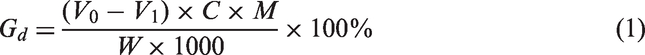Abstract
With increasing demands on the high-performance and environmental-friendly melt-blown nonwovens (MBs) for separation and filtration, herein, we reported a poly(lactic acid)/polyamide 11 (PLA/PA11) 100% bio-based MBs in situ compatibilized by dual-monomer glycidyl methacrylate–styrene melt-grafted poly(lactic acid) (PLA-g-(St-co-GMA)). The morphology, crystallization, thermal and rheological behaviors of reactive extruded PLA/PA11/PLA-g-(St-co-GMA) melt blends were investigated, and the structure and filtration performance of corresponding MBs were fully characterized. The results indicated that the interface between PLA and PA11 were effectively improved due to the compatibilizer of PLA-g-(St-co-GMA). PA11 promoted the cold crystallization and enhanced the thermal stability of PLA matrix. The reactive blends displayed a higher viscosity with unchanged rheological behaviors compared with PLA. In comparison with PLA MBs, the average fiber diameter and pore size of PLA/PA11/PLA-g-(St-co-GMA) MBs were slightly increased, whereas the strength and toughness were significantly improved. All the PLA/PA11/PLA-g-(St-co-GMA) MBs presented high air permeability and low filtration resistance. High filtration efficiency (>99.8%) could also be obtained especially for the target particles with diameters larger than 2.5 µm.
Introduction
Melt-blown (MB) is a simple, versatile, and industrialized one-step process for producing superfine fiber in micrometer and smaller scale [1]. In comparison with other techniques for producing superfine fiber materials, such as electrospinning [2], phase separation method [3], centrifugal spinning [4], MB is regarded as a more environmental friendly, economical, and efficient process as it requires no solvent [5]. Melt-blown nonwovens (MBs) also has been applied in various areas such as disposable, medical, health care, absorbing and filtration materials owing to its fluffy, porous structure with larger surface area. With tremendous developing of the bio-based polymers since this century, sustainable bio-based MBs also displays great potential as fibrous filters in the area of disposable filtration.
Poly(lactic acid) (PLA), as the most well-known bio-based and biodegradable polymer, has been widely studied and applied in many fields such as packaging, disposable and medical products [6–8], and PLA MBs also shows a huge commercial market prospect [9]. However, due to some inherent drawbacks of PLA such as brittleness and low thermal stability, the comprehensive performance of PLA products as well as PLA MBs are also limited. In order to obtain fully bio-based and toughened PLA materials, melt blending of PLA with another flexible bio-based polymer is a simple and effective way to develop PLA composites with improved or balanced properties, and this method was also considered to be greener and more economical [10]. Whereas, most of the PLA/bio-based or PLA/biodegradable polymer melt blends are immiscible from the thermodynamic standpoint.
Polyamide 11 (PA11) is one of the commercialized and bio-based polyamide, whose monomer derives from castor oil. PA11 displays superior toughness, high thermal stability, and lower crystalline melting temperature (180–190℃) than PA6 (≈220℃) and PA66 (≈253℃) [11–13]. In recent years, PA11 was also used to improve the thermal and mechanical properties of PLA in a binary blend. Stoclet et al. [14] dealt with the structural and mechanical studies of PLA/PA11 blends, self-compatibility of PLA/PA11 immiscible binary blends was found without using a compatibilizing agent, but the mechanical property of PLA/PA11 blends was not desirable. FatmaWalha et al. [15] also demonstrated that PLA and PA11 were immiscible polymers from the rheological study.
Reactive extrusion is an effective and widely used method to compatibilize the immiscible polymer blends, which could combine the efficient compatibilization with high-speed processing in a continuous way [16]. During the melting and extrusion, compatibilizer such as grafting polymer, copolymer, block polymer, polymer with functional group or catalyst are often added as the third component to induce or promote the reaction, so as to improve the interface between target polymer blends. With regard to PLA/PA11 blends, compatibilizer including reactive rubber [ethylene glycidyl methacrylate-graft-styrene-co-acrylonitrile (EGMA-g-AS)] [17], chain extender (Joncryl ADR®-4368) [15], thermoplastic elastomer (poly(ether-b-amide) (PEBA)) [18], and catalyst (titanium tetraisopropoxide (TTIP) [19] and p-toluenesulfonic acid (TsOH)) [10] were studied. To some content, the reactive PLA/PA11 blends with compatibilizer mentioned above presented enhanced toughness and strength due to the improved interface between PLA and PA11. However, it is still a challenge to design or prepare a kind of compatibilizer in a time-saving and effective way.
The method of melt-grafting functional monomers on polypropylene (PP) [20,21] and PLA [22,23], such as maleic anhydride (MA) and glycidyl methacrylate (GMA), had been frequently used because of the simplicity and cost efficiency. The grafted PP and PLA were always further used as the compatibilizer for their blends. However, the extensive degradation of PP and PLA was a common and unavoidable phenomenon during the grafting reaction due to the existence of the initiator (peroxide). The thermal degradation decreased the mechanical properties of the target polymer, especially in single-monomer grafting process. Multi-monomer grafting technique was earliest applied in the PP in order to inhibit the degradation during the grafting reaction [21]. Xie et al. [24,25] studied the melt-grafting of GMA onto PP with styrene (St) added as a comonomer during the grafting reaction. It was found the dual-monomer grafting systems could significantly reduce the degradation of PP and enhance the grafting efficiency of GMA. Inspired by that, we also considered to use this dual monomer to be grafted onto PLA during extrusion, which was further used as the compatibilizer of PLA/PA11 blends. More importantly, most of the above compatibilized PLA/PA11 blends were focused on toughened film or injection modeling, few research works were focused on the superfine fiber or MBs.
In this study, we aimed to obtain a kind of toughened and environmental friendly 100% bio-based PLA/PA11 MBs for application in separation and filtration. A new compatibilizer for PLA/PA11 blends was firstly developed using free-radical melt-grafting of dual-monomer glycidyl methacrylate-styrene (GMA-co-St) onto poly(lactic acid) (PLA-g-(St-co-GMA)) by reactive extrusion. PLA/PA11/PLA-g-(St-co-GMA) melt blends were reactive extruded and investigated by the morphology, crystallization, thermal, and rheological behaviors. The corresponding PLA/PA11/PLA-g-(St-co-GMA) MBs were also manufactured and fully characterized by the morphology, fiber diameter, pore size, mechanical properties, and filtration efficiency. The fabricated PLA/PA11/PLA-g-(St-co-GMA) toughened bio-based MBs have the potential to be used as air filtration media especially for the Particulate Matter 2.5 (PM 2.5) air pollution.
