Abstract
Low temperature plasma technology has the characteristics of economy, pollution-free and high efficiency. The pioneering works were carried out by applying low temperature plasma to surface modification of natural leather and chrome tanning process, to reduce water pollution. The effects of oxygen low temperature plasma treatment on the micro-structure, chemical compositions and active groups of leather fiber were studied in this paper. The optimal low temperature plasma treatment time was 10 min, which had the maximal chrome exhausting value. The SEM results showed that the leather surface was etched rougher with time increasing. The contact angle measurements showed that the hydrophilic property of leather surface increased after low temperature plasma process. The XPS data showed that the O1s area ratios increased from 19.49% to 26.45%, the content of COOH roughly tripled after O2 low temperature plasma treatment for 10 min, and the surface chrome content increased from 1.09% to 1.31% after chrome tanning. Based on the above results, low temperature plasma technology may provide a new exploring method for high-exhaustion chrome tanning technology.
Introduction
Tanning process is very important for natural leather to gain good structural stability and humidity resistance. Chrome tanning dominated leather tanning process more than one hundred years, because it could improve the humidity resistance, softness and chemical stability of leather. However, chromium absorption rate in tanning process is just about 60 to 80%, which means nearly one-third chrome tanning agents are left in the waste water. It is not only a horrendous waste of resources, but also a big pollution to our environment [1,2]. Therefore, it is very important to improve the total chromium absorption of leather to reduce the amount of chromium in effluent. In order to reduce the environmental pollution in the leather industry, many different solutions have been proposed, such as recycling the tanning floats [3,4], combination tanning methods [5], and process improvement [6]. However, these methods are based on traditional wet-chemical processing method which has some drawbacks. For example, consuming substantial volumes of water and chemical, cumbersome process, harmful effect and cost effect [7,8].
Low temperature plasma (LTP) technology became a research focus because of corresponding with green and clean chemical technique principle [9,10]. It is a widely used approach to the surface modification of polymer materials recently [11–14]. The modification on polymer materials by LTP may result in surface etching [15], forming cross-linked structure and introducing specific functional groups to change the surface characteristics such as hydrophilicity [16,17], adhesion [18], dyeing [19], and biological compatibility [20]. Therefore, applying this technique to chemical engineering realm of leather-making will bring new methods to the traditional industry of leather making. In the tanning process, the natural leather treated by LTP may introduce active groups such as hydroxyl, carboxyl and amino, etc. [21], which could not only improve leather surface energy, but also enhance its cross-linking ability with inorganic metal tanning agents. Previous studies of applying LTP treatment to leather surface and other textile modification mainly focused on the surface microstructure [22], wettability [23], hydrophobicity [24,25], hydrophilicity [26,27], dyeing [28–30] and mechanical properties [31], but researches on the effect of the active groups change on chrome tanning performance have seldom been reported yet.
In this paper, the natural leather samples were treated by O2 LTP for different time. The effects of O2 LTP treatment on the micro-structure, active groups and chrome tanning ability of leather were studied. This study aims to apply environment friendly LTP method on leather making to improve the absorption of chromium, thus to reduce chrome pollution to the environment.
Experimental section
The bare goat skin obtained from Ruixing Leather Co., Ltd (Haining, China) was acid pickled in the solution of NaCl and H2SO4 in our laboratory. NaCl, H2SO4, dehydrated alcohol and NaHCO3 were supplied by Chengdu Kelong Chemical Reagent Co. Ltd (Chengdu, China).
Experiment method
Preparation of samples
The samples (10 cm × 15 cm) taken from pickled goat leather (lab made) were dipped into dehydrated alcohol and cleaned with ultrasonic washer for 5 min. Then the samples were taken into a vacuum drying oven and dried at 40℃ before LTP treatment and tests.
Preparation of samples treated with LTP
The experimental procedures of LTP treatment conducted by a plasma generator (DT-O2S, OPS Plasma Inc., China) are shown in Figure 1. The dried pickled goat leather was processed in LTP, generated by a plasma generator (40 kHz) at low gas pressure (1–100 Pa) in the vacuum chamber (260 mm × 260 mm × 260 mm). O2 gas controlled by a flow meter was used to modify leather.
Schematic view of plasma reactor.
The samples were placed on the tray of LTP equipment. O2 plasma gas was introduced into the reaction chamber when the vacuum chamber was vacuum pumped to less than 20 Pa. Then process parameters were set out including pressure, discharge power (150 W), flow rate (30 SCCM – Standard Cubic Centimeter per Minute) and treatment time (0, 5, 10, 15, 20, 25, 30 min). After the plasma treatment, O2 gas was still introduced for 5 min into the chamber until the chamber was connected to the atmosphere. Then the treated samples of different treatment time were obtained.
