Abstract
1. Introduction
Tissue engineering is a multi-disciplinary field that involves biologists, materials scientists, mechanical engineers, physicists and chemists. This therapeutic strategy offers an attractive solution that can overcome the shortcomings of the current treatment options for diseased organs. The underlying concept of tissue engineering is to regenerate the diseased tissue with new tissue. The emergence of nanotechnology has radically improved tissue engineering approaches, as it has now been recognized that biological systems respond well to nano-dimensional structures [1]. Cellular response occurs at multiple levels - at the microscopic and nanodimensional levels as well as at the molecular level. The extracellular matrix abounds with nano-dimensional structures that control the cell-fate processes. Therefore, providing a nano-scale topography mimicking the native extracellular matrix can lead to the better growth and proliferation of cells [2]. The integration of nanoscale carrier systems that release signalling molecules in a preprogrammed manner to produce a specific response from the cells adds one more dimension to the role of nanotechnology in tissue engineering. The manipulation of the mechanical properties of the ECM mimics is another aspect that has been found to profoundly influence the functional expression and survival of cells. Different strategies integrating geometry, materials, mechanics and chemical moieties have been developed to promote the effective regeneration of cells. Electrospinning, solvent casting, particulate leaching, microsphere-based sintering, freeze drying, phase separation and self-assembled ensembles have all been attempted to manipulate the geometry of the substrate that acts as a scaffold to support cells [3]. In many cases, a single geometry may not be ideal for achieving functional tissue regeneration. For instance, a combinational strategy of electrospinning and freeze-drying was successfully utilized for the preparation of a poly(glycolic acid) (PGA) nanofibre-reinforced collagen sponge for cardiac tissue regeneration [4, 5]. Apart from the improvement in its mechanical properties, the nanofibre content reduced the shrinkage of the collagen sponge when it was taken to the culture medium. The incorporation of nanoparticles to improve a specific property of an ECM mimic has also been attempted. For example, titania nanoparticles (TiO2) have been found to improve the adhesion of cells to the surface of the substrate material. Human mesenchymal stem cells (hMSC) have demonstrated very good adhesion on nanoporous titania with a particle size within the range of 12 nm. The adhesion and proliferation rate of hMSC over TiO2 nanoparticles surpassed their performance on natural and synthetic polymer coatings. Likewise, titania-coated microwell-based microfluidic devices represent an exciting approach for stem cell culturing because the proliferation rate is high [6]. Designing appropriate experimental models to assess the efficiency of tissue engineering strategies is also important in arriving at unbiased conclusions. The results obtained from static cell cultures for assessing the potential of an ECM mimic have been found to be of little use in biological conditions, as it does not mimic the
2. The evolution of tissue engineering
Three main components have been identified as crucial for successful tissue engineering. These components are popularly referred to as the ‘tissue engineering triad’, and comprise the scaffolds, cells and growth factors [8]. The earliest efforts in tissue engineering placed emphasis on the choice of material used as a scaffold. Later, tailoring the surface functionalities and the geometry of the scaffold garnered much attention in developing a scaffold that mimics the topography of the extracellular matrix. The integration of the nano-geometrical features and appropriate functional groups on the biomimetic scaffold produced better results when compared to the two-dimensional cultures initially attempted [9, 10, 11]. This particular aspect continues to be the most investigated facet in tissue engineering at present. The next generation of tissue-engineered constructs incorporated growth factors, as it was realized that the availability of the right quantity of chemical factors at the appropriate time was imperative for maintaining the functionality of the cells seeded in the scaffolds. In order to ensure the availability of these factors for a longer duration, the integration of nanocarriers containing these growth factors in the scaffold came into vogue. These attempts were especially effective for cartilage [12, 13], bone tissue engineering [14] and angiogenesis [15]. The type of growth factors and nanotopographical cues required for the successful regeneration of a tissue are also dependent on the cell type, number and age [16]. The emergence of stem cell technologies has opened up new vistas in tissue engineering, as the differentiation of these cells can be regulated through the suitable control of the culture
In recent years, a fourth facet has emerged as a crucial factor determining the functional regeneration of a tissue. It has now been recognized that the pressure and mechanical forces acting on a cell determine its functional expression by activating cell signalling pathways by a process now referred to as ‘mechanotransduction’ [18]. This knowledge has resulted in the development of composite scaffolds - cross-linked and gradient scaffolds that possess unique mechanical properties aiding the functional regeneration of the tissue. Such strategies are becoming increasingly popular for the regeneration of cartilage [19], cardiac tissue [20] and bone [21]. Thus, the original tissue engineering triad has now transformed into a ‘tetrad’ with the introduction of mechanical cues and mechanotransduction for effective tissue engineering. The field of tissue engineering continues to evolve, with more external stimuli such as ultrasound and electric impulses being explored as additional factors that can stimulate and activate cells to grow, proliferate and establish cell-cell communication for effective functioning [22]. A new paradigm in tissue engineering that has emerged in recent years is the concept of the ‘living scaffold’. The cells are normally seeded on a 3D scaffold and allowed to permeate and infiltrate into the interior. However, according to the concept of ‘living scaffolds’, the cells are entrapped within the scaffold and allowed to extend in all dimensions [23]. Such attempts have yielded especially encouraging results in the field of bone [24] and liver tissue engineering [25]. The living scaffolds that integrate the concepts of nano-geometry, growth factor delivery, stem cells and mechanotransduction may very well represent the next generation of tissue engineering constructs.
