Abstract
Unfigured sea-island superfine fiber PA/PU non-woven (USFSLB) is used to mimic leather’s microstructure as the base of artificial leather. USFSLB has many characteristics and advantages resembling those of natural leather. However, compared with natural leather, the wearing comfort of artificial leather is inferior due to its poor moisture adsorption and permeability. In this work, a “two-step” method of chemical treatment is proposed, in which collagen/chromium-vegetable tannin (C-CrT) is immobilized on nylon fiber of USFSLB to improve its moisture adsorption and permeability (“breath” property). The two-step surface modification involved sulfuric acid hydrolysis and modifying the C-CrT on nylon fiber. Compared with the pristine USFSLB, the tensile strength, the elongation at break, the anti-static performance, the thickness, and the uniformity of C-CrT-treated USFSLB were improved at different levels. Importantly, the C-CrT-treated USFSLB showed excellent moisture adsorption and permeability, especially the liquid wicking rate (LWR) improved by 344%. The self-assembly mechanism of collagen/chromium-vegetable tannin (C-CrT) modified on nylon fibers was analyzed and discussed.
Introduction
The sustainable development and the use of biomass have been studied constantly by researchers. The hides of livestock from pig, cattle and sheep are animal husbandry by-products and contain more than 90% collagen. They are considered to be the largest renewable animal biomass resources in the world. Besides, these resources tend to be increasing with the increase in living standard. Although they are mainly used for leather-making, the application rarely exceeds 50%. China is known as a country with leather mass production of leather. Consequently, the production inevitably gives rise to leather solid wastes. It is estimated that more than 1.4 million tons of leather solid wastes are produced every year. However, more than 80% of collagen exists in wastes [1]. Therefore, it is very meaningful that collagen in solid wastes is recycled. There are abundant polar groups in collagen, such as –OH, –COOH, –CONH2 and –NH2. These polar groups can improve the moisture adsorption and transfer properties of USFSLB materials.
The natural polyphenols, called the tannins, are extensively distributed in the root, bark, stalk, and fruits of plants as metabolism products [2,3]. Based on their chemical structure, they are classified as hydrolysable tannins, condensed tannins, and complex tannins. The hydrolysable tannins yield gallic acid or ellagic acid when they are hydrolyzed with acids, bases, or some enzymes [4]. The condensed tannins are the polymerized products of flavan-3-ols and/or flavan-3,4-diols [5]. The complex tannins contain both the structures of condensed tannins and hydrolysable tannins. The tannins used in this research are the condensed tannins. Precipitating proteins and precipitating metal ions are the two characteristic capabilities of tannins. The ability of tannins to precipitate metal ions is due to their multiple adjacent phenolic hydroxyl groups, which can form stable complexes with many metal ions, such as chromium [6–11]. In this regard, the collagen is a possible choice due to the availability of large number of groups, such as –CO–, –CO–NH–, and –NH2. These groups can interact with tannins.
USFSLB is used to simulate leather microstructure as the base of artificial leather because it has many characteristics and advantages as compared with natural leather like its mechanical strength, chemical resistance and homogeneity. However, compared with natural leather, the wearing comfort of artificial leather is inferior due to its poor moisture adsorption and permeability. Therefore, the sanitation performance of superfine fiber artificial leather has to be improved [12]. In structure, USFSLB is composed of two components – of nylon fibers and polyurethane. Nylon is an inert polymer, and the reactive group of nylon fiber is only about 10% compared with that of collagen fibers found in natural leather [13].
There have been a number of domestic researches focusing on improving the sanitation performance of artificial leather, and include studies involving formic acid [14,15], sulfuric acid [16], triethanolamine [17], protease [17], pancreatic enzymes hydrolysis [17], which can damage the fibers due to hydrolysis. For getting better moisture adsorption and permeability, the degree of hydrolysis should been improved. According to some previous reports, the collagen material filling [17] can improve the moisture permeability to as high as 70%. However, it is the common physically adsorption and filling, which makes its properties to be unstable. The use of soluble chitosan derivatives (CS-HCA) [18] had also been reported, which improved the moisture permeability up to around 58%. The phosphine and collagen [19,20] were reported to improve the moisture permeability and moisture adsorption by around 65% and 179.72%, respectively. These two methods must hydrolyze the fibers before for the production of active groups. Therefore, the number of chitosan derivatives and collagen on the fibers depended on the degree of hydrolysis. However, the degree of hydrolysis must be controlled to proper levels. Therefore, the degree of improvement was limited. The glutaraldehyde and polyamidoamine dendrimers [21–23] were used to improve the moisture permeability by around 96.72%. However, controlling the proper generation of polyamidoamine dendrimers is difficult. Therefore, the application of this method on commercial scale is very difficult.
Based on some recent studies, a method of using collagen-chrome vegetable tannins (C-CrT) was proposed to modify the nylon fibers of USFSLB. Additionally, nylon has the similar structure as of collagen. In theory, it is feasible that the collagen can be immobilized on nylon fibers using chrome vegetable tannins. This novel method has more advantages than the previously reported methods. This is due to the reason that the collagen and the chromium-vegetable tannins can be immobilized using layer-by-layer self-assembly, which has the similar effect as of polyamidoamine dendrimers, and is also obtained easily. In this work, the modification of USFSLB has been reported using chemical etching of fiber surfaces and modification of fibers using biomass materials, collagen and chromium-vegetable tannins, as shown in Figure 1. Various parameters, including moisture adsorption and permeability, tensile strength, elongation at break, anti-static performance, thickness, and uniformity of the modified USFSLB were measured. The self-assembly mechanism of chemical treatment of collagen/chromium-vegetable tannin (C-CrT) immobilized on nylon fibers of USFSLB was analyzed and discussed.
Schematic illustration of C-CrT modified nylon fiber of USFSLB. C-CrT: collagen/chromium-vegetable tannin.
Experimental
Materials
The materials and instruments used in the experiments are as follows: Collagen (G1) (Tech, Mw3690, Haining Debang Co., Ltd, China), chromium vegetable tannins (Tech, Mw2374, Sichuan De Cai Co., Ltd, China), sulfuric acid (AR, 98%, Guoyao Co., Ltd, China), sodium bicarbonate (AR, 99.5%, Guoyao Co., Ltd, China), PA/PU superfine fiber non-woven with the mass per unit area of 536.7 g/m2 and thickness of 1.4 mm (Yantai Wanhua Co., Ltd, China).
