Abstract
This paper reports surface hydrophilizing modification of polytetrafluoroethylene/glass fiber fabric by employing bio-inspired polydopamine functionalization. The modification process was accelerated by introducing sodium periodate (NaIO4) as oxidant. Surface morphology and chemical composition of the modified polytetrafluoroethylene/glass fiber fabric were characterized by using scanning electron microscopy, atomic force microscopy (AFM), and Fourier transform infrared spectroscopy, respectively. Hydrophilicity of the polytetrafluoroethylene/glass fiber fabric was investigated through water contact angle measurement. It was found that polydopamine successfully deposited on the surface of polytetrafluoroethylene/glass fiber fabric and the uniformity of the polydopamine coating increased with increasing modification time. Water contact angle of polytetrafluoroethylene/glass fiber fabric decreased after polydopamine modification and reached 29° when the sample was treated for 120 min, indicating an improved hydrophilic performance. The formed polydopamine coating was also demonstrated to have reliable chemical and mechanical stability.
Introduction
Polytetrafluoroethylene (PTFE) is a kind of material with excellent chemical stability, thermal resistance, high mechanical strength, and erosion resistance [1]. Due to these characteristics, it has been extensively utilized in the field of water treatment [2], separators of lithium-ion batteries [3], pervaporation [4], and blood purification [5]. Glass fiber (GF) fabric has the advantages of high strength and good thermal stability. PTFE coated glass fiber (PTFE/GF) fabric, which combines the advantages of PTFE and GF, is a new type of high performance and multipurpose composite material that are widely used in civil engineering, environment protection, chemical engineering, food processing, and so on [6]. However, the intrinsic non-polar linear molecular configuration of carbon and fluorine atoms in PTFE lead to a low surface energy of PTFE/GF fabric, which restrict their applications. Therefore, surface hydrophilic modification to PTFE/GF fabric is generally required in order to conquer this problem. Many endeavors have been made to improve surface hydrophilicity of PTFE by using various methods including chemical etching [7], plasma [8], and ion beam treatments [9]. The chemical etching method via changing surface chemistry by using a sodium naphthalene solution has been used in industry for many years [10]. However, it caused serious environmental pollution and exposed human body to a hazard chemical atmosphere. Plasma and ion beam treatments were efficacious methods to improve the wettability and adhesion of the fluoropolymer surfaces. However, sample shapes and the high cost impeded their further applications [11]. Recently, a simple, versatile, and environmental friendly surface modification method based on dopamine has been widely used in the hydrophilic modification of various materials, such as polyvinylidene fluoride [12], PTFE membrane [13], polylactide [14], and so on. Dopamine is a main component of adhesive proteins in mussel and is able to form polydopamine (PDA) coatings on various material surfaces by oxidative polymerization [15–17]. Despite the widespread interest in surface modification by taking the advantages of dopamine, the oxidation and polymerization process of dopamine (DA) normally take more than one day to form a uniform layer by using air as the oxidant [18,19]. It was demonstrated by some researchers that the reaction rate of dopamine polymerization can be greatly accelerated with the addition of strong oxidants such as ammonium persulfate, NaIO4, and copper ions [20,21]. However, there are few reports on the hydrophilic modification of PTFE/GF fabric based on rapid deposition of PDA. Furthermore, PTFE/GF fabrics are generally used in harsh conditions, they are inevitably contact with various solvents, and this makes it necessary to investigate the resistance of the deposited PDA coatings to these solvents, but research on this has rarely been reported.
In this paper, PTFE/GF fabric was modified by employing rapid dopamine deposition with the aim to improve its surface hydrophilic property. The effect of treatment time on the surface morphology, chemical composition, and hydrophilic property of the modified PTFE/GF fabric was investigated. Chemical and mechanical stability of the deposited PDA coatings on PTFE/GF fabric were also evaluated.
