Abstract
In this study, the functionalization of nanofibers by incorporating different particles and additives into the polymeric matrix for developing ultraviolet protective materials was investigated. Titanium dioxide was selected because of its known characteristic of being a widely used material for obtaining ultraviolet protection. Besides, 1,3,7-Trimethyl xanthine, which was used previously in medical applications to decrease the harm of ultraviolet radiation on DNA, was used to see its potential in obtaining ultraviolet protective nanofibers. By using these two different additives in different concentrations, functionalized polyamide 6.6 nanofibers were produced, their ultraviolet protection property and ratings were recorded, and the results were evaluated comparatively. Addition of titanium dioxide in all concentrations significantly improved ultraviolet protection of polyamide 6.6 nanofibers for both UVA- and UVB-type radiations. Moreover, ultraviolet protection factor value of polyamide 6.6 nanofibers was considerably enhanced by incorporation of 1,3,7-Trimethyl xanthine in the concentrations of 15 and 20% w/w of the polymer; which of their protection ratings were defined as “excellent protection” for both.
Keywords
Introduction
The materials that are used for textile and clothing applications can be functionalized in various ways including the application of different particles and additives by using either conventional production processes or electrospinning technology. Using electrostatic forces, electrospinning is a method that can produce polymer fibers in nano-scale diameters [1]. It is a simple and versatile method to produce nanofibers and membranes, and to demonstrate the effect of incorporating different particles and additives, which can improve the mechanical, thermal, optical, electrical characteristics of the nanofibers and provide some properties to them such as antibacterial protection, inflammability, self-cleaning ability, ultraviolet (UV) protection, etc. [2].
Investigations on the UV protection property have gained importance for quite some time due to the increase in awareness about UV radiation and its effects on human health. UV radiation, which corresponds to the 5% of solar energy reaching to the earth surface, is categorized into three groups as UVA, UVB, and UVC. Having different wavelengths, these three types of UV radiation have several impacts on human health. UVA-type radiation (315–400 nm) creates melanin on the skin and this can cause temporary redness; UVB-type radiation (280–315 nm) can cause sunburn, erythema, and further skin cancer; while UVC-type radiation (100–280 nm) is the most damaging type of UV radiation for skin and eyes; however; it is filtered by the stratospheric ozone layer and does not reach to the earth’s surface [3–5].
In an attempt to lessen the impact of the UV radiation on human health, materials used for textile and clothing applications including the conventional and nano-materials are functionalized for enhancing their UV protection. For obtaining UV protection, UV absorbing additives are applied, which can be in two different types as organic and inorganic materials. UV absorbing additives are the colorless compounds with the strong ability to absorb UV radiation between 290 and 360 nm wavelengths [6].
The most commonly used inorganic UV absorbing additives are titanium dioxide (TiO2) and zinc oxide (ZnO) [7]. TiO2exists in three forms, which are anatase, rutile, and brookite. Brookite cannot be used for UV protection practically since it is stable only at low temperature; whereas rutile can absorb the UV light and it can be transparent at visible light wavelengths; and finally anatase can be used for UV protection and it can also provide photochemical activity [8]. TiO2is a semiconductor oxide with a large band gap. When TiO2is illuminated by light with wavelength shorter than the absorption edges, the electrons absorb energy and be excited to cross the band gap creating the pairs of electrons and holes. These pairs of electrons and holes will then either combine with the other holes or the electrons providing UV protection property or will be captured and initiate reduction and oxidation reactions providing the light catalyzer property to TiO2[9].
Other than the inorganic UV absorbing additives, there are organic UV absorbing additives, which are mainly the derivatives of o-hydroxy benzophenones, o-hydroxy phenyl triazines, o-hydroxy phenyl hydrazines [6]. The organic UV absorbing additives are classified into two groups including benzophenones, anthranilates, and dibenzoylmethanes for UVA protection and PABA derivatives, salicylates, cinnamates, and camphor derivatives for UVB protection [10].
