Abstract
Recently, textile technologies have gained a huge attention for fabricating the tissue constructs. In tissue engineering, the development of biodegradable polymer scaffolds which resemble the native function of the extra cellular matrix is a great challenge. In this research, a novel electrospun scaffold based on polyurethane, propolis and neem oil was developed for tissue-engineering applications through textile technology like electrospinning. The morphology of the electrospun polyurethane/propolis and polyurethane/propolis/neem oil exhibited reduced fibre and pore diameter than the pristine polyurethane. The change in the chemical structure of the electrospun composites was identified by peak shifting of CH band peak with hydrogen bond formation.The contact angle measurements revealed that the PU/propolis mat was found to be super hydrophilic (0°) in nature and the electrospun polyurethane/propolis/neem oil mat was observed to be hydrophilic (41° ± 1) in nature. Thermogravimetric analysis and atomic force microscopy analysis showed that the prepared polyurethane/propolis and polyurethane/propolis/neem oil mats exhibited higher thermal stability and decreased surface roughness than the pristine polyurethane. Moreover, the developed polyurethane/propolis (activated partial thromboplastin time – 144 ± 4 s and prothrombin time – 85 ± 3 s) and polyurethane/propolis/neem oil (activated partial thromboplastin time – 147 ± 3 s and prothrombin time – 86 ± 2 s) composite displayed decreased blood clotting time than the pristine polyurethane (activated partial thromboplastin time – 153 ± 3 s and prothrombin time – 89 ± 3 s). The results of the haemolytic assay indicated that the electrospun polyurethane/propolis (1.21%) and polyurethane/propolis/neem oil (1.30%) mats exhibited better safety to red blood cells than the pristine polyurethane (2.48%). Hence, these desirable characteristics of the newly developed scaffold might be used as a potential platform for tissue engineering applications.
Introduction
Nowadays, tissue engineering has emerged as a prominent method for the remodeling of the damaged tissue. The aim of tissue engineering is to develop an alternative substitute for the damaged tissue and also to improve the function of human tissues. Till now, the tissue engineering methods were successfully applied in different tissue organs of the human body. Tissue engineering applications extend their horizon to organs like bone, cartilage, skin, pancreas, vascular and cardiac [1]. Scaffold is a vital component in tissue engineering as it facilitates and supports the regeneration of new tissue growth [2]. The first and foremost criterion for the successful utilization of scaffold in tissue engineering is its compatibility with the blood. The blood compatibility plays a vital role in deciding the application of the fabricated scaffolds in the tissue engineering applications [3]. The fibres made from polymers hold great promise in mimicking the native function of the extra cellular matrix (ECM) [4]. The natural ECM consists of two macromolecules, namely proteoglycans and fibrous proteins. These macromolecules have fibres with diameters ranging from 50 to 150 nm, respectively [5,6]. Recent studies reported that the fibres exhibited better cell adhesion and proliferation for new tissue growth [7,8]. Textile technologies have been widely utilized for fabricating the fibrous scaffolds for different tissue engineering applications [9]. The structures obtained by these technologies offer huge versatility through controlling various parameters such as size and orientation of fibres, pore size, geometry and interconnectivity and surface morphology. Further, these properties play a vital role in influencing the physical properties and cellular performance of the fabricated constructs [10]. The contribution of medical textiles to tissue engineering is very diverse and several textile technologies are widely applied in tissue engineering. One of the textile technologies which has demonstrated potential is electrospinning.
Electrospinning is a widely used technique to extract the fibres from the synthetic and natural polymers [11]. Electrospinning is a versatile and the least expensive technique in which ultra-fine fibres are fabricated by applying high voltage. On applying a high voltage to the polymer jet, the ultra-fine fibres are drawn from the syringe needle and collected on the drum [12,13]. The ultra-fine fibres have high surface area and porosity which enhance adherence, migration and growth of the fibroblast cell responses [14]. Till date, a wide number of biodegradable polymers have been employed in the tissue engineering application for fabricating the scaffold. In this study, the biodegradable polyurethane (PU) is utilized for fabricating the scaffolds. As a wound dressing material, the PU is used in the form of foams, membranes and films [15]. Moreover, it is widely used in biomedical applications owing to its flexibility, better barrier properties and oxygen permeability [16]. Although, PU possesses good biocompatibility and mechanical characteristics, its cellular adhesion is low [17]. There are various techniques utilized such as ion beam irradiation, protein immobilization, grafting of peptides, plasma induced grafting and direct plasma treatments to improve the cellular responses by altering the wettability of PU [18–22].
