Abstract
Cosmeto-textiles, which allow the administration of molecules when in contact with the skin, are increasingly being developed by cosmetic industries. We have designed an innovative approach for cosmeto-textile products, based on the impregnation of textile fibers with chitosan hydrogels, which have been cross-linked with genipin and loaded with dihydroxyacetone, which is an active component that induces sunless tanning. Dihydroxyacetone-loaded chitosan hydrogels have been prepared and characterized by means of cryogenic scanning electron microscopy (cryo-SEM). The images showed that genipin cross-linking decreases the mesh distance of hydrogels. The release of dihydroxyacetone from these cross-linked genipin chitosan hydrogels has been studied by a dialysis membrane method. These dihydroxyacetone-loaded chitosan hydrogels have been incorporated to polyamide textiles by a simple padding technique. The presence of dihydroxyacetone on these textiles has been detected by hyperspectral imaging on a dark field high resolution optical microscope. Finally, the performance of fabrics as cosmeto-textiles, with a tanning effect, has been evaluated by skin-colorimetry measured with an evaluation panel of 10 people. The results have demonstrated that dihydroxyacetone-loaded textiles produce a tanning effect on skin, and incorporation of dihydroxyacetone-loaded chitosan hydrogels into polyamide fabrics represents a friendly and appropriate strategy to obtain a cosmeto-textile with tanning effect.
Introduction
Functionalized textiles and especially cosmeto-textiles are increasingly used for cosmetic applications [1]. A cosmeto-textile is a textile article containing a substance or a preparation intended to be durably released on the skin [2]. A cosmeto-textile allows the controlled release of molecules to the outer skin layers. The amount of compound transferred has to be sufficient to ensure that cosmetic benefits are possible [3]. There are several approaches to achieve specific textile functionalization; one of them is based on the impregnation of textile fibers with hydrogels. This technique presents two major advantages: the easy encapsulation of actives, of hydrophilic nature, into the hydrogel matrix by a simple pre-mixing step, and the easy impregnation of textiles with preformed hydrogels, by using a padder machine. Polyamide fabrics were selected for this study, for being a synthetic fiber with an increasing presence in medical material and healthcare/hygiene products with a lipophilic character [4].
Chitosan-based hydrogels have attracted considerable attention for biomedical applications [5–7] due to its low toxicity, good biocompatibility, and biodegradability. Chitosan is a natural cationic polysaccharide composed of randomly distributed β(1→4)-linked N-acetylglucosamine and glucosamine. Among natural polymers, chitosan, an amino polysaccharide obtained by the deacetylation of chitin, has gained considerable attention from researchers worldwide. This polymer has been used as textile finishing conferring antibacterial properties [8,9], as adsorbent of anionic dyes [10,11] or as shrink-resist agent in wool [12,13], among other applications. In the present work, this polymer has been selected because is highly available and it has been already used in textiles.
Hydrogels are hydrophilic three-dimensional (3D) cross-linked networks of highly hydrated molecules, which can swell in the presence of water, retaining a large amount of aqueous solution within their macromolecular structure. Chitosan hydrogels can also be cross-linked to provide stability to their 3D network. Several reagents, such as glutaraldehyde, formaldehyde or epoxy compounds, have been used for cross-linking chitosan [14,15]. However, they present toxicity problems, which can be overcome by using genipin, a natural product reactive to amino groups [16]. Genipin has been used to cross-link collagen and gelatin [17,18], and also chitosan [19,20]. Genipin is a low-toxicity cross-linking agent, which is highly active on polymers with amino functional groups, such as chitosan, forming covalently cross-linked networks. Basically, the reaction mechanism goes first through a nucleophilic attack of the amino group of chitosan to a carboxymethyl group of genipin, leading to the formation of amide linkages. Subsequently, a slower second reaction is the nucleophilic substitution of the ester group of genipin by another amino group of chitosan forming a secondary amide link. This process is quite complex, since genipin can also react with itself forming active dimers and trimers [21]. Eventually, genipin forms covalent bridges between two adjacent chitosan chains. Moreover, as polyamide fiber contains amino terminal groups, genipin can form covalent unions between these terminal groups and the amino groups of chitosan. Therefore, genipin can fix chitosan hydrogels on polyamide fibers, as well as cross-linking chitosan molecules. Thus, a stronger polymer network could be incorporated into the fabric.