More recently, it has been realized that the use of natural bio-compounds can play an effective role in UV protection. Studies on that issue include oral consumption or topical application of these compounds in the form of a skin care product. Recent studies that were conducted for investigating the protective effect of green tea polyphenols, which has antioxidant, anti-inflammatory, and anticarcinogenic properties, through oral consumptions and topical applications against UV radiation, have revealed that they reduce UV-induced damage on DNA, oxidation, immune suppression, and tumor initiation [11–22]. It was also pointed out that the topical application of green tea and white tea extracts had comparable protective effects against UV-induced harm on human skin [22]. Similar findings were also detected with caffeinated black tea, caffeinated green tea, or 1,3,7-Trimethyl xanthine itself, which had an inhibitory effect on the formation of skin tumor multiplicity and the suppression of UV-induced tumorigenesis [23–25]. The literature survey revealed that the polyphenols, 1,3,7-Trimethyl xanthine and caffeinated compounds can be used as chemoprevention agents, whether orally or topically, against the UV-induced skin diseases.
On the other hand, the number of studies regarding the applicability and functionality of the mentioned substances within textile and clothing applications is quite limited. Although there is no study covering the scientific base, there are a few commercial materials available on the market. As an example, it is claimed that SKIN Performance S.Cafe® fabric, a commercial product that is produced from the yarns including coffee beans, provides quick drying, UV protection, and odor control [26]. In addition, coffee grounds are converted into nano-powders and incorporated into polymers for producing UV-protective eco-friendly textile yarns [27]. In fact, our preliminary trials including the measurement of UV absorption percentage of 1,3,7-Trimethyl xanthine solution showed that it can provide UV protection especially within the wavelengths around 260–320 nm as seen in Figure 1.
One of the most common types of polymer used in nanofiber production is polyamide 6.6 (PA6.6). PA6.6 has attributes such as biocompatibility as well as providing more environment-friendly process in comparison to other polymers since it can be dissolved in formic acid or formic acid/ acetic acid solution instead of DMF, which is a commonly used toxic solvent for the other polymers [28]. PA6.6 is generally preferred in many sportswear and active wear applications owing to its strength, elasticity, resistance to abrasion, and durability characteristics and its lighter weight than polyester caused by its lower specific viscosity [29–31]. Hence, electrospun membranes of this fiber functionalized by incorporation of additives can be fabricated for being used as one of the layers of the sportswear and active wear clothing system.
The purpose of this study was to functionalize electrospun PA6.6. nanofibers for developing UV protective materials and to investigate the usability of 1,3,7-Trimethyl xanthine in UV protection. To this aim, TiO2and 1,3,7-Trimethyl xanthine were individually incorporated into polymer matrix at different concentrations. The UV protection property of the nanofibers including 1,3,7-Trimethyl xanthine was assessed comparatively with nanofibers including TiO2.
Experimental part
Materials
PA6.6 (C12H26N2O4, molecular weight: 262.35 g/mol, Sigma Aldrich) was used as the polymer, TiO2(IV) anatase (molecular weight: 79.87 g/mol with the particle size approximately 325 mesh (<44 µm) and ≥ 99% purity, Sigma Aldrich) and 1,3,7-Trimethyl xanthine (C8H10N4O2, molecular weight: 194.19 g/mol, ≥ 99% purity, Sigma Aldrich) were used as the absorbers for UV protection. Acetic acid and formic acid were used as the solvents. All materials were used as received without any purification.
Electrospinning of nanofibers
PA6.6 was dissolved in acetic acid–formic acid solution (AA/FA: 1/1: v/v) at room temperature for 24 h. PA6.6 polymer solutions were prepared at the concentration of 10% w/v [28,32]. The concentrations of UV absorbing additives were selected as (10, 15, and 20% w/w of polymer) based on the results obtained in the earlier studies in the literature and preliminary trials carried out by the authors [2,33,34]. In addition, 0.02 g NaCl was added to increase the conductivity and charge density of the solutions. Because addition of salt to the electrospinning solution has potential to positively influence the morphology of the nanofibers and electrospun webs by resulting in eliminating the beads or altering their structure making them more spindle-like, decreasing the average fiber diameter, fabricating the smoother nanofibers and forming the multilayer spider network within the electrospun nanofiber webs, and enhancing their mechanical properties as already reported in the literature [35–41]. All solutions were magnetically stirred at room temperature for 24 h, and they were exposed to homogenization in the ultrasonic bath for 1 h before electrospinning.
