Abstract
Polyimide/organic montmorillonite nanofibrous membranes were fabricated through electrospinning of polyamic acid/organic montmorillonite precursors followed by a thermal imidization process. Scanning electron microscopy micrographs showed small amounts of organic montmorillonite addition (<3%) and increase in the diameter of polyimide nanofibers. However, higher organic montmorillonite fraction generated microspheres due to the aggregation of clay nanoplatelets. Transmission electron microscopy images indicated that both exfoliated and intercalated organic montmorillonites existed in polyimide nanofibers when low quantity of clay was loaded (<3%). As the clay amount increased, exfoliated structure disappeared and stacked clay layers were observed. The optimum concentration of organic montmorillonite is 3%. By incorporating 3% clay, the tensile strength and modulus of membrane increased by 92.1 and 48.6%, respectively. Moreover, the limiting oxygen index value arose from 29.2 to 30.4%. Higher thermal stability was also achieved because of the heat barrier effect of clay layers. The onset decomposition temperature increased by 17℃ by adding 5% clay. These results demonstrate that tiny organic montmorillonite incorporation is relatively effective for improving the mechanical properties as well as flame retardancy of polyimide nanofibrous membrane.
Introduction
Electrospinning is a facile technique that can directly produce continuous nanofibers with diameters ranging from 10 to 1000 nm. The obtained electrospun nanofibrous (ESNF) membranes possess many attractive features, such as high surface area and variable porosity [1–3]. Recently, ESNF membranes have attracted great attention due to their potential applications in tissue engineering scaffolds [4], optoelectronic devices [5], reinforcement fillers [6], and water purification [7], to name a few. Many synthetic and natural polymers have been processed to ESNF membranes that offer a great advantage in terms of the easy handling of the materials.
Polyimides (PIs) are an important group of high-performance polymers with excellent electrical insulation, good chemical resistance, as well as outstanding thermal stability. They are widely used in many technical aspects, such as aero shuttle, vehicle, and electronics [8,9]. The effective properties of PI-based materials are not only decided by the inherent chemical structures but also depend on the processing methods. PI-based ESNF membranes combine large surface area, high porosity with excellent performance, which makes these materials good candidates as lithium ion battery separators [10], high-temperature gas filters [11], catalyst support [12], thermal insulating sponge [13], etc. As a material used in high-temperature fields, excellent mechanical properties and higher thermal stability as well as lower flammability are expected for PI ESNF membranes. Great efforts have been made to improve the mechanical performance of PI ESNF membranes by adjusting spinning parameters [14–17]. Several works have been also conducted to endow the PI ESNF membranes with higher thermal stability by incorporating inorganic nanoparticles [18,19].
Montmorillonite (MMT), a type of layered silicate, is commonly used in polymer nanocomposites as reinforcing fillers due to its low cost and high aspect ratio as well as their unique intercalation/exfoliation characteristics [20–22]. The previous study showed that a small quantity of MMT loading resulted in pronounced improvements in thermal stability and mechanical properties [23]. This positive effect was observed in polyamide/clay ESNF membranes due to the improved orientation and crystallization of polymer nanofibers induced by MMT [24,25]. A similar phenomenon is expected in PI-MMT ESNF membranes. In addition, plate-like MMTs have excellent barrier properties that could improve the flame-retardant properties of polymer nanocomposite [26]. Nevertheless, rare work was reported about the MMT effect on the flammability of modified ESNF membranes.
In the present work, PI-MMT composite nanofibrous membranes were fabricated by electrospinning. The effects of clay concentration on the morphologies, mechanical properties, thermal stability, and flame behavior of PI ESNF membranes were investigated.