The physical properties of chitosan hydrogels have enabled their use in drug delivery applications, in which the active ingredients or drugs are loaded in the hydrogel matrix. The porous structure of these hydrogels can be modulated by the polymer concentration and by the degree of cross-linking. This can modify the swelling extent of the network and therefore, have a direct impact on release of active ingredients [22].
Dihydroxyacetone, DHA, a three-carbon sugar, is the most-often used sunless tanning agent and appears commercially in skin creams, lotions, conditioners, aerosol foams, sprays, and fogs [23–25]. Commercial formulations typically contain from 1 to 10 wt% of this agent. DHA can react with free amino acids derived from peptides, in skin horny layer, via the Maillard reaction resulting in brown pigments known as melanoidins [26,27]. Their formation can give the appearance of a natural skin tan without the risks associated with long-term exposure to sun radiation. In this context, textiles containing DHA may allow an increase in tanning when in contact with the skin.
The aim of this study is designing an innovative approach for skin tanning based on polyamide fabrics coated with DHA-loaded chitosan hydrogels. To this end, we have first synthesized and characterized DHA-loaded chitosan hydrogels, by means of cryogenic electron microscopy and release studies. Then, these DHA-loaded chitosan hydrogels have been successfully incorporated to polyamide fabrics, and its efficacy as a cosmeto-textile with tanning effect has been verified by an evaluation panel of 10 people.
Skin tanning by a cosmeto-textile coated with DHA-loaded chitosan hydrogels could be more comfortable and easy compared to other application methods. Possible commercial applications could be on elastic leggings, which are in homogeneous contact with skin.
Experimental
Materials
Chitosan medium molecular weight (190–310 kDa, determined by viscosity measurements) with a deacetylation degree of 85% was purchased from Sigma-Aldrich in a powder form. Genipin (crystal-like powder, reagent grade) with an average molecular weight of 226 g/mol, and purity of 98% determined by HPLC, was supplied by Challenge Bioproducts Co. (Taiwan). Polyamide fabric 6.6 and DHA (DHA) were both supplied by AITEX (Spain). Glacial acetic acid was obtained from Panreac (Spain) with a purity of 99.5%. Milli-Q deionized and filtered water and phosphate buffer solution (PBS, at pH 7.4 or 5.5) were used for preparing solutions. Chitosan, genipin, and DHA are shown in Figure 1.