Tailoring wettability is an important criterion as it is involved in protein adsorption on the surface. Various postulates have been made, and there is a consensus that wettability in the optimum range is essential for the cell adhesion and proliferation [23–25]. In this research, for tailoring the wettability of the scaffolds, components like propolis and neem were selected based on the literature [26,27] and motivation to investigate the combined role of these components.Adomavičiūtė et al. [26] fabricated electrospun PLA scaffold added with propolis and silver nanoparticles. It was observed that the addition of propolis favoured the decrease in the wettability behavior and also promoted the viability of keratinocytes as indicated in the in vitro testing. Further, Kim et al. [27] fabricated wound dressing scaffold based on PU loaded with propolis. The PU scaffold added with propolis exhibited hydrophilic behavior. Further, it was reported that the addition of propolis into the PU membrane improved the fibroblast cells adhesion and proliferation. Propolis is a resinous substance obtained from bees through their salivary secretions [28]. Propolis is reported to have various bioactive constituents like flavonoid aglycones, phenolic components, amino acids and also some inorganic compounds [29]. As a composition, it contains 50% of resin which are flavonoids and related phenolic acids, 30% are wax, 10% are essential oils, 5% are pollen and the remaining 5% includes the various organic compounds [30]. Moreover, it possesses various biological activities such as antibacterial, antifungal, antiviral, antioxidant and anti-inflammatory [28]. The plant extracts were used to treat cancers owing to their active anti-cancer properties. Apart from their anti-cancer activities, the plant extracts were also utilized for wound healing effects [31]. Certain reports had suggested that the medicinal plants accelerate the various phases of the wound healing process [32–35]. Both in vivo and in vitro testing of plant extracts from Annona squamosa [36], Ageratina pichinchensis [37], Andrographis paniculata [38], Butea monosperma [39] and Calendula extracts [40] depicted accelerated wound healing. Jin et al. [41] fabricated wound dressing scaffold based on polycaprolactone (PCL) loaded with different plant extracts. The plant extracts loaded with PCL exhibited favourable fibroblast adhesion and proliferation. Although there are several plant extracts available, neem oil attracted us due to its versatile medical properties that have been widely documented. Neem oil is obtained from the seeds of neem or the Margosa trees (Azadirachta indica). Neem or the Margosa trees (Azadirachta indica) are commonly found in India, Pakistan, Bangladesh and Nepal and reported to have many biological constituents like nimbin, nimbidin, nimbolide and limonoids. These constituents play an important role in curing various diseases in clinical applications [42]. Further, it possesses polyphenolic and flavonoid components which are observed to have several medicinal activities such as antibacterial, antifungal, antiviral, antioxidant and anti-inflammatory activities [43–45].
Wettability is an important criterion as it majorly influences the protein adhesion which is a decisive factor for cell adhesion. Although use of propolis has been experimented in the formation of hydrophilic scaffolds in tissue engineering, an appropriate control of the wettability to the optimum range has never been reported. This study for the first time explores the use of propolis in combination with neem oil with the intention of rendering appropriate wettability for the developed composite. In this work, a novel tissue engineering scaffold is electrospun based on PU blended with propolis and neem oil. Besides, the physico-chemical properties, blood compatibility and cytocompatibility assays of the newly developed composites were investigated in order to assess their effectiveness for tissue engineering applications.
Materials and methods
Materials
Tecoflex EG 80A was purchased from Lubrizol, USA. It contains 35% of hard segments (Shore A hardness = 72, specific gravity = 1.04) with a melting point of soft segment at 22℃. Its constituent formulation contains hard segment of methylene bis(cyclohexyl) diisocyanate (hydrogenated MDI, H12MDI), the soft segment of polytetramethylene oxide with molecular weight = 1000 g/mol and 1,4 butane diol as a chain extender.. The propolis used in this study is Brazilian Green Propolis procured from Eu Yan Sang, Johor, Malaysia. Similarly, the neem oil was obtained locally from Renga Trading and Manufacturing, Malaysia. DMF solvent was purchased from Merck, Malaysia. Phosphate buffered saline (Biotech Grade) and sodium chloride physiological saline (0.9% w/v) were obtained from Sigma-Aldrich, Malaysia. All reagents used for activated partial thromboplastin time (APTT) and prothrombin time (PT) assays were obtained from the Diagnostic Enterprises, India.
Solution preparation
Initially, the PU with a weight fraction of 9 wt% was obtained by dissolving the calculated amount of PU in DMF and stirred for 12 h maximum. Similarly, the homogeneous solution of propolis and neem oil was prepared at a weight fraction of 8 wt% which was obtained by mixing the required volume of propolis and neem oil in DMF and stirred for 1 h maximum. To prepare 8 wt% of propolis and neem oil, 0.240 g of propolis and 240 µl of neem oil were mixed in 3 ml of DMF, respectively. Last, PU/propolis and PU/propolis oil/neem oil were prepared at a ratio of 7:2 (v/v%) and 7:1:1 (v/v%), respectively, and stirred for 2 h for even dissolution. Further, the membranes were electrospun at room temperature with humidity of 55%.