Chrome tanning test
Samples untreated and treated with LTP were chrome tanned according to the routine chrome tanning process respectively to investigate the impact of LTP on the chrome tanning performance of leather. Chrome tanning process [17] was presented as follows: samples were weighed and placed in 500 mL glass bottle. According to the weight of the leather samples, 200 wt.% deionized water, 0.5 wt.% formic acid, 1.0 wt.% sulfuric acid and 7 wt.% NaCl were added to each bottle. Then these bottles were fixed in a thermostatic water bath oscillator to oscillate for 2 h. Then, 6 wt.% chrome tanning agents (Cr2O3, 24 ± 1%. TJ-T125-2, Sichuan TingJiang new material Co., Ltd, China) was added to each bottle by two times in 3 h. Then the pH value of the solution was adjusted to 3.8 with NaHCO3 (aq) and kept reaction at 40℃ for 8 h. The obtained tanned leather samples were dried in a vacuum oven for 24 h at room temperature. All experiments were carried out in triplicate, and average values with standard deviation errors were studied.
Characterization and performance test
Inductively coupled plasma-atomic emission spectrometry
The chromium content in the waste tanning solution was diluted into suitable concentration and tested by ICP-AES (IRIS ADV, Thermo Electron Corporation, American); thus, the concentration of chrome (c) in the waste water can be obtained directly from the results of inductively coupled plasma-atomic emission spectrometry (ICP-AES). The chrome mass (mw) in waste water can be calculated with the formula: mw = c × V (V: the volume of waste water). Then chrome exhaustion (CE) can be calculated with the formula: CE = (ma−mw)/ma (ma: the mass of chrome added to the sample in the tanning process).
Scanning electron microscopy
The morphology of the LTP treated and untreated leather samples was obtained using a scanning electron microscope (JSM-5900LV, JEOL) to investigate the etching effect of O2 LTP treatment.
Contact angle measurements
Contact angle meter (OCAH200, Data Physics Corporation, Germany) was used to measure the water contact angles on natural leather before and after O2 plasma treatment. A droplet of deionized water (0.3 ul) was dripped on the leather surface. Four readings were taken from each sample and mean value was calculated.
X-ray photoelectron spectroscopy
The leather samples were dried in vacuum oven for 24 h at room temperature before X-ray photoelectron spectroscopy (XPS) test. The surface composition of C, N and O of leather samples (0, 5, 10, 20, 30 min) was investigated using an XSAM800 (Kratos, England). Preliminary data analysis and quantification were performed using XPSPEAK 4.1 software. The binding energies (BEs) were determined by reference to the BE of the C1s peak at 284.6 eV prior to peak fitting. The untreated and O2 LTP-treated leather (10 min) were taken to conduct chrome tanning. The surface composition of C, N, O and Cr of the obtained chrome tanned leather samples was also tested as above.
Results and discussion
The effect of LTP treatment on chrome exhaustion of leather
To study the effect of O2 LTP treatment on the chrome tanning ability of leather, dried pickled goat leather samples were processed by O2 LTP under different conditions. The leather’s chrome exhaustion after different O2 LTP treatment time (0, 5, 10, 15, 20, 25, 30 min) was calculated through the results of ICP-AES.
It is seen from Figure 2 that, with the O2 LTP treatment time increasing, the chrome exhaustion of leather increases sharply (from 0 to 10 min) and decreases gradually with further treatment (from 10 to 30 min). The optimal treatment time is 10 min, which shows the maximum value of chrome exhaustion. In the tanning process, the natural leather treated by LTP may have introduced more active groups, which can enhance its cross-linking ability with tanning agents. The decline of chrome exhaustion after 10 min may be the result of over-etching, which will be studied in the following parts.
The effect of LTP treatment time on chrome exhaustion of leather. LTP: low temperature plasma.