Experiment
Materials
Pyromellitic dianhydride (PMDA, 99%) and 4,4-oxydianiline (ODA, 99%) were purchased from Sinopharm Group (Tianjin). Anhydrous N, N-dimethylformamide (DMF, 99.9%) was supplied by Kemeiou Chemical Reagent Co., Ltd (Tianjin, China). Organically modified montmorillonite (organic montmorillonite (OMMT), Nanomer®1.34TCN) obtained by modifying MMT surface with octadecylamine was bought from Nanocor (USA). All chemical reagents were used as received without further treatment.
Preparation of PAA solution and PAA/OMMT precursor suspension
Aromatic PI was synthesized in two steps following the previous work [9]. Polyamic acid (PAA) precursor solution was prepared through the polycondensation of PMDA and ODA. Typically, 4.0048 g of ODA (0.02 molar) was dissolved in 50 ml of DMF by mechanically stirring in a three-necked round-bottom flask located in ice bath with a temperature of −2℃ in N2 atmosphere. Then 4.3624 g of PMDA (0.02 molar) was added and fully reacted with ODA for 8 h. Transparent yellowish PAA solutions were finally obtained and kept in fridge (−4℃).
PAA/OMMT precursor was prepared as illustrated in Figure 1. Dry OMMTs (0.08, 0.25, 0.42, and 0.59 g, respectively) were first dispersed in 50 ml of DMF through sonication for 10 min. Then 0.02 molar of PMDA and 0.02 molar of ODA were added as described previously. After 8 h of polycondensation reaction in N2 atmosphere, homogenous PAA/OMMT precursor suspensions were formed.
Scheme of preparation of PI/OMMT ESNF membranes. DMF: N-dimethylformamide; ODA: 4,4-oxidianiline; OMMT: organic montmorillonite; PAA: polyamic acid; PI: polyimide.
Preparation of PI and PI/OMMT ESNF membranes
Electrospinning process was performed using a 19 kV electrical potential applied to a 20 cm gap between a spinneret (inner diameter: 0.45 mm) and a rotating disc collector. The PAA solutions or PAA/OMMT precursor suspensions were fed at a speed of 1.2 ml/h and the collecting time was fixed for 4.5 h. The prepared PAA and PAA/OMMT ESNF membranes were overnight dried at 60℃ to remove the residual solvent. PI or PI/OMMT ESNF membranes were then obtained by a thermal imidization process described in previous work [9]. In this process, PAA or PAA/OMMT ESNF membranes were first heated to 120℃ at temperature ramp rate of 3℃/min in a high-temperature oven and annealed for 30 min. Then the temperature of oven was increased to 300℃ at the same temperature ramp rate. Finally, PI or PI/OMMT ESNF membranes were obtained after 120 min annealing. The prepared samples were noted as PI/M0, PI/M1, PI/M3, PI/M5, and PI/M7, respectively. The values followed the MMT (M) were their corresponding mass percentage (mOMMT/mPI × 100%). The mass of PI in composites was thought to be weight sum of PMDA and ODA used (8.3682 g).
Characterizations
The morphologies of the pure PI and PI/OMMT nanofibrous membranes were observed by a thermal field emission scanning electron microscopy (Gemini SEM500, ZEISS, Germany) at an acceleration voltage of 15 kV. Prior to tests, samples were sputter coated with gold. The chemical composition of the prepared membranes was characterized by energy-dispersive spectrum (EDS) analysis.
Fourier transform infrared (FT-IR) spectra were conducted in Nicolet iS50 Fourier infrared spectrophotometer (Thermo Fisher, USA). Each sample had a spectral result with a resolution higher than 0.09 cm−1 across a wavenumber interval between 4000 and 400 cm−1.
A H7650 transmission electron microscope (TEM, Hitachi, Japan) was used to observe the exfoliation of OMMT at an acceleration voltage of 120 kV. A thin fiber web was mounted on a copper grid in advance. X-ray diffraction (XRD) patterns were recorded in a D8 Discover X-ray diffractometer (Bruker, Germany) with CuKα radiation (λ = 0·154 nm) to determine the interlayer spacing of clay platelets.