A typical electrospinning system with a closed chamber consisting of three major components as high voltage power supply, a collector, and a needle (with a diameter of 0.8 mm) was used. Nanofibers were deposited on a very lightweight (15.4 g/m2) polyethylene/polypropylene (20/80) spunbond nonwoven covered on the rotating collector with a speed of 0.28 m/s. Although increase in the speed of the rotating collector has positive effects on the fiber alignment [42], the speed of the collector was intentionally adjusted to low level since the purpose of using rotating collector was only to distribute the fibers over a large area homogeneously and eliminate the effect of drawing on fiber uniformity. The distance between the needle tip and the rotating collector was set to 6 cm which was the optimum parameter in terms of distance demonstrated experimentally in the preliminary trials. According to these trials, also, the voltage was set to 18 kV and the feeding rate was set to 0.15 ml/h at which a stable spinning process was obtained. All of the conditions of the process parameters were kept constant during the electrospinning process.
The composition of the electrospinning solutions.

AA–FA: acetic acid–formic acid; PA6.6: polyamide 6.6.
Characterization
Morphology of produced nanofibers was observed by using a scanning electron microscope (SEM), Zeiss EVO® LS 10. Average diameter of the nanofibers from seven different samples was investigated by taking measurements from 50 different regions of each. FT-IR spectra of nanofibers were recorded by using Perkin Elmer FT-IR C99089 within the wavelength range of 500–4000 cm−1. By FT-IR spectra, the bond structure and the bonding energy of the nanofibers were examined and the functional groups were identified. Thus, by FT-IR technique, the presence of UV absorbing additive on the functionalized electrospun PA6.6 nanofibers was investigated.
Assessment of UV protection
The ability of the nanofiber samples to provide UV protection was assessed by measuring the ultraviolet protection factor (UPF) and mean UVA and mean UVB transmissions with Camspec M350 UV/Visible Spectrophotometer (SDL/ATLAS) according to Australian/New Zealand Standard AS/NZS4399:1996 [43]. Based on the UPF values, the samples were also rated according to the relevant standard [43]. The standard defines that UPF of the textile products is classified into three categories such as “good” (15–24 UPF), “very good” (25–39 UPF), and “excellent” (40–50+ UPF) [44]. Textile products with UPF value below 15 are not labeled as UV protective [45].
The mean UVA and UVB transmittances are determined as the arithmetic mean of the transmittance in the wavelengths of the UVA (315–400 nm) and UVB (290–315 nm), respectively, as shown in equations (1) and (2) [43]


UVR values are calculated as the weighted average of UVA and UVB values within the interval of 290–400 nm.
The UPF values are determined by using equation (3)[43]

Results and discussion
Morphological analysis
In Figure 2, SEM images of the nanofibers were illustrated. It was observed that nanofibers containing UV absorbing additives were successfully produced free of beads and in continuous form.