Experimentation
Materials
PTFE/GF fabric with a thickness of 0.2 mm was provided by Nanjing E-Thread Polymer Materials Co., Ltd, China, the fabric was composed of plain weave GF fabric impregnated with PTFE emulsion. Dopamine hydrochloride (DA·HCl), Tris (hydroxymethyl) aminomethane (Tris), NaIO4, HCl, and NaOH were purchased from Macklin, China. All the reagents were used as-received without further purification.
Sample preparation
PTFE/GF fabric was pre-treated with abrasive paper (1200 mesh) and then cleaned with alcohol followed by distilled water before modification to remove impurities on the surface and increase the surface roughness; the corresponding sample was denoted as pre-treated PTFE/GF. For surface modification, a 2.0 g/L aqueous solution of dopamine was prepared by dissolving 0.26 g DA·HCl powder in 100 mL distilled water, pH of the solution was adjusted to 8.5 by adding Tris; NaIO4solution with a concentration of 30 × 10−3M was added to the above dopamine solution to form a mixed solution of DA/NaIO4. PTFE/GF fabric with a size of 40 mm × 40 mm was immersed loosely in the above mixed solution for different time periods (40 min, 60 min, 80 min, 100 min, and 120 min). Subsequently, the samples were washed in distilled water and dried in vacuum oven at 60℃ overnight. The obtained samples were denoted as M1−40, M1−60, M1−80, M1−100, and M1−120, respectively. For comparison, PTFE/GF fabric was also treated with dopamine solution by using air as oxidant for 24 h, and the obtained sample was denoted as M2.
Characterization
The effect of NaIO4on dopamine polymerization was determined by the color change of DA solution and DA/NaIO4solution with reaction time, which was quantitatively investigated by using ultraviolet/visible (UV/Vis) spectrometer (Lambda 750, PerkinElmer, America).
The thickness of PDA coating was determined using spectroscopic ellipsometry (M-2000V, J. A. Woollam, USA). The ellipsometry measurements were performed with the wavelength ranging from 370 to 1700 nm at incident angel of 70° on five different locations for each sample and the average thickness was used.
Surface morphology of the samples was investigated by scanning electron microscopy (SEM, Phenom Pro, Netherlands) and atomic force microscopy (AFM, CSPM 5500, Being Nano, China). Surface functional groups of the PDA-modified PTFE/GF fabric were characterized by using Fourier transform infrared spectroscopy (FTIR) (Bruker TENSOR27, Germany).
To study the effect of PDA modification on surface hydrophilicity of the PTFE/GF fabric, water contact angle (WCA) was measured by using a contact angle goniometer (JY PHb, Chengde Jinhe, China). The measurement was carried out at the temperature of 25℃ and relative humidity of 40–50%. A drop of water (2.0 µL) was placed on the fabric surface using a micropipette and contact angles were measured using a circle fitting method by the drop shape analysis software. The value of WCA was measured after leaving the droplet on its surface for 10 s. Three tests were conducted for each sample and the average value of WCA was used. For dynamic WCA measurement, the water droplet was left on the PTFE/GF fabric for up to 120 s and the WCA was measured at a time interval of 10 s.
Chemical stability of the PDA coating was evaluated by immersing the surface-modified PTFE/GF fabric in different solvents including distilled water, 1 M HCl, 1 M NaOH, and 1 M ethanol, and then UV/Vis spectrum of the solvents after various immersion time periods (up to 72 h) was investigated by using an UV/Vis spectrophotometer (Lambda 750, PerkinElmer, USA).
The adhesion strength between PDA coating and the PTFE/GF fabric was investigated by utilizing an MST microscratch tester (CMS Instrument, Switzerland). The diameter of the needle used was 100 µm, the applied force was from 30 mN to 20 N, and the maximum scratch length was 4 mm.
Results and discussion
The effect of NaIO4on dopamine polymerization
The dopamine oxidation reaction results in a solution that turns from clear and transparent to dark brown and opaque as the reaction proceeds to completion [22]. Figure 1shows the UV/Vis spectrum of DA solution and DA/NaIO4solution at various reaction time periods. It can be seen that the absorbance at around 420 nm, attributed to the polymerization of dopamine [23], increased with increasing reaction time for both DA and DA/NaIO4solution. By contrast, it was found that the absorbance of DA/NaIO4was much higher than that of DA at the same reaction time, indicating a rapid oxidation reaction rate in the presence of NaIO4.