The thickness of the nanofibrous membranes was also measured using a digital thickness gauge (Chuan Lu 0-12.7, China). Tensile tests were performed on a universal tensile machine (Instron 3369, USA) using a load cell of 1 kN and a crosshead rate of 5 mm/min. The clamping distance was 20 mm. Specimens for tensile test were cut into 40 × 8 mm2. Five specimens were tested for each sample. All tests were carried out at 23℃ and 65% humidity.
Thermogravimetric analysis (TGA) was carried out to evaluate the thermal stability of nanofibrous membranes using a STA449F3 thermogravimetric analyzer (Netzsch, Germany). All the specimens were tested in a nitrogen atmosphere from room temperature to 800℃ at a heating rate of 10℃/min.
Limiting oxygen index (LOI) values were measured on an Oxygen Index Meter (ATSFAAR 20008600, Italy) according to standard GB75454-1997. The film sample was cut into strips (58 mm× 15 mm) that were then placed in the combustor where a mixture of oxygen and nitrogen flowed upwards. The volume content of the oxygen was adjusted to a lowest oxygen concentration that could sustain burning. LOI was expressed in volume percentage.
Results and discussion
Morphologies
The morphologies of electropsun membranes are displayed in Figure 2. Pure PI showed a typical nonwoven structure composed of nanofibers with diameters ranging from 93 to 536 nm, as shown in Figure 2(a). With 1 wt% addition of OMMT, diameter of nanofiber increased due to a greater viscosity of the precursor suspension [25]. When the amount of OMMT exceeded 3%, microspheres connected with nanofibers were observed (Figure 2(c) to (e)). Generally, microspheres with larger size were generated when higher content of clay was loaded, as indicated by the arrows in Figure 2(c) to (e). High quantity of clay incorporating in polymers usually results in the aggregation of the nanoplatelets [27] that were wrapped by PI macromolecules and formed the visible microspheres after spinning. In addition, an extra band appeared near 1040 cm−1 in the composite membranes which corresponded to Si–O bending vibration of clay [28], indicating that OMMT was incorporated into PI nanofiber.
Scanning electron microscopy (SEM) micrographs of PI/OMMT nanofibrous membranes. (a) PI/M0, (b) PI/M1, (c)PI/M3, (d) PI/M5, (e) PI/M7, and (f) FT-IR spectra of PI/OMMT ESNF membranes. OMMT: organic montmorillonite; PI: polyimide.
The EDS analysis was performed to further confirm the distribution of OMMT in the nanofibrous membranes. The corresponding elements contents distribution is shown in Figure 3. C and O elements were mainly found in pure PI nanofibrous membranes (Figure 3(a1) and (a2)). However, slight amounts of Si elements were observed possibly attributed to the dust floating on the surface of the sample. With the increasing of OMMT amounts, the contents of Si elements in the composite membranes increased obviously. This result fits well with the observation of SEM and FT-IR analysis as discussed previously.