UV–visible absorbance spectra of 1,3,7-Trimethyl xanthine. SEM images of nanofibers (SEM photographs magnification: 10,000X). (a) Sample no. 1 (10% PA6.6), (b) sample no. 2 (10% PA6.6/10% TiO2), (c) sample no. 3 (10% PA6.6/15% TiO2), (d) sample no. 4 (10% PA6.6/20% TiO2), (e) sample no. 5 (10% PA6.6/10% 1,3,7-Trimethyl xanthine), (f) sample no. 6 (10% PA6.6/15% 1,3,7-Trimethyl xanthine), and (g) sample no. 7 (10% PA6.6/20% 1,3,7-Trimethyl xanthine).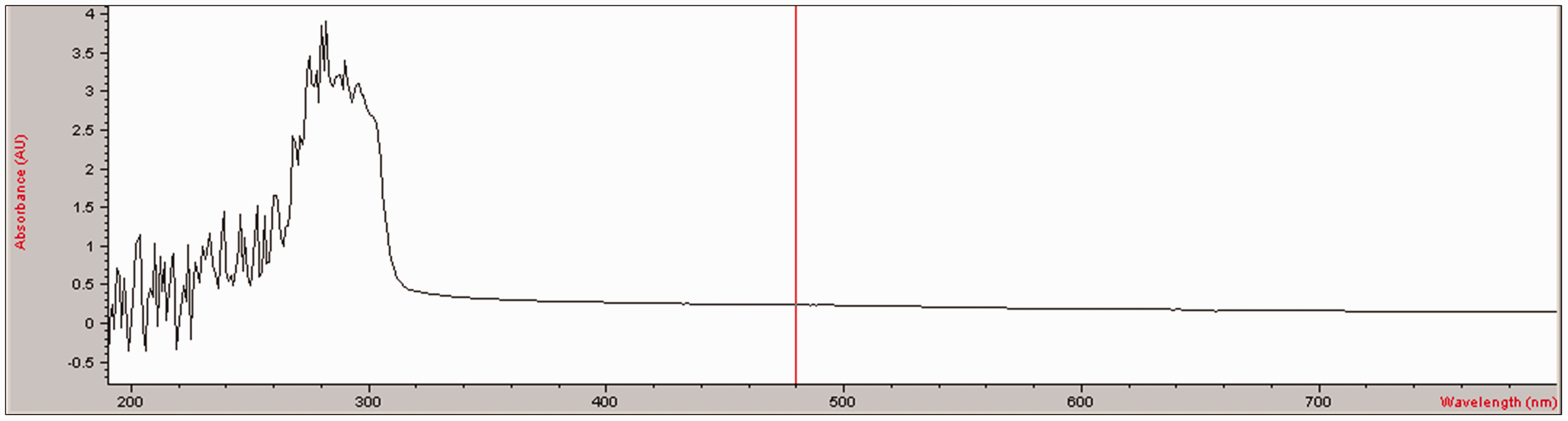

In SEM images, spider-like webs among nanofibers were observed in some parts of most of the samples including UV absorbing additives. This may be attributed to the fast evaporation of formic acid, which was caused by well-dispersed additives through the electrospinning solution and increased ionization of the polymer solution [2,46,47].
Average fiber diameter of the electrospun nanofiber samples.

PA6.6: polyamide 6.6.
FT-IR spectrum analysis
The chemical structure of PA6.6 and 1,3,7-Trimethyl xanthine are shown in Figures 3 and 4 respectively. The FT-IR spectra for PA6.6 polymer, TiO2, and 1,3,7-Trimethyl xanthine additives, as well as the spectra of electrospun PA6.6 nanofibers in the pure form (without the additives), with 1,3,7-Trimethyl xanthine and with TiO2additives in different concentrations, were recorded within the range of 500–4000 cm−1, and they are shown in Figures 5to 7.
