Abstract
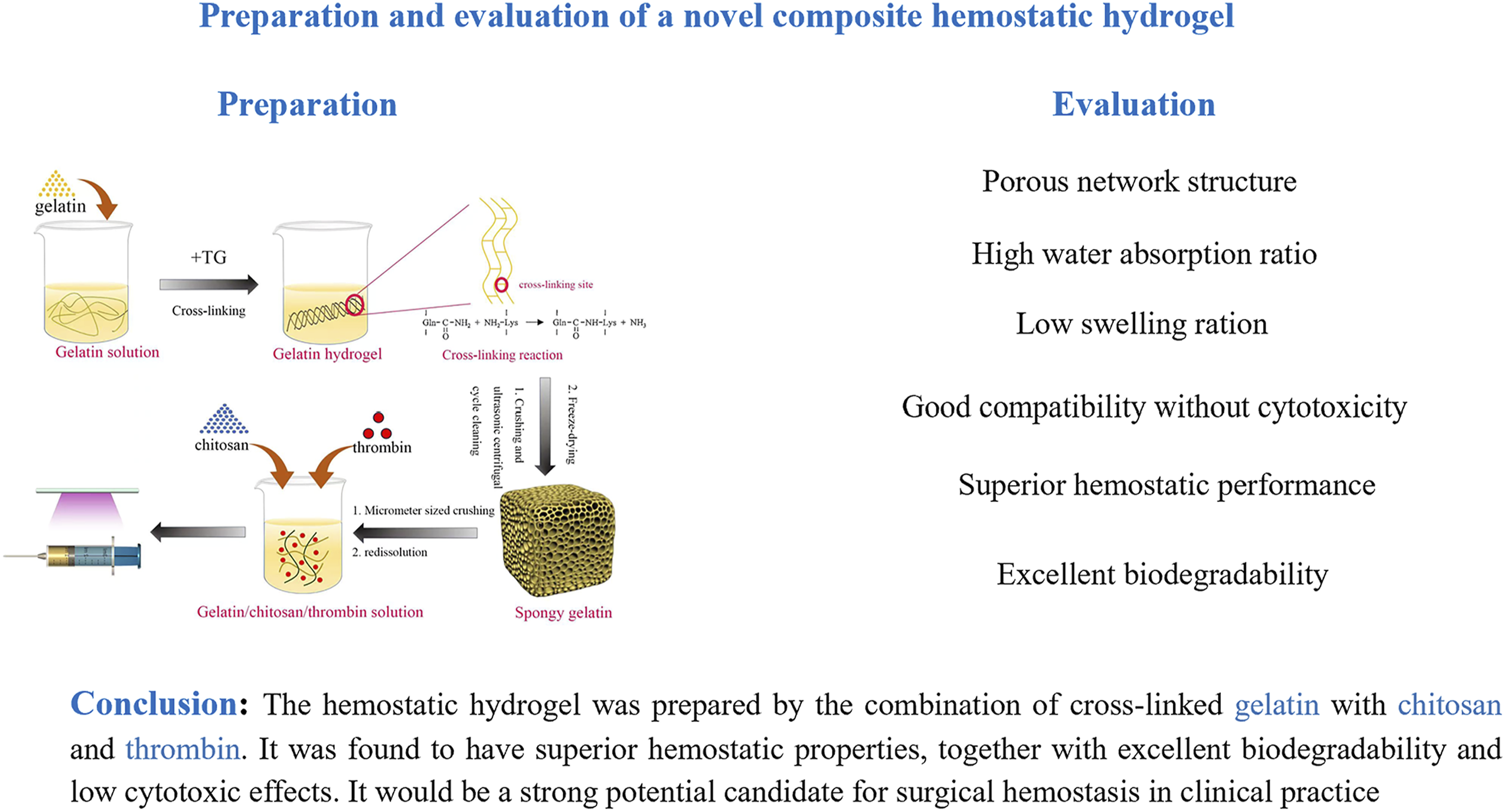
Hemostasis is critical for ensuring surgical success over the past few decades. Various topical hemostatic agents have been developed to promote hemostasis in various surgeries, particularly in cases where traditional surgical techniques are not applicable. However, the hemostatic performance of most agents is often limited by their reliance on a single component. Therefore, it is necessary to develop composite hemostatic agents that integrate multiple materials from diverse sources to enhance hemostatic efficacy. In addition, existing hemostatic agents in solid forms are not often effective in scenarios involving irregularly shaped or deep wounds, as well as endoscopic surgical procedures. In this study, a gelatin-chitosan-thrombin (GCT) composite hemostatic hydrogel was prepared using cross-linking method. The agent’s properties, including morphology, water absorption ratio, swelling ratio, and cytotoxicity were systematically evaluated. A rabbit spinal laminectomy model and a rat live injury model were used to evaluate the hemostatic efficacy of GCT agent. Histological assessment was performed to investigate its biocompatibility. The three-dimensional porous structure of the GCT agent endows it with a high absorption capacity and a low swelling ratio. The GCT agent demonstrates superior hemostatic performance in terms of blood loss and bleeding time compared to existing agents in vivo. In addition, the GCT agent exhibits excellent biodegradability and biocompatibility in vivo, and minimal hemolytic and cytotoxic effects in vitro. Therefore, the novel composite hemostatic hydrogel would be a strong candidate for surgical hemostasis especially when precise application is required.
Get full access to this article
View all access options for this article.