Schemes showing molecular structures of (a) chitosan; (b) genipin; and (c) DHA.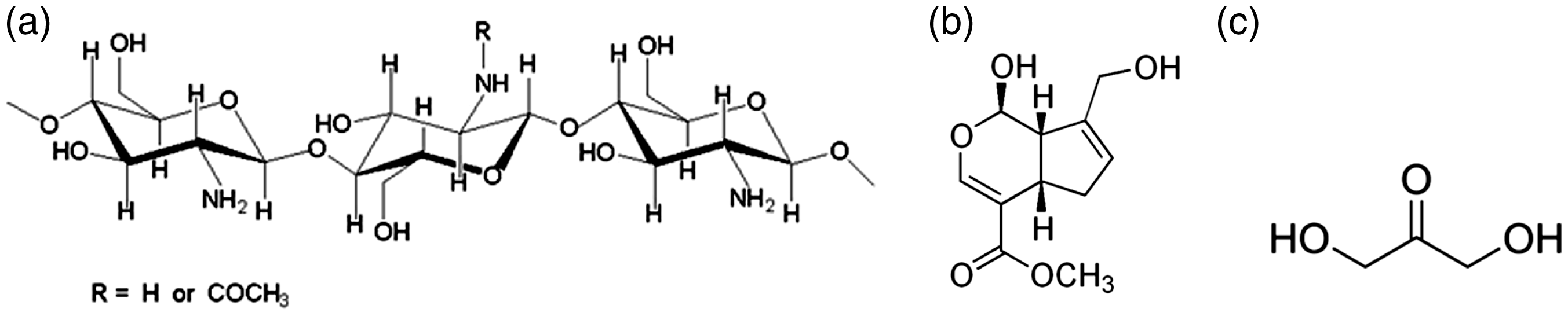
Preparation of DHA-loaded chitosan hydrogels
Chitosan solution at 1 wt% was prepared by dissolving chitosan powder in 1 vol% acetic acid by stirring overnight (magnetic stirring). The chitosan solution was then filtered, in order to remove chitosan insoluble impurities. Fresh genipin solutions were prepared by dissolving genipin powder in phosphate buffer at pH 7.4 to produce solutions with genipin loadings in the range of 0.01–0.5 wt%. This range of genipin concentrations was selected because it allowed cross-linking in a few hours of reaction time at 25℃, as described in a previous publication [28]. Cross-linking reactions of chitosan with genipin occur after mixing both solutions at 1:1 (w/w) ratio. DHA was added to the resulting pre-hydrogel solution in 3 wt%. The molecular structure of cross-linked chitosan is illustrated in the scheme shown in Figure 2.
Example of the molecular structure of three chitosan polymer chains, cross-linked by genipin bridges, which might consist of either one single genipin monomer or short gepinin oligomers (adapted from Mi et al. [29]).
Cryogenic scanning electron microscopy
Cryogenic scanning electron microscopy (cryo-SEM) was performed using a Hitachi S-3500N scanning electron microscope (Hitachi, Japan) equipped with a cryo-chamber Quorum PP3000T. Prior to imaging, the hydrogel samples were rapidly frozen in liquid nitrogen and fractured inside a cryo chamber at −130℃. After sublimation for 60 s at −100℃ in vacuum, samples were sputtered with a thin platinum layer.
DHA release studies
Diffusion experiments were carried out in a dissolution instrument (Hanson Vision Elite 8TM) set with automatic sampling (Hanson Research Autoplus Maximixer TM and MultifillTM), by using the dialysis membrane method. It consists of eight dissolution vessels with receptor solution immersed in a thermostated bath [30]. Dissolution testing is an extensively applied method which allows the release study without any kind of manipulation. Moreover, this in vitro method has been used because it is a simple method for comparing formulations and evaluating the release of DHA from hydrogels, despite the fact it does not represent in vivo conditions [31]. In the present case, a dialysis bag was used as sample holder, given the soft nature of hydrogels, even though such bags might cause a short delay in drug release.
DHA was incorporated in the pre-hydrogel matrix, namely the solution of all components before gelation, at 3 wt% concentration. An aliquot of each hydrogel (3 g) was placed in a pre-swollen dialysis bag with a 12,000–14,000 Da molecular weight cutoff (Spectra/Por dialysis membrane; Spectrum Laboratories, Auckland, NZ), and immersed into 500 ml of receptor PBS solution at pH 5.5 and temperature 32℃ (scheme shown in Figure 3). The membrane molecular weight cut off is appropriate to allow DHA release but preventing chitosan diffusion, which has a much larger molecular weight than DHA.
Scheme describing the experimental setup of release essays.