3. Scaffold design strategies
One of the prime objectives of tissue engineering is to identify an ideal scaffold that will support the adhesion, growth, expansion and functional expression of cells. However, there is no universal scaffold or scaffold design that can support all cell types, as each cell requires a specific environment to be functionally active. The selection of the scaffold material primarily depends on the cell type, though several other factors may also influence the choice. Non-biodegradable scaffolds are not very effective in achieving the functional regeneration of cells, as the persistent scaffold will retard the ability of the cells to generate and remodel their own extra-cellular matrix. Hence, biodegradable materials are preferred for scaffold fabrication. Some of the popular polymers in this category include poly(lactide-co-glycolide) [26], poly(L-lactide) [27], poly(glycolide) [28], poly(caprolactone) [29], poly(ethylene oxide) [30], chitosan [31] and alginate [32], etc. The key is to choose a polymer whose rate of degradation will match the proliferation rate of the cells on the scaffold. If the scaffold degrades faster than the cells can divide and cover the entire scaffold, then it may lead to loss of cell adhesion. If the scaffold degrades very slowly, then the functions of the cells may be altered. Many parameters influence the degradation rate of a polymer. These include molecular weight, hydrophobicity, crystallinity and the porosity of the scaffold materials [33, 34]. As most of the biodegradable polymers degrade via hydrolysis, regulating the accessibility of water to the scissile bond can serve to control the degradation rate of the polymeric scaffold. The incorporation of segments with greater hydrophobicity serves to retard the degradation rates of polymers. For example, increasing the lactide content in PLGA copolymer results in reduced degradation rates due to the enhanced hydrophilicity imparted to the polymer chains by the methyl side-group of the lactide moiety [35].
The degradation mode may be either through surface erosion or bulk erosion. In the case of surface erosion, the degradation starts from the surface layer and gradually proceeds towards the core. This results in continuous dimension changes. In the case of bulk eroding systems, water permeation into the core is quick, and as a result the degradation occurs throughout the matrix leading to a rapid loss of its mechanical properties. For tissue engineering applications, surface erosion is preferred due to gradual reduction in the scaffold stiffness/water permeability ratio. The surface degradation can be promoted by introducing enzyme cleavable cross-links into the scaffold material. As enzyme-catalysed hydrolysis requires the proper orientation and binding of the scissile group to the active site, the degradation tend to take place through the surface erosion mode. A dextran hydrogel scaffold for nerve tissue engineering, cross-linked with MMP-2 (matrix metalloproteinase 2) enzyme-specific sequences, was found to exhibit surface degradation due to the confinement of the MMP-2 to the surface of the hydrogel [36]. Similar strategies have been employed with poly(ethylene glycol) polymers containing plasmin or collagenase-specific sequences [37].
One of the concerns in using biodegradable polymeric scaffolds is that they are susceptible to infections by pathogens. Graphene, a carbon nanomaterial that has a sheet like arrangement of carbon atoms in a monolayer, has recently been reported to be an excellent scaffold material [38]. Graphene is flexible, tough with excellent mechanical properties, and can be easily transferred to any substrate [38]. It is also reported to be biocompatible and produces no toxic products. In addition, the large-scale production of graphene is possible and no further fabrication techniques are required to pattern the graphene scaffold [39, 40]. Human mesenchymal cells, when cultured on a graphene scaffold, were found to differentiate into osteoblasts even without the introduction of growth factors such as bone morphogenic factor (BMP) [41]. The presence of graphene did not alter the morphology of the stem cells. Interestingly, it was observed that amorphous carbon films and highly-oriented pyrolytic graphite also supported the proliferation of the mesenchymal stem cells but did not induce their differentiation in the absence of growth factors. This unique property of graphene to induce the differentiation of stem cells has been attributed to its high Young's modulus and its ability to undergo out-of-plane deformations and sustain high lateral stress, unlike other carbon-based materials [41]. The mechanical properties of graphene contribute the optimum amount of cytoskeleton tension, leading to the formation of focal anchor points. Recently, 3D graphene foam was employed as a scaffold for neural tissue engineering using neural stem cells. The electroactive property of graphene was exploited to differentiate the neural stem cells into astrocytes and neurons [42]. The 3D construct was found to be superior to the 2D graphene sheets in the functional expression of proteins in the differentiated cells [43]. Carbon nanotube-based hydrogel scaffolds have also been successfully used to create a beating cardiac tissue in the laboratory. The excellent mechanical and electrical properties of carbon nanotubes enable them to guide, orient and stimulate the cardiomyocytes [44]. Apart from the chemical nature of the polymer, the geometry, topography and porosity of the scaffold determine the accessibility to water and, hence, the degradation rate.
The geometry of the scaffold also plays an important role in the oriented growth of the cells. The native extracellular matrix contains a complex meshwork of collagen fibrils, structural proteins with specific adhesive motifs like laminin, fibronectin, elastin, etc., and glycosaminoglycans [45]. These components regulate the oriented cell growth as well as the various cell-fate processes. The fibrous mesh-like network formed by the collagen is considered crucial for providing contact guidance to the cells and direct the orientation of cells in a particular direction. Accordingly, mimicking the fibrous morphology of the collagen network is one of the most widely explored strategies in scaffold design. The fabrication of a fibrous scaffold is mainly carried out using a simple and versatile technique known as ‘electrospinning’ that was originally developed in the early 1900s [46]. A typical electrospinning process involves the application of a high potential to a polymer solution taken in a spinneret to overcome the surface tension and elongate the polymer jet into fine fibres that are deposited on a metallic collector. The dimensions of the fibres can be tuned by suitably altering the system as well as the process parameters. The system parameters that influence the nature of the electrospun fibres are the molecular weight of the polymer, the volatility of the solvent system employed, the viscosity of the polymer solution, the dielectric constant of the solvent and the conductivity of the solution. The process parameters that can regulate the nature of the fibres are the applied potential, the distance between the needle tip to the collector (tip-to-target distance), the flow-rate of the polymer solution, the needle gauge size, temperature and humidity [47]. Electrospun scaffolds from a wide-range of polymers, metals and even ceramics, have been reported in the literature [48]. Figure 1 shows a typical electrospinning set-up and different orientations of electrospun nanofibres.