Modification methods
Two steps of C-CrT modification were involved. In Step 1, the non-woven fabric was cut into 19×9 cm2 with 10.7 ± 0.2 g weight. The specimens were immersed in a container with the addition of deionized water (5000 wt.% liquor ratio; the percentage of dry USFSLB mass). Then, the container was sealed and mounted on an infrared-ray heating machine, and heated to 70℃ for 30 min. After being hydrolyzed for 4 h at 15 wt.% sulfuric acid with 5000 wt.% liquor ratio, the samples were washed in water for 30 min using ultrasonic. The samples were then left to dry. In Step 2, the hydrolyzed samples were immersed in a container with the addition of 15 wt.% collagen (1500 wt.% liquor ratio; the percentage of dry USFSLB mass). The container was sealed and mounted onto an infrared-ray heating machine, and heated to 45℃ for 3 h. Then, the samples were removed, and the pH of collagen solution was adjusted to 4.0 using formic acid. Later, they were immersed in a container containing chromium-vegetable tannins. The container was sealed and mounted onto an infrared-ray heating machine, and heated to 60℃ for 3 h. Finally, the samples were washed for 30 min using ultrasonic and left to dry. Eventually, the C-CrT-treated USFSLB was obtained.
Characterization
The water contact angle of the surfaces was determined using a static contact angle measuring device (OCA20, Dataphyscs, Germany). The water contact angle was measured using the sessile drop before and after the modification. Each stated contact angle is the average of 11 measurements from various positions on the surface. In order to observe the possible changes in the surface morphology of the material, scanning electron microscope (SEM) was used. The samples were placed in an oven (105℃) and dried until constant weight. After Au spraying, the micrographs of the surfaces of samples were taken using a field emission SEM (FEI Q45 + EDAX Octane Prime, USA). The SEM and energy disperse spectroscopy (EDS) were used to determine the elements on the surface of nylon before and after the treatment. Attenuated total reflectance Fourier transform infrared (ATR-FTIR) spectra were acquired using a Fourier-transform infrared spectrometer (VECTOR-22, Bruker, Germany). For each spectrum, a resolution of 4 cm−1 was applied and 32 scans were gathered. Furthermore, X-ray photoelectron spectroscopy (XPS) was used to investigate the surface chemical composition of the samples before and after the treatments. All XPS spectra were collected using an Al-Kα X-ray photoelectron spectrometer (Thermo Fisher Scientific, USA), which used monochromatic X-rays focused on a 300 µm spot size. The collagen and collagen-chromium tannins solution was detected using transmission electron microscopy (TEM) (FEI Tecnai G2 F20 S-TWIN, FEI, USA). The collagen and collagen-chromium tannins solution was diluted to a mass fraction of 2%, deposited on carbon-coated copper grids and inverted on a drop of aqueous sodium phosphotungstate solution, which is a solution of phosphotungstic acid with a mass fraction of 2%. The phosphotungstate solution was neutralized using sodium hydroxide to pH of 7. Then, the shapes of collagen and collagen-chromium tannins solution were analyzed and compared using conventional TEM. When the value of positive and negative charges on the surface of collagen was equal, that is, when the degree of ionization of acidic and basic groups in the substance was the same, the pH of the solution was equal to isoelectric point (pI); collagen (10 mg) was dissolved in 100 ml of acetic acid (0.05 mol/L), and centrifuged. The 10-mL supernatant of collagen was removed, and analyzed using automatic potentiometric titrator (905 Titrando, Mtrohm, Switzerland).
Determination of moisture permeability
Static water-vapor transmission (SWVT) rate [23] was measured. In short, the samples were cut into circular pieces having the diameter of 55 mm, and put into a constant temperature and humidity equipment for air conditioning. The temperature and relative humidity were maintained at values of 20±2℃ and 65±2% for 24 h, respectively. After air conditioning, 30 mL of distilled water was poured in the test cup. The sample and the rubber gasket were put onto the test cup. The aluminum cover was tightened. Then, the total mass of the test cup including the sample was measured and recorded as m1. The test cup was then placed in a desiccator with 98% sulfuric acid as the dryer. The desiccator was kept in the constant temperature and humidity equipment (GT-7005, Gao Tie, Taiwan) with the temperature and relative humidity maintained at 20±2℃ and 65±2% for 24 h, respectively. After water-vapor transmission, the test cup was removed, weighed again, and recorded as m2. All the measurements were performed four times to ensure the reproducibility of data.
Liquid wicking rate (LWR) [24] was also determined. Liquid wicking rate is the measurement of the capillarity of the test material and represents the rate at which the liquid is transported to fabric through capillary action. Furthermore, the liquid wicking height was achieved per unit time in the textile material through capillary action and is used to assess the moisture adsorption capability of fabric. The specific measurement procedure is as follows. The sample was first cut into 30 mm wide, and 50 mm long pieces. It was then placed into an oven (105℃) and dried to constant weight. The end of sample was immersed in water for about 15 mm, and was timed until the liquid wicking height reached 30 mm. The value was recorded as time (s) and calculated as the liquid wicking rate (mm/s). All the measurements were performed four times to ensure the reproducibility of data.
Physical property testing
The tensile strength and the elongation at break of USFSLB were determined (refer to QB/T 2710-2005) [25] using a tensile tester (PT-1171, Dongguan Baoda International Co., Ltd, China). All the measurements were performed at least five times to ensure the repeatability of results.
The softness of modified USFSLB was determined (refer to ISO 17235:2011, IULTCS IUP/36 TEST METHOD) [26] using a softness tester (GX-5039, Dongguan gaoxin Co., Ltd, China). The actual diameter of the circular aperture was selected to be 24.975 ± 0.025 mm. All the measurements were performed thrice to ensure the repeatability of results.
The antistatic property was determined according to GB/T 12703-91 [27] using an anti-static tester (FY403E, Wenzhou Fangyuan Co., Ltd, China). The timing method was selected, whereas the decay time was 10 s. The friction time was 60 s. The peak voltage (V) was recorded. All the measurements were performed thrice to ensure the repeatability of results.
