palmitic acid.
FTIR spectra of matrix and the NCFs prepared using various concentrations of aq. AgNO3 source solution.
From Figure 3, it is evident that the intensity of most of the bands in the NCFs was higher than that of the matrix. The common prominent bands present in both the matrix and the NCFs observed at 3324 cm−1 (OH stretching)corresponds to the cellulose molecules present in cotton fabrics. Also, the peaks at 1738 cm−1 (CO of ester), 1363 cm−1 (CH bending), 1219 cm−1 (Ar–O–R) of lignin of fabric and flavanoids of leaf extract, 1103 cm−1 (C–O stretch), 1045 cm−1 (CO stretch in alcohols) and 998 cm−1 (inplane C–OC symmetric) stretching vibration were common in the matrix and the NCFs. The band at 895 cm−1 in matrix and NCFs indicates the presence of β-glucosidic linkage in the cellulose structure. In the case of NCFs, the intensity of the bands corresponding to both OH and CO was higher than that of the matrix indicating the formation of new OH and CO groups. Such new formation is possible due to the oxidation of alkane and flavonoids of leaf extract by the generated AgNPs in the NCFs. As no new bands were observed in the case of NCFs, the structural features of the matrix were retained. Hence the interaction between the matrix and the AgNPs may be due to electrostatic forces between them. Such an observation was also made in the case of cellulose/AgNP composite films made using C. alata [28], Terminalia catappa [29] and Ocimum sanctum [30].
XRD analysis
To examine the effect of AgNPs on the crystallinity of the composites, the X-ray diffractograms of the matrix and the NCFs with in situ-generated AgNPs using 1 to 5 mM aq. AgNO3 source solutions were recorded and are presented in Figure 4(a). For clarity, the diffractograms of the matrix and the NCF with in situ-generated AgNPs using 5 mM aq. AgNO3 solution are presented in Figure 4(b).
X-ray diffractograms of matrix and NCFs developed with 1 to 5 mM aq. AgNO3 solutions (a); matrix and NCF developed with 5 mM aq. AgNO3 solution (b) and diffractogram of NCF developed with 5 mM aq. AgNO3 solution expanded in 2θ = 30° to 80° range (c).
From Figure 4(a) and (b), it is evident that the intensity of the diffractogram of the matrix was higher than that of the NCFs. It indicates that AgNPs generated inside the NCFs rearranged the molecular chains of the matrix component randomly thus lowering its crystallinity. Such an observation was also made by Sadanand et al. [30], Muthulakshmi et al. [29] and Sivaranjana et al. [28] in cellulose/AgNP composite films. The main peaks observed in both matrix and the NCFs at 2θ = 14.9°, 16.4° and 22.8° belong to the reflections from the (1−10), (110) and (200) planes of cellulose I structure. Due to the higher intensity of these three peaks, the feeble peaks corresponding to AgNPs were hard to identify. To visualise them, the diffractogram of the NCF made using 5 mM aq. AgNO3 was expanded in 2θ = 30°–80° (Figure 4(c)). From Figure 4(c), it is evident that there are several peaks in the diffractogram. The prominent peaks observed at 2θ = 38°, 42.1°, 45.4°, 64° and 77.4° belong to the reflections of (111), (200), (211), (220) and (311) planes of AgNPs [29]. The other prominent peaks observed at 2θ = 27.8° and 32.1° were assigned to the reflections from (110) and (111) planes of Ag2O nanoparticles. Hence the NCFs had both AgNPs and Ag2O nanoparticles. Such a possibility cannot be ruled out as silver is a good oxidising agent and some of the AgNPs might have been oxidised to Ag2O nanoparticles.