Experimental
Materials
PLA (grade 6252D especially for MB processing) with a density of 1.24 g/cm3 was a commercially available product purchased from NatureWorks LLC (USA), whose optical purity of LLA content was 93.14%. The PA11 (grade BMNO TLD) with a density of 1.03 g/cm3 was also a commercially available product purchased from Arkema (France). GMA with molar weight of 142.16 g/mol, styrene (St) with molar weight of 104.15 g/mol (purity 99% with 10–15 ppm stabilizer) and dicumyl peroxide (DCP) with molecular weight of 270.37 were supplied by Shanghai Aladdin Bio-Chem Technology Co., LTD, China.
Processing flow of PLA-g-(St-co-GMA), PLA/PA11/PLA-g-(St-co-GMA) melt blends and MBs.
Synthesis of PLA-g-(St-co-GMA) and compounding of PLA/PA11/PLA-g-(St-co-GMA) blends
Processing flow of PLA-g-(St-co-GMA), PLA/PA11/PLA-g-(St-co-GMA) melt blends are shown in Figure 1. Both of PLA and PA11 pellets were dried in a vacuum oven at 80℃ for 12 h. DCP, as an initiator, was firstly dissolved in the GMA/St solution blends at the weight ratio of GMA/St/DCP (1/1/0.05) [24] at room temperature. The GMA/St/DCP solution was then blended with PLA pellets using a mechanical mixer (SHR series high-speed mixer, Giant Company, China, rotate speed 1000 r/min) at the weight ratios of PLA/GMA/St/DCP (100/3/3/0.15). The grafting reaction of blends was carried out in a twin-screw extruder (TSE-30A, Nanjing Ruiya Extrusion System Limited, China, internal diameter =30 mm, L/D = 40) from 150℃ to 190℃, and the screw speed was 60 r/min. The reactive extruded plastic were cut into pellets and dissolved in dimethylbenzene at 130℃ for 2 h with magnetic stirring, the solution was then poured into a large excess of methanol to get the white precipitate. The white precipitate was also washed by acetone and methanol for several times to remove the residual GMA, St monomers, GMA-St copolymer, GMA and St homopolymer. The washed white precipitate, PLA-g-(St-co-GMA), was then dried in a vacuum oven at 80℃ for 2 h.
PLA-g-(St-co-GMA), PLA, and PA11 were also intermittently blended by the mechanical mixer at the weight ratios of 100/0/0, 90/5/5, 80/10/10, 70/15/15, 62/20/20, and the mechanical blends were reactive extruded in the twin-screw extruder from 150℃ to 215℃ at 100 r/min, and the PLA/PA11/PLA-g-(St-co-GMA) melt blends for MB were prepared.
Preparation of PLA/PA11/PLA-g-(St-co-GMA) MBs
PLA/PA11/PLA-g-(St-co-GMA) MBs (Gram weight ranged from 40 to 45 g/m2) were fabricated using a lab-scale MB machine (Jiaxing Longman Measurement and Control Technology Co., Ltd, China) with a single-hole die, the processing model and principle are shown in Figure 1. The diameter of orifice is 0.3 mm and the length/diameter (L/D) = 40. The blends were molten in the heated pot, and continuous nitrogen (N2) gas with constant pressure (0.2 MPa) was injected in order to guarantee the melt to be extruded in a fixed quantity. Meanwhile, the drawing air (0.3 MPa) was also heated to 270–280℃. The spinned MB fibers were cooled at room temperature and collected on a stainless steel roller (collector) with a suction set. The collecting speed and the die-to-collector (DCD) were optimized in order to get uniform MB webs. Except for the temperatures (PLA at 230℃ and PLA/PA11/PLA-g-(St-co-GMA) blends at 235–245℃), all the MBs samples were prepared using the same processing parameters.
Characterization
Characterization of PLA-g-(St-co-GMA)
Grafting degree calculation
Chemical titration method was used to calculate the grafting degree of GMA onto PLA. One gram dried PLA-g-(St-co-GMA) was dissolved in 50 mL dichloromethane (CH2Cl2), 0.5 ml 37% HCL was added into the solution and hold for 30 min in order to fully open the epoxy group of GMA. 0.05 mol/L NaOH methanol solution was then used to titrate with phenolphthalein set as an indicator. Grafting degree of GMA was calculated as following equation (1) [26]
Here, Gd is the grafting degree (%), V0 is the titrant volume of NaOH methanol solution used for PLA (ml), V1 is the titrant volume of NaOH methanol solution used for PLA-g-(St-co-GMA) (ml), C is the molarity of NaOH methanol solution, M is the molecular weight of GMA (142.15), W is the dried sample weight of PLA-g-(St-co-GMA)(g).
Fourier transformation infrared spectroscopy (FTIR) analysis
FTIR spectra of PLA and PLA-g-(St-co-GMA) were recorded using Nicolet (Thermo Fisher, USA) at room temperature. PLA and PLA-g-(St-co-GMA) samples were hot pressed (200℃) into a film with the thickness of 0.4 mm, and each spectrum was obtained by co-adding 64 consecutive scans with a resolution of 4 cm−1 in the range of 4000–600 cm−1.
Characterization of PLA/PA11/PLA-g-(St-co-GMA) melt blends
Phase morphology
Phase morphology of PLA/PA11/PLA-g-(St-co-GMA) melt blends were observed by using a field emission scanning electron microscopy (FE-SEM) (Ultra 55, Carl Zeiss, AG, Germany) at an accelerating voltage of 2–3 kV. The extruded plastic strips were immersed in liquid N2 for 30 min and broken immediately, the cryo-fractured surface of the melt blends were gold coated (2–3 nm) before observation.
Thermal-crystallization properties
Differential scanning calorimetry (DSC) tests were carried out by using a Perkin-Elmer DSC 8000 (USA) under N2 atmosphere (20 mL/min). Samples of 5 ± 0.5 mg were sealed in aluminum crucibles. Samples were heated from 25℃ to 230℃ at 10℃/min (first heating), equilibrated at 230℃ for 3 min to eliminate the thermal history and cooled to 25℃ at 10℃/min, then heated again from 25℃ to 230℃ at 10℃/min (second heating). The crystal weight fraction (Xc) of PLA and PA11 was calculated using the following equations (2) and (3)
Here,
Thermal stability
Thermogravimetric analysis (TGA) was performed by using a Perkin-Elmer TGA 4000 (USA) under the N2 atmosphere (50 mL/min). Samples of 5 ± 0.5 mg were weighted and put into a ceramic crucible, samples were heated from room temperature to 550℃ at 20℃/min.
Rheological behaviors
Rheological behavior was measured using a small amplitude oscillatory shear method on a stress-controlled rotational rheometer (MCR 301, Anton Paar GmbH, Austria) with a plane–plane configuration, a sheet diameter of 25 mm and a gap of 0.4 mm. Measurements were carried out at 210℃ in N2 atmosphere, the angular frequency ranged from 0.1 to 400 s−1 with a shear strain of 0.5%.
Characterization of PLA/PA11/PLA-g-(St-co-GMA) MBs
All the samples were balanced in a standard laboratory for 24 h with temperature (25 ± 2)℃ and relative humidity 65% before test.