Results and discussion
Characterization of modified USFSLB
There was a slight change between the contact angle of hydrolyzed USFSLB and that of the untreated USFSLB (Figure 2(a) and (b)). After the collagen was coated on the fiber of USFSLB, the contact angle obviously decreased (Figure 2(c)). It became much lower after the treatment using chromium-vegetable tannins (Figure 2(d)). These changes suggested that the hydrophilicity of USFSLB had improved after C-CrT treatment. This was further confirmed by comparing the digital images of dyed water droplet on untreated and C-CrT-treated USFSLB (Figure 3).
Water contact angles of untreated, hydrolyzed, collagen-coated and C-CrT-treated USFSLB. C-CrT: collagen/chromium-vegetable tannin. Digital images of dyed water droplet on (a) untreated, (b) C-CrT-grafted after 2 s. C-CrT: collagen/chromium-vegetable tannin.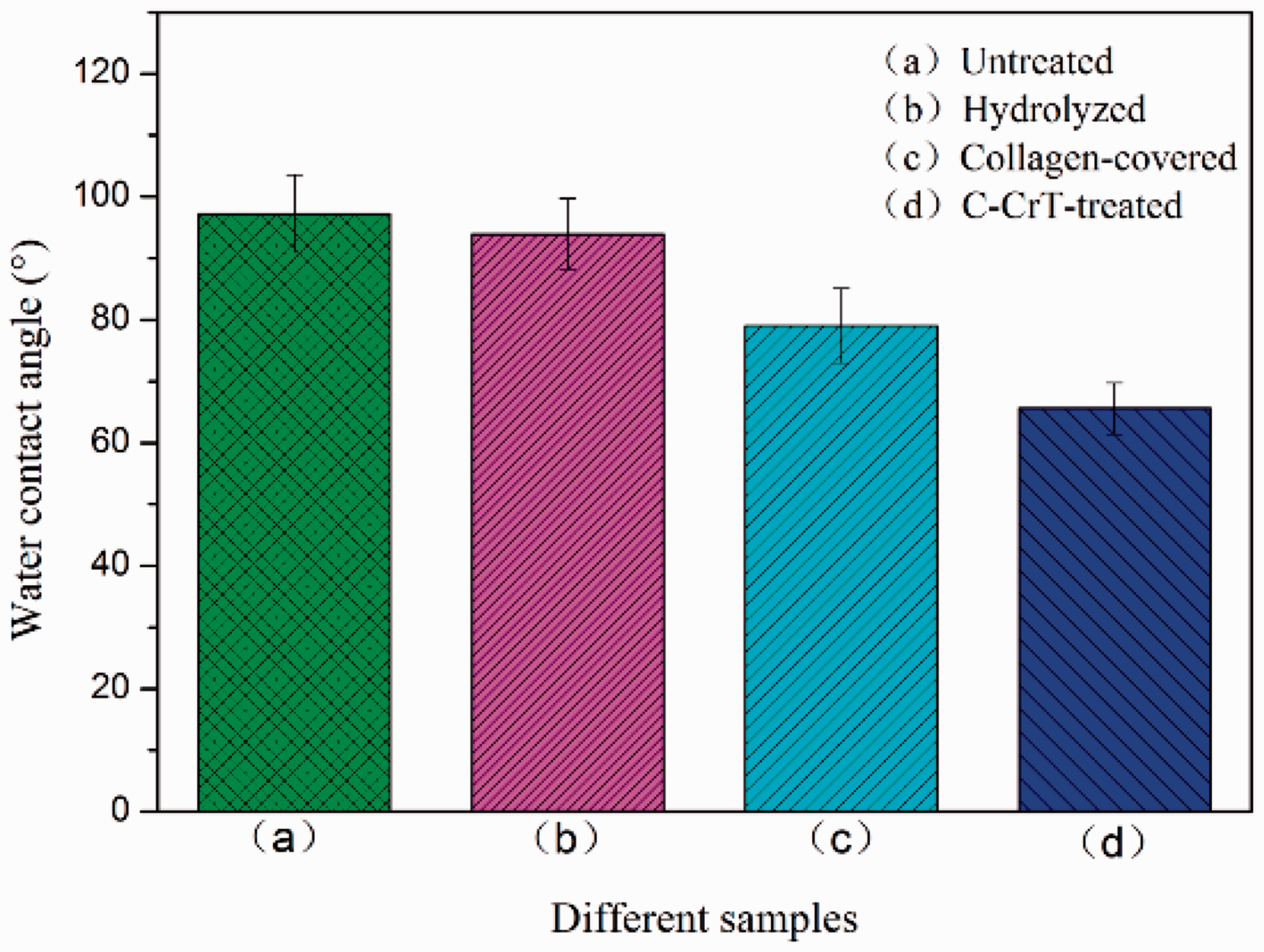

The SEM images of the top view of untreated, hydrolyzed, collagen-coated and C-CrT-treated USFSLB are shown in Figure 4. Compared with the untreated USFSLB, the nylon fibers of USFSLB became loose after sulfuric acid treatment (Figure 4(b)). After being coated with collagen, the surface morphology of nylon fiber hardly changed (Figure 4(c)). However, after the addition of chromium vegetable tannins, a lot of microspheres were observed on the nylon fibers (Figure 4(d)).
SEM images of the top view of (a) untreated (b) hydrolyzed (c) collagen-coated (d) C-CrT-treated (CrT 7%) USFSLB. C-CrT: collagen/chromium-vegetable tannin.