UV/Vis spectrum of DA solution (a) and DA/NaIO4solution (b) at different reaction time periods, the spectrum of DA/NaIO4was taken after the solution was diluted three times.
Surface morphology
Figure 2shows digital photos of PTFE/GF fabric before and after PDA modification, and surface micro-morphology of the corresponding sample is presented in Figure 3. It can be noted from Figure 2that the pristine and pre-treated PTFE/GF fabric was white; after PDA modification, the color of PTFE/GF fabric changed from shallow brown to dark brown as the immersion time increased. SEM images in Figure 3showed that the pristine and pre-treated PTFE/GF fabric had a porous microstructure. By contrast, micropores in PTFE/GF fabric were covered to some degree after PDA deposition. With the increase in immersion time, the formed PDA coating on the surface of PTFE/GF fabric became denser and more uniform. The surface color of M2was comparable to that of M1−100.
Digital images of pristine PTFE/GF (a), pre-treated PTFE/GF (b), PDA-modified PTFE/GF using NaIO4as oxidant with different immersion time periods: 40 min (c), 60 min (d), 80 min (e), 100 min (f), 120 min (g), PDA-modified PTFE/GF using air as oxidant for 24 h (h), and the relationship between coating thickness and treatment time (i). SEM images of pristine PTFE/GF (a), pre-treated PTFE/GF (b), PDA-modified PTFE/GF using NaIO4as oxidant with different immersion time periods: 40 min (c), 60 min (d), 80 min (e), 100 min (f), 120 min (g), and PDA-modified PTFE/GF using air as oxidant for 24 h (h).

AFM was used to analyze three-dimensional surface morphology of the PTFE/GF fabric before and after PDA modification and the resultant images are shown in Figure 4. The surface of pristine and pre-treated PTFE/GF fabrics was relatively smooth with a surface roughness ( AFM images of pristine PTFE/GF (a), pre-treated PTFE/GF (b), PDA-modified PTFE/GF using NaIO4as oxidant with different immersion time periods: 40 min (c), 60 min (d), 80 min (e), 100 min (f), 120 min (g), and PDA-modified PTFE/GF using air as oxidant for 24 h (h).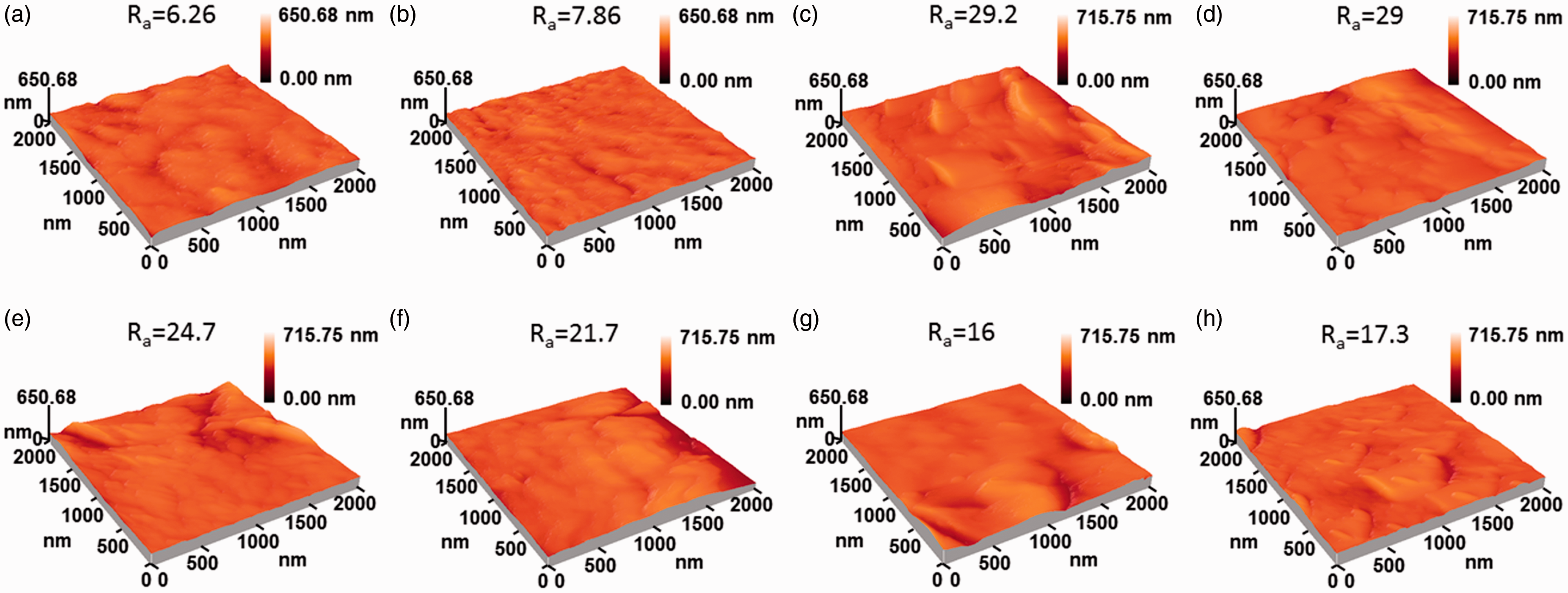
FTIR analysis
Figure 5(a)shows the FTIR spectra of PTFE/GF fabric before and after PDA modification. It can be seen that the FTIR spectrum of all samples showed the characteristic absorption peak at around 1153 cm−1and 1207 cm−1which corresponded to the CF2stretching in PTFE [24]. Compared with unmodified PTFE/GF fabric, a new peak located at 1650 cm−1appeared in the spectra of PDA-modified PTFE/GF