Abstract
In this paper, a flame-retardant blend yarn was designed with flame-retardant viscose fiber and polyester. Flame-retardant viscose fiber was blended with polyester by core-spun method. Polyester was set as a core and the flame-retardant viscose fiber was set as a sheath to change the flame retardancy and disadvantage of moisture permeability. After the flame-retardant viscose fiber was spun into roving, polyester was added in spun yarn. The core yarn samples with different ratios were prepared by adjusting the amount of flame-retardant viscose fiber. The flame retardancy of core-spun yarn was evaluated by limiting oxygen index, scanning electron microscope, thermogravimetric analysis, and thermogravimetric/Fourier transform infrared techniques. Limiting oxygen index of the yarn with 50% flame-retardant viscose fiber and 50% polyester was 27.6% and just decreased slightly to 27.1% after 30 washing cycles. Polyester begins to melt from inside core-spun yarn after heating and the molten polyester flows to outer layer of yarns by the diversion effect of higher temperature. Then it was carbonized by the polymetaphosphate which was generated by the flame-retardant viscose fiber. In thermal processes, the major product of thermal decomposition was CO2.
Introduction
Polyester–viscose blended yarns were widely recognized in the market for its excellent complementary properties. It not only maintains the firmness, wrinkle resistance, dimensional stability, and wearability of polyester, but also improved the permeability of the fabric and reduced fabric pilling and antistatic phenomenon by blending with viscose fiber (VF).
PET stands for polyethylene terephthalate, which is a cost-efficient thermoplastic material with beneficial properties and easy processability, and is part of the polyester family, widely used in various industries [1]. PET fiber although of excellent durability can be affected by hydrolysis, aminolysis, and various kinds of radiation. The flammability of PET is a major drawback, which limits its applications in some important fields, such as special clothing and electrical materials. Many researches in this direction have been conducted by the polyester producers and textile research institutes. Despite the presence of benzene groups in the main polymer chain, thermoplastic polyesters show very limited tendency to char, but instead, aromatic-containing polymer fragments volatilize and feed the flame. To overcome this limitation, many approaches have been tested to improve its flame retardancy. Fire-retardant additives, although they usually facilitate decomposition of the polyesters at lower temperature, also usually promote charring and therefore suppress combustion [2]. In doing so, however, the thermal stability and mechanical properties are generally compromised [3–5].
Phosphazene derivatives are effective flame retardants due to the high level of phosphorus and nitrogen elements [6–10]. Phosphazene derivatives are a class of nitrogen–phosphorus-based linear polymers or small cyclic compounds with alternating single and double bonds. Phosphazenes can be used as special rubbers, low-temperature elastomers, flame-retardant materials, etc. due to their excellent performances such as low- and high-temperature resistance, nonflammability. These derivatives are widely used in polymers as flame retardant [11]. Therefore, the phosphazene derivatives may be used in PET by selecting various functional groups. Chlorine-containing phosphazene derivatives (chlorocyclophosphazene, general structural formula: [N=PCl2]n) have been extensively studied for many years [6]. Phosphazene derivatives have excellent biocompatibility and they can be degraded to nontoxic small molecules easily. The chlorine atom in chlorine-containing phosphazene derivatives can be substituted by nucleophiles such as amines. Hexachlorocyclotriphosphazene (HCCTP) is a chlorine-containing phosphazene derivative which possesses excellent flame-retardant properties [7], but the thermal decomposition temperature of HCCTP is too low to melt spinning. Some phosphazene derivatives with better performance have been prepared using different nucleophiles and HCCTP [8–10]. However, when the phosphazene derivatives were used in PET fibers through blended melt spinning, it caused serious damage to mechanical properties and the antidripping action has not changed.
In previous publication [12], phosphazene derivatives were used as flame retardant to product flame-retardant viscose fiber (FRVF), the flame retardancy of VF was greatly improved, and the typical intumescent flame-retardant mechanism was shown.
Therefore, we design a flame-retardant blend yarn with FRVF (the phosphazene derivatives were used as flame retardant) and PET to make up for gaps in the market. In this study, PET was blended with FRVF by the core-spun method to develop a flame-retardant fabric. PET fiber filament was set as a core, and the FRVF was set as a sheath to overcome the disadvantage of moisture permeability and flammability. The flame retardancy was evaluated through limiting oxygen index (LOI) and 45-degree ignition test. Thermal stability and decomposition reaction of the yarn were evaluated through thermogravimetric analysis (TGA) and scanning electron microscope (SEM). Real-time quantitative analyses of volatile products generated during thermal oxidation of fibers were evaluated through thermogravimetric/Fourier transform infrared spectroscopic analyses (TG-FTIR) technique. The three-dimensional infrared (IR) spectroscopy spectrum was given to have an in-depth study of thermal decomposition mechanism.