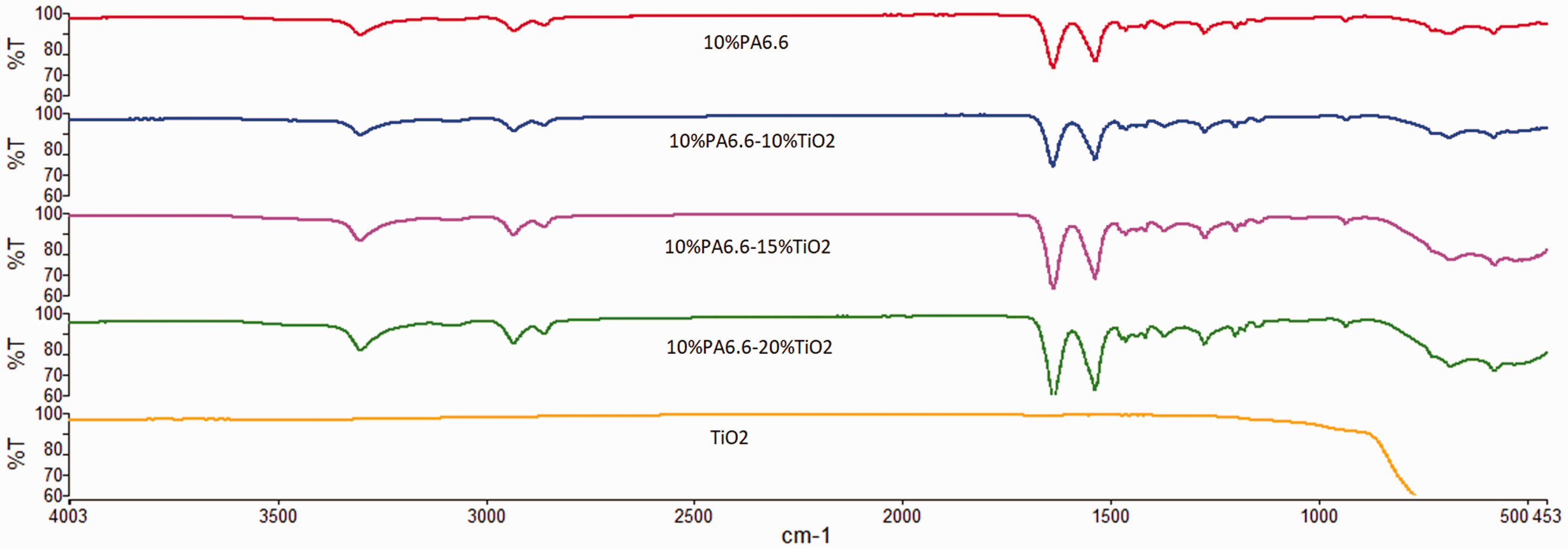
Chemical structure of PA6.6 [50]. Chemical structure of 1,3,7-Trimethyl xanthine [51]. FT-IR spectra of PA6.6 polymer, TiO2, and 1,3,7-Trimethyl xanthine. PA6.6: polyamide 6.6. FT-IR spectra of 10% PA6.6, 10% PA 6.6/10% TiO2, 10% PA 6.6/15% TiO2, and 10% PA 6.6/20% TiO2nanofibers. PA6.6: polyamide 6.6. FT-IR spectra of 10% PA6.6, 10% PA 6.6/10% 1,3,7-Trimethyl xanthine, 10% PA6.6/15% 1,3,7-Trimethyl xanthine, and 10% PA 6.6/20% 1,3,7-Trimethyl xanthine nanofibers. PA6.6: polyamide 6.6.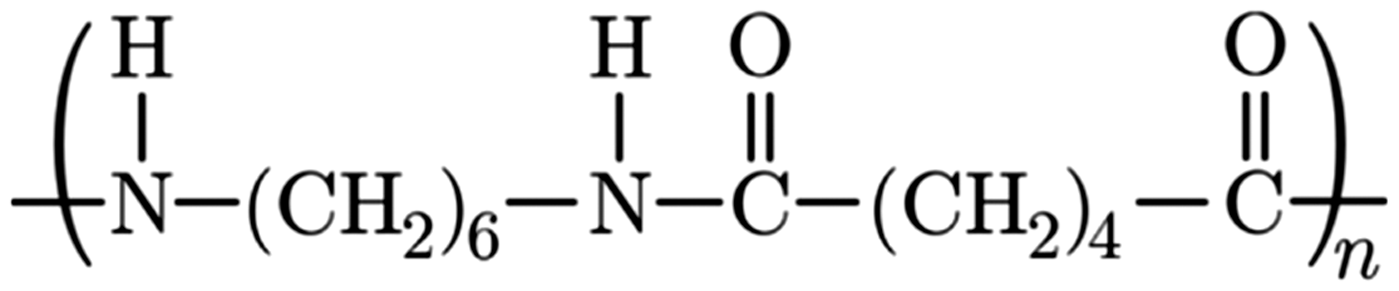



In the spectrum of PA6.6 polymer that is shown in Figure 5, the peaks regarding secondary amide groups (Figure 3) were recorded around 3300 cm−1and around 1540–1640 cm−1, which corresponded to the bending vibrations of N–H bond and the stretching vibrations of carbonyl bond C=O, respectively. The stretching and bending vibrations of C–H bonds of CH2groups were revealed around 2800 and 2900 cm−1, respectively [48]. The peaks around 1100–1400 cm−1were caused by the vibrations of C–N bonds [49].