Preparation of the membranes
Electrospinning method was used in this study to fabricate PU, PU/propolis oil and PU/propolis/neem oil fibres. Briefly, the prepared PU, PU/propolis and PU/propolis/neem oil solutions were loaded in a 10 ml syringe and fitted in the syringe pump.The electrospun fibres were attained at a flow rate of 0.5 ml/h with an applied voltage of 10.5 kV. Then, the obtained fibres were collected on an aluminum foil which was placed 20 cm away from the tip of the needle. The residual DMF content present in the deposited fibres was removed by drying under vacuum at room temperature. The thickness of the electrospun membranes was measured through TMI 49-56 digital micrometer. The mean thickness of the electrospun PU, PU/propolis and PU/propolis/neem oil scaffolds was 0.190, 0.111 and 0.044 mm, respectively.
Physio-chemical characterizations
SEM analysis
Scanning electron microscope (SEM) unit was used to study the morphology of the electrospun fibres. The obtained electrospun fibres were gold-coated prior to examining the morphology of the fibres. From the captured SEM images, the average fibre dimeter and pore diameter (distance between two fibres) were calculated using Image J software by choosing 50 locations randomly.
Contact angle measurements
To measure the contact angle of the developed membranes, the water drop method was utilized. A distilled water droplet (0.5 µl) was placed on the obtained fibrous membrane and the droplet image was captured within 2 s using a video cam. Finally, from the captured image, the manual contact angles were measured through computer integrated software.
FTIR analysis
Nicolet FT-IR spectrometer was utilized to investigate the chemical bands of the electrospun fibres. The spectra were obtained by recording a small piece of electrospun fibre mat from a wavelength of 600–4000 cm−1 at 4 cm−1 resolution with 32 scans/min.
Thermal analysis
Thermogravimetric analysis (TGA) unit was used to study the integrity of the electrospun membranes. The samples were heated from 30 to 1000℃ at a rate of 10℃/min under nitrogen atmosphere.
AFM analysis
Atomic force microscopy (AFM) equipment was used to calculate the average roughness of the electrospun fibres. The AFM analysis of the samples was conducted in normal atmosphere and scanned in the size of 20 µm × 20 µm. Finally, the 3D images with 256 × 256 pixels were obtained via JPKSPM data processing software.
Mechanical testing
The deformation rate of the electrospun membranes was tested using a uniaxial testing machine. The samples (40 mm × 15 mm) were fixed in the grip ends of the tensile machine, and the mechanical test was carried at a strain rate of 5 mm/min with the load cell of 500 N. Last, the deformation rates were determined through machine-generated stress–strain curve.
Coagulation assay
APTT and PT assay
The PT and APTT assays were used to examine the anticoagulant nature of the electrospun membranes. To begin the assay, the electrospun scaffolds with a size of 0.5 cm × 0.5 cm were cut and incubated with 50 µl of human blood plasma for 1 min at 37℃. Then, they were further mixed with 50 µl of rabbit brain cephaloplastin followed by 50 µl of CaCl2 (0.025 M) solution which registers the APTT. For PT assay, the samples added with 50 µl of human blood plasma were incubated with 50 µl of NaCl–thromboplastin reagent for 3 min at 37℃ and PT was registered. The blood clotting initiation was done by stirring and the time was measured by chronometer [46].
Haemolysis assay
To evaluate the safety of the electrospun fibres with red blood cells (RBCs), the haemolysis assay was carried out in Thermoset Scientific Multiskan™ FC (Waltham, MA, USA). To begin the assay, the samples (1 cm × 1 cm) were soaked in 5 ml of saline (0.9% w/v) at 37℃ for 30 min. After soaking, the samples were incubated with a 3 ml mixture of citrated blood and saline (4:5 v/v%) for 1 h at 37℃. Untreated 0.9 wt% NaCl aqueous solution and distilled water serve as negative and positive controls, respectively. Then, the samples were retrieved and centrifuged and absorbance was measured. The absorbance was recorded by aspirating the clear supernatant at 542 nm which implies the amount of haemoglobin released. The haemolytic percentage was calculated using the formula as discussed previously [46].
Statistical analysis
All experiments were performed three times independently. One way ANOVA was conducted followed by Dunnett post hoc test to analyse the statistical significance (p < 0.05). The obtained results from all experiments are expressed as mean ± SD. In case of qualitative experiments, an illustrative of three independent images is shown.
Result and discussion
Morphological analysis
The morphological images of the electrospun PU, PU/propolis and PU/propolis/neem oil are indicated in Figure 1(a) to (c).
SEM images of (a) polyurethane, (b) polyurethane/propolis composites, (c) polyurethane/propolis/neem oil composites, fibre diameter distribution of (d) polyurethane, (e) polyurethane/propolis composites, (f) polyurethane/propolis/neem oil composites, pore size distribution of (i) polyurethane, (j) polyurethane/propolis composites and (k) polyurethane/propolis/neem oil composites.