The dialysis bag was placed at the bottom of the vessel and the distance between the paddle and the sample was constant in all experiments. The receptor solution was PBS at pH 5.5 at 32℃. The stirring speed of the paddles in each dissolution vessel was 25 r/min. This slow stirring speed was considered appropriate, given the volume of the vessel (500 ml), and allowing a fast homogenization of the receptor solution. Samples were withdrawn at predetermined time intervals over an 8 h period. Concentration of DHA was calculated by measuring the absorbance at 271 nm. All absorbance determinations were carried out by using an Epoch Microplate Spectrophotometer (BioTek, USA), controlled with Gen5 Data Analysis software interface. All diffusion experiments were repeated at least twice. Although the results obtained with the dialysis bag method are not comparable to those resulting in physiological conditions, the setup is useful for comparative studies between formulations.
To investigate the kinetics of DHA release from hydrogels, Higuchi model was used for fitting the results [32]. Higuchi proposed in 1961 the first example of a mathematical model aimed to describe drug release from a matrix system.
Hydrogel application to polyamide fabrics: Nano-scale optical and spectral characterization
Chitosan hydrogels cross-linked with genipin were applied on polyamide fabrics by the padding technique. Fabrics were immersed in hydrogel solutions and run three times through squeeze rolls with an HVF laboratory padder (Mathis, Switzerland), at 3 m/min padding speed and 3 Bar of squeeze roll pressure. Finally, the fabrics impregnated with hydrogels were left to dry at room temperature. Chitosan hydrogels become attached to polyamide fibers. Polyamide contains amino terminal groups, and consequently, genipin can form covalent bridges between chitosan and polyamide fibers.
In order to verify that DHA was successfully incorporated in the textile, polyamide fabrics coated with a hydrogel with 1% chitosan, 0.1% genipin, and 3% DHA, were characterized by means of hyperspectral imaging and mapping of this compound by using an optical microscope with enhanced dark field (EDF) and a powerful illumination source (Cytoviva, Auburn, AL, USA). The hyperspectral image was obtained with a concentric imaging visible and near-infrared (VNIR) spectrophotometer (400–1000 nm) with integrated CCD camera, and is controlled by ENVI 4.8 software for hyperspectral image analysis. A reference spectral library of the active was created prior to analysis of experimental samples, and such samples were compared to that reference, evaluating the presence of DHA.
Efficacy as cosmeto-textiles: Tanning studies
DHA skin tanning effect, on a panel of 10 people, was assessed visually as well as quantified with a skin-colorimeter (CL400, Courage & Khazaha), which characterizes the surface color of an object from the colorimetric measures L*, a*, and b*, which result from the CIE tristimulus values X, Y, Z [33]. L* is referred to as the metric lightness and measures how light or dark a color is on an achromatic basis (relative to a matching shade of gray), ranging from L* = 0 for black to L* = 100 for white. Hue is a color appearance parameter defined in terms of the two chromaticity coordinates a* (a* > 0 indicates redness) and b* (b* > 0 indicating yellowness). The desire to better characterize the color as well as the lightness/darkness of the treated skin in terms of factors more relevant to human skin led to adoption of the individual typologic angle (ITAo), proposed by Chardon et al. [34] as a quantitative and objective means of describing the intensity of skin pigmentation. The ITA values reported are the average measurements of 10 people. Then, the measured L* and b* were converted to the corresponding ITA°, expressed in degrees as ITA° = 57.3 ArcTangent ((L* − 50)/b*). The change in ITA° as a result of treatment, ΔITA°, is the quantity reported and was taken in such a way that larger values signify an increase in tan intensity.
Textiles coated with hydrogels with 1% chitosan, 0.1% genipin, and 3% DHA were applied on the skin (on the forearm), and after four days, tanning was determined by the change in ITA° angle values as compared with control skin regions, which were exposed to the same textile but in absence of tanning agent.
Results and discussion
Preparation of DHA-loaded chitosan hydrogels
Hydrogels were formed by cross-linking chitosan with different genipin concentrations, in the range 0.01–0.5 wt% in weight, at 25℃. In the case of DHA-loaded hydrogels, 3 wt% respect to hydrogel total weight was incorporated. Its gelation process was followed over time (Figure 4). Initially, both non-loaded and DHA-loaded hydrogels were liquid and optically clear. For non-loaded hydrogels, after 4 h of its preparation, gelation was achieved for genipin concentrations of 0.1 wt% and higher. However, for DHA-loaded hydrogels, gelation required higher genipin concentrations, achieved at 0.25 and 0.5 wt% genipin.