fabrics, this peak was ascribed to aromatic C=C resonance vibrations originated from PDA structure [25]. In addition, a broad absorption between 3100 cm−1and 3600 cm−1was observed for PDA-modified PTFE/GF fabrics, and this peak was assigned to the N–H/O–H stretching vibrations in PDA [19]. This indicates that the PTFE/GF fabric has been successfully modified by PDA by using both air and NaIO4as oxidants. Figure 5(b)shows the relationship between relative intensity of the peak at 1650 cm−1and reaction time. The relative intensity of the peak at 1650 cm−1was obtained by calculating the ratio between the height of the peak at 1650 cm−1and 1207 cm−1which was the characteristic peak of PTFE. It can be found that the relative intensity of the peak at 1650 cm−1increased with prolonged reaction time, indicating that the amount of the deposited PDA increased with increasing immersion time.
FTIR spectra of pristine PTFE/GF, pre-treated PTFE/GF, PDA-modified PTFE/GF using NaIO4as oxidant with different immersion time periods and PDA-modified PTFE/GF using air as oxidant for 24 h (a) and relative intensity of the peak at 1650 cm−1versus reaction time (b).
Surface hydrophilicity
The influence of PDA modification on the hydrophilicity of the PTFE/GF fabric was characterized by WCA and the results are shown in Figure 6. It can be seen that the WCA of pristine and pre-treated PTFE/GF fabric was about 117°, indicating a hydrophobic property of the materials and this was due to the intrinsic small surface free energy of PTFE. After being modified by PDA, WCA of the PTFE/GF fabric decreased drastically due to the presence of many functional groups, such as the carboxy, amino, imine, and phenol groups in the chemical structure of PDA [26]. For PDA-modified PTFE/GF fabric by using air as oxidant for 24 h, the WCA was 55°. For PDA-modified PTFE/GF fabric by using NaIO4as oxidant, the WCA reached 57° at an immersion time of 40 min, and further decreased with increasing immersion time. The lowest WCA was about 29° after modified with the rapid method for 120 min. The results of dynamic WCA are shown in Figure 6(c). It can be noted that the WCA of pristine and pre-treated PTFE/GF fabric was almost unchanged with prolonged time. Whereas in contrast, the WCA of PDA-modified PTFE/GF fabric showed a decrease with increasing time, and the change of WCA was more evident for samples treated with longer times. These results also confirmed that the hydrophilicity of PTFE/GF fabric was improved after PDA modification.