The morphological images showed that the electrospun membranes exhibited randomly oriented fibres without any beads. The analysis of the electrospun PU/propolis and PU/propolis/neem oil revealed that their average diameter was decreased with the addition of the propolis and neem oil into the matrix of PU. The average fibre diameter of the electrospun PU was found to be 890 ± 117 nm, while the electrospun PU/propolis and PU/propolis/neem oil showed fibre diameter of 550 ± 148 nm and 642 ± 148 nm, respectively (mean differences were significant compared with pure PU (p < 0.05)), and the corresponding fibre distribution curve of the electrospun PU, PU/propolis and PU/propolis/neem oil is shown in Figure 1(d) to (f). The addition of propolis to the PU resulted in the reduction of viscosity of the solution. It has been widely reported that a reduction in viscosity leads to decrease in the fibre diameter [47,48]. Hence, we attribute the fibre diameter reduction of the PU/propolis scaffold to the viscosity reduction. This observation contradicts the recently reported work of Kim et al. [27] where they observed an increase in the fibre diameter with the addition of propolis. This may be owing to the fact that the PU is of different class compared with the Tecoflex 80 A which was used in this experiment. Hence, the components of the propolis are expected to have a different chemical environment. Also, the content of propolis may vary according to geographical location, botanical origin and bee species [49–53] which could be one of the major cause of this conflicting behavior. Since electrospinning is a multifactorial process which not only depends on viscosity but also on other factors like solution conductivity, choice of the solvent etc., this property would have also resulted in this outlying behavior. Interestingly, the addition of neem oil resulted in a minor improvement in the viscosity, resulting in the slight increase of fibre diameter but still possessing smaller diameter than the pure PU. Chen et al. [54] developed scaffold for tissue engineering application utilizing PU blended with collagen. It was observed that the PU/collagen scaffold showed reduced fibre diameter in the range of 700–800 nm compared to pristine PU which showed the average diameter of 1330 nm. Further, the developed PU/collagen scaffold with reduced fibre diameter exhibited enhanced cell adhesion and proliferation. Hence, the reduced fibre diameter of PU/propolis and PU/propolis/neem oil may be beneficial for the enhanced cell adhesion and proliferation of new tissue growth.
Using Image J software, the pore size of the electrospun fibrous mats was calculated and depicted in Figure 1(i) to (k). From pore size measurements, it was observed that the pore size of the PU membrane was decreased with the incorporation of propolis and neem oil. The PU/propolis and PU/propolis/neem oil composite membrane showed pore size of 622 ± 111 nm and 654 ± 106 nm (mean differences were significant compared with pure PU (p < 0.05)), while pristine PU showed pore size of 1064 ± 74 nm, respectively. The pore size is dependent on the fibre diameter distribution. It has been recently postulated that a decrease in fibre diameter coincides with the decrease in pore size [55]. Prabhakaran et al. [55] electrospun fibrous scaffold loaded with gelatin for cardiac tissue engineering. It was observed that the incorporation of gelatin into the poly(
FTIR analysis
The IR spectra for the electrospun PU, PU/propolis and PU/propolis/neem oil are shown in Figure 2(a). In the spectrum of PU membrane, the three main characteristics peaks seen at (i) 3323 cm−1, (ii) 2939 and 2854 cm−1 and (iii) 1730 and 1703 cm−1 were assigned to NH stretching, CH stretching and carbonyl stretching, respectively. Further, the other peaks at 1597 cm−1 and 1531 cm−1, 1413 cm−1 and 1221 cm−1 and 1104 cm−1 were attributed to vibrations of NH stretching, CH stretching and CO stretch corresponding to alcohol group [39]. FTIR analysis of the electrospun PU/propolis and PU/propolis/neem oil depicted no new peak formation but the CH peak intensity was decreased in PU/propolis and increased in PU/propolis and neem oil composites owing to hydrogen bond formation as shown in Figure 2(b) [56]. Further, a peak shift occurred in the PU/propolis and PU/propolis/neem oil indicating the presence of propolis and neem oil in the PU matrix. The CH peak at 2939 cm−1 in PU was slightly moved to 2937 cm−1 in PU/propolis and 2925 cm−1 in PU/propolis/neem oil mats which indicates the interaction of PU with propolis and neem oil [57].
(a) FTIR spectra of polyurethane, polyurethane/propolis composites and polyurethane/propolis/neem oil composites and (b) CH peak intensity of the electrospun membranes.