A typical electrospinning set-up. The polymer to be spun is loaded as a solution in a syringe and its flow is controlled using a syringe pump. A high voltage supply is connected between the tip of the needle and the metal collector placed at a definite distance away from the syringe.
The orientation of the fibres can be controlled by using a dynamic collector instead of a static collector, which generally leads to the formation of random fibres. In the case of the dynamic collector, the speed of rotation is an additional parameter that needs to be optimized to obtain uniform defect-free highly-oriented fibres. Different collector geometries have been employed to obtain defect-free fibres with the desired dimensions and orientations. These include rotating drum, rotating wire drum, rotating tube, parallel collectors, knife-edge collectors, disc, electrode arrays, ring collector and multiple spinneret designs. Each of these designs possesses unique advantages as well as disadvantages. A few design geometries that have been reported in the literature are depicted in Figure 2.
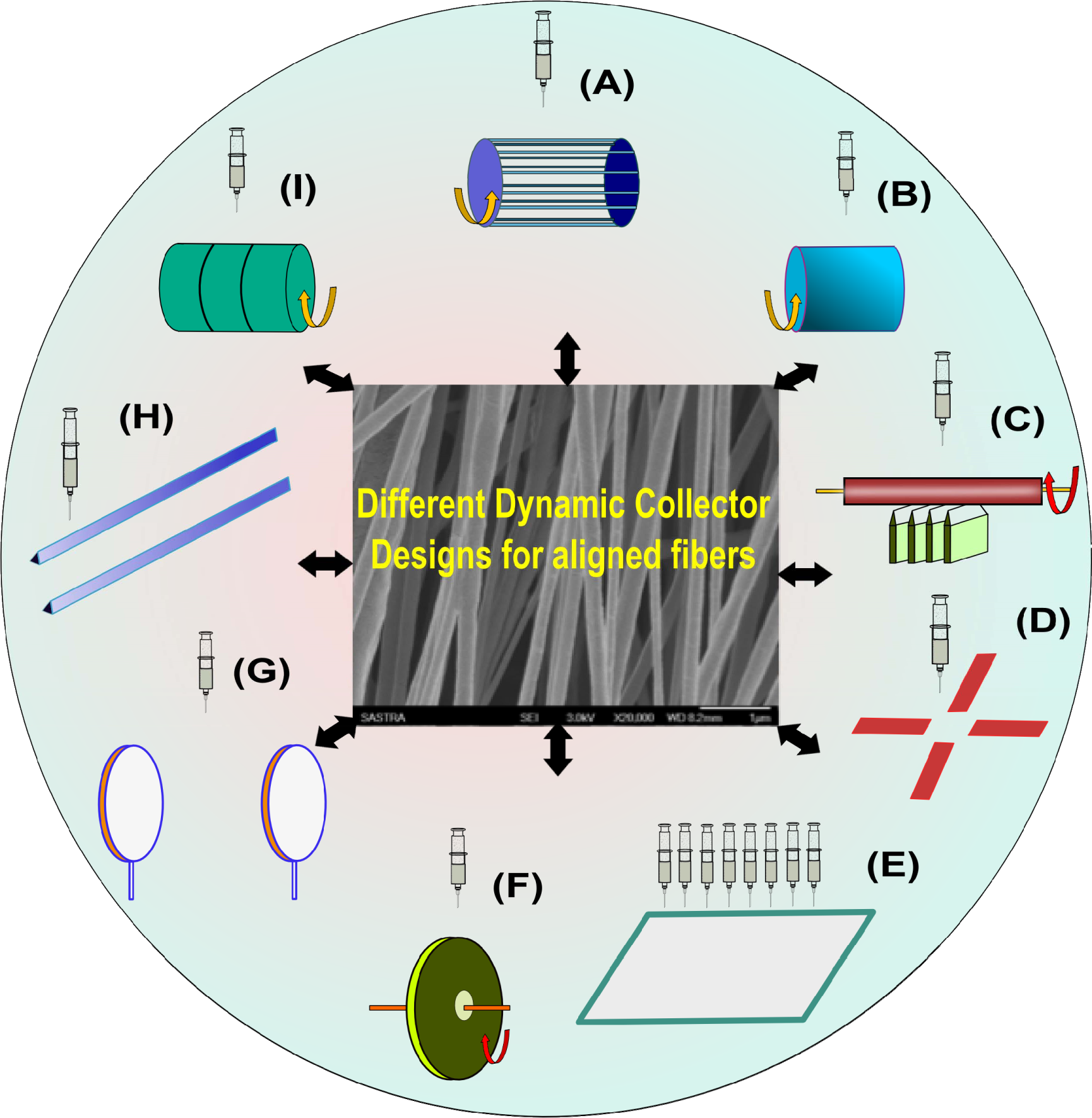
Different dynamic collectors employed for obtaining aligned fibres: (A) rotating wire drum collector; (B) rotating drum collector; (C) rotating tube collector on knife-edge; (D) counter electrode array; (E) multiple spinneret; (F) disc collector; (G) parallel ring collector; (H) parallel electrode collector; (I) rotating drum with wire wound over.
The aligned nanofibrous scaffolds have been demonstrated to promote oriented cell growth along the direction of alignment. This has been particularly effective in the case of endothelial cells [49], cardiomyocytes [50] and nerve cells [51]. Apart from the orientation of the cells, aligned fibres have also been reported to enhance the expression of functional proteins. An aligned nanofibrous scaffold of poly(caprolactone) seeded with Schwann cells was found to enhance the expression levels of early myelination markers, like myelin associated glycoprotein as well as Schwann cell maturation such as myelin protein zero [52]. Though the exact relationship between fibre alignment and gene expression is yet to be deciphered, the results obtained using aligned scaffolds suggest that the cell orientation and functional expression of proteins by the cells is superior on these scaffolds when compared with randomly-aligned scaffolds. The fabrication of multi-layered nanofibrous scaffolds has also been achieved through co-axial electrospinning [53]. Two coaxial spinnerets filled with different polymer solutions are subjected to an applied electric field to collect the fibres. The system and process parameters can be optimized to achieve the desired fibre dimensions and orientation.