Experimental
Materials and sample preparation
The PET fiber (from Sinopec Yizheng Chemical fiber Co, Ltd, P.R. China, finess: 145 dtex, tenacity: 2.8 CN/dtex, elongation at break: 56.8%) was blended with FRVF (made by our lab, finess: 1.5 dtex, tenacity: 1.8 CN/dtex, elongation at break: 22.9%) by core-spun method. Ring spinning method was used to spinning. PET fiber filament was used as a core, and the FRVF was used as a sheath. After the FRVF was spun into roving, PET fiber filament was added in spun yarn. The core yarn samples were prepared with different weight ratios by adjusting the amount of FRVF. Final samples labeled as CSY-1 (PET:FRVF=60:40), CSY-2 (PET: FRVF=55:45), CSY-3 (PET:FRVF=50:50), CSY-4 (PET:FRVF=40:60), CSY-5 (PET:FRVF=30:70), respectively. The specific spinning process is shown in Figure 1.
The preparation process flow sheet of core yarn. FRVF: flame-retardant viscose fiber; PET: polyethylene terephthalate.
Characterization of prepared sample
The LOI tests of the samples were carried out by a M606B digital LOI meter (Qingdao Shanfang, P.R. China) according to the ASTM D2863-17 standard. Flammability of the sample was tested on a GT-C32 45-degree flammability tester (GESTER, China), according to 45-degree slope burning method in Japan industry standard JIS L 1091-1999. The samples were subjected to 30 home laundering cycles using a reference detergent according to the ISO 6330:2012 standard. Properties of the fibers before and after the washing were compared.
The SEM images were taken using a S4800 II cold-cathode field-emission SEM (Hitachi, Japan) at various accelerating voltages of 10–15 kV. It should be noted that the accelerating voltage needs to be adjusted in order to prevent the breakdown of nonconductive VF. The SEM samples were coated with gold.
The TGA and DSC of the samples were carried out using a STA449C thermal analyzer (NETZSCH, Germany) at a heating rate of 10℃/min from room temperature to 900℃ in air. The TG-FTIR of the samples was carried out using a STA449C thermal analyzer (NETZSCH, Germany) interfaced to an E55 FTIR spectrometer (Bruker, Germany) in which the spectral range was 4000–400 cm−1, the resolution can achieve 0.2 cm−1, wavenumber accuracy was better than 0.01 cm−1, transmittance accuracy was better than 0.1%. The heating rate was set as 10℃/min from room temperature to 900℃ in air. The airway was maintained at a constant temperature of 200℃ in the TG-FTIR test to ensure that the gases do not condense. One end of airway was connected to the thermal analyzer directly. The 3D diagrams from the TG-FTIR technique can provide a qualitative picture of the overall information of sample thermal decomposition and the absorbance corresponding to the vibrational modes of the different bonds or functional groups that was represented versus the wavenumber and the temperature [13].
The mechanical properties of the samples were tested on an YG020D-50 yarn electronic tension meter (Changzhou Huafang, P.R. China) at 25℃ and relative humidity 65%.
Results and discussion
Physical and flame retardancy of samples
Mechanical properties of samples.

FRVF: flame-retardant viscose fiber; PET: polyethylene terephthalate.
CSY-1, 2, 3 samples have good strength and elongation at break, so they have good weaving properties. But the strength and elongation at break of CSY-4 and CSY-5 were relatively low.
The LOI values of the samples are shown in Figure 2. The LOI value of PET was only 21% before and after washing, and the test found that PET fibers have molten drop phenomenon, which means PET has no flame-retardant properties. However, the LOI of CSY-3 was still >27% after 30 washing cycles, indicating that phosphazene derivatives impart durable flame retardancy to the fibers. The LOI value of samples CSY-1, CSY-2, and CSY-3 was bigger than that of VF and PET samples, and the higher the FRVF content of yarn, the bigger its LOI value, which present the flame-retardant property of CSY can be greatly improved by adding FRVF. This should be mainly caused by the addition of phosphazene derivatives. It can also be found that the LOI value of yarn decreases slightly after 30 washes, indicating that the wash has no significant effect on the flame retardancy.