In the chemical structure of 1,3,7-Trimethyl xanthine (Figure 4) C–H bonds, C=O bonds, which were carbonyl groups on cyclic structure, C=C and C–C bonds on cyclic structure, C–NH3and C–N bonds were existing.
The vibrations around 2800–3020 cm−1might be assigned to C–H bonds. The stretching of N–CH3bond was observed in the FT-IR spectrum of 1,3,7-Trimethyl xanthine around 920–1025 cm−1. C=O vibration bands were observed around 1567–1658 cm−1. The bands were observed in the region 580–680 cm−1due to the deformation vibrations of C–N–CH3and C=C–N bonds [52].
FT-IR spectrum of TiO2showed no specific peaks throughout the entire measurement range. A rapid decline in transmission around 800 cm−1was recognized due to the vibration of Ti–O bond [53,54].
The FT-IR spectra of 10% PA6.6, 10% PA6.6/10% TiO2, 10% PA6.6/15% TiO2, and 10% PA6.6/20% TiO2nanofibers are shown in Figure 6together for comparison. The characteristic peaks of PA 6.6 were observed in all nanofiber samples including TiO2. The peaks observed in the range of 570 and 690 cm−1, which were the vibration peaks of Ti–O bonds, are clearly visible for the nanofibers including 15 and 20% (w/w of the polymer) TiO2concentrations [53]. These showed that TiO2was successfully imparted into the electrospun PA 6.6 nanofibers for those concentrations.
The FT-IR spectra of 10% PA6.6, 10% PA 6.6/10% 1,3,7-Trimethyl xanthine, 10% PA6.6/15% 1,3,7-Trimethyl xanthine, and 10% PA 6.6/20% 1,3,7-Trimethyl xanthine nanofibers are plotted in Figure 7. With the incorporation of 1,3,7-Trimethyl xanthine into the structure, the amount of C=O bonds increased, which increased the peak intensity around 1540–1640 cm−1. Besides, the peaks around 580–680 cm−1became apparent for samples including 1,3,7-Trimethyl xanthine due to deformation vibrations of N=C–H and N–C–H bonds.
UV protection property
UPF values and UV protection rates of electrospun nanofibers.

PA6.6: polyamide 6.6; UPF: ultraviolet protection factor; UV: ultraviolet.
In Table 3, considering the mean UVA, mean UVB values of PA6.6 nanofibers in the pure form (without the additives), with 1,3,7-Trimethyl xanthine and with TiO2additives in different concentrations, it can be stated that the transmission is lower in the lower wavelengths (290–315 nm), which correspond to UVB spectrum and then it increases for the wavelengths (315–400 nm), which correspond to UVA spectrum. Being the weighted average of UVA and UVB transmissions, the UVR values were calculated as being close to UVA values.