Surface morphology and fiber diameter statistics
Surface morphology of MBs was observed by using FE-SEM at an accelerating voltage of 1–2 kV. MBs samples (1 cm × 1 cm) were gold coated (2–3 nm) before observation. Fiber diameter was measured by using image analysis software (ImageJ 1.48v, National Institutes of Health, USA) according to the method of Ellison et al. [29]. One hundred fibers were randomly measured from five SEM micrographs of each MBs sample. The statistical data, such as average fiber diameter (dav), standard deviation (σ), and coefficient of variation (CV) were calculated, and the fiber diameter distributions were also fit based on a polynominal fitting function (polynominal order = 5), R2 value represented the fitting degree of the experimental data.
Mechanical properties
Tensile behaviors of single-layer MBs were operated by using a universal tensile machine (Instron-3369, USA). Samples were selected in machine direction (MD); the tensile speed was 50 mm/min with the samples size of 50 mm × 20 mm. All the results were calculated as average of five specimens.
Pore size distribution
Pore size distribution of single-layer MBs was performed using the Pore Size Meter (PSM-165, TOPAS, Germany) according to ASTM F 316-03. Topor™ liquid with well wet-table properties was used as the test fluid. Circular samples with diameter of 30 ± 5 mm were randomly cut from the MBs. Bubble point method was adopted to calculate the pore size of samples.
Air permeability, filtration efficiency and resistance
Air permeability of single-layer MBs was tested by an Automatic Air Permeability Apparatus (YG 461E-III, Fangyuan Instrument, China) according to ASTM D737. A pressure difference △P = 200 Pa was loaded in a single-layer MBs with area of 20 cm2. All the results were calculated as average of five specimens.
Filtration efficiency and resistance (pressure drop) of single-layer MBs without any electret treatment were carried out by the Series AFC Particle Filter Media Test Rig (AFC 131, TOPAS, Germany). The flow rate of air was fixed at 32 L/min and circular MBs samples with diameter of 15 cm were tested. Fractional efficiency of aerosols from 0.3 µm up to 20 µm was measured with an optical particle sizer, and filtration resistance was also recorded. All the results were calculated as average of five specimens.
Result and discussion
Mechanism of dual-monomer melt grafted on PLA
Figure 2 shows the reaction mechanism for melt grafting of St and GMA onto PLA. It was well known that free radical melt grafting started with free radicals capable of abstracting H from PLA molecules. PLA macro radicals were highly reactive, which could react with other monomers in a melt system. However, it was a competition between the grafting and chain scission (β-scission) of PLA macro radicals especially in a single-monomer grafting system. PLA macro radicals, in comparison with PP macro radicals, underwent more serious β-scission because of the “bite” reaction with the terminal hydroxyl group (–OH) or carboxy group (–COOH). Whereas in dual monomer (St-co-GMA) grafting system, carbon–carbon double bonds (C=C) in St monomers was confirmed to be more reactive than those in GMA, thus St monomers were assumed preferentially to react toward PLA macro radicals according to the Q-e semiempirical equation [24], and then the GMA could be grafted on PLA-g-St macro radicals. The order of the whole reaction of PLA-g-(St-co-GMA) is shown in Figure 2, and the grafting degree of GMA onto PLA was 1.2% by calculation according to the equation (1).
Reaction mechanism of melt-grafting of St and GMA onto PLA.