LOI result of the samples before and after washing. LOI: limiting oxygen index.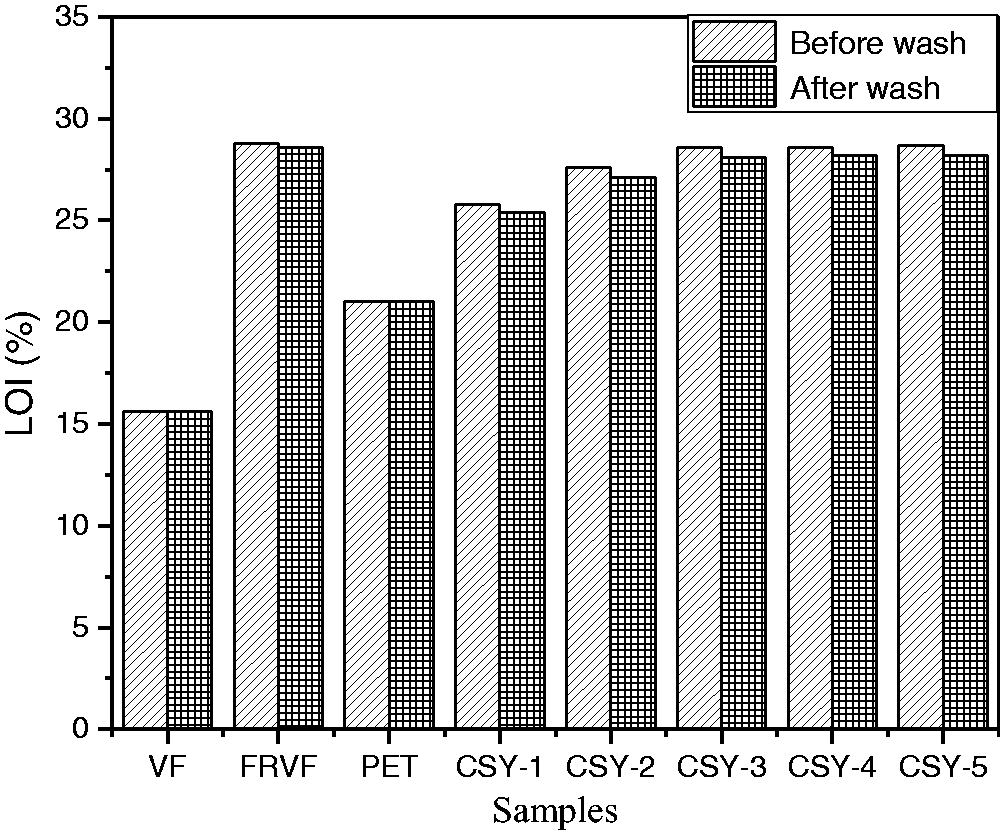
Forty-five-degree ignition test result before and after washing.

FRVF: flame-retardant viscose fiber; LOI: limiting oxygen index; PET: polyethylene terephthalate.
Microstructure of the fiber sample
The SEM images of the FRVF (before and after burning), PET (before and after burning), and residues of CSY-3 (after burning) are shown in Figure 2. Before burning, FRVF (a) and PET (c) both have a smooth structure. After burning, in the residues of FRVF the fiber structure was maintained, and the surface of fibers was carbonized as shown in Figure 3(b). And in the residues of PET the fiber structure was lost completely, and the surface of PET was melting into a mass as shown in Figure 3(d). However, CSY-3 has shown a totally different phenomenon to FRVF and PET, as shown in Figure 3(e) and (f). Most of the fibers of CSY-3 still maintain the tight yarn structure of the core yarn after burning. As shown in Figure 3(e) most of the residual fibers have the characteristics of FRVF from the fiber’s shape, which presents a flat fibrous structure instead of the cylindrical structure. And we can see a large number of irregular bulges appeared on the surface of the CSY-3 combustion product in Figure 3(f) also, which is clearly different from Figure 3(b).
SEM images of the FRVF (a) and PET (c); residues of FRVF (b), PET (d), and CSY-3 (e and f) after burning behavior test.