Wettability analysis
The water wettability for the electrospun PU, PU/propolis and PU/propolis/neem oil was measured. From the water wettability results, it was observed that the surface of PU was found to be hydrophobic (100° ± 1) and with the addition of propolis, the wettability was decreased to 0° indicating superhydrophilic nature. Further, with the blending of neem oil, the wettability shifted to 41° ± 1 insinuating hydrophilic nature of the developed composites (mean differences were significant compared with pure PU (p < 0.05)). Hydrophobic surfaces are incapable of cell growth when it comes in contact with the living systems. Till now, various attempts such as physicochemical and surface modification techniques have been utilized to solve the issues associated with the hydrophobic surfaces [58,59]. Propolis is reported to possess several hydrophilic phenolic chemical constituents which would have imparted hydrophilicity to the composite. A similar trend of increasing hydrophilicity was reported by Adomavičiūtė et al. [26] and Kim et al. [27]. Adomavičiūtė et al. [26] observed that their contact angle was around 30° with the addition of the 10% propolis. However, Kim et al. [27] did not find their wettability range shifting towards superhydrophilic as reported in their work. As stated previously, the choice of the polymer and constituents present in the propolis [49–53] might be attributed to this behavior. Although the wettability properties of the PU/propolis were deemed to be improving, it was found to be extremely hydrophilic. This extreme nature may not be conducive for the certain tissue engineering applications. As the surface is superhydrophilic, it produces extensive repulsive force leading to the lower protein adhesion. Protein adhesion is an important criterion to promote cell adhesion and hence it leads to lower the cell adhesion on the surface [25]. For this reason, surface with moderately hydrophilic is generally preferred to selectively promote the cell adhesion behavior [23–25]. With these intentions, we had looked for another component which can shift the contact angle to the moderate level. Although there are several options to accomplish this, neem oil attracted us due to its versatile medical properties that have been widely documented [42–45]. The addition of neem oil to this found to alter the wettability to the moderate level (41° ± 1.155) which is found to be relevant to the tissue engineering applications [23–25]. Further, it would be interesting to examine how the varying concentration of neem oil affects the wettability of the scaffolds.
Thermal analysis
Certain studies reported the thermal analysis of the fabricated composites in tissue engineering applications to predict the degradation behavior [60,61]. Thermal analysis clearly depicts the interaction of added constituents [62], thermal stability [63] and integrity of the developed materials [64]. Hence, in this study, TGA analysis was used to determine the stability of the electrospun composite and also interaction of propolis and neem oil with the PU. TGA analysis showed that the incorporation of propolis and neem oil improved the thermal behavior of the pristine PU. It was observed that the initial onset temperature (276℃) of PU was increased to 316℃ and 312℃ in PU/propolis oil and PU/propolis/neem oil mat, indicating the enhanced thermal behavior of the electrospun composite as shown in Figure 3. Adomavičiūtė et al. [65] investigated the thermal behavior of the melt-spun polypropylene (PP) fibres added with propolis. It was observed that the PP fibres added with propolis showed similar thermal behavior to the pure PP. In our study, the addition of propolis showed slight improvement in the thermal stability which might be due to bioactive components present in the propolis. In the work of Kim et al. [27], the addition of propolis into the PU matrix showed reduction in the onset thermal degradation which might be due to different grade of polymer and varying constituents of the propolis [49–53] used in their study. Further, the DTG curve for the electrospun PU and its composites is shown in Figure 4. The degradation in PU occurs in three stages, namely first loss observed at 223–348℃, the second loss shown 348–446℃ and third loss occur at 557–684℃, respectively. These three losses were attributed to the decomposition of rigid and flexible segments of the PU. In the electrospun PU/propolis composite, it showed three weight loss in which the first loss observed at 232–379℃, the second loss begins at 379–496℃ and the third loss was observed at 496–675℃. It was noted that in PU/propolis composite, the weight loss intensity was increased compared to PU membrane indicating enhanced weight loss. It was reported that the propolis has phenolic and flavonoids which might have melted resulting in the enhancement of the weight loss. In the case of electrospun PU/propolis/neem oil composite, the weight loss occurs in two stages, namely first weight loss occur at 245–377℃ and the second loss at 377–523℃, respectively. With the addition of neem oil, the intensity of the second major weight loss peak in PU/propolis/neem oil was observed to be decreased than PU and PU/propolis indicating reduced weight loss. The reason might be that the bioactive compounds present in the neem oil must possess thermally stable constituents which have resulted in the reduced weight loss. Manikandan et al. [66] fabricated electrospun wound dressing scaffold based on PU added with murivennai oil. It was observed that the addition of murivennai oil favoured the improvement in the thermal stability and reduced weight loss which correlates with our findings.
TGA analysis of polyurethane, polyurethane/propolis composites and polyurethane/propolis/neem oil composites. Weight residue percentage of polyurethane, polyurethane/propolis composites and polyurethane/propolis/neem oil composites.

Surface roughness measurements
The AFM images for the electrospun PU, PU/propolis and PU/propolis/neem oil are shown in Figure 5(a) to (c). From the calculated surface roughness from the AFM images, it was observed that the surface roughness value for PU/propolis and PU/propolis/neem oil was decreased when compared to the pristine PU. The average Ra for the PU membrane was calculated to 576 nm, while the electrospun PU/propolis and PU/propolis/neem oil showed Ra of 327 and 363 nm, respectively (mean differences were significant compared with pure PU (p < 0.05)). Vincent et al. [67] reported in their study that the smaller fibre diameter will result in the reduced surface roughness. Hence, in our study, the fibre diameter of the fabricated composites was smaller than the pristine PU which might have resulted in the smoother surfaces. Further, with the addition of the neem oil, a slight increase was observed in the surface roughness which correlates with the observations of the Manikandan et al. [66]. Manikandan et al. [66] fabricated wound dressing scaffold comprising PU incorporated with murivennai oil. It was observed that the addition of murivennai oil resulted in the improvement of the surface roughness which is in agreement with our observations.