From Table 3, it can be obviously seen that the presence of both TiO2and 1,3,7-Trimethyl xanthine in various concentrations provided a consistent reduction in the transmissions of UVA, UVB, and UVR when compared to that of PA 6.6 nanofibers which were determined as 11.10, 8.7, and 10.43%, respectively. For the samples including TiO2as the UV absorber, the mean UVA transmission decreased from 9.20 to 3.30%; the mean UVB value decreased from 2.15 to 0.40%, and the mean UVR decreased from 7.24 to 2.49%. This result was consistent with the previous findings that nanofibrous materials including TiO2could effectively provide UV protection against UVA- and UVB-type radiations. The reduction in mean UVA and UVB transmissions increased as the concentration of the TiO2in the nanofiber increased [2,55–57]. Regarding the UV protection of nanofibers containing 1,3,7-Trimethyl xanthine, the results revealed that the samples containing 1,3,7-Trimethyl xanthine resulted in the decline of both mean UVA and UVB transmissions getting the values similar to TiO2added nanofiber samples. The mean UVA value for the samples including 1,3,7-Trimethyl xanthine decreased from 8.45 to 5.45% whereas the reduction occurred from 2.65 to 1.10% and from 6.84 to 4.56% in the mean UVB value and in mean UVR value, respectively.
The mean UVA, mean UVB and UPF values of samples were together demonstrated in Figure 8. It can be seen that the mean UPF values of the nanofibers including the additives, TiO2and 1,3,7-Trimethyl xanthine, increased in parallel to the decrease in mean UVA, mean UVB, and mean UVR values.
UV protective ability of nanofibers. UPF: ultraviolet protection factor.
The UPF value of PA6.6 nanofiber was 11.7 which corresponded to UPF rating of 5. This indicated that the material had no protection against UV radiation. As was evident from the results, the increase in the UPF values of the TiO2added nanofibers was observed to be significant, especially for the sample with 20% (w/w of the polymer) concentration of TiO2.
UPF ratings for the samples including TiO2as an absorber improved from “good protection” to “excellent protection” gradually from 10 and 20% (w/w of the polymer) TiO2concentrations.Moreover, UPF value of PA6.6 nanofibers was considerably enhanced by incorporation of 1,3,7-Trimethyl xanthine in the concentrations of 15 and 20% w/w of the polymer; which of their protection ratings were defined as “excellent protection” for both. On the other hand, since the materials used in textile and clothing must have a UPF rating of at least 15 to be rated as protective against UV radiation, the nanofiber sample with 10% w/w 1,3,7-Trimethyl xanthine concentration has hardly any UV blocking property.
Conclusions
In the present study, PA6.6 nanofibers with the addition of TiO2and 1,3,7-Trimethyl xanthine were successfully electrospun at the concentrations of 10, 15, and 20% w/w of the polymer solution of 10% w/v. The study demonstrated that, as expected, nanofibers including TiO2showed good to excellent UV protection depending on its concentration. On the other hand, the nanofibers including 1,3,7-Trimethyl xanthine showed UV protection at higher concentrations.
Confirming the UV protection property of TiO2, the results revealed the improvement in the UV protection of the materials with the presence of 1,3,7-Trimethyl xanthine and they may provide a basis for the potential use of this substance for developing UV protective textile and clothing materials. In fact, TiO2electrospun nanofibers can be used as a model in protective outdoor sportswear and active wear with easy-care ability applications as a part of layered fabric system due to its UV protection and self-cleaning property [33,58–63]. Based on the findings, novel PA 6.6. electrospun nanofibers with 1,3,7-Trimethyl xanthine can be used for the same purpose as an alternative to TiO2in the applications where self-cleaning effect is not particularly required or 1,3,7-Trimethyl xanthine can also be used in combination with TiO2in the electrospun nanofibers to take the advantage of self-cleaning property of TiO2. On the other hand, the results can lead to the examination of the UV protection of the other additives including 1,3,7-Trimethyl xanthine such as tea, coffee, and coffee ground or the consideration of the use of UV absorbing additives in combination to obtain a higher performance in UV protection.
Nonetheless, further investigations are necessary to evaluate the potential of this material for the application in different textile production processes. Moreover, tensile properties of the materials developed for UV protection should be evaluated for specific uses in future works.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the ITU Scientific Research Fund (ITU-BAP, Project no: 39038) for the financial support of this study.