In the heating process, PET melted at about 280℃ and flowed to FRVF due to the guiding effect of heat flow just like a wick effect. Before PET was decomposed, it was adsorbed by FRVF. Phosphazene derivative which was used as a flame retardant in FRVF (∼10%) was heated during the fire. The decomposition of phosphazene derivative occurred in the range of 177–350℃ [14]. The phosphorus present formed the liquid-state membranes of phosphoric acid when heated and then dehydrated to form polymetaphosphate. A covering layer was formed on the surface of the fiber by polymetaphosphate to prevent further thermal decomposition of FRVF. In the thermal decomposition of phosphazene derivative, nitrogen, nitrogen oxides, and other nonflammable gases were continuously generated. These gases and the liquid film formed a swelling bubble-like structure as shown in Figure 3(b); this air-free coat is more conducive to the heat insulation of the interior of the fiber. It can also be seen that as the inside of the fiber still continues to degrade slowly after carbonization and gas was generated, the carbonized layer is continuously stressed, therefore, cracks appeared on the surface of the carbonized layer.
Since PET was adsorbed by FRVF before being decomposed, the polymetaphosphate not only covered the surface of the FRVF, but also covered PET, and formed a cover layer. PET’s dehydration was accelerated by polymetaphosphate and it formed a char layer before being decomposed. With the release of nonflammable gases, the surface of the sample formed an inflated air-free coat. The rugged surface structure was formed as we can see in Figure 3(e) and (f). This showed a typical intumescent flame-retardant mechanism.
Thermal behavior of the fiber compositions
Thermogravimetry (TG) and derivative thermogravimetry (DTG) curves of PET, FRVF, and CSY-3 are shown in Figure 4. Differential scanning calorimetry (DSC) curves are shown in Figure 5. The thermal degradation process of PET can be divided into two major stages: FRVF can be divided into three major stages, and the CSY-3 can be divided into four major stages. The rates of weight loss were reflected on DTG curves.
TG and DTG curves of PET, FRVF, and CSY-3. DTG: derivative thermogravimetry; FRVF: flame-retardant viscose fiber; PET: polyethylene terephthalate; TG: thermogravimetry. DSC curves of samples. DSC: differential scanning calorimetry; FRVF: flame-retardant viscose fiber; PET: polyethylene terephthalate.

For CSY-3, stage 1 is the dehydration process in the range 50–140℃ with a weight loss of ∼3.7%. As shown in Figure 4, the moisture content of CSY-3 was higher than PET (∼0.4%) and less than half of the FRVF (∼10%). This is caused by the twist of reduced specific surface area.
Stage 2 began at about 150℃ (same as FRVF) and ended at ∼370℃, and the weight loss was 35.6%. As shown in Figure 4, PET has no weight loss before 360℃, and the weight loss of FRVF was 47.4% at this range. Theoretically, CSY-3 should lose about 23.7% of weight loss in this phase if no interaction occurs. The actual test result was 35.6%, which is much larger than the theoretical value. This means that, when the temperature up to ∼280℃ (melting point of PET), PET melted and flowed to FRVF due to the guiding effect of heat flow and accelerated dehydration by polymetaphosphate (produced by the decomposition of FRVF). It is clear that blending with FRVF caused the lower decomposition temperature and more weight loss of primary decomposition for PET.
The DSC results suggest that the process is much complicated with the blend of FRVF and PET. In this stage, the DSC curve of FRVF has two endothermic peaks at ∼310.3 and ∼375℃, and PET gives no exothermic peak. The DSC curve of CSY-3 gives only one exothermic peak at 337.7℃. It is hard to identify each reaction in this process. However, it is clear that the major decomposition reaction of PET was accelerated by the blend of FRVF.
Stage 3 began at about 370℃, a little lower to FRVF and PET, ended at ∼470℃, and the weight loss was 23.6%. In this stage, the DSC curves of CSY-3 and FRVF give no endothermic peak. The DSC curve of PET gives the only one endothermic peak at 464.4℃. This stage mainly corresponds to the decomposition of PET that was not carbonized in the previous stage. Compared to the weight loss temperature of PET in this range, the weight loss temperature of CSY-3 at this stage is advanced by ∼10℃. However, the weight loss is far less than half of the weight loss of PET in this temperature range (81.5%). This is still due to the polyphosphoric acid produced by the decomposition of FRVF carbonizing PET, causing a portion of the melted PET to be carbonized at a lower temperature. The carbonized products were first formed on the outer layer of the yarn due to the core yarn spinning method. The formed carbonized layer tightly covers the fibers inside the yarn isolating heat and oxygen; thus, the fibers inside the yarn were protected and the structure of the yarn was preserved.