SEM images of untreated leather and LTP-treated leather
The optimal reaction time of O2 LTP treatment was found by chrome exhaustion in the above tanning experiment. Figure 3 shows SEM images of untreated leather and O2 LTP-treated leather at 10 min and 30 min. It can be found that the grain surface of untreated leather (Figure 3(a1) and (a2)) was smoother. As shown in Figure 3(b1) and (b2), the leather grain surface became rougher and some tiny voids were produced around the soft pores after LTP treated for 10 min. When the plasma treatment time was 30 min, the leather surface was etched rougher and some filamentous fibers appeared on the grain surface and inside the pores (Figure 3(c1) and (c2)). These changes are due to the etching effect of plasma on the surface and the melting effects on the pore borders, which have already been discussed in the former researches [21–26]. The longer the leather gets processed, the rougher the surface is etched. The above results indicate that plasma treatment has a certain role in the separation and the degradation of fibers. That change will be beneficial to leather tanning to combine with more chrome, which would result in better chrome absorptivity. However, too long plasma treatment will cause over-etching, which may destroy crosslinking molecules [23], and that is disadvantageous for improving the chromium absorption rate of the leather sample.
SEM images of untreated and LTP treated leather: (a1, a2) untreated; (b1, b2) O2 LTP (10 min); (c1, c2) O2 LTP (30 min). SEM: Scanning electron microscopy; LTP: low temperature plasma.
Contact angle measurements
The contact angle results and the images of water-drop are shown in Figure 4(a) to (c). As the plasma treatment time increased, contact angle decreased from 78.7° (Figure 4(a), untreated leather) to 47.4° (Figure 4(b), 10 min) and 44.7° (Figure 4(c), 30 min), which indicated the improved hydrophilic property of leather surface. The hydrophilicity change is believed mainly due to oxidization of atomic oxygen dissociated from the oxygen gas in the plasma [23]. The oxidization may introduce many oxygen-containing functional groups, which was taken for further explanation from X-ray photoelectron spectroscopy.
The contact angle results of untreated leather and LTP treated leather: (a) untreated; (b) O2 LTP (10 min); (c) O2 LTP (30 min). LTP: low temperature plasma.
XPS analysis of natural leather and tanned leather
The effect of LTP on contents of oxygen and carbon of natural leather
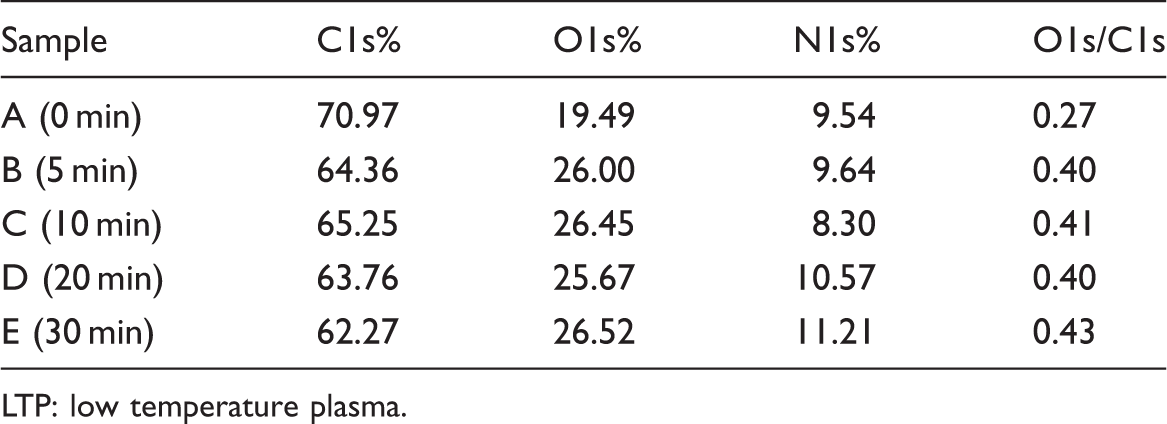
The contents of C, N, O of the untreated and O2 LTP-treated leather can be calculated by XPS data [30–33]. The XPS results of the untreated leather and O2 LTP-treated leather are shown in Figure 5 and Table 1. It can be found that, compared with untreated leather, the C1s% value decreased from 70.97% to 62.27%, the O1s% value increased from 19.49% to 26.52%, and the O1s/C1s ratio increased from 0.27 to 0.43. Oxygen content increasing indicated that more oxygen-containing groups were generated on the leather surface after LTP treatment. It is believed that there must be oxidation reaction which adds surface functionality and introduces more oxygen containing groups such as carboxyl, carbonyl and hydroxyl to the treated substrate.
The XPS spectrum of untreated and LTP treated leather: (a) untreated; (b) O2 LTP (5 min); (c) O2 LTP (10 min); (d) O2 LTP (20 min); (e) O2 LTP (30 min). LTP: low temperature plasma; X-ray photoelectron spectroscopy. Relative peak area contents of C1s, O1s and N1s of untreated and LTP-treated leather. LTP: low temperature plasma.