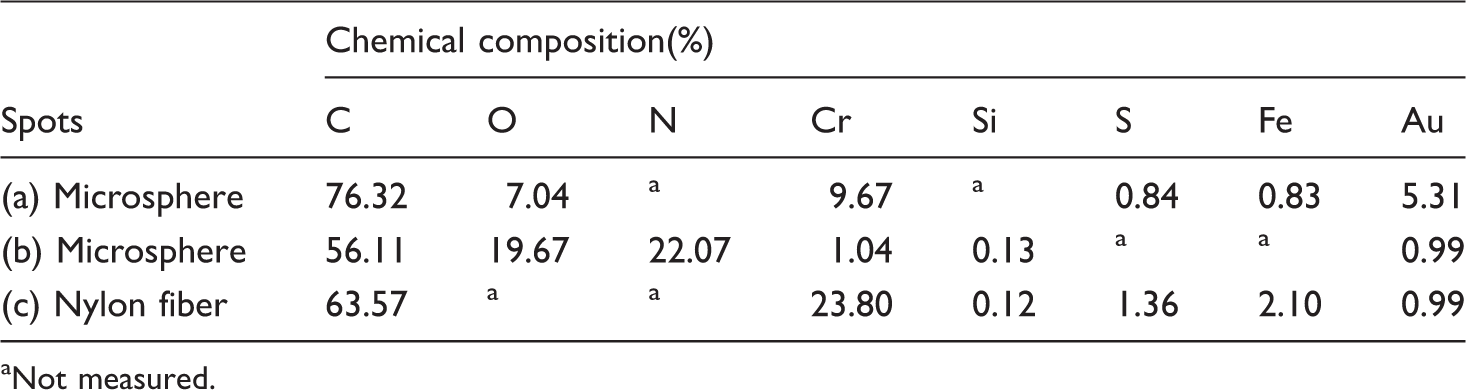
Figure 5 shows the chromium-vegetable tannins, untreated, collagen-coated and C-CrT-treated USFSLB. Figure 5(a) shows the FTIR spectra of chromium vegetable tannins. The peak at 3357 cm−1 was assigned to –OH of vegetable tannins, while that at 1731 cm−1 was attributed to C=O of PU (–NH–COO–R), which was due to the reason that USFSLB was the composite of PA/PU. The peaks at 1676, 1660, 1646, and 1638 cm−1 were attributed to amide I, while those at 1558, 1540, and 1516 cm−1 were assigned to amide II. Furthermore, the peak at 3300 cm−1 was assigned to –NH2 of PA (Figure 5(b) to (d)). Compared with the untreated USFSLB, in the C-CrT-treated USFSLB, an additional peak at 1038 cm−1 appeared (Figure 5(d)), which was attributed to cyclic ether that belonged to vegetable tannins (Figure 5(a)). The peak at 3300 cm−1 was attributed to N–H stretching vibration and was broadened (Figure 5(d)) due to the increase in number of –NH2 coming from collagen and –OH from vegetable tannins. Therefore, it was confirmed that collagen and chromium-vegetable tannins were successfully immobilized on nylon fibers of USFSLB. Meantime, compared with the untreated USFSLB (Figure 5(b)), the FTIR spectra of collagen-coated USFSLB did not change (Figure 5(c)), which was due to the reason that a significant portion of collagen was washed out without the chrome-vegetable tannins during the washing process. The FTIR-ATR evidence of surface changes is inconspicuously positive. However, the improvements in the adsorption of moisture and permeability (“breath” property) were very obvious. It may be because the dosage of chrome-vegetable tannins was little (5% of dry USFSLB). Otherwise, according to Figure 14 and Table 2, there were two different outer layers, namely (a) and (b). The (a) layer was mainly made up of chrome-vegetable tannins. The (b) layer was primarily made up of collagen.
FTIR spectra of (a) chromium-vegetable tannins and (b) untreated, (c) collagen-coated (d) C-CrT-treated USFSLB. C-CrT: collagen/chromium-vegetable tannin. Elemental analysis of USFSLB before and after the treatment. Elemental analysis of different spots. aNot measured.

The chemical composition of the sample surface was investigated using the XPS with high sensitivity. According to the XPS analysis and narrow scanning spectra of N1s, O1s and C1s (Figure 6), in N1s and O1s spectra, the peak intensity of C-CrT-treated USFSLB increased significantly compared with the (a) untreated, (b) hydrolyzed and (c) collagen-coated USFSLB. This was attributed to increased amino nitrogen from collagen and phenolic oxygen from chromium-vegetable tannins. Additionally, in the C1s spectra, the peak of C-CrT-treated USFSLB had an asymmetrical tail (Figure 6 C1s), which was attributed to higher content of carbon with the binding energy of 287.2 and 288.7 eV, corresponding to the presence of functional groups (O=C–N, and O=C–O species) on the collagen surface [7,28–32].
XPS survey and narrow scanning spectra of C1s, N1s, O1s on (a) untreated, (b) hydrolyzed, (c) collagen-coated, (d) C-CrT-treated USFSLB. C-CrT: collagen/chromium-vegetable tannin.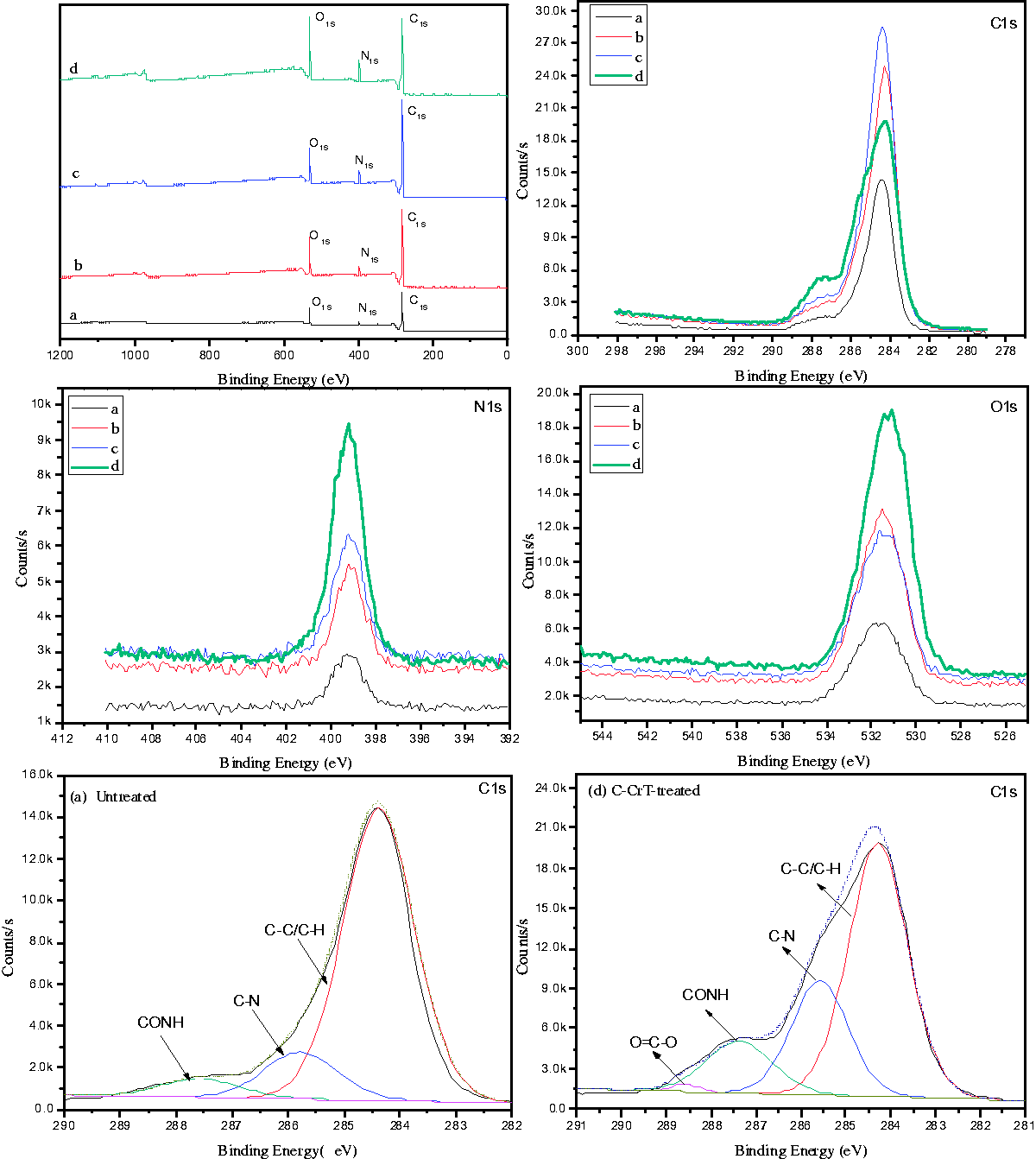