Elemental mapping analysis of C element (a1 to e1), O element (a2 to e2), Si element (a3 to e3), and their corresponding intensity (a4 to e4) in PI/OMMT ESNF membranes: (a1 to a4) PI/M0; (b1 to b4) PI/M1; (c1 to c4) PI/M3; (d1 to d4) PI/M5; (e1 to e4) PI/M7.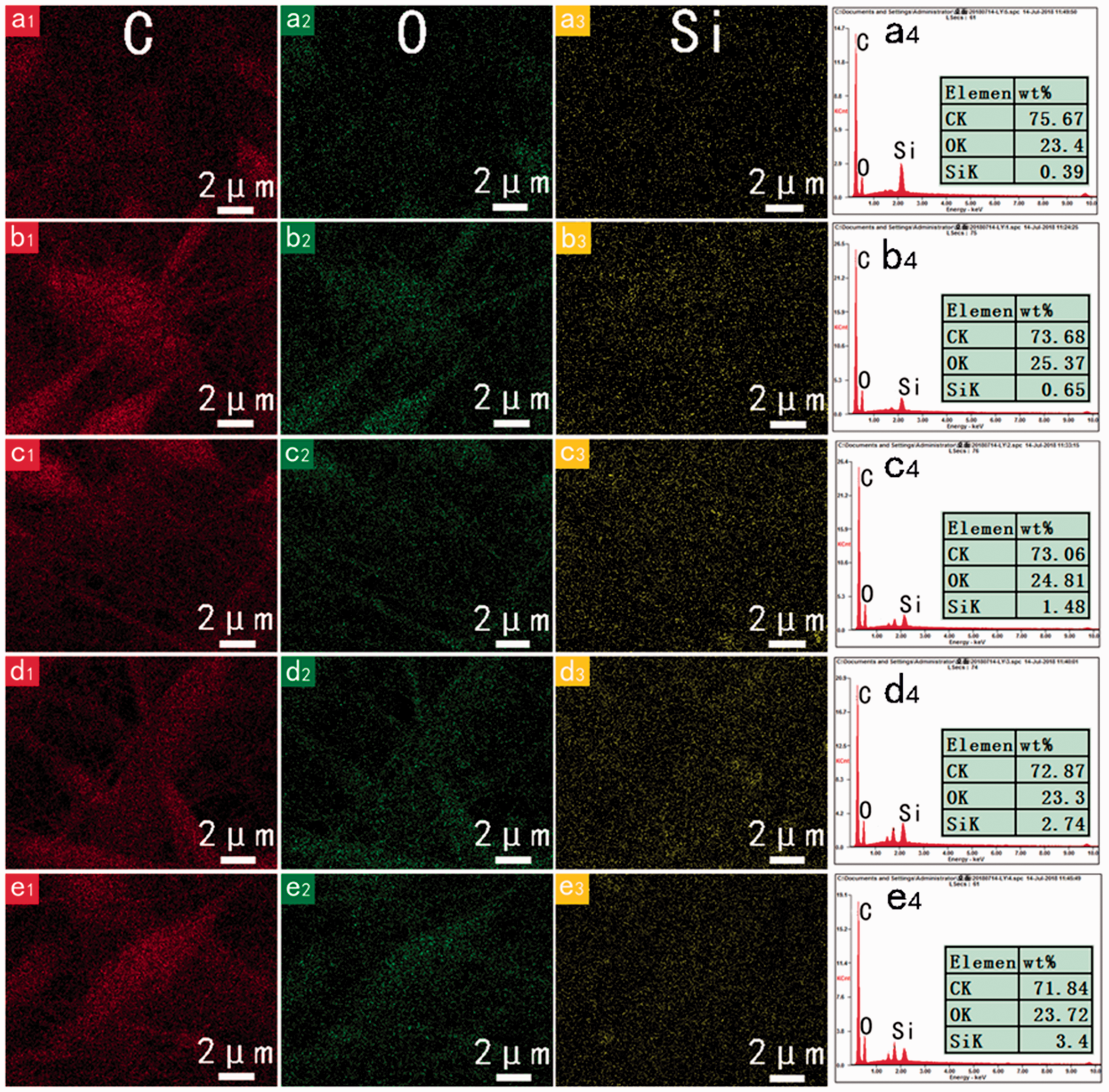
TEM cross-sections for PI/OMMT composite ESNF membranes are depicted in Figure 4(a) to (e). OMMT in PI nanofibers displayed ultrathin sheets with wrinkled, folded, and silk-like morphology (Figure 4(a) and (b)) when the OMMT content was 1 or 3%. Layered lattice structure of OMMT was also observed in some regions. These indicate the OMMT nanoplatelets maintain their highly exfoliated state in PI nanofibers; however, intercalation and unlaminated structure are also present in PI/M1 or PI/M3 [21]. Increasing the clay content to 5% and more intermediate organization of both intercalation and phase separation were observed in Figure 4(c). Further increment of clay amounts brought in the disappearance of intercalated structure and stacked clay layers increased in TEM micrographs (Figure 4(d)). This tendency is consistent with the previous study [29].