The fibre dimensions can also influence the cell proliferation, migration and functional expression. Nanofibres can support cell adhesion, growth and proliferation. Hence, a majority of electrospun scaffolds prefer achieving nano-dimensional fibres. One of the challenges in using a nanofibre scaffold is the reduction in porosity due to the compact packing of the nanofibres. This problem is especially pronounced in the case of aligned scaffolds compared with the case of randomly-oriented nanofibrous scaffolds. Adequate porosity is essential for sustaining the nutrient and oxygen requirements of the cells seeded on the scaffold. Interconnected pores will facilitate the infiltration of the cells to establish cell-cell communication leading to a functional tissue network. Microfibers have been found to have larger pores, thereby enabling cell infiltration and migration. As such, several approaches have attempted to integrate the advantages of both nanofibres and microfibers in a single scaffold by developing a hybrid scaffold comprising both nanofibres and microfibers. Such strategies have been found to be effective in inducing angiogenesis [54] as well as bone tissue engineering [55] using nano-microfibrous scaffolds fabricated from starch-poly(caprolactone). A unique combination of a low temperature solid freeform fabrication (SFF) technique and electrospinning was used to fabricate a multi-layered 3D collagen scaffold [56]. This structure had alternating layers of micron-sized fibres obtained through SFF followed by an electrospun nanofibrous layer. This hierarchical scaffold supported the adhesion, spreading, proliferation and penetration of mesenchymal stem cells. Modifying the electrospinning parameters to achieve better porosity and larger pore dimensions has also been attempted. Increased flow-rates were found to increase the pore dimensions of synthetic human elastin nanofibrous scaffolds that were found to be superior to those scaffolds fabricated using low flow-rates for skin tissue engineering applications [57]. The difference in the cell response to both scaffolds was attributed to the differences in the pore dimensions.
The aligned fibrous scaffolds provide superior contact guidance but are limited by poor cell infiltration. In order to overcome this disadvantage, several modified electrospinning strategies have been employed. ‘Sacrificial fibres’ are employed to create additional pores in the scaffold architecture with the passage of time. In this strategy, an aligned fibrous scaffold comprising a physical blend of two different polymers is fabricated. One of the polymers, termed as the ‘sacrificial polymer’, should exhibit good solubility in water. When the cells are seeded onto this scaffold, they initially adhere and spread on the surface. With the passage of time, the sacrificial fibres start degrading, resulting in the creation of additional pores that facilitate the penetration of the cells into the interior, thereby forming a 3D cellular network. Figure 3 depicts the principle of sacrificial fibres.

Generation of larger pores through sacrificial fibres. The cells are seeded on the surface of a nanofibrous scaffold made from a water-insoluble and a water-soluble polymer. On immersion in water, the water-soluble polymer dissolves, leading to an increase in pores that facilitate cell infiltration.
This strategy was successfully employed to culture mesenchymal stem cells using a blend of poly(caprolactone), acting as the structural component, and poly(ethylene oxide) as the sacrificial component [58]. The blend scaffold showed the proliferation of cells throughout the scaffold, whereas the scaffold comprising poly(caprolactone) alone had cells only on the periphery of the scaffold. Yet another group used an aligned nanofibrous scaffold, comprising the same blend of polymers, to successfully repair rotator cuff tendon tears in rat models [59]. Similarly, poly(ethylene glycol) was employed as a sacrificial template in combination with glutaraldehyde cross-linked gelatin fibres. These scaffolds exhibited excellent penetration of the fibroblasts cells compared with conventional electrospun scaffolds [60]. A composite scaffold comprising poly(caprolactone), collagen I and hydroxyl apatite was successfully employed for bone tissue engineering using poly(ethylene oxide) as the sacrificial nanofibres [61]. In another variation of the sacrificial fibre concept, polymer solutions containing water-soluble particles such as sucrose and sodium chloride are electrospun followed by leaching in an aqueous medium, when the water soluble particles dissolve away leaving behind a porous matrix. This strategy was successfully demonstrated using poly(glycolic acid)-sucrose electrospun nanofibres that supported the deeper infiltration of fibroblasts into the scaffold [62].
A recent innovation in the field of electrospinning is the development of focused, low-density, uncompressed nanofibre (FLUF) scaffolds that resemble cotton balls. This scaffold retains the mechanical properties and the structural framework, but has in addition larger pores when compared with the nanofibrous mats obtained through conventional electrospinning. These FLUF scaffolds are made using poly(caprolactone) and have been demonstrated to exhibit the superior spreading, infiltration and proliferation of INS-1, a pancreatic beta insulinoma cell line [63]. The cotton-ball morphology was achieved by using a multiple needle probe array on a spherical disc as the collector instead of the traditional flat-plate collectors.
The ultrasonication of electrospun nanofibres of poly(L-lactic acid) at 4°C before cell seeding has been reported to enhance the pore dimensions of the scaffold and resulted in the increased infiltration and density of the fibroblasts when compared with the untreated scaffold [22]. However, the optimization of the ultrasound frequency, power and duration may be necessary for each type of scaffold as there exists a risk of the fragmentation of the polymer fibres on exposure to ultrasound.