Figure 3 showed the FTIR spectra of PLA and PLA-g-(St-co-GMA). As for PLA, peak at 3500 cm−1 was assigned as the –OH absorption in stretching mode; 2996, 2946 cm−1 were attributed to –CH3 stretching mode; 1753 cm−1 was the absorption of C=O stretching, 1453 cm−1 was the asymmetric bending absorption of –CH3; 1382, 1362 cm−1 could be assigned as the symmetric bending absorption of –CH3, –CH; 1268, 1183, 1129,1087, 1046 cm−1 were stretching absorption of C–O–C; 868 cm−1 was assigned as the absorption of O–CH–CH3 and 756 cm−1 as the wagging absorption of α-CH3 [30]. FTIR analysis of PLA-g-(St-co-GMA) was further carried out to verify the reaction result. Several new peaks appeared and some peaks were shifted in comparison with those of PLA. Peak at 3472 cm−1 corresponding to –OH absorption became much wider due to some hydrogen bond; peaks corresponding to –CH3 stretching mode of PLA were red shifted to lower wave numbers (2926, 2824 cm−1) because of the grafting reaction. New peaks observed at 1601, 1583, and 1493 cm−1 were ascribed to the absorption of C=C in aromatic rings, implying that the St had been grafted on the PLA. A new band was also observed at 916 cm−1, which could be designated to epoxide group, suggesting the GMA had been grafted. Peaks corresponding to the absorption of C=O and C–O–C kept unchanged, indicating that the main chain of PLA remained stable.
FTIR spectra of PLA and PLA-g-(St-co-GMA).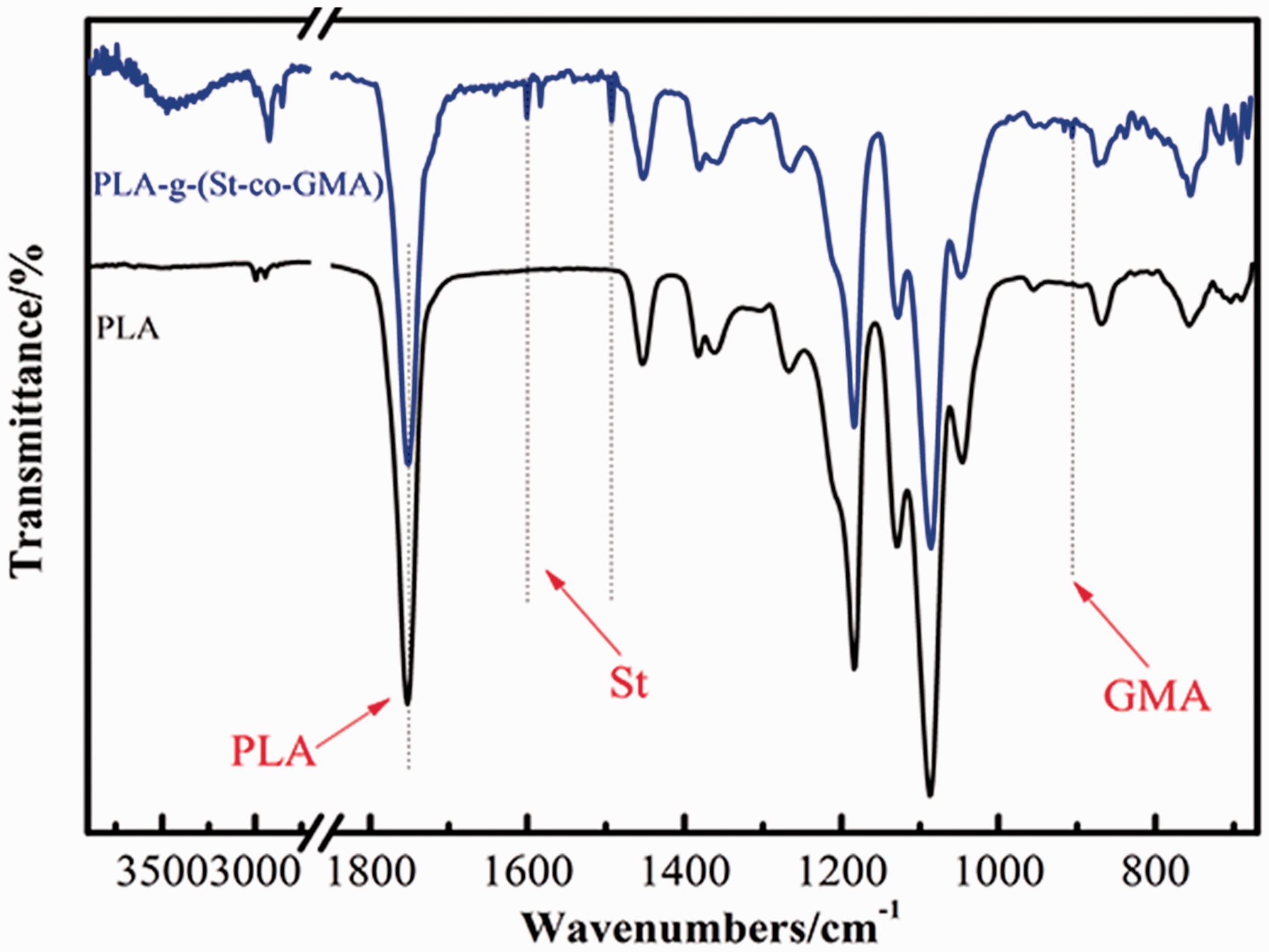
Morphology of PLA/PA11/PLA-g-(St-co-GMA) blends
SEM images of cryo-fractured PLA/PA11/PLA-g-(St-co-GMA) blends are shown in Figure 4. The phase morphology in binary polymer blends was generally affected by the weight and viscosity ratios of blending components. It was obvious to find that PLA/PA11/PLA-g-(St-co-GMA) blends still presented a “sea-islands” structure as PLA/PA11 blends showed. PA11 phases were uniformly dispersed in PLA matrix as globules shape roughly 0.5–2 µm in diameters, and some dispersed phases began to contact and even compress with each other when increasing the blending weight ratio of PA11. However, the interface between PLA and PA11 phases in PLA/PA11/PLA-g-(St-co-GMA) blends was effectively intensified in comparison to the PLA/PA11 blends reported in previous studies [14,15,17], and the compatibilized blends also displayed a rougher surface. It should be noted that some voids and debondings still existed, and the interface between PLA and PA11 was not completely “wetted” by the compatibilizer, especially when the blending proportion of PLA-g-(St-co-GMA) was low i.e. PLA/PA11/PLA-g-(St-co-GMA) (90/5/5). With increasing the blending ratio of PLA-g-(St-co-GMA) and PA11 i.e. PLA/PA11/PLA-g-(St-co-GMA) (70/15/15) and (60/20/20), the PA11 phases contacted tightly with PLA matrix and the binding effect from PLA matrix became more obvious. It was attributed to the compatibilizing effect of PLA-g-(St-co-GMA), which was selectively located on the interface between PLA and PA11 phases.
SEM images of PLA/PA11/PLA-g-(St-co-GMA) blends (A) 90/5/5, (B) 80/10/10, (C) 70/15/15, and (D) 60/20/20.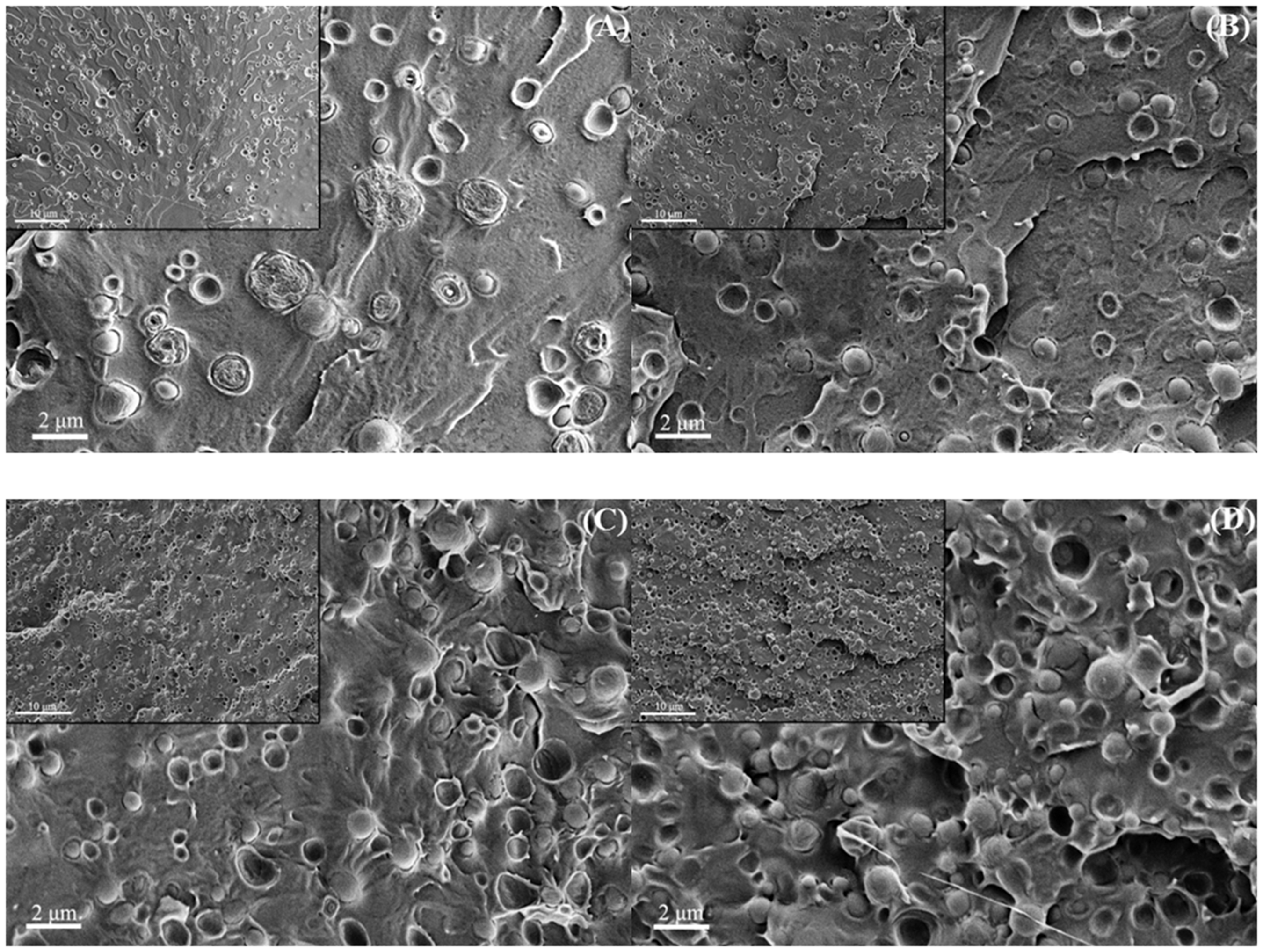
The schematic diagram of compatibilized interface between PLA and PA11 is shown in Figure 5, owing to the polarity and reactivity of the GMA on grafted chains, PLA-g-(St-co-GMA) could react with the terminal amino group of PA11, and the produced PLA-g-PA11 by reaction could be acted as the compatibilizer for PLA/PA11 blends [31]. PLA-g-(St-co-GMA) was inclined to transfer and locate on the surface of PA11 phases, and the reaction between PLA-g-(St-co-GMA) and PA11 would increase the interfacial adhesion. Meanwhile, the main chain of PLA-g-(St-co-GMA) and PLA could be well entangled between each other due to the good miscibility.