AFM images of (a) polyurethane, (b) polyurethane/propolis composites and (c) polyurethane/propolis/neem oil composites.
Mechanical testing
The tensile strength of electrospun fibrous scaffolds is considered as important factor for tissue engineering applications. The tensile strength of pristine PU and their blended composites is shown in Figure 6. It revealed that the addition of propolis and neem oil resulted in the improvement of the tensile strength. The pristine PU showed tensile strength of 3.7 ± 0.73 MPa, while the PU added with propolis and propolis/neem oil showed tensile strength of 4.97 ± 0.34 MPa and 5.88 ± 1.27 MPa, respectively (mean differences were significant compared with pure PU (p < 0.05)). Further, the elongation at break for the pristine PU, PU/propolis and PU/propolis/neem oil was found to be 105, 107 and 200%, respectively. The enhancement of the elongation at break for the electrospun PU/propolis and PU/propolis/neem oil was observed to be 2 and 95% more compared to the pristine PU. Kim et al. [27] fabricated PU scaffold added with propolis for biomedical applications. It was observed that the addition of propolis resulted in the enhancement of the tensile strength and concluded that the adhesive property of the propolis favoured the improvement in the tensile strength which resembles our findings [27]. Unnithan et al. [68] fabricated PU scaffold incorporated with emu oil. It was observed that the addition of emu oil into PU resulted in the enhancement of the tensile strength, and it was also considered that the reason for improvement might be the adhesive property of the emu oil which also correlates with our findings [68]. Hence, the improvement in the tensile strength might be owing to the combining effect of adhesive property possessed by the propolis and neem oil. Qian et al. [69] observed that their PLCL scaffold blended with the gelatin and chitosan fibres exhibited higher tensile strength than the control. They suggested that the improvement in the tensile strength was due to the small fibres of the fabricated composite membranes [69]. In this study, the PU added with propolis and neem oil showed smaller fibre diameters than the pristine PU which might have favoured the enhanced tensile strength.
Mechanical testing of polyurethane, polyurethane/propolis composites and polyurethane/propolis/neem oil composites.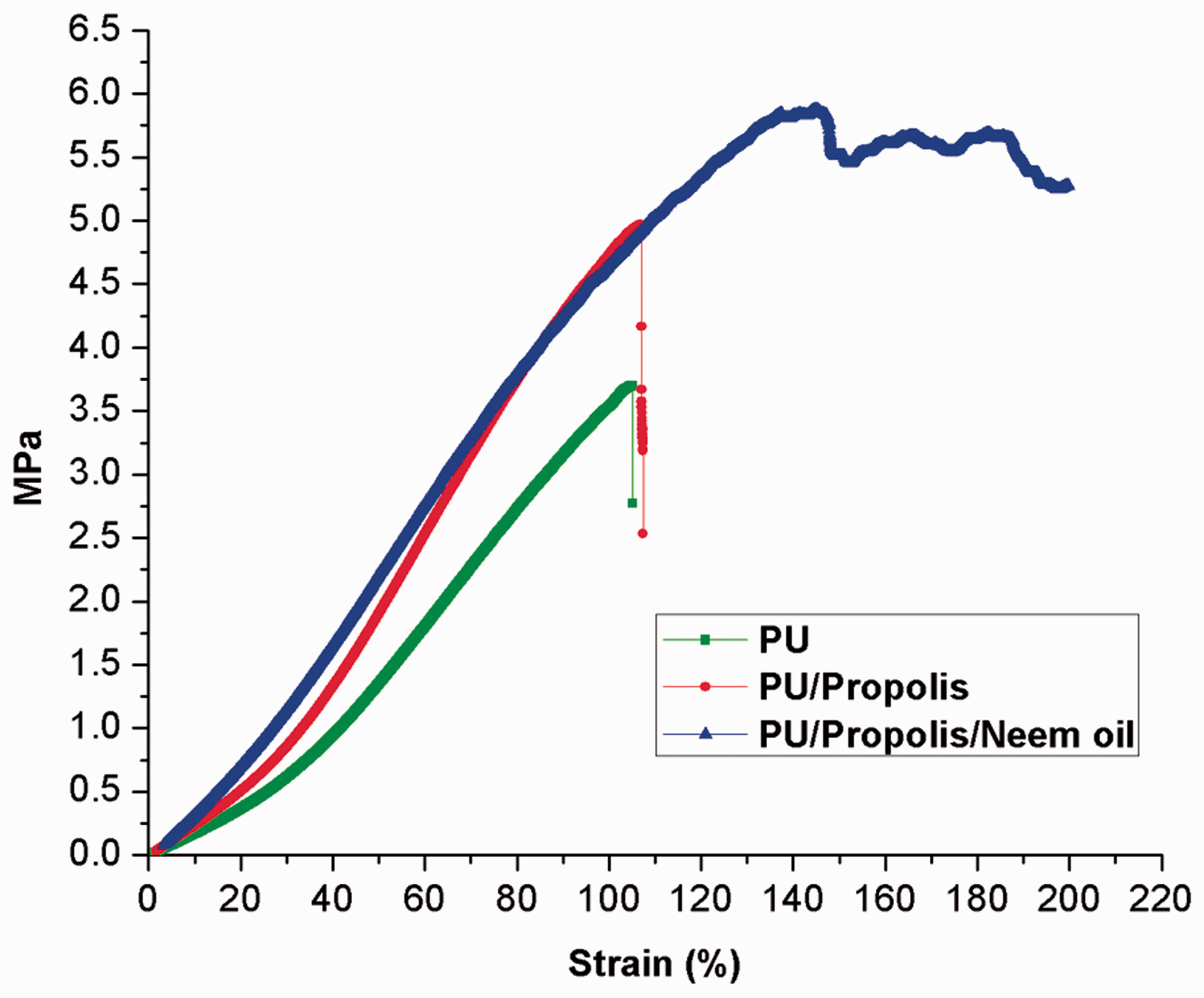
Coagulation assay results
The blood clot formation time for the electrospun PU, PU/propolis and PU/propolis/neem oil is shown in Figure 7(a) and (b). The obtained blood compatibility results revealed that the blood clotting time for the electrospun PU/propolis and PU/propolis/neem oil was decreased than the pristine PU.