Cryogenic electrospinning is another modification developed to improve the pore dimensions in electrospun nanofibrous scaffolds. The principle involved in this technique is to deposit polymer nanofibres and ice crystals over a cold collector. The removal of the ice crystals results in pores with large dimensions [64]. However, it is important to exert a high degree of control on the temperature and humidity of the system to achieve the porous structures. In a variation of the same concept, the polymer nanofibres were frozen in liquid nitrogen, thereby inducing a phase separation between the polymer and the solvent. Accordingly, this technique is also referred to as ‘thermal-induced phase separation’ (TIPS) and is driven by low temperatures. The removal of the solvent through vacuum evaporation resulted in highly porous fibres. This variant of electrospinning was successfully used for obtaining highly porous nanofibres of poly(acrylonitrile) and poly(caprolactone) [65]. Phase separation-induced porous fibre generation has also been achieved using a non-solvent, and the technique is referred to as ‘non-solvent-induced phase separation’ (NIPS) [66]. To achieve porous fibres, the electrospun polymer is collected in a trough containing a non-solvent for the polymer resulting in polymer precipitation. Porous poly(caprolactone) fibres were fabricated by this technique using water as the non-solvent. The presence of water vapours during the NIPS process has also been reported to contribute to phase separation, the process being referred to as ‘vapour-induced phase separation’ (VIPS) [67]. An extension of the NIPS - or immersion precipitation - has been used to generate hollow poly(acrylonitrile) nanofibres with a porous outer layer. This was achieved by electrospinning an outer layer of poly(acrylonitrile) over an inner core of poly(methyl methacrylate). The solvent in the collector was a solvent for poly(methyl methacrylate) and a non-solvent for poly(acrylonitrile). This resulted in the selective dissolution of poly(methyl methacrylate) leading to formation of a hollow fibres. This technique was named ‘core-shell electrospinning’ and could be explored for a myriad of applications [68].
Though a major portion of the research on tissue engineering constructs has focused on electrospinning, other techniques such as particulate leaching, freeze thaw and sintering, have also been investigated for the development of non-fibrous scaffolds for tissue engineering applications. Particulate leaching involves the incorporation of porogen (i.e., water-soluble particles of desired dimensions in a polymer solution), which is insoluble in water. This sol is cast into a film and the solvent is allowed to evaporate, following which the film is immersed in water for leaching off the porogen. This results in the formation of pores. The porogen:polymer ratio can be varied to achieve interconnectivity. Commonly-employed porogens are sodium chloride, sugar and ammonium carbonate, etc. [69]. Supercritical carbondioxide has also been employed for creating pores [70]. In spite of the simplicity of the technique, the randomness of the pores formed and the rapid loss in the mechanical properties have been major limiting factors. Another technique employed in the fabrication of porous non-fibrous scaffolds is the freeze-thaw method, where a polymer is subjected to alternate cycles of freezing and thawing. The temperature shocks induce the phase separation of the polymer chains resulting in the formation of pores. The number of freeze-thaw cycles, the duration and the temperatures employed can be varied to achieve desired porosity, pore dimensions and interconnectivity [71]. Poly(vinyl alcohol) has been extensively employed for the fabrication of scaffolds using the freeze-thaw technique. For tissue scaffolds that require sufficient compressive strength, as in the case of hard tissue engineering, the technique of employing sintered microspheres has been popular. This method involves the preparation of polymer microspheres using the emulsion method. Microspheres of uniform dimensions are filled in a mould that is then heated to a temperature just above the glass transition temperature of the polymer. This results in a fusion of neighbouring spheres, thereby forming a porous network with excellent compressive strength. Sintered scaffolds of poly(lactide-co-glycolide) have been found to be excellent substrates for bone tissue engineering [72]. Figure 4 shows the scanning electron micrographs of several non-fibrous scaffolds that have been used for engineering various tissues.

Non-fibrous scaffolds obtained by: (A) Particulate leaching; (B): Freeze-thaw; (C) Sintering.
4. Incorporation of growth factors
Cells respond to the presence of growth factors and soluble factors, such as cytokines for growth, proliferation and migration. Therefore, the incorporation of these factors in the scaffold can aid the accelerated growth and maturation of the cells. However, the constant presence of growth factors may not be prove beneficial, as cells require a particular amount of growth factors at a specific time. This can be regulated by introducing drug delivery systems into the scaffolds that enable the gradual release of the growth factors. A hydrogel system delivering insulin-like growth factor-1 was found to promote the survival and function of cardiomyocytes [73]. The introduction of hydroxy apatite into microspheres has been shown to be effective in bone tissue engineering applications [74]. Similarly, vascular endothelial growth factor and platelet-derived growth factors have been found to be effective in promoting angiogenesis [74]. Gene delivery systems such as poly(ethylene imine) have been employed to deliver bone morphogenic proteins to scaffolds seeded with cells to promote bone regeneration [75]. A major challenge is to identify the right markers and correct rate of delivery of these markers for accelerated cell proliferation. The use of peptide motifs to elicit specific responses from cells has been explored by employing self-assembled peptide amphiphilic fibres as scaffolds or by electrospinning peptides into fibres (Figure 5). This strategy retains the benefits of the nanofibrous topography along with the additional advantage of having a distribution of cell-specific recognition motifs on the surface [76].

Peptide-incorporated polymer nanofibres. The white dots on the fibre surface represent the peptide structures distributed throughout the surface.
Yet another facet in incorporating growth factors in tissue engineering constructs is the localization of these factors. A homogeneous distribution of the factors throughout the scaffold may prove ineffective, as the cells prefer anisotropic distribution. Hence, a combination of biofabrication techniques along with drug delivery is the emerging paradigm in the field of tissue engineering.