Schematic diagram illustrating the compatibilizing effect of PLA-g-(St-co-GMA) on the interface between PLA and PA11.
Melting and crystallization of PLA/PA11/PLA-g-(St-co-GMA) blends
DSC heating and cooling curves of PLA, PLA-g-(St-co-GMA), PA11, and PLA/PA11/PLA-g-(St-co-GMA) blends are shown in Figure 6, the corresponding parameters are listed in Table 1. In the heating trace of PLA, the glass transition (Tg) at 57.4℃, cold crystallization (Tcc) at 121.6℃, and two obvious separated melting peaks (Tm) were observed. Cold crystallization was the typical phenomenon for PLA during the heating due to the mobility and rearrangement of PLA macromolecules, and the double melting peaks could be attributed to the mechanisms of melt-recrystallization, multiple lamellae or crystal structures [32]. With regard to the PLA-g-(St-co-GMA), Tcc was increased and the corresponding peak became much wider, which indicated that the mobility of PLA-g-(St-co-GMA) macromolecules were limited due to the grafting components, meanwhile the double Tm peaks also got unapparent. In contrast with PLA and PLA-g-(St-co-GMA), PA11 displayed only two double Tm peaks from 181.9℃ to 188.4℃, the relative low Tm of PA11 could efficiently decrease the thermal degradation of PLA during the melt-blending process. No obvious Tg or Tcc peaks were found due to its quick crystallization by the strong hydrogen bond of N–H bonds between molecular chains. As for PLA/PA11/PLA-g-(St-co-GMA) blends, the Tcc of all blends were shifted to a lower temperature by comparing with PLA, suggesting the crystallization ability of PLA was enhanced. The crystallinity degree of PLA (Xc1) in the blends was also slightly increased, and it could be attributed to the nucleating effects of the well-dispersed PA11 phases. PA11 in PLA/PA11/PLA-g-(St-co-GMA) blends all presented only one Tm peak, and the Xc of PA11 was regularly increased from 12.7% to 20.9% with increasing the blending content. But all the Xc of PA11 in the blends were lower than that of neat PA11 (24.8%), implying that the PLA matrix also had an inhibitory effect on the crystal formation of PA11.
DSC (A) heating and (B) cooling curves of PLA/PA11/PLA-g-(St-co-GMA) blends (a) 100/0/0 (b) 0/0/100 (c) 90/5/5 (d) 80/10/10, (e) 70/15/15, (f) 60/20/20, (g) 0/100/0. DSC parameters of PLA/PA11/PLA-g-(St-co-GMA) blends. PLA: poly(lactic acid); PA11: polyamide 11; GMA: glycidyl methacrylate.

In the cooling process of PLA and PLA-g-(St-co-GMA), no obvious crystallization peak (Tc) was observed because of the slow crystallization, and it also indicated that the grafting reaction would not promote the crystallization of PLA. However, PA11 exhibited a high Tc2 at 165.6℃, the sharp and narrow Tc2 peak suggested its fast crystallization rate. With regard to the PLA/PA11/PLA-g-(St-co-GMA) blends, it was interesting to find that the Tc2 of PA11 at high temperature (165.6℃) became unapparent, while those Tc1 at low temperature (≈100℃) got more obvious. With increasing the blending ratio of PA11, the exothermic enthalpy and the Tc1 were both increased, and the Tc2 also became more apparent. This phenomenon was regarded as the typical confined crystallization and it was common in the crystalline/amorphous polymer blends with sea–island structure [33]. It was reported that PA11, as dispersed phases, underwent super cooling in the cooling process, the crystallization at Tc2 was corresponding to a relative large volume and more perfect crystals, while those at Tc1 were corresponding to unperfect crystals with small size, and this phenomenon was found in other polymer blends like polyammide 6 (PA6), isotactic polypropylene (iPP), linear low density poly ethylene (LLDPE) and poly(ethyleneoxide) (PEO) [34–36].
Thermal stability of PLA/PA11/PLA-g-(St-co-GMA) blends
Biodegradable polymers generally showed poor thermal stability, which restricted its processing at high temperature. To evaluate the effect of PA11 on the thermal performance of PLA, TG curves of PLA/PA11/PLA-g-(St-co-GMA) blends are presented in Figure 7. The corresponding thermal parameters are summarized in Table 2, T5wt%, T50wt%, and T95wt% referred to the thermal decomposition temperatures at 5%, 50%, and 95% weight loss, respectively. A single-degradation process was observed in both of PLA and PLA-g-(St-co-GMA), the close T5wt% of PLA (321.5℃) and PLA-g-(St-co-GMA) (318.8℃) indicated that the dual-monomer melt-grafting would not reduce the thermal stability of PLA significantly. Non-free and free radical theories were always used to explain the thermal degradation mechanism of PLA. The former considered that the thermal degradation of PLA was owing to the random chain scission of molecular chains, intramolecular and intermolecular ester exchange reaction; while the latter deemed that the ester pyrolysis at high temperature would produce free radicals such as alkyl and acyl oxygen, these free radicals further accelerated the thermal degradation of PLA [37,38]. In comparison with PLA and PLA-g-(St-co-GMA), PA11 displayed much better thermal stability with the T5wt% at 394.8℃. Thermal degradation mechanism of PA11 was similar to PA6, PA66, and PA12, which was due to the crosslinking between lactam and dehydration process, C–N bond rupture also existed because of the comparative low bond energy, and the produced water molecules by crosslinking could also promote the hydrolysis of amide group.