TEM cross-section images of (a) PI/M1, (b) PI/M3, (c) PI/M5, and (d) PI/M7, and (e) XRD patterns of OMMT and (f) PI/OMMT nanofibrous membranes.
XRD was used to confirm the intercalated structures and patterns of representative ESNF membranes are shown in Figure 4(f). The pure OMMT has a 2θ peak at 4.9° (Figure 4(e)) corresponding to a d001 lattice spacing of 1.78 nm [26]. In the XRD patterns of PI/OMMT nanofibers, the diffraction peak shifted to lower angles as shown in Figure 4(f). This indicates the intercalation of the PI macromolecular chains, increasing the interlayer spacing of clay platelets. On the other hand, a decrease in the degree of coherent layer stacking (i.e. a more disordered system) results in peak broadening and intensity loss in XRD patterns [30].
Tensile properties
Tensile curves of PI and PI/OMMT ESNF membranes are presented in Figure 5 and the parameters defining their tensile mechanical behavior are summarized in Table 1. Generally, the pure PI ESNF membrane behaved soft and brittle, showing a low modulus as well as a low breaking elongation. This is attributed to the low crystallinity of the electrospun nanofibers that are lack of stretching during the electrospinning process. [31]. By adding 3% clay, PI/M3 had the highest tensile strength (1.71 MPa) and the greatest Young’s modulus (6.91 MPa). This is attributed to the reinforcement effect of exfoliated clay nanoplatelets (Figure 4(b)) [26]. When the OMMT content was over 3%, clay layers tended to maintain a stacked stage in the PI fibers and then a structure of phase separation appeared as shown in Figure 4(c) and (d). As a result, sample PI/M5 and PI/M7 decreased in mechanical properties in comparison to sample PI/M3. On the contrast, clay nanoplatelets were intercalated or exfoliated and well dispersed in PI/M1 and PI/M3. The tensile strength increased with the clay content due to interfacial bonding between exfoliated clay layers and PI matrix. This is in accordance with the works reported in other cases of electrospun polyamide/MMT and polyvinyl alcohol/MMT ESNF membranes [32,33]. However, the change of break elongation of composite membranes did not display a regular tendency. PI ESNF membrane with 1% clay possessed the highest elongation at break. As is well known, the nanofibers in the ESNF membranes exhibit curved shapes and entangled organization [34,35]. When an external load is applied, the curved nanofibers tend to be first straightened and the membranes would then exhibit elongated behaviors. First, exfoliated clay increases the fiber diameter and therefore decreases the entanglement of fibers. The break elongation of PI/M1 increased to 16.21% because the shear slip increased during the tensile tests. Second, microspheres induced by higher amount of clay in the membrane (PI/M5) cause a local increase of stress, resulting in a lower break elongation. These two competing factors contribute to the random changes on the break elongation of ESNF membranes. Moreover, the thickness of tested ESNF membranes plays an important role in determining their tensile mechanical properties. It was reasonable to observe that PI/M7 showed larger break elongation than PI/M5. These results indicate that the material itself and the geometric arrangement of fibers as well as the membrane thickness could largely affect the mechanical property of nanofibrous membrane [18].
Tensile curves of PI and PI/OMMT ESNF membranes. PI: polyimide. Tensile properties of PI and PI/OMMT nanofibrous membranes. OMMT: organic montmorillonite; PI: polyimide

Thermal stability
The thermal stability of ESNF membranes was studied by TGA and the corresponding thermograms are depicted in Figure 6. The onset decomposition temperature (Td5%), the highest weight decomposition temperature (Tdmax), the maximum weight loss rate (dW/dTmax), and the residue amount at 800℃ are listed in Table 2.