Furthermore, the curve fitting to C1s core-level spectrum of the untreated and C-CrT-treated nylon surface of USFSLB was also conducted, as shown in Figure 6. The results show that the three peaks of untreated (a) components with the binding energies (BEs) of about 284.4, 285.8, and 287.2 eV were attributed to C–H/C–C, C–N, and O=C–N species, respectively. The C1s core-level spectrum of C-CrT-treated nylon surface also exhibited four peaks with the BEs of about 284.4, 285.8, 287.2, and 288.7 eV, which were attributed to C–H/C–C, C–N, O=C–N, and O=C–O species, respectively. The remarkable increase in C–N and O=C–N indicates that a lot of collagen is grafted on the nylon of USFSLB.
Compared with the (a) untreated, (b) hydrolyzed and (c) collagen-coated USFSLB, the [C]/[O] ratio of C-CrT-treated USFSLB significantly decreased (see Table 1), which was attributed to the presence of phenolic hydroxyl groups from the vegetable tannins. Therefore, it was confirmed that the C-CrT was successfully immobilized on the nylon fibers of USFSLB.
Figure 7 shows the results of XPS Cr in the chromium-vegetable tannins and the C-CrT-treated surface. The Cr in the chromium-vegetable tannins gave distinct peaks at: 577.6 eV (Cr 2p3/2) and 587 eV (Cr 2p1/2), whereas the nylon fiber of C-CrT-treated USFSLB gave distinct peaks at: 576.6 eV (Cr 2p3/2) and 586.5 eV (Cr 2p1/2). Compared with the Cr in the chromium-vegetable tannins, the binding energy of Cr in USFSLB decreased and its oxidation state decreased. Furthermore, chromium presented lower valence state in the C-CrT-treated USFSLB. The oxo-functional groups (denoted as CxOH), including –OH and –COOH from the collagen, vegetable tannins and hydrolyzed nylon surface, significantly influenced the valence states of Cr. The Cr of chromium-vegetable tannins with higher positive redox potential was unstable in the presence of electron donor functional groups (–OH and COOH). Therefore, the Cr species was bound to phenolic hydroxy and carboxyl groups through complexation reaction [31].
XPS narrow scanning spectra of Cr in the C-CrT-treated USFSLB (a) and the chromium-vegetable tannins (b). C-CrT: collagen/chromium-vegetable tannin.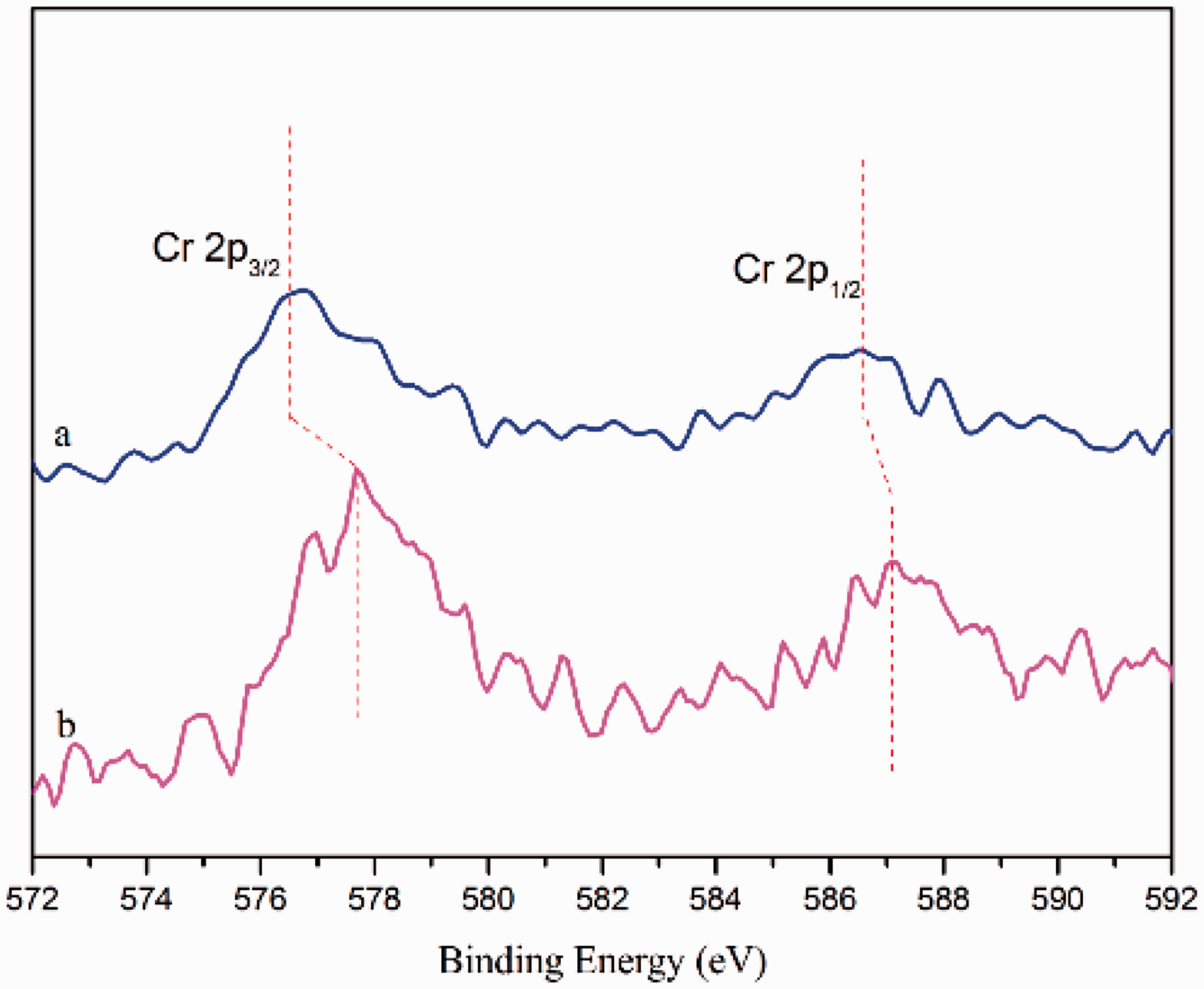
Self-assembly mechanism of collagen with tannins
The tannins were the condensed tannins, as confirmed from the results presented in Figure 8. These compounds were formed of flavan-3-ol monomer units based on resorcinol A-rings and pyrogallol B-rings (as shown in Figure 8), which was linked together through C4–C8 bonded to oligomers and high molecular weight polymers [33]. It is worth mentioning that condensed tannins are hydrophobic. [7] The condensed tannins used in current experiments were grafted on C6 of A-ring using sulfonic acid groups. The sulfonic acid groups can improve the water-solubility of tannins. Therefore, the vegetable tannins can be divided into two parts consisting of hydrophilic and hydrophobic moieties (as shown in Figure 9). In consideration of the diversity of molecular structures among vegetable tannins and many different functional groups in the collagen, all of the following interaction types would, in principle, take place: hydrogen bonding, ionic bonding, hydrophobic