TG curves of PLA/PA11/PLA-g-(St-co-GMA) blends (a) 100/0/0 (b) 0/0/100 (c) 90/5/5 (d) 80/10/10, (e) 70/15/15, (f) 60/20/20, (g) 0/100/0. TG parameters of PLA/PA11/PLA-g-(St-co-GMA) blends. PLA: poly(lactic acid); PA11: polyamide 11; GMA: glycidyl methacrylate.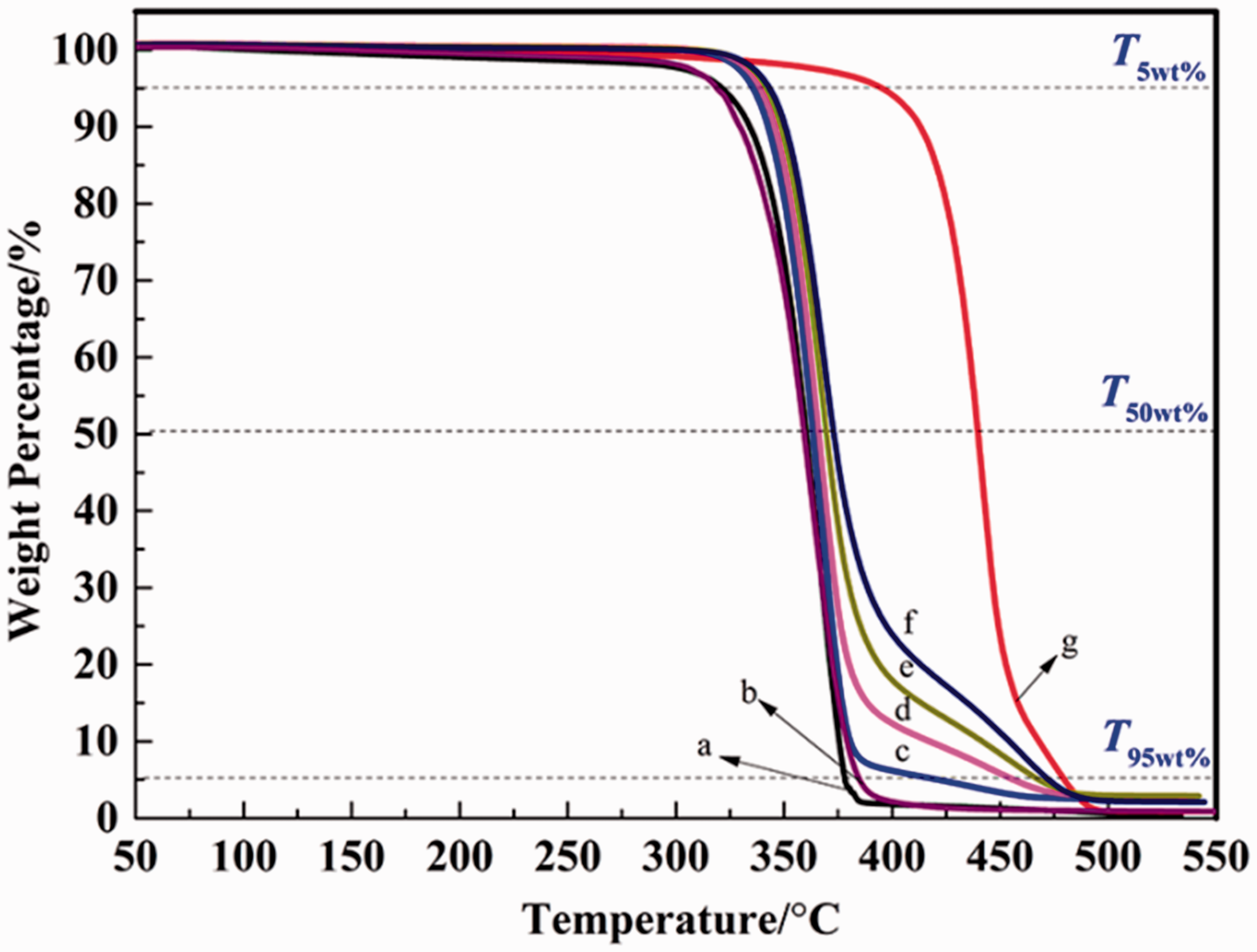
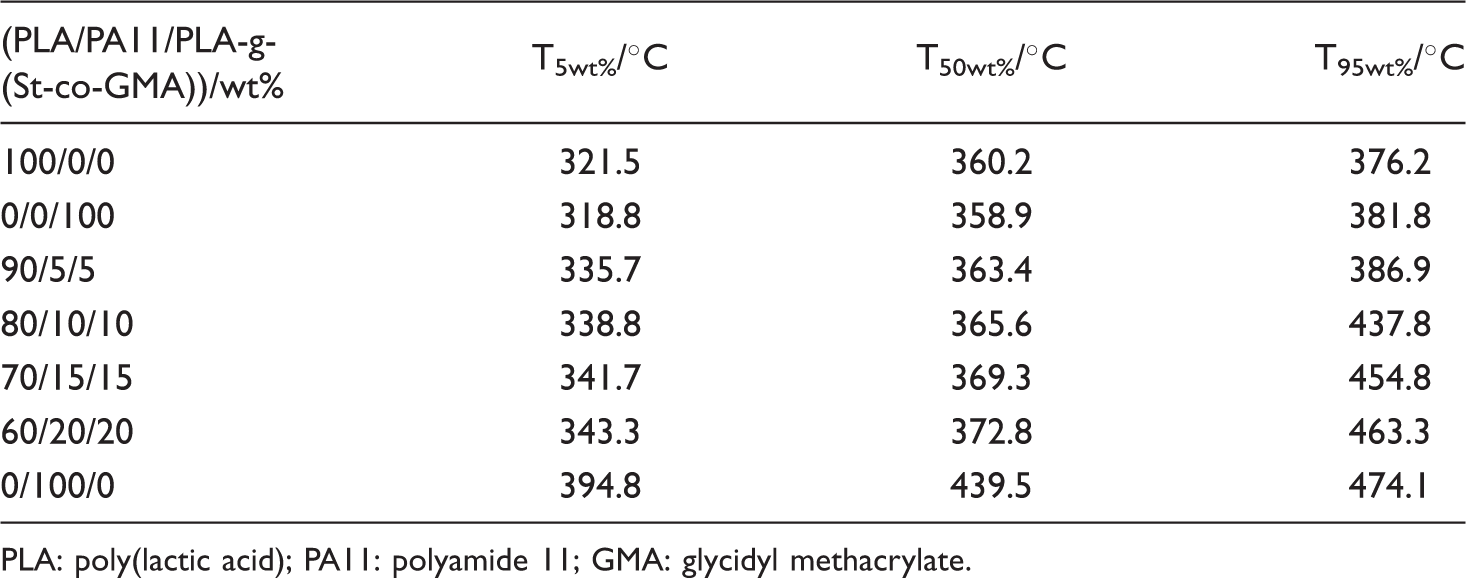
Two obvious thermal decomposition stages occurred in the PLA/PA11/PLA-g-(St-co-GMA) blends. Owing to the better thermal stability of PA11 and reactive compatibilization, T5wt% of PLA/PA11/PLA-g-(St-co-GMA) blends were higher than that of PLA by 14.2–21.8℃. The possible reason for this phenomenon was that the end groups in PLA, like ghydroxyl and carboxyl, as well as the formed free radicals in thermal decomposition, could react with the amide group in PA11. Hydrogen bonds, polycondensation reaction, and slight increase of the Xc1 could also inhibit the thermal degradation of PLA. With increasing to a higher temperature (>350℃), the thermal decomposition of PLA in blends was intensified as well as PA11, and T50wt% of PLA/PA11/PLA-g-(St-co-GMA) blends were higher than PLA only by 3.2–12.6℃, which indicated that the improvement of thermal stability was weakened.