5. Self-assembled peptide nanofibres for tissue engineering
Another interesting strategy that has emerged in the recent years is the use of self-assembled peptide constructs as tissue engineering scaffolds. The ease of formation and reproducible structures obtained as a result of self-assembly have been instrumental in the widespread interest generated for this category of scaffolds. It is also possible to introduce biorecognition motifs in these scaffolds to achieve the desired response from the cells. The self-assembly of peptides into fibres was an accidental discovery by Zhang during the course of his research with a novel protein zuotin, which has a highly repetitive sequence, n-AEAEAKAKAEAEAKAKAKAK-c [77]. Based on this observation, two more similar, repetitive sequences were identified from zuotin, namely RADA16 with a sequence RADARADARADARADA and EAK16 with a sequence EAKAEAKAEAKAEAKA, both of which formed nanofibres rich in β sheets [77]. Since the nanofibrous architecture formed by these peptides resembles the extracellular matrix, they can be used as scaffolds for 3D tissue cultures and tissue engineering. The nature of the self-assembled structures formed depends on the amino acid sequence as well as on the self-assembling conditions. Bio-inspired sequences have been synthesized and such synthetic sequences have been successfully employed for tissue engineering. Hartgerink et al., in a seminal work, synthesized a peptide amphiphile with an N-terminus modified with an alkyl chain that self-assembled in an aqueous medium to form cylindrical micelles [78]. The peptide sequence consisted of several cysteine residues that imparted structural stability to the self-assembled structure through the formation of disulphide bridges. Glycine residues imparted some level of flexibility to the structure, while a phosphoserine moiety in the sequence aided mineralization by serving as a nucleating centre for the formation of calcium phosphate. An RGD (arginine-glycine-aspartate) motif aided the adhesion of cells through binding interactions with the cell surface receptor integrin. This synthetic peptide amphiphile promoted osteoblast adhesion, growth, proliferation and bone formation. A synthetic peptide amphiphile with RGD-glutamine-alanine-glycine was synthesized by Hosseinkhani et al., to load the angiogenic growth factor basic fibroblast growth factor (bFGF) for sustained release during tissue regeneration [79]. The same peptide amphiphile system was demonstrated to enhance the proliferation and differentiation of mesenchymal stem cells into an osteogenic lineage [80]. This study was further extended with the use of poly(glycolic acid)-incorporated collagen sponge along with the same synthetic peptide amphiphile system cultured in a perfusion bioreactor to form a 3D hybrid scaffold. The hybrid scaffold, when implanted subcutaneously in rat models, was found to form bone homogenously throughout the scaffold [81]. The incorporation of the growth factor bFGF in the hybrid scaffold enhanced the levels of the bone marker osteocalcin and alkaline phosphatase activity, which was not observed in the hybrid scaffolds without bFGF [79]. In a recent work, mucosal cells loaded in a self-assembling peptide scaffold Puramatrix® (RADA16) were successfully employed for middle ear tissue engineering [82]. A recent review of self-assembling proteins and peptides for regenerative medicine has discussed in detail the wide-ranging applications of self-assembling peptide scaffolds for cartilage, bone, nerve, cardiac and tooth tissue engineering [83–87].
6. Mechanical cues
In the biological milieu, cells are constantly exposed to different stresses – tensile, compressive and shear. The magnitude, frequency and duration of these stresses dictate the cell response. Tissues, like bone, cartilage, arteries and the heart, have been found to express specific proteins in response to the type of stress they experience [20]. The process of converting mechanical stimuli into biochemical signals is known as ‘mechanotransduction’ and is responsible for cell-fate and function [13]. Recently, it has been identified that the stiffness of the extracellular matrix drives the cell responses [88] and that the cells remodel their extracellular matrix in order to modulate their responses. Biomimic scaffolds also have been shown to contribute to the differential expression of genes due to the variation in their stiffness [89]. Thus, the gene expression of cells in a highly cross-linked or stiff scaffold will be different from those cultured on a more flexible scaffold of the same polymer and geometry. The elastin expression of MC3T3 cells cultured on poly(hydroxy butyrate-co-hydroxy valerate) scaffolds with different stiffness was found to be higher in the stiffer scaffold, probably to offset the stiffness of the scaffold [90]. The cells cultured on static and dynamic cultures have been found to differ in their functional expression [91]. The concept of dynamic culture employing bioreactors has emerged in recent years to exploit the beneficial aspects of mechanical stimuli on cell proliferation [92]. Such dynamic systems have been employed extensively for engineering liver, bone, cartilage and cardiac tissues [93, 94]. Embryonic stem cells cultured under dynamic conditions were found to exhibit accelerated hematopoietic differentiation that has been attributed to the increased expression of cell adhesion-related genes, extracellular matrix proteins and growth factors [91]. Similarly, laminar flow and cyclic flexure accelerated the formation of cardiac tissue from bone marrow-derived mesenchymal stem cells [95]. Alternately, strategies employing the extensive cross-linking of the scaffolds [96], the incorporation of reinforcing nanoparticles such as carbon nanotubes [97], creating a thickness gradient in the scaffolds [98], have also been found to be beneficial for cell growth and expression. Though many pathways have been suggested to be involved in response to mechanical stimuli, this area remains wide open as the complete mechanism involved in mechanotransduction remains to be unravelled, thereby leaving room for further research.