The reason is suggested to be that, O2 LTP treatment can introduce more active oxygen ions generated from oxygen gas dissociated in the plasma, its interaction with leather fiber will directly result in bringing about more oxygen-containing groups such as carbonyl and carboxyl [23,34]. The dissociative reaction of the oxygen gas is as below: O2 + e →O + O + e; O2 + e → O + O−. For O2 LTP treatment, atomic oxygen, charged particles and UV radiation mainly function as reactive species. The atomic oxygen mainly contributes to surface oxidation, the charged particles are the main cause of surface etching, and the UV radiation induces the increase of oxidation rate [35,36].
The C1s peaks were proceeded with peak-fit processing, which leads to several component Gaussian peaks, including C–C (284.6 eV), C–O (285.8 eV), C = O (287.6 eV) and COOH (288.6 eV). [22] Figure 6 shows the peak fitting results of the C1s under different LTP treatment time. Table 2 shows the relative area ratio of different peaks of leather samples. The O2 LTP-treated data showed obvious increase of COOH group after LTP treatment for 10 min (2.23% to 6.90%), and change little from 10 min to 30 min. It is believed that various oxidative reactions occurred during LTP treatment. New C = O groups are also believed to have formed on C–C and C–O bonds through intermolecular reorganization with radicals. New COOH bonds are believed to have formed on the C = O bonds with the active oxygen atoms. After further oxidation and stabilization with proton transfer, COOH groups could be generated. The results indicate that O2 LTP treatment can cause the leather surface oxidization, which introduces more COOH groups [37]. The 10 min treated sample has relatively high level of COOH, which results in high-chrome exhaustion value. The samples after 10 min also have more COOH. However, the over-etching effect causes the degradation of surface fibers as shown in SEM pictures, which leads to the decline of C = O content and chrome exhaustion value in 30-min sample.
XPS spectra of C1s of the untreated and LTP treated leather: (a) untreated; (b) O2 LTP (5 min); (c) O2 LTP (10 min); (d) O2 LTP (20 min); (e) O2 LTP (30 min). LTP: low temperature plasma; X-ray photoelectron spectroscopy. Relative subpeak area of C1s of the untreated and LTP-treated leather. LTP: low temperature plasma.

The effect of LTP on chromium content on the surface of tanned leather
In chrome tanning process, trivalent chromium ions mainly complex with COOH groups to generate coordination bonds. This section mainly focuses on the surface chrome tanning ability before and after O2 LTP treatment for 10 min, which can indicate the COOH change on leather surface. The untreated and O2 LTP-treated leather samples were chrome tanned in this step, and then XPS test was used to calculate the content of Cr on the leather surface; the results are listed in Figure 7 and Table 3.
The XPS spectrum of untreated and LTP treated leather after chrome tanning. LTP: low temperature plasma; X-ray photoelectron spectroscopy. Surface chromium content of the untreated and LTP-treated leather after chrome tanning. LTP: low temperature plasma.

The relative mole fraction of chrome was 1.09% and the carbon-chrome ratio was 64.84 on the surface of untreated sample. Compared with the untreated sample, the relative mole fraction of chrome of the treated sample increased from 1.09% to 1.31%, and the carbon-chrome ratio decreased from 64.84 to 50.91. The mechanism model of LTP treatment and chrome tanning of leather are shown in Figure 8. After O2 LTP treatment, more COOH groups were generated on the leather surface, which resulted in more chromium absorption.
The mechanism model of LTP treatment and chrome tanning of leather. LTP: low temperature plasma.
Conclusions
Surface modification of leather was conducted using O2 LTP at different treatment time. The best treatment time was 10 min which had the highest chrome exhaustion rate. The SEM results showed that O2 LTP-treated leather was etched relatively rougher and generated some micro-pores on the leather surface. The results of contact angel measurements and XPS showed an increase of hydrophilicity and oxygen content of leather surface treated by O2 LTP, and the surface oxidization introduced more oxygen-containing groups, especially COOH groups. The XPS data of chrome tanned leather proved more chrome on the surface after O2 LTP treatment.
Conclusion can be drawn from the above results that O2 LTP treatment under appropriate condition can cause the leather surface oxidation and introduce more oxygen-containing groups, which can improve the chrome tanning ability of leather. The results may bring new clean technique and method to the traditional industry of leather making.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors wish to acknowledge the financial support to the project from National Natural Science Foundation of P.R. China (Item No.21376152) and Department of Science and Technology of Sichuan Province (Item No.04G009-021).