Rheological behaviors
The viscosity behaviors of PLA/PA11/PLA-g-(St-co-GMA) blends are demonstrated in Figure 8 plotted as complex viscosity (η*) versus angular frequency (γ) at 210℃ and 230℃. PLA for MB processing displayed a low η* due to its low molecular weight (Mw ≈ 100,000–120,000) and weak intermolecular force. Non-Newtonian shear-thinning behaviors of PLA was observed in the range of 0.1–1 and 100–400 s−1 due to the unwrapping and orientation of molecular chain, but the η* remained stable as a result of the balance between the unwrapping and re-entanglement of molecular chain. PLA-g-(St-co-GMA) presented a higher η* than PLA due to the grafting reaction, the grafted segment facilitated the entanglement of PLA macromolecular chain, and some cross-linking reaction could also inhibit the molecular chain movement. PA11 also displayed higher η* even than PLA-g-(St-co-GMA) owing to its stronger hydrogen (H) bonding between amide groups. As for PLA/PA11/PLA-g-(St-co-GMA) blends, the η* ranged logically between those of PLA and PA11, and increased slightly with blending more amount of PA11 and PLA-g-(St-co-GMA). The η* of PLA/PA11/PLA-g-(St-co-GMA) blends were also sensitive to the change of γ especially when the γ exceeded 100 s−1. When the temperature was increased from 210℃ to 230℃, the rheological behavior of shear thinning kept unchanged, whereas the corresponding η* of the melts was decreased due to the accelerated movement of molecular chains. The higher temperatures enlarged the intermolecular distances, and more “free volume” allowed the molecular chains moved. The increase of processing temperatures was helpful to improve the melt-flowing index (MFI) of the melt, whereas the thermal degradation was also accelerated.
The complex viscosity (η*) versus the angular frequency (γ) at 210℃ and 230℃ of PLA/PA11/PLA-g-(St-co-GMA) blends (a) 100/0/0, (b) 0/0/100, (c) 90/5/5, (d) 80/10/10, (e) 70/15/15, (f) 60/20/20, (g) 0/100/0.
Morphology and fiber diameter distribution of PLA/PA11/PLA-g-(St-co-GMA) MBs
Surface morphology of PLA/PA11/PLA-g-(St-co-GMA) MBs is shown in Figure 9, and the corresponding statistical analysis results of fiber diameter distribution are presented in Table 3. During a typical MB process, the fibers were formed by the elongation of the polymer streams coming out of the orifice by hot air, and collected in a suitable surface to be a web [1]. It was observed that the PLA MBs displayed a entangled fiber structure whose fibers were smooth with the diameters ranged from sub-micrometer to 7 µm, and the average fiber diameter (dav) of PLA MB fibers was 3.07 µm as a typical commercial MB technology produced (2–5 µm) [39]. The nonuniform fiber distribution of MB fibers was attributed to the turbulent drawing air flow, which resulted in the irregular drawing force of hot air on the extruded polymer melt.
SEM images of PLA/PA11/PLA-g-(St-co-GMA) MBs and corresponding fiber diameter distribution (A), (A)′ 100/0/0, (B), (B)′ 90/5/5, (C), (C)′ 80/10/10, (D), (D)′ 70/15/15, (E), (E)′ 60/20/20. Fiber distribution parameters of PLA/PA11/PLA-g-(St-co-GMA) MBs. PLA: poly(lactic acid); PA11: polyamide 11; GMA: glycidyl methacrylate.

Owing to the compatibilizing effect of PLA-g-(St-co-GMA), the MB processibility of PLA/PA11/PLA-g-(St-co-GMA) blends was improved with less defects, such as deficient stretched MB fibers and molten polymer drops, compared with that of PLA/PA11, as well as the MB fibers were smooth and uniform along the fibers’ length. The dav of PLA/PA11/PLA-g-(St-co-GMA) MBs was increased from 3.58 µm to 5.24 µm with increasing the blending ratios of PA11 and PLA-g-(St-co-GMA). MB fibers with larger diameters (>10 µm) occurred and the diameter distribution also became wider which were fitted by the polynomial fitting function (polynomial order = 5). It was also found that increasing the melt viscosity led to an increase in fiber diameter of MB fibers which was consistent with the research of Gupta et al. [8]. The increase of dav in PLA/PA11/PLA-g-(St-co-GMA) MBs could be attributed to the extruded swell behaviors, which resulted in increasing the initial diameter of the MB fibers. Some curled, ribbon-shaped, and branching fibers in PLA/PA11/PLA-g-(St-co-GMA) MBs were also observed owing to the spiral flow, irregular violent whipping of hot drawing air, which led to the insufficient separation and cooling of the some fibers before reaching to the collector [29]. In comparison with PLA melt, the stability of extruding process for PLA/PA11/PLA-g-(St-co-GMA) blends from the orifice was decreased, which also contributed to the defects of MB web, such as nonuniform fiber diameters, broken fibers, polymer particles, and adhesion between fibers.
Mechanical properties of PLA/PA11/PLA-g-(St-co-GMA) MBs
In order to evaluate the toughening effect of PA11 on the PLA MBs after compatibilized by PLA-g-(St-co-GMA), the tensile mechanical properties of PLA/PA11/PLA-g-(St-co-GMA) MBs are presented in Figure 10, and the corresponding tensile parameters are listed in Table 4. It was well known that MBs, with porous and fluffy structures, consisted of many superfine fibers and cohesive points, both of which provided the whole mechanical strength. When MBs underwent stretching, the tensile force firstly made the fabric get narrower, and the fibers began to compress each other. At the same time, the tensile force transferred to the adhesive points, these points would be disbanded until the tensile force accumulated to the maximum bearing capacity. An equilibrium stage generally occurred when the tensile breakage of fibers and disbandment of adhesive points were balanced with the contribution of new fibers and cohesive points. At last, the MB web was destroyed and the strength decreases.
Tensile stress–strain curves of PLA/PA11/PLA-g-(St-co-GMA) MBs (a) 100/0/0, (b) 90/5/5, (c) 80/10/10, (d) 70/15/15, (e) 60/20/20. Tensile properties of PLA/PA11/PLA-g-(St-co-GMA) MBs. PLA: poly(lactic acid); PA11: polyamide 11; GMA: glycidyl methacrylate; MB: melt-blown nonwoven.