(a) APTT assay of polyurethane, polyurethane/propolis composites and polyurethane/propolis/neem oil composites (mean differences were significant compared with pure PU (p < 0.05)) and b) PT assay of polyurethane, polyurethane/propolis composites and polyurethane/propolis/neem oil composites (mean differences were not significant compared with pure PU (p < 0.05)).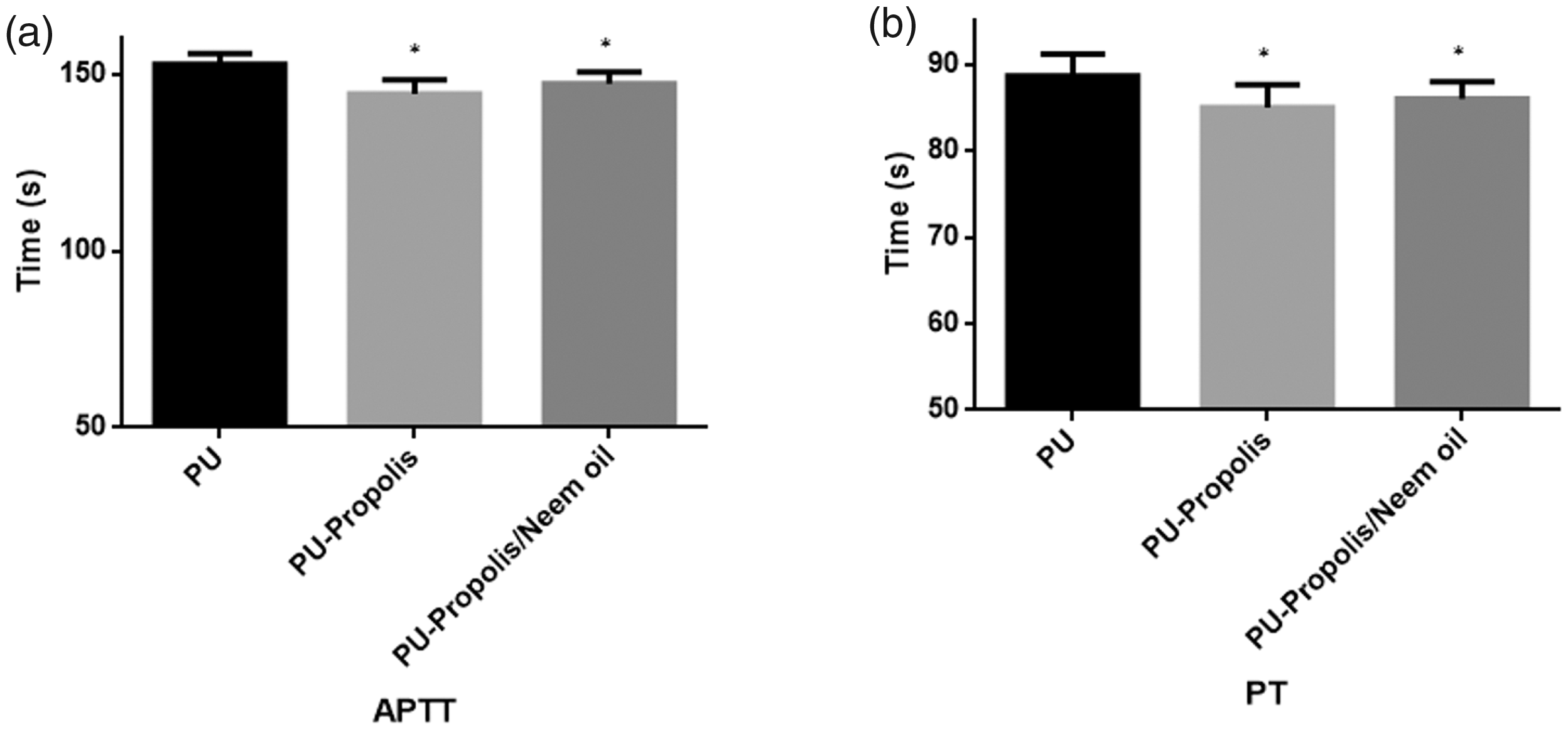
In the measurement of APTT assay, the blood clot formation time for PU membrane was observed to be 153 ± 3 s, and for electrospun PU/propolis and PU/propolis/neem oil mats, the blood clot formation time was found to be 144 ± 4 s and 147 ± 3 s, respectively. Similarly, in the measurement of PT assay, the blood clot formation time for PU membrane was observed to be 89 ± 3 s and for electrospun PU/propolis and PU/propolis/neem oil mats, the blood clot formation time was found to be 85 ± 3 s and 86 ± 2 s, respectively. Further, the APTT and PT values without any electrospun membranes were observed to be 143 ± 2 s and 85 ± 2 s. The decrease in blood clotting time was due to the existence of propolis and neem oil in the PU matrix. The reduction for the reduction in the blood clotting time was due to the trade-off between the polar and apolar regions of the PU/propolis and PU/propolis/neem oil scaffold [70].
In order to determine the safety of electrospun membranes with RBC, haemolysis assay was performed, and the obtained haemolytic percentage results are indicated in Figure 8. The calculated haemolytic percentage for PU membrane was found to be 2.48% and for the PU/propolis and PU/propolis/neem oil mats, the measured haemolytic percentage was 1.21 and 1.30%, respectively. The positive and negative controls displayed 100% and 0%, respectively. Hence, the toxicity of electrospun PU/propolis and PU/propolis/neem oil to RBC was very low compared to the pristine PU. Further, the electrospun composite mats were reported to as non-haemolytic materials because of measured haemolytic percentage was below 2%. In acute wound healing, the first stage involves inflammatory phase in which blood-clotting cascade takes place in order to prevent excessive blood bleeding and also to provide protection to the affected wound area [71]. In our study, the fabricated composites showed decreased blood clotting time and safe haemolytic index which might be promulgated for the acute wound healing.
Haemolytic assay of polyurethane, polyurethane/propolis composites and polyurethane/propolis/neem oil composites (mean differences were significant compared with pure PU (p < 0.05)).