bonding, and covalent bonding. However, the latter represented an irreversible change, which required molecular oxygen and was favored by high pH or by the presence of polyphenol oxidases [34], while the charged groups were absent in the condensed tannins. Proanthocyanidins became charged only at high pH due to the dissociation of phenolic hydroxyl groups along with the formation of phenoxide ions [35]. Therefore, the interaction types of ionic and covalent bonds between the collagen and vegetable tannins were not considered. The presence of hydrogen donors in the form of phenolic hydroxyl groups in the vegetable tannins and of hydrogen acceptors in the form of carbonyl functions of the collagen would naturally lead to a possibility of formation of hydrogen bonds [36–40]. The hydrophobic amino acid side chains on collagen, such as aromatic ring, pyrrolidine ring, and aliphatic chain are prone to become concentrated and form a hydrophobic pocket [32]. Since both the groups (the aromatic nuclei of vegetable tannins and the aliphatic side chains of collagen amino acids) contain hydrophobic regions, it would seem equally possible that these would participate in the interaction phenomena (as shown in Figure 9). Considering that the collagen has very complex structures with a variety of chemical groups of different affinities [41], it was in the hydrophobic micro domains of collagen molecules with amide that hydrogen bonding would occur, which reinforced the hydrophobic bonding (as shown in Figure 9). In addition, when collagen was dissolved in distilled water with the pH of 6.0 in Step 2, it could not form microspheres. However, when the chromium vegetable tannins were added, the microspheres formed (Figure 10). Therefore, the chromium vegetable tannins played an important role in the formation of microspheres. Self-assembly mechanism involves chromium-vegetable tannins, whose hydrophobic part could crosslink with the collagen through hydrogen bonding and hydrophobic bonding, after which the hydrophilic part of chromium-vegetable tannins could crosslink with –COOH, and –NH2 on the collagen through coordination bond [42,43]. These ways of cross-linking were alternated repeatedly until the microspheres were formed (Figures 9 and 10). Therefore, the mechanism of self-assembly between chromium-vegetable tannins and collagen was in favor of hydrogen bonding, hydrophobic bonding and coordination bonds.
Probable flavonoid constitutional unit in the condensed tannins. Self-assembly mechanism of collagen and chromium-vegetable tannins. TEM images of (a) collagen solution, (b) collagen and chromium-vegetable tannins solution.


Modification mechanism analysis
Figure 13 shows the method of modifying C-CrT on nylon fiber. The C-CrT microspheres were formed through hydrophobic bonding, hydrogen bonding and coordination bonds.
Potentiometric titration curves of collagen. Surface electric charge of collagen and nylon fiber under different pH values. Modification mechanism of C-CrT grafted onto nylon fiber. C-CrT: collagen/chromium-vegetable tannin. SEM/EDS of C-CrT-treated USFSLB on (a) microsphere, (b) microsphere, (c) nylon fiber. C-CrT: collagen/chromium-vegetable tannin.