7. Cells
The discovery of the therapeutic implications of stem cells has contributed to the emergence of regenerative medicine as a promising candidate for healthcare. The ability of stem cells to differentiate into multiple lineages has been used in tissue engineering different tissues by merely altering the culture conditions. The ethical issues associated with the use of embryonic stem cells had dampened the progress made in cell-based therapies, but the discovery of the potency of adult stem cells and induced pluripotent cells have opened up an entirely new era of regenerative medicine [99]. As the direct introduction of stem cells in a specific region of the body results in the poor retention of stem cells, their integration into 3D scaffolds has served to realize its potential for tissue engineering applications [100]. Most of the work employing stem cells has focused on evaluating the differentiation potential of the stem cells [101]. Few attempts have been reported of employing scaffolds to ensure the expansion of stem cells over a period of time without undergoing any differentiation [102]. However, the long-term evaluation of such stem cell-based constructs has to be investigated
8. Living scaffolds – the emerging paradigm
Though a wide-range of strategies have been employed to develop an ideal scaffold that possesses the optimum dimensions, porosity, topography and mechanical properties, the clinical success of such constructs remains elusive. The use of a pre-fabricated scaffold followed by cell seeding has several shortcomings. The seeding is done on the surface and the penetration of the cells into the scaffold is random, non-homogenous, and does not usually span the entire thickness of the scaffold. Multiple steps involved in seeding and incubation increase the time-span as well as the cost of the process [16]. Another major shortcoming of conventional solid scaffolds is that it does not mimic the multi-cellular architecture found in organs. For example, each organ has its own network of blood vessels. This aspect is difficult to reproduce in current tissue engineering scaffolds. To overcome some of these difficulties, the concept of developing cell-encapsulated tissue constructs was introduced in the early 2000s. These techniques are collectively referred to as ‘biofabrication tools’, and the cell-encapsulated scaffolds are referred to as ‘living scaffolds’. The biofabrication techniques encompass two major categories – jet-based and non-jet-based techniques. The jet-based techniques that have been explored include cell electrospinning, cell printing, laser-guided cell writing, biojetting and threading and bio-electrospraying. Apart from these, microfluidic techniques have also been used to obtain living scaffolds.
Cell electrospinning was first reported in 2005 and it attempts to integrate the advantages of a biomimetic fibrous scaffold and the uniform distribution of cells throughout the scaffold that is the hallmark of biofabrication [103]. The typical experimental set-up used for electrospinning is slightly modified to achieve cell electrospinning. The cell-laden fibres are collected in a grounded rotating collector that is immersed in a cell-culture medium to keep the cells alive. Earlier trials had involved an additional intermediate step of immersing the collector in a cross-linking solution to maintain the structural integrity and retain the cells [104], but current attempts employ thermo-responsive polymers that undergo a sol-gel transition at 37°C, the temperature at which the cell culture medium and collector are maintained [105]. Variations in the form and number of spinnerets employed (single, coaxial or multiple) and the type of cells (human cells or micro-organisms) have also been reported [106, 107]. The use of co-axial spinnerets facilitates the fabrication of distinct layers that contain different cell types, thereby leading to a functional organ. For example, the reconstruction of a blood vessel would require an inner endothelial layer followed by a smooth muscle cell layer, which can be achieved through this technique. The choice of the biopolymers is restricted to alginate, xanthan gum and carrageenan, etc. [108], which are considered cell-friendly and can be electrospun without the use of harsh organic solvents that may destroy the cells. Concerns as to the ability of the cells to survive the high-applied field also remain. Preliminary investigations have, however, revealed that the cells retain their viability after cell electrospinning, implying the potential of this technique for regenerative medicine applications. Further in-depth investigations on the gene expression and functional aspects of the cells need to be carried out to establish the efficacy of this technique.
Bio-electrospraying - or bio-electrohydrodynamic jetting - is another technique that employs a high electric field to form micro-droplets containing cells using a cell suspension passed through a large bore needle [109]. One advantage of this method is that it can be used for low-density as well as high-density cell suspensions, unlike other methods of biofabrication. In spite of the high electric field employed, it is reported that the viability of the cells largely remains satisfactory and is about 70% [110]. However, the reduction of the viability of the bio-electrosprayed cells has not been conclusively ascribed to either necrosis or apoptosis. Recently, it has been reported that bone marrow stem cells that have been subjected to bio-electrospraying not only exhibited good viability at around 85% but also retained their osteogenic, chondrogenic and adipogenic differentiation potential [111]. However, potentials beyond 15 kV were found to be detrimental to the survival of the cells. Aerodynamically-assisted biojetting is an analogous biofabrication technique that uses pressure instead of an electric field to generate the cell-containing droplets and has been successfully demonstrated to deposit hematopoietic stem cells without compromising their viability [112]. As this method avoids the use of a high electric field, it can compete with the electric field-driven techniques, such as bio-electrospraying and cell electrospinning, for supremacy in biofabrication. A hybrid technique involving a combination of bio-electrospraying and aerodynamically-assisted biojetting has been used to pattern human embryonic kidney cells expressing green fluorescent protein [113]. The hybrid protocol enabled the achievement of a stable jet due to the combined effect of an electric field and pressure. In an extension of the aerodynamically-assisted biojetting, a new technique known as pressure-assisted spinning (PAS) has been reported. In this method, coaxial nozzles are employed with one containing the cell suspension and the other a polymer solution. The application of pressure drives the formation of the cell-encapsulated fibres of the polymer. This method was demonstrated for the first time employing rat aorta smooth muscle cells encapsulated in medical grade poly(dimethoxysilane) [114].
Cell printing techniques employing jet formation have been explored for more than a decade now. Inkjet printing is among the earliest cell printing techniques to be developed. The advantage of this technique is that it is possible to achieve the precise positioning of cells in any location on a substrate. Hence, this method is also referred to as ‘bio-printing’. With this method, a cell suspension is loaded into the print head and a droplet containing a cell is created either through piezoelectric actuation or a thermal or acoustic actuator. The viability of the cells has been reported to be in the vicinity of 90% [115]. A key parameter that needs to be regulated is the viscosity of the cell suspension. Highly viscous solutions may affect the flow through the nozzle. Thus, very high cell densities cannot be handled with this technique, which remains its major limitation. Figure 6 depicts the formation of a cell containing a droplet through different actuating mechanisms.

Inkjet printing employing thermal, piezoelectric and acoustic wave actuators for the formation of a droplet. The left panel depicts a typical thermal or piezoelectric actuator system, while the right panel depicts an acoustic wave actuator system.