Based on our observations, the following putative mechanism for the interaction of fibres with propolis and neem oil was proposed. Hydrophobic surfaces are incapable of cell growth when it comes in contact with the living systems. With the addition of propolis to the PU, the surface became superhydrophilic due to the hydrophilic constituents present in the propolis. The superhydrophilic surface may repel the protein adsorption resulting in the reduced cell adhesion. To tailor the wettability to a moderate level, neem oil was added to the PU/propolis scaffold. The addition of neem oil converted the superhydrophilic surface to moderately hydrophilic. Hence, PU/propolis/neem oil scaffold may attract the protein adhesion resulting in the enhanced cell adhesion for new tissue growth.
Conclusion
In this study, PU scaffold containing propolis and neem oil was developed using electrospinning technique. The morphology of the electrospun PU/propolis and PU/propolis/neem oil mats exhibited reduced fibre and pore diameter than the pristine PU control. The change in the chemical structure of the electrospun composites was identified by peak shifting of CH band peak with hydrogen bond formation. The contact angle measurements revealed that the PU/propolis mat was found to be super hydrophilic in nature and the PU/propolis/neem oil mat was observed to be hydrophilic in nature. Further, TGA and AFM analysis showed that the prepared PU/propolis and PU/propolis/neem oil mats exhibited higher thermal stability and decreased surface roughness than the pristine PU. Moreover, the developed composites displayed decreased in blood compatibility through a decrease in blood clotting time. The results of haemolytic assay observed that the electrospun PU/propolis and PU/propolis/neem oil mats exhibited better safety to RBC than the pristine PU. Hence, this study deciphered that the desirable characteristics of the newly developed scaffold might be used as a potential candidate for tissue engineering applications. However, the limitations involved in the study are the components that are found to be changed with origin, and this may affect the quality of the fibres developed. Although the developed scaffold has desirable physicochemical and blood compatible properties needed for the tissue engineering applications, its cytocompatibility properties against the host tissues are to be studied to analyse its potential. This is important for knowing how the added constituents influence the target cells for new tissue growth.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author(s) received financial support from the Ministry of Higher Education Malaysia with the Grant nos. Q.J130000.2545.17H00 and Q.J130000.2545.20H00.