Appearance of non-loaded and DHA-loaded chitosan hydrogels at different genipin concentrations, just after their preparation, and 4 and 24 h after their preparation. Vials have been turned upside down, to show the slow flowing of viscous samples.
By increasing aging time to 24 h, for non-loaded hydrogels, gelation is achieved at lower genipin concentrations, from 0.05 wt%. It also should be noted that chitosan hydrogels become dark blue, in presence of genipin. This color change is attributed to the formation of genipin derivatives produced by reactions of genipin with primary amines [21,29,35]. Therefore, color change is associated with cross-linking reactions.
In the case of DHA-loaded hydrogels, at the same aging time of 24 h, gelation is only achieved at genipin concentrations of 0.1 wt% and higher. Consequently, the presence of DHA retards gelation of hydrogels, showing a slower gelation kinetics if compared to non-loaded DHA hydrogels at the same genipin concentration. This is due to the existence of DHA-chitosan interactions, which reduces cross-linking, as described in a previous paper [36].
A Maillard reaction would be expected to occur between carbonyl groups of DHA and free amino groups of chitosan, losing a mole of water and forming an N-substituted glycosylamine, similar to that occurred between skin aminoacids and DHA. Dehydration leads to the formation of a Schiff base and then a rearrangement reaction produces a Heyns product [27] (Figure 5).
Maillard reaction between the DHA carbonyl group and a free amino group. The initial step is a condensation, and a Schiff base is formed after dehydration. Finally a Heyns product is released. (Reproduced from Carnali et al. [27] with permission from the American Chemical Society.)
Cross-linking of chitosan by genipin goes, firstly, through a nucleophilic attack of the amine group of chitosan to a carboxymethyl group of genipin, leading to the formation of amide linkages. Subsequently, a nucleophilic substitution of the ester group of genipin by the amine group of chitosan takes place, forming a secondary amide link [21]. However, when DHA is present in the system, the nucleophilic attack of the amine group of chitosan could be produced over the carbonyl group of DHA, producing an active-polymer interaction which would extend the gelation time in DHA-loaded hydrogels.
Morphology of hydrogels by cryo-SEM observation
Structure and morphology of synthesized hydrogels were examined by using a cryo-SEM [37]. Both the effects of the cross-linking density and presence of DHA were investigated. Figure 6(a–c) shows the effect of the amount of cross-linker added on the structure of the hydrogels. Hydrogels without cross-linker form a laminated structure (Figure 6(a)). By increasing genipin concentration to 0.1 wt% (Figure 6(b)) and to 1 wt% (Figure 6(c)), the mesh distance decreases. On the other hand, Figure 6(d) shows the cryo-SEM image of hydrogels at 0.1 wt% genipin concentration with 3 wt% loaded DHA. This sample shows a larger mesh distance (Figure 6(d)) in comparison with the same hydrogel composition but in absence of DHA (Figure 6(b)).
Cryo-SEM images of chitosan hydrogels without genipin (a), with 0.1 wt% genipin concentration (b), and with 1 wt% genipin concentration (c). These samples did not contain DHA. In contrast, (d) shows the cryo-SEM image at 0.1 wt% genipin concentration in presence of 3 wt% loaded DHA.
Interpretation of cryo-SEM images should be cautious, since artifacts could be confused with real morphological features. However, in the present images one can clearly observe that the characteristic distance between structures becomes shorter as genipin concentration is increased (Figure 6(a), (b) and (c)). Certainly, this is an indication of a reduction of mesh distance with genipin concentration. Moreover, the larger structures observed in the DHA-loaded chitosan hydrogel (Figure 6(d)) are in very good agreement with the slower gelation velocity shown by DHA-loaded chitosan hydrogels (Figure 4). This is due to a lower cross-linking degree, caused by weak non-covalent interactions between DHA and chitosan, as reported before [36], which was attributed to hydrogen bonds.