The final stage begins at 550℃ until the end. At this stage, the weight loss of CSY-3 was 22.0%, and the final residual weight was 15.1%. In this stage, the DSC curve of CSY-3 gives two exothermic peaks at 501 and 529℃. The DSC curve of FRVF peaked at 530℃ and the curve of PET peaked at 568.9℃. The residual weight of CSY-3 at this stage is far greater than the average of PET and FRVF residuals (18.7 + 1.3)/2 = 10%. In this range, the carbonized product of FRVF and PET was heated together; all these carbonized products were further oxidized to produce small molecular gases such as CO2 and H2O. In the previous temperature range, PET was carbonized by the poly(meta-phosphoric acid) decomposed by FRVF. The carbonized layer caused a lot of PET fail to completely decompose at previous temperature range, but decomposition and carbonization after further heating, which caused a high weight loss rate for CSY-3 at this stage.
Real-time qualitative analysis of volatile products generated during thermal oxidation
The 3D IR spectroscopy of thermal decomposition products of PET (a), FRVF (b), and CSY-3(c) is shown in Figure 6. From the figure, we can find many discontinuous peaks, which is due to the fact that some small particles partially block the airway during the TG-FTIR test. After comparing with the standard pattern (EPA Vapor Phase No. 2911), it was confirmed that 2630 cm−1 was the main characteristic peak of CO2 and 3725, 3694, 3624, 3602 cm−1 were also the characteristic peaks of CO2. The main product in the thermal decomposition process of all these three samples is CO2. A series of small absorption peaks appearing at 3500–3900 and 670 cm−1 are characteristic absorption peaks of gaseous H2O.
Three-dimensional diagram of the gases evolved during the thermal pyrolysis of PET (a), FRVF (b), and CSY-3(c).
For PET, the C=O stretching peak of phthalic anhydride appeared at 1762 cm−1 at 468℃, which means PET produced a large amount of phthalic anhydride when decomposed by heat. The peak gradually disappears at 611℃, which shows that phthalic anhydride was further oxidized and decomposed at high temperatures. The COO antisymmetrical vibration peak appeared at 1540 cm−1 at 380℃ and gradually disappears after 380℃, which means decomposition products of PET at lower temperature have carboxylate COO appearing.
The thermal decomposition products of CSY had only a small amount of CO2 and H2O before 300℃, the CO2 concentration gradually increased after 300℃. Simultaneously, the peaks of the stretching peaks of ester C=O at 1740 cm−1 and the characteristic absorption peaks of small molecules of alkanes at 1080 cm−1 appeared, which indicates that at this temperature CSY-3 decomposes violently and produces a large number of small molecular gases. These gases are partially oxidized to CO2 and H2O, and some of them are not oxidized and enter the detection chamber. This corresponds to the second and third stage weight loss in the TGA. When the temperature reached 350℃, the characteristic absorption peak of the small molecule gas disappeared, leaving only the characteristic absorption peaks of CO2 and H2O. When the temperature reaches 600℃, the absorption peak of CO2 also becomes insignificant. This is due to the char layer coated on the surface of the yarn preventing the oxidation and decomposition inside CSY-3; only a small amount of H2O absorption peak indicates that the dehydration carbonization reaction of the polymetaphosphoric acid inside the yarn is still in progress. The main products in the thermal decomposition of CSY-3 were H2O and CO2.
Conclusions
The flame-retardant yarn was made of PET and FRVF by core-spun method. PET fiber filament was set as a core, and the FRVF was set as a sheath to change the disadvantage of moisture permeability and flame retardancy. The LOI value of CSY was bigger than that of PET, and the flame-retardant property of CSY can be greatly improved by adding FRVF. The LOI value of CSY-3 (PET:FRVF=50:50) decreases slightly after 30 washes, indicating that wash has no significant effect on the ignition property. Since PET was adsorbed by FRVF before being decomposed, the polymetaphosphate not only covered the surface of the FRVF, but also covered PET and formed a cover layer. PET’s dehydration was accelerated by polymetaphosphate and formed a char layer before being decomposed. With the release of nonflammable gases, the surface of the sample formed an inflated air-free coat. The carbonized products were first formed on the outer layer of the yarn due to the core yarn spinning method. The formed carbonized layer tightly covers the fibers inside the yarn isolating heat and oxygen; thus, the fibers inside the yarn were protected and the structure of the yarn was preserved.
The carbonization of the PET was promoted by the decomposition of phosphazene derivative in FRVF. The temperature of decomposition reaction was reduced and a lot of carbide protective layers were formed at low temperature zone. The main products in the thermal decomposition of CSY-3 were H2O and CO2.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