Antibacterial activity
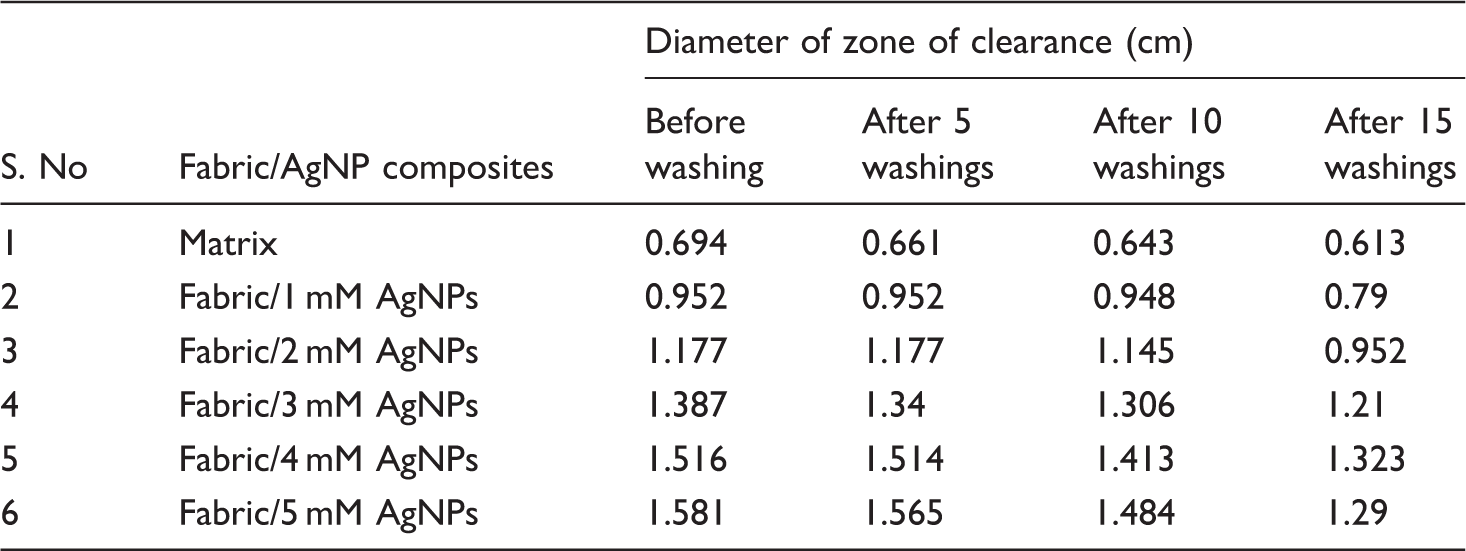
To examine the antibacterial activity of the matrix and the NCFs with in situ-generated AgNPs using different concentrated aq. AgNO3 source solutions, the antibacterial test was conducted. To prove the stability of the AgNPs generated, the test was conducted before and after 5, 10 and 15 washings. The images of the clear zones formed are shown in Figure 5(a–d). From Figure 5, it is evident that in all the cases, the matrix did not show any activity against E. coli bacteria. However, the NCFs exhibited good antibacterial activity.
Antibacterial activity against E. coli bacteria of the NCFs with in situ-generated AgNPs using Cassia alalta leaf extract as a reducing agent after different washings – before washing (a); after five washings (b); after 10 washings (c) and after 15 washings (d). The numbers indicate – 2. Matrix; 3, 4, 5, 6 and 7 the NCFs with in situ-generated AgNPs using 1, 2, 3, 4 and 5 mM aq. AgNO3 source solutions, respectively.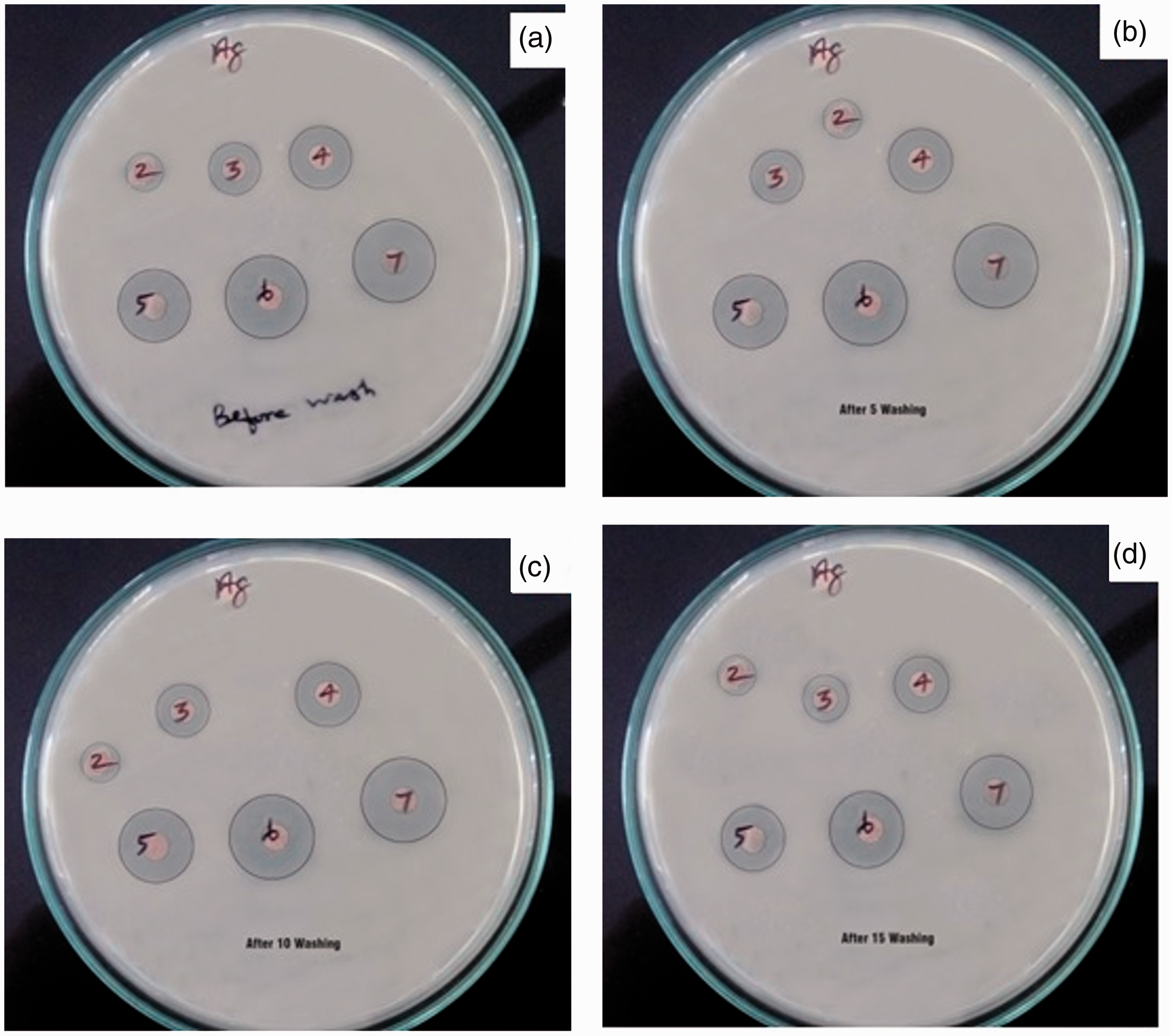
Antibacterial performance of matrix and nanocomposites against E. coli bacteria after different washings.
To probe, whether the NCFs prepared in this study can be utilised for inhibiting bacteria in the water, the antibacterial test was conducted for sewerage water. The zones of the clearance formed for the NCFs placed in sewerage water were photographed, and the digital photographs are presented in Figure 6. From Figure 6(a), it is evident that when the matrix fabric was placed in black sewerage water, no zone of the clearance was formed indicating the absence of antibacterial activity. However, when the NCFs were placed in the sewerage water, zones of clearance were formed indicating that the NCFs were able to inhibit the bacteria present in the sewerage water (Figure 6(b–f)). Further, the zone size increased with increasing concentration of the source solutions. However, the diameters of the zones of clearance for each concentration could not be measured as the zone was formed irregularly as the