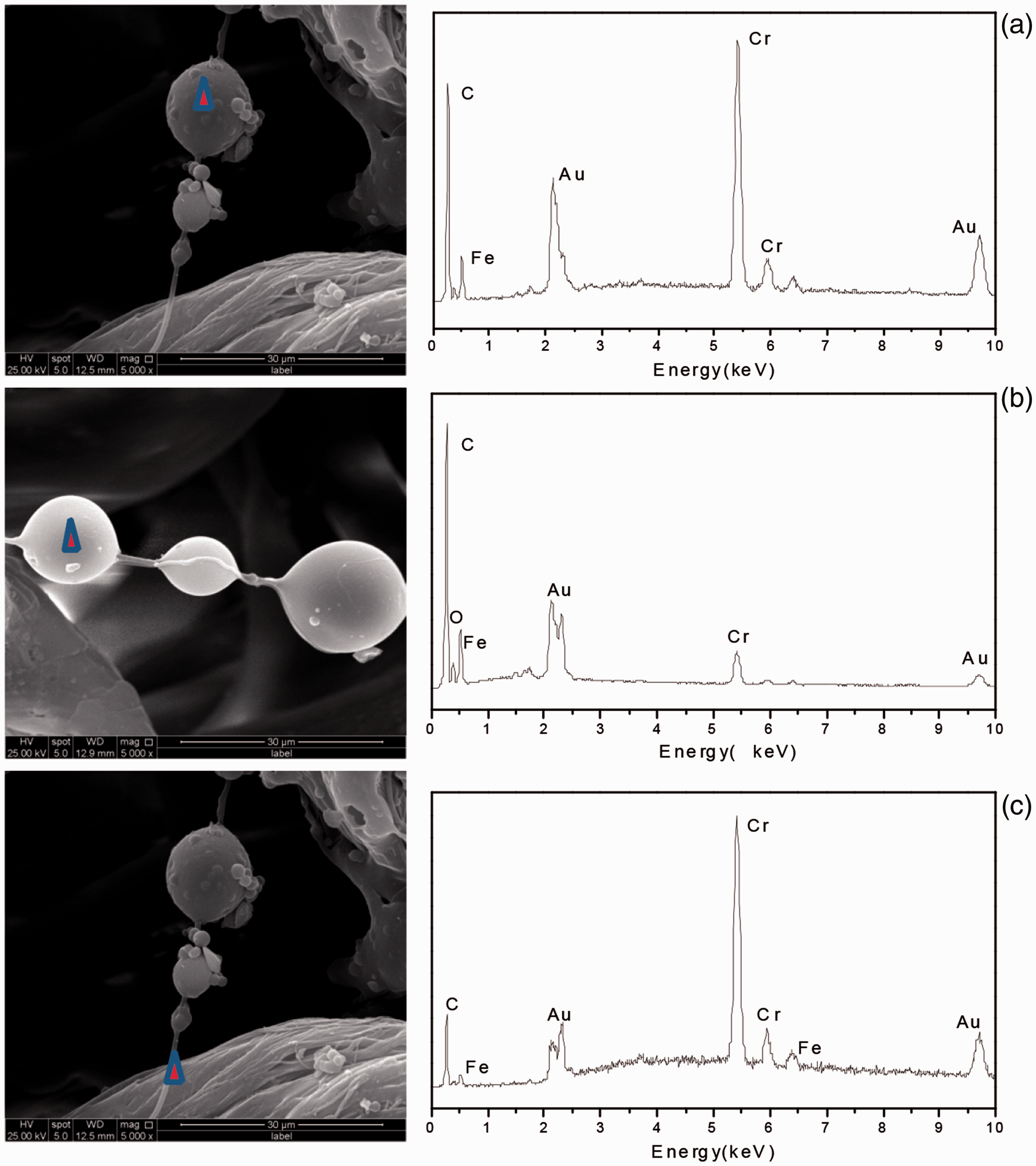
In addition, it is well-known that nylon has a small amount of residual COOH and NH2 end groups. Ionizations of the residual amino and carboxyl end groups in nylon played an important role in the observed pH-sensitive permeation. The nylon formed high barrier to the permeation of cationic substance in the acidic medium (below the pH of 5.0), but not in the basic medium (above the pH of 9.0) (relative to the neutral-pH region of 6.0–9.0) [44]. The isoelectric point (pI) of the collagen, applied in this study, was about 7.4 (Figure 11). Therefore, the collagen was cationic, and the nylon was neutral in the initial solution with the pH of 6.0. The collagen was fully permeated in the USFSLB for a given time (Figure 12). Then, the chromium-vegetable tannins were added, and the pH of the collagen solution was adjusted to 4.0 to prevent the precipitation of chromium. For the pH of 4.0, the collagen and nylon had cationic charge, while the vegetable tannins had anionic charge scarcely. The nylon rejected the collagen, while the vegetable tannins could come close to the nylon fiber (Figure 12). Therefore, the chromium-vegetable tannins, which are the hydrophilic part, could preferentially form the complex with –COOH of nylon fibers through coordination bonds. Then, the chromium-vegetable tannins, which are the hydrophobic part, could crosslink with the collagen through hydrogen bonding and hydrophobic bonding (Figure 13). Additionally, the hydrophilic part of the chromium-vegetable tannins could crosslink with –COOH and –NH2 on collagen through coordination bonds. These ways of cross-linking were alternated repeatedly until the microspheres were formed on the nylon fibers (Figure 13). Therefore, the outer surface of microspheres presented in two forms was surrounded by chromium tannins layer and by collagen layer, and was confirmed by SEM/EDS analysis (as shown in Figure 14).
SEM/EDS results of C-CrT-treated USFSLB for different spots are shown in Figure 14.
After the elemental analysis of different microspheres (Table 2), it was confirmed that there were two forms of the outer surface of microspheres. One was surrounded by chromium tannins layer (as shown in Figure 14(a)), for which the relative contents of elemental C, O and Cr were 76.32%, 7.04% and 9.67%, respectively (Table 2). The other was surrounded by collagen layer (as shown in Figure 14(b)), for which the relative contents of elemental C, O and N were 56.11%, 19.67% and 22.07%, respectively (Table 2).
Moisture permeability
SWVT and LWR of the hydrolyzed USFSLB were found to be 658 g/m2ċ24 h and 0.513 mm/s, respectively, while the corresponding values for C-CrT-treated USFSLB were 986 g/m2ċ24 h and1.323 mm/s, respectively (as shown in Figure 15). Compared with the pristine USFSLB, the SWVT and LWR of hydrolyzed USFSLB improved by 27% and 72%, respectively, while those for C-CrT-treated USFSLB improved by 90.35% and 344%, respectively. This was due to the reason that sulfuric acid hydrolyzed the nylon fiber of USFSLB to break the amide bonds (–CO–NH–). As a result, carboxyl (–COOH) and amino (–NH2) groups were exposed on the nylon fibers of USFSLB. These polar groups not only improved the moisture permeability, but also provided active groups for subsequent modification. This means that the chromium vegetable tannins reacted with these active groups through coordination bonds (from chromium complexes) and hydrogen-bonding (from phenolic hydroxyl of tannins). Therefore, the collagen and chromium vegetable tannins with a lot of polar groups, such as –OH, –NH2, and –COOH were immobilized on the surface of nylon fiber of USFSLB and improved the moisture permeability of USFSLB. The hydrophilic properties of USFSLB fabric, modified by collagen and chromium vegetable tannins, were improved, although the air permeability decreased a bit. This was due to the reason that air permeability is related to the porosity of USFSLB. However, USFSLB is the semi-finished product of artificial leather. Furthermore, PU coating proceeds on the surface of USFSLB. Therefore, compared with the PU coating, the decrease in air permeability for C-CrT-treated fibers was negligible.