Typical WCA images (a), WCA versus modification time for PTFE/GF fabric (b), and the change of WCA with time (c).
Coating stability
Coating stability is one of the most important features for PDA-modified surface in practical applications. To investigate the chemical stability of the PDA coating, PDA-modified PTFE/GF fabrics were immersed in distilled water, strong acid (1 M HCl), strong alkali (1 M NaOH), and reducing agent (1 M ethanol) solutions, and UV/Vis spectrum of the solution after different immersion time periods was taken. The absorbance at 420 nm, which is the characteristic peak of PDA, after different immersion time periods is shown in Figure 7. The image at 0 min in Figure 7(a)was taken after the sample was immersed into the solution and then taken out immediately. Correspondingly, the initial point of the curves in Figure 7(b) to (e)was the absorbance of the solution after the sample was immersed in and then taken out immediately. The results were compared between M1−120 and M2. It can be seen that, for both M1−120 and M2, the absorbance increased slightly with the increase in immersion time for all solvents, but the increase was most significant in the first 24 h and became slower afterwards. This indicates that some of the PDA coatings were damaged with prolonged immersion time. However, the increase in absorbance was lower for M1−120 than that of M2, suggesting a better chemical stability of PDA coating formed by using NaIO4as oxidant. In addition, it can be noted from Figure 7that the increase in absorbance was most significant for alkaline aqueous solution. This was in accordance with previous findings and can be ascribed to the inherent decomposition of PDA in strong alkaline [21].
The color change of different solvents with increasing immersion time of PDA-modified PTFE/GF fabric (a) and the relationship between UV/Vis absorbance at 420 nm with immersion time for different solvents: deionized water (b), 1 M HCl (c), 1 M NaOH (d), and 1 M ethanol (e).
Figure 8shows typical curves of the change in acoustic emission with force and scratch length, which was used to evaluate the coating strength of M1−120 and M2. It can be noted that the acoustic emission was low and remained unchanged until the force reached a critical value, then fluctuated with increasing force. The critical value of the force at which point the acoustic emission reached a maximum at the first time can be used as an indication of the initial damage of the surface coating. It was found that the critical force was 3.16 ± 0.06 N for M1−120 and 2.35 ± 0.08 N for M2, indicating a better adhesion strength between PDA coating and the PTFE/GF fabric for M1−120.
Acoustic emission as a function of force and scratch length for M1−120 (a) and M2(b).
For mechanical stability evaluation, the fabric was firstly subjected to ultrasonic treatment (with a power of 2500 W and frequency of 28 kHz) for 1 min, three times and then WCA of the sample was measured as shown in Figure 9. It was found that the ultrasonic treatment had an insignificant influence on the WCA of PDA-modified PTFE/GF fabric, indicating a good mechanical stability.