circular pieces of fabric specimens got distorted when placed in the sewerage water. But the images in Figure 6 visually indicate the increased size of the zone with increasing concentration of the source solutions as seen in Figure 6(b–f). Table 2 shows the measured values of irregular area of clearance zone formation using Image J software for all the nanocomposites. Our preliminary tests indicated that these low-cost nanocomposites can also be used for water treatment applications.
Antibacterial activity against sewage water of the matrix (a); NCFs with in situ-generated AgNPs using 1 (b), 2 (c), 3 (d), 4 (e) and 5 mM (f) aq. AgNO3 source solutions. Antibacterial clear zone area of matrix and nanocomposites against sewage water condition.

Comparison of the antibacterial activity against the different bacteria of NCFs prepared by different methods.

From Table 3, it is evident that though several methods were used in making NCFs, some of the routes are complicated and expensive. In the present case, we used an environmental friendly bioreduction method in generating the AgNPs in the NCFs which showed antibacterial activity against E. coli bacteria even after 15 washings. Further, the prepared NCFs inhibited the growth of bacteria even in sewerage water.
Conclusions
NCFs with in situ-generated AgNPs were prepared using 1 to 5 mM aq. AgNO3 source solutions and C. alata leaf extract in water as a reducing agent. The NCFs had roughly spherical AgNPs with size range of 20 to 119 nm as evidenced in SEM analysis. The X-ray analysis indicated the generation of both AgNPs and Ag2O nanoparticles in the NCFs. The NCFs exhibited antibacterial activity against E. coli bacteria and retained the activity even after 15 washings. The NCFs with in situ-generated AgNPs inhibited the growth of bacteria even in sewerage water. Further, the antibacterial activity of the NCFs increased with increasing concentration of the aq. AgNO3 source solutions. The NCFs can be considered for medical and water treatment applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