Recently, fast photocuring acrylate-based polymers containing fibrosarcoma cells have been printed using this technique, with precise positioning on the designated wells [116]. This method can also be employed to develop 3D arrays of cells of the same type or different types, DNA, proteins and other biomolecules. In a novel strategy, a commercial inkjet printer was employed for bioprinting fibroblasts on a 3D substrate. The shear stress applied during the droplet formation led to the development of transient pores about 10 nm in diameter that were used to introduce fluorescent actin into the cells that aided in the visualization of the cytoskeleton dynamics [117]. The results from this study have opened up new avenues for the nanopatterning of extracellular matrix proteins onto specific locations on a substrate, which can provide unique cellular microenvironments for tissue engineering applications.
Laser-assisted bioprinting (LAB) techniques are nozzle-free techniques that have been successfully employed to transfer cell-containing droplets onto a substrate [118]. The fabrication set-up comprises a laser source, a quartz substrate (ribbon) coated with the cell suspension on a hydrogel, and a substrate that is positioned below the ‘ribbon’. The choice of quartz is based on its transparency to the laser wavelengths employed. There are many variants of this technique. In the matrix-assisted pulsed laser evaporation (MAPLE) method, the laser power is absorbed directly by the hydrogel coating containing the cells, and results in the formation of cell-containing droplets that possess sufficient kinetic energy imparted by the laser power and which are collected on the substrate [119]. With the biological laser processing (BioLP) method, the laser power is absorbed by a thin intermediate metal layer between the quartz and cell containing hydrogel, causing it to undergo thermal expansion resulting in the displacement of a small volume of the hydrogel containing the cells to the substrate [120]. A wide range of cells, such as osteosarcoma, astroglial cells, neural stem cells, endothelial cells and smooth muscle cells, etc., have been patterned on substrates employing the numerous variants of laser-assisted printing [121, 122]. The laser-assisted printing technology was successfully used to introduce endothelial cells for angiogenesis in a 3D matrix containing hepatocytes [123]. The cell viability in this technique is reported to be about 95% [124]. This method exhibits a high resolution for patterning cells but is also limited by several shortcomings. The risk of damage to the cells due to the use of a high power laser is high. The uniform thickness of the cell suspension layer is important in achieving uniform cell output. The scale-up of the system remains a challenge. The drying of the cell coating is another limiting factor. Efforts to address these challenges are underway and, if successful, such techniques may represent the future of scaffold fabrication for tissue regeneration. Figure 7 shows the principle of laser-assisted bioprinting.

Laser-assisted bioprinting, where the laser is focused on a cell-loaded layer to form droplets that are deposited on the substrate.
A new advancement in the field of cell printing has been the development of an organotypic construct obtained through printing tissue spheroids instead of a single. Each spheroid consists of several cells which, when placed sufficiently close to each other, start establishing cell-cell contacts leading to the formation of a network that resembles an organ. This technique has been labelled ‘tissue fragment printing’ and holds much promise for the future [125].
Apart from jet-based biofabrication, several non-jet based techniques have also been reported in the literature. However, these are yet to receive the same kind of attention as the jet-based techniques. A cryogelation technique was reported to encapsulate bovine arterial smooth muscle cells in a poly(vinyl alcohol) hydrogel [126]. The repeated freeze-thaw cycles were optimized to achieve satisfactory cell density. The viscosity of the polymer, solvent and serum content, were all factors that influenced the cell viability. Attempts to cross-link the hydrogel for additional stability resulted in the localization of the cells at the periphery of the scaffold. In a recent report, a unique self-assembled tubular scaffold was developed using peptide amphiphiles [127]. The peptide amphiphiles can self-assemble to form nanofibres with diameters of about 10 nm, and can grow several microns long. These peptide amphiphiles can be modified with cell-specific sequences that will elicit appropriate responses from the cells. At low concentrations, these peptide amphiphiles exist as hydrogels, when cells can be introduced. The self-assembled fibres can be aligned in a desired direction by the application of shear stresses. These cell-entrapped self-assembled tubular peptide constructs were found to support the proliferation and orientation of endothelial cells, thereby demonstrating its potential to promote angiogenesis. The polyelectrolyte complex of the anionic alginate and cationic chitosan were used to encapsulate human mesenchymal stem cells [128]. This construct ably supported the growth, proliferation and multilineage differentiation into chondrocytes and osteocytes. However, the poor mechanical properties of the scaffold remained a drawback, though it might be overcome by the use of different polymers or higher molecular weight polymers.
9. Cell sheet engineering
The use of scaffolds for tissue regeneration has some pitfalls that has limited its translation into clinical practice. In the case of thick constructs, an oxygen gradient exists in the scaffold as one moves from the periphery to the interior. This results in poor cell viability in the interior [129]. In addition, the presence of a scaffold can trigger immune and inflammatory responses. If the cells have to be detached from the scaffold, trypsinization is frequently used. This, unfortunately, also results in the disruption of cell-cell contact. Hence, a new scaffold-free technique known as ‘cell sheet engineering’ has emerged in recent years. This technique employs a thermoresponsive polymer, poly(N-isopropyl acrylamide) (PNIPAM), coated on tissue culture plates. The cells are seeded on the PNIPAM surface and allowed to proliferate at 37°C [130]. After the attainment of the desired cell density, the temperature is reduced to 20°C, which is lower than the critical solution temperature of PNIPAM. This polymer undergoes a sol-to-gel transformation above its critical solution temperature of 32°C. Therefore, under the culture conditions, the polymer is hydrophobic and thus supports the adhesion, growth and proliferation of the cells. When the temperature is lowered, it transforms into a hydrophilic moiety and swells due to the entry of water. This results in the formation of a water layer below the cells, which now lose their affinity for the polymer. This facilitates the removal of the cell sheets without use of any harsh conditions, and they can be used for
10. Regenerative engineering – the road ahead
The plethora of techniques and fabrication tools that have emerged in recent years have signified new beginnings in the field of tissue engineering. However, there are still challenges in realizing the success of
Footnotes
11. Acknowledgements
The authors gratefully acknowledge financial support by the Nano Mission, Department of Science & Technology and SASTRA University.