DHA release studies
DHA release from chitosan hydrogels was studied by the dialysis membrane method, as described in detail in the experimental section. Figure 7(a) shows the cumulative amount of DHA measured in the compartment of PBS receptor solution, as a function of time. Various donor systems, with 3 wt% DHA, were compared: (a) DHA released from an aqueous solution; (b) from a non-cross-linked chitosan hydrogel, (c) from cross-linked chitosan hydrogels with genipin cross-linker concentration of 0.1 wt%, and (d) with 0.5 wt% genipin concentration.
DHA release studies from an aqueous solution, chitosan solution, and hydrogels cross-linked with different genipin concentrations of 0.1 and 0.5 wt%. (a) Cumulative release as a function of time. The line is a visual guide, and the error bars indicate the standard deviation; (b) relative released amount, Mt/Mmax, showing the released amount Mt relative to the total amount Mmax at short times (t ≤ 3 h). The lines indicate fits to Higuchi equation. Release of non-cross-linked hydrogel cannot be distinguished from release of aqueous solution and most points overlap.
A marked reduction in release velocity was observed when genipin was added for cross-linked chitosan, in comparison with DHA in aqueous solution or non-cross-linked chitosan hydrogels. Moreover, release rate velocity decreases with cross-linker concentration. This slower DHA diffusion from cross-linked chitosan hydrogels could be attributed to a denser and more viscous network. This reduction in diffusion velocity was expected and it has also been observed elsewhere for cross-linked hydrogels [30]. Interestingly, the two samples without genipin (aqueous solution and uncross-linked hydrogel) produced almost identical results. In conclusion, the results confirm that DHA is not retained in the hydrogel and their release velocity can be controlled by tuning the degree of cross-linking.
With regards to the mechanism of DHA release, plotting the amounts of tanning agent released against the square root of time, straight lines were obtained with a rather good fit at short times (Figure 7(b)). These linear plots might indicate that DHA release in these systems is diffusion controlled, in accordance with the equation developed by Higuchi [32] for diffusion controlled release of active ingredients from semi-solid matrices. Nevertheless, it should be noted that DHA interacts with amino groups, forming an intermediate Schiff base, as described in Figure 5. Therefore, release of DHA has a very complex mechanism, which cannot be explained by Higuchi equation based on a simple diffusion process.
Results of fitting Higuchi equation (Mt/Mmax = kh t1/2) to the release of DHA from various systems at short times (t ≤ 3 h). The Higuchi kinetic parameter and the correlation factor (R2) are shown.

The results of Higuchi kinetic parameter clearly show that cross-linking slows down release kinetics. The values of this parameter decrease from 0.20 to 0.15, by increasing genipin from zero to 0.5 wt%. These values can be compared to that of DHA in simple aqueous solution in absence of both chitosan and genipin, which produces exactly the same velocity of release, with kh = 0.20.
Detection of DHA in textiles
DHA-loaded chitosan hydrogels were applied on textiles by the padding technique. In order to understand if this active component was homogenously incorporated to the textile, treated textiles were examined using the CytoViva VNIR hyperspectral imaging system coupled to high resolution darkfield microscopy. The tanning agent has a significantly different refractive index from the textile, which leads to greater scattering of light in the sections containing it, and hence, its easy detection. Figure 8(a) illustrates a spectral scan of DHA in powder form at 10X. From this image, different pixels were selected to build a spectral library (shown in Figure 8(b)), which is used to confirm the presence of DHA. Figure 8(c) corresponds to a spectral scan of the textile fibers at 10X. Finally, Figure 8(d) shows the mapping of DHA. Pixels in red map the presence of DHA spectra, illustrating that DHA was successfully and homogeneously incorporated in the textile.