WCA of PDA-modified PTFE/GF fabric after three times of ultrasonic treatment.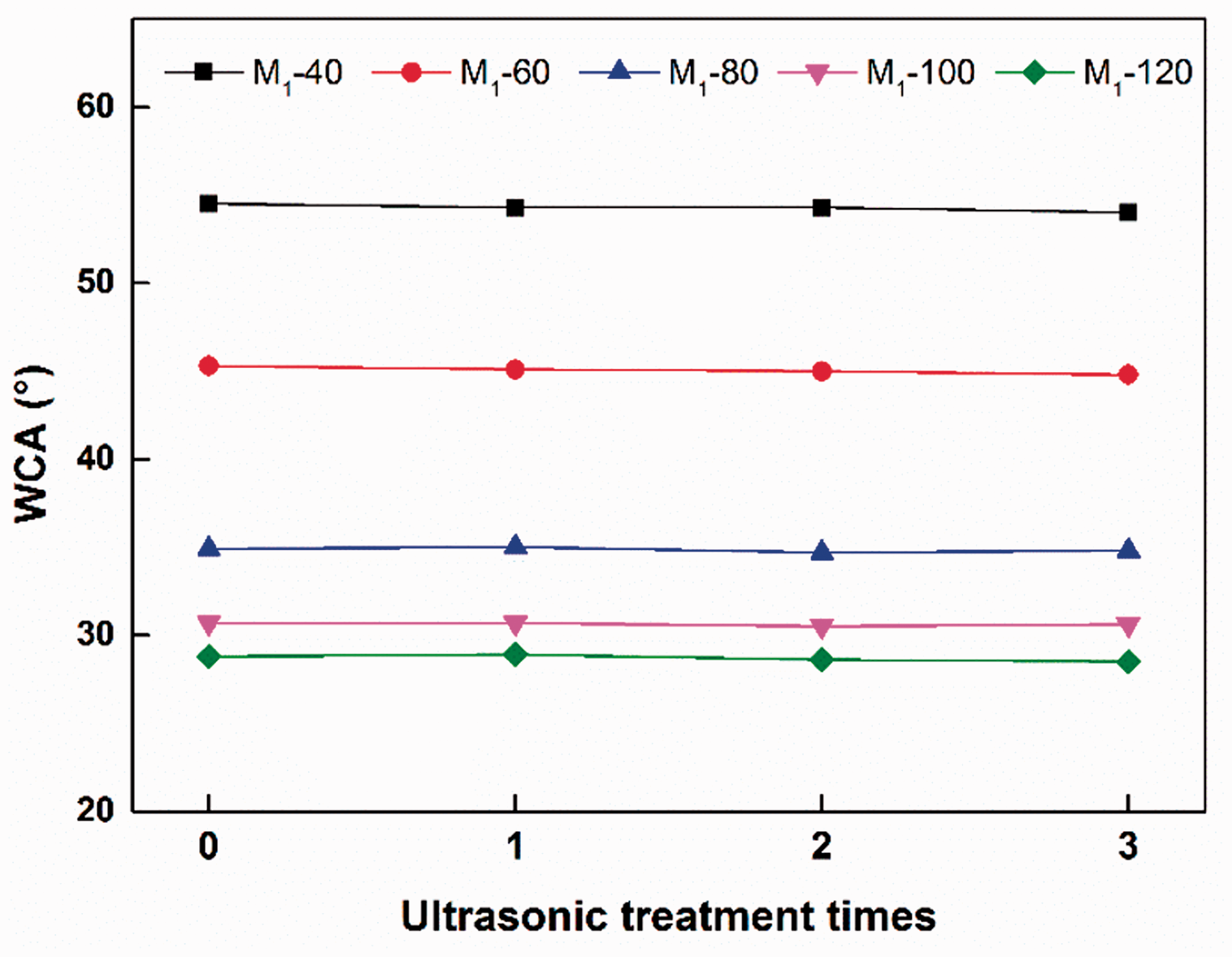
Conclusions
PTFE/GF fabric was modified by employing rapid PDA functionalization in order to improve its surface hydrophilicity. Compared with traditional dopamine modification using air as oxidant, it was found that the oxidation reaction rate of dopamine was greatly accelerated by introducing NaIO4as oxidant. The hydrophilicity of PTFE/GF fabric was greatly improved after PDA modification with the WCA reduced from 117° to 29° after a modification time of 120 min. Moreover, it was demonstrated that the formed PDA coating on the PTFE/GF fabric surface had a good chemical and mechanical stability. Combing good hydrophilicity and coating stability, the PDA-modified PTFE/GF fabric would be a good candidate for the application in oil/water separations.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to thank the financial support from “The National Key Research and Development Program of China” (Grant Number 2017YFB0309805-2) and “The Key Research and Development Program of Shandong Province” (Grant Number 2018GGX108003).