SWVT and LWR of (a) untreated, (b) hydrolyzed, (c) collagen-coated, (d) C-CrT-treated USFSLB.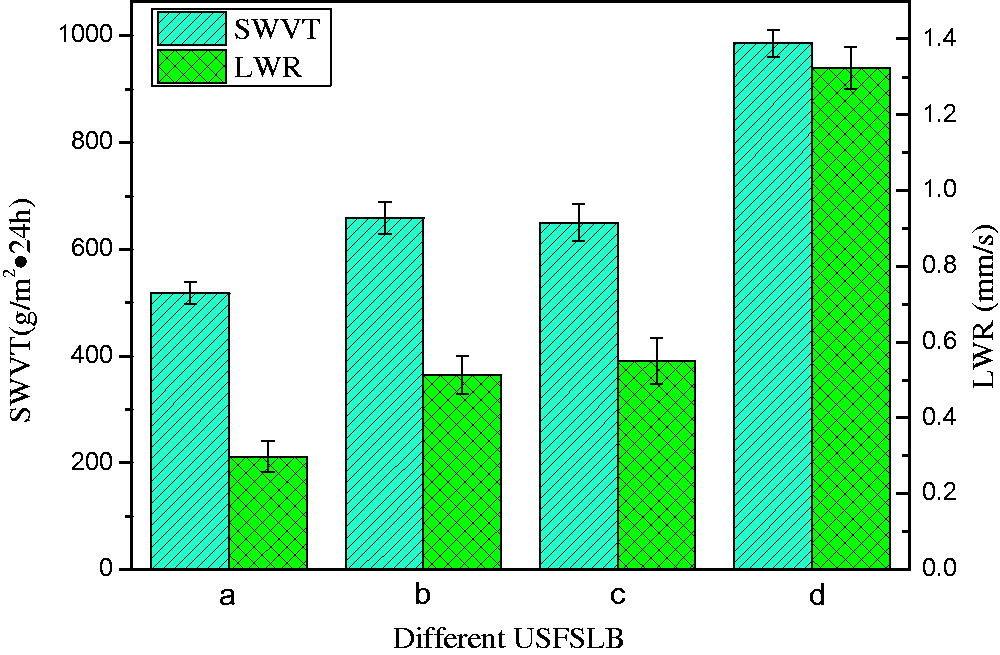
Physical properties
Tensile strength and elongation at break of USFSLB improved after the modification (Figure 16). This was due to the reason that the cross-linking between nylon fibers occurred. The thickness and uniformity of C-CrT-treated USFSLB obviously increased (Figure 17) due to the good selective filling properties of collagen and tannins. The softness of hydrolyzed USFSLB improved (Figure 18), indicating that the sulfuric acid made the USFSLB loose. However, the softness of C-CrT-treated USFSLB obviously declined compared to that of pristine USFSLB.
Tensile strength and elongation at break of (a) untreated, (b) hydrolyzed, (c) collagen-coated, (d) C-CrT-treated USFSLB. C-CrT: collagen/chromium-vegetable tannin. Thickness and CV value of (a) untreated, (b) hydrolyzed, (c) collagen-coated, (d) C-CrT-treated USFSLB. C-CrT: collagen/chromium-vegetable tannin. The softness of (a) untreated, (b) hydrolyzed, (c) collagen-coated, (d) C-CrT-treated USFSLB. C-CrT: collagen/chromium-vegetable tannin.


(Figure 18). This was due to the reason that a lot of collagen was immobilized on the surface of nylon fibers by chromium vegetable tannins. Polar functional groups, such as OH, NH2 and COOH of collagen would cross-link each other through hydrogen bonding during the dehydration of USFSLB. Therefore, the molecules did not slip and the softness of C-CrT-treated USFSLB declined. Furthermore, the chromium vegetable tannins were grafted on the nylon fibers of USFSLB, which resulted in a decline of softness of C-CrT-treated USFSLB.
Anti-static performance also improved after the modification, especially the collagen-coated USFSLB. However, the anti-static performance of C-CrT-treated USFSLB was lower than that of collagen-coated USFSLB (Figure 19). This was due to the reason that the collagen covered the nylon fiber and the polar functional groups were fully exposed on the surface of nylon fiber. These polar functional groups easily adsorbed water molecules, due to which the conductive water film would be formed on the surface of nylon fiber. As a result, the anti-static performance improved. However, on the fiber of C-CrT-treated USFSLB, a lot of different sizes of microspheres were observed (as shown in Figure 4). The microspheres composed of collagen and chromium-vegetable tannins had a large number of polar groups. These polar groups formed the conductive water film on the surface of microspheres and on the nylon surface. Therefore, the conductive water film was discontinuous on the surface of nylon fiber and the anti-static performance of the C-CrT-treated USFSLB was lower than that of collagen-coated USFSLB.
The anti-static performance of (a) untreated, (b) hydrolyzed, (c) collagen-coated, (d) C-CrT-treated USFSLB. C-CrT: collagen/chromium-vegetable tannin.
Conclusions
In this work, a novel and general strategy, namely the “two-step” modification has been demonstrated for immobilizing C-CrT on nylon fibers and producing “breath” USFSLB. It was shown that the tensile strength, elongation at break, anti-static performance, thickness, and uniformity of the C-CrT-treated USFSLB improved to different levels, while the softness decreased. Importantly, the C-CrT-treated USFSLB showed excellent moisture adsorption and permeability. In addition, the collagen and vegetable tannins were successfully immobilized on nylon fibers in the form of microspheres. The hydrogen bonding, coordination bonds and hydrophobic bonding were mainly considered as the self-assembly mechanism of microspheres.
This method might be suitable for production–manufacturing because it is simple to operate, involves non-expensive, and readily available materials. More significantly, the PA/PU superfine non-woven fiber was modified to “breath” base for artificial leather and the biomass resources were also recycled during the process.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Young Scientists Fund of National Natural Science Foundation of China (Grant No. 21808136), National Key R&D Program of China (Grant No. 2017YFB0308500), and Doctoral Startup Fund of Shaanxi University of Science and Technology (Grant No. 2017BJ-25).