TGA (a) and DTG (b) curves of PI/OMMT ESNF membranes. PI: polyimide. Thermal characteristics of PI/OMMT ESNF membranes. ESNF: electrospun nanofibrous; OMMT: organic montmorillonite; PI: polyimide

In the TGA curves, a slight weight loss below 250℃ was possible due to the decomposition of oligomers that were generated during the polymerization of PMDA and ODA. The following rapid mass loss occurred above 530℃ for all the samples because of the breaking of the PI macromolecular chains. This indicated that the synthesized PI nanofiber had a relatively high thermal stability. A further increase in temperature (above 650℃) led to a carbonization process of PI nanofibers. A high residue percentage (above 52%) was observed for each sample, which was greater than common polymers such as polyolefins, polyurethane at same temperature [36]. With the addition of clay, the thermal stability of ESNF membranes was enhanced. The initial decomposition temperature (Td5%) increased with the increment of clay fraction up to 5%. Moreover, Tdmax and residue amounts rise with the increasing contents of clay added. This is because clay nanoplatelets work as heat barrier, protecting PI from being decomposed rapidly [37]. However, when clay amount in PI ESNF membranes was further increased, Td5% decreased mildly. This is possibly related to the higher thermal conductivity resulting from clay platelets aggregation in PI matrix as shown in Figure 4(d).
LOI testing
According to the testing, the LOI value of synthesized PI ESNF membrane was 29.2. When 3% OMMT was added, LOI value of composite ESNF membranes increased to 30.4. However, further increase of clay amount lowered the LOI value of membranes. This change could be attributed to the different clay distribution when various amounts of clay were added. The stacked clay layers aggregate when high fraction of clay was loaded as shown in Figure 4(d), making the failure of clay’s barrier effect on heat [26]. Due to the low quantity of OMMT in composite membranes (<1 phr), the LOI values did not improve significantly in this work. Nevertheless, the LOI test results suggested that clay addition had a positive effect on the flame retardancy of PI ESNF membranes. The heat blocking effect of PI/OMMT membranes was evaluated following the scheme shown in Figure 7. A bunch of cotton was placed on the top of a prepared membrane (PI/M3) that suffered a flame with a temperature of ∼520℃. Cotton should be burned vigorously in a fire; however, only mild charring occurred on the surface touching the membranes. It indicates the excellent heat-resistant performance of the PI/OMMT ESNF membrane in this work.
(a) Heat blocking test, (b) cotton after test, (c) the back surface of the membrane after the test, and (d) LOI values of PI/OMMT ESNF membranes. LOI: limiting oxygen index; PI: polyimide.
Conclusions
This work prepared PI/OMMT nanofibrous membranes via electrospinning a precursor of PAA/OMMT followed by a thermal imidization process. The diameter of nanofiber increased with the incorporation of OMMT. Unlaminated, intercalated, and exfoliated clay were present in PI/OMMT ESNF membranes. With the increase of clay loading amount, exfoliated structure disappeared whereas intercalated architecture and stacked clay layers increased. When clay content reached 7% of PI mass, clay nanoplatelets tended to aggregate in the fibers. Three percent was the optimum concentration for OMMT to reinforce the mechanical properties and flame-retardant properties of PI ESNF membranes. It caused the tensile strength and modulus of ESNF membranes to increase by 92.1 and 48.6%, respectively. Improved LOI values were also obtained by incorporating low quantities of clay. Due to the heat barrier effect of OMMT, 17℃ increments on the initial decomposition temperature were observed by the presence of 5% clay. It is believed the PI/OMMT ESNF membranes have a potential application as a fire-resistant separator.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (Grant No. 51573133) and Tianjin Education Ministry S&T Project (2017KJ069) as well as Program for New Century Excellent Talents in University (NCET-12-1063).