(a) Spectral scan of DHA, (b) DHA spectral library, (c) spectral scan of polyamide textile, and (d) mapping of DHA in the polyamide textile.
Efficacy as a cosmeto-textiles: Tanning studies
The cosmeto-textile tanning efficacy was evaluated by assessing change in skin color. Textiles coated with DHA-loaded chitosan hydrogels were applied on the skin (forearm) of a 10 people panel, and both a qualitative observation and quantitative determination (by using a colorimeter) were assessed during a time period of four days. Regarding the qualitative observation, it was based on the opinion of the 10 panel people. After four days of the contact of the textile with the skin, six of the panellists considered that a tanning effect was produced. In this group of volunteers, four of them considered that the tanning was aesthetic, with no yellowness. Much more precise evaluation was performed by quantitatively measuring the change of skin color, by using a colorimeter applied on the skin and comparing before and after the application of DHA-loaded fabrics. The results were compared to those of skin regions that were treated with the same hydrogel-coated textiles, but in absence of DHA (control regions). After 8 h and four days, ITA° values were obtained. In Figure 9, it is shown the evolution of ITA° values for the control region and the treated region as a function of time. The lower the ITA° value obtained, the higher the tanning effect. After four days, a decrease of a 7.3% in the ITA° parameter for the treated region was observed and, hence, we could quantitative demonstrate that skin contact with the cosmeto-textile produces a tanning effect.
Quantitative ITA° values, calculated from skin-colorimetry determinations, show the evolution for the control and treated regions as a function of time.
The tanning effect was also evaluated on a volunteer panelist wearing polyamide leggings for different times, shown in Figure S1, enclosed as supporting information. Leggings were treated with a pre-hydrogel solution (1% chitosan, 0.1% genipin, 25% DHA) by the padding technique. The DHA concentration was increased to evaluate the tanning effect in a shorter period of time.
Conclusions
In this study, we have designed an innovative approach for tanning of the skin based on polyamide fabrics coated with DHA-loaded chitosan hydrogels. DHA has been successfully incorporated in chitosan hydrogels, and characterized by means of cryo-SEM. A reduction of hydrogel mesh distance was detected by increasing genipin concentration. Moreover, the release velocity of DHA from hydrogel can be controlled by tuning the degree of cross-linking. These DHA-loaded chitosan hydrogels were applied into polyamide fabrics, by using a simple padding technique and it was confirmed that DHA was successfully and homogeneously incorporated in the textile. The efficacy of this cosmeto-textile as a tanning product when in contact with the skin has been measured by skin colorimetry. The results clearly proved that DHA-loaded textiles had a tanning effect. Thus, we can conclude that the incorporation of DHA-loaded chitosan hydrogels into polyamide fabrics represents a friendly and appropriate strategy to obtain a cosmeto-textile with tanning effect.
Supplemental Material
Supplemental material for Polyamide fabric coated with a dihydroxyacetone-loaded chitosan hydrogel for a cosmeto-textile application
Supplemental Material for Polyamide fabric coated with a dihydroxyacetone-loaded chitosan hydrogel for a cosmeto-textile application by Isabel Solé, Susana Vílchez, Núria Montanyà, Maria José García-Celma, Marcela Ferrándiz and Jordi Esquena in Journal of Industrial Textiles
Footnotes
Acknowledgments
The optical microscopic observations have been performed by the Nanostructured Liquid Characterization Unit, located at the Institute of Advanced Chemistry of Catalonia (IQAC), belonging to the Spanish National Research Council (CSIC) and affiliated to the NANBIOSIS ICTS of the Biomedical Networking Center (CIBER-BBN).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CDTI (INNPACTO program), grant IPT-300000-2010-0026. S. Vílchez and J. Esquena also acknowledge the Spanish Ministry of Science, Innovation and Universities for CQT2014-52687-C3-1-P and CTQ2017-84998-P grants.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