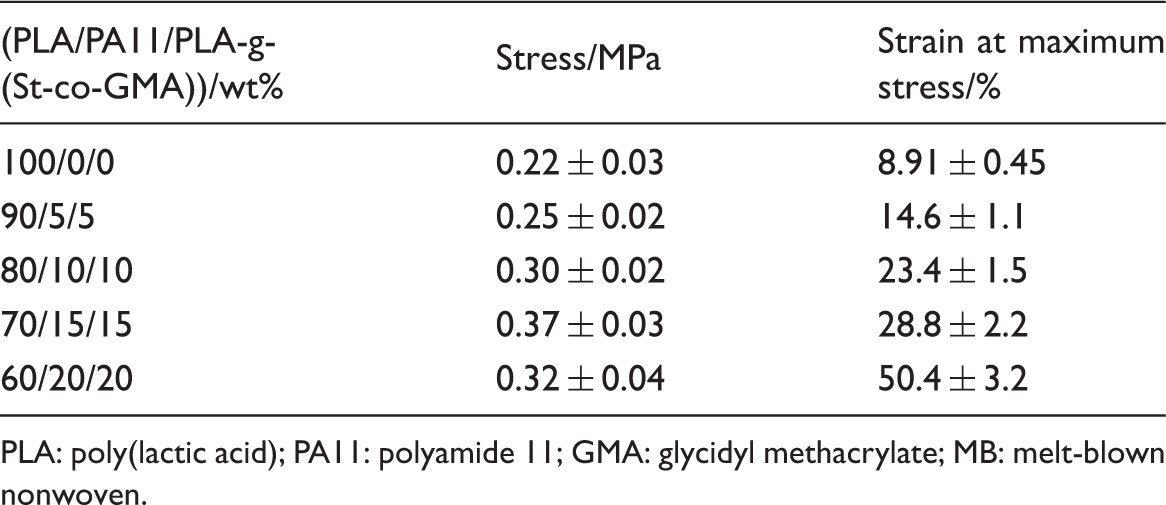
As shown in Figure 10, the strain of PLA MBs at maximum stress was about 8.91 ± 0.45%, the equilibrium stage was short, the peak was sharp and narrow due to the inherent brittleness of PLA fibers and low adhesive force. Most of the elongation of PLA MBs was attributed to its porous structure because the breaking elongation of PLA fiber or film was less than 4% in general. However, with regard to PLA/PA11/PLA-g-(St-co-GMA) MBs, the stress and the strain at maximum stress were both effectively enhanced. The stress and the strain at maximum stress of PLA/PA11/PLA-g-(St-co-GMA) (70/15/15) MBs were increased by 68.2% and 223.23% in comparison with those of PLA MBs, which indicated that the PLA/PA11/PLA-g-(St-co-GMA) (70/15/15) MBs could hold a higher strength for a longer time and withstand greater deformation due to the improved toughness. The toughening effect of PA11 on PLA MBs could be attributed to the great toughness of PA11 and the improvement of compatibility. The “rubber-like” PA11 could absorb energy and the deformation of voids locating on interface of PLA and PA11 could both increased the toughness of PLA MB fibers [18,40]. More MB fibers would be involved in the stretching process owing to the improvement of toughness, thus both of the strength and elongation were increased. Whereas, the stress of PLA/PA11/PLA-g-(St-co-GMA) (60/20/20) MBs was less than that of PLA/PA11/PLA-g-(St-co-GMA) (70/15/15), although the strain was further increased (50.4 ± 3.2%). It was mainly due to some defects in the MB web caused by declining spinnability of PLA/PA11/PLA-g-(St-co-GMA) (60/20/20) melt, such as some deficient stretched MB fibers or molten drops.
Pore size distribution of PLA/PA11/PLA-g-(St-co-GMA) MBs
Pore size distributions of PLA/PA11/PLA-g-(St-co-GMA) MBs are shown in Figure 11. Due to the random location and broad fiber distributions of MB fibers, the pore size of PLA MBs ranged from 5 to 45 µm which also displayed broad fiber distribution with double peaks. With increasing the dav in PLA/PA11/PLA-g-(St-co-GMA) MBs as shown in Figure 9, the pore size distribution of PLA/PA11/PLA-g-(St-co-GMA) MBs became broader with a few larger pores (50–65 µm) appeared. The Pav of PLA/PA11/PLA-g-(St-co-GMA) MBs was also regularly increased from 19.4 ± 1.7 µm to 31.2 ± 4.1 µm, which were larger than that of PLA (16.1 ± 1.7 µm). Two important aspects should be taken into account to explain this phenomenon. On the one hand, it was the density change of the fiber location in MB fibrous web. The increase of the fiber diameter indicated that the gaps between fibers became wider, because the extruded quantity of the polymer melt and rolling speed of collector were fixed in the MB processing. On the other hand, the increase in the degree of the fiber entanglements also had an influence to largen the pore size in PLA/PA11/PLA-g-(St-co-GMA) MBs.
Pore size of PLA/PA11/PLA-g-(St-co-GMA) MBs (a) 100/0/0, (b) 90/5/5, (c) 80/10/10, and (d) 70/15/15, (e) 60/20/20.
Air permeability and filtration efficiency of PLA/PA11/PLA-g-(St-co-GMA) MBs
MBs is widely applied in filtration and separation and absorption materials due to the specific large surface area of fluffy bulky web and superfine fibers, thus air permeability as well as filtration properties are important indexes for MBs. The results of air permeability and filtration resistance of PLA/PA11/PLA-g-(St-co-GMA) MBs are shown in Figure 12(a). In accordance with the results of pore size distribution shown in Figure 9, the air permeability of PLA/PA11/PLA-g-(St-co-GMA) MBs increased from 308.4 ± 8.2 mm·s−1 to 405.2 ± 17.3 mm·s−1, which were larger than that of PLA (273.9 ± 6.2 mm·s−1). The filtration resistance i.e. the filtration pressure drop of PLA/PA11/PLA-g-(St-co-GMA) MBs also decreased regularly compared with the PLA MBs. All the PLA/PA11/PLA-g-(St-co-GMA) MBs presented high air permeability and low filtration resistance.
(A) Air permeability and (B) filtration efficiency of PLA/PA11/PLA-g-(St-co-GMA) MBs (a) 100/0/0, (b) 90/5/5, (c) 80/10/10, (d) 70/15/15, (e) 60/20/20.
Air permeability and filtration performance parameters of PLA/PA11/PLA-g-(St-co-GMA) MBs.

PLA: poly(lactic acid); PA11: polyamide 11; GMA: glycidyl methacrylate; MB: melt-blown nonwoven.
Conclusion
In this study, a new kind of compatibilizer (PLA-g-(St-co-GMA)) with GMA content of 1.2% was developed using free-radical melt grafting of dual-monomer (GMA-co-St) onto PLA by reactive extrusion method. The PLA/PA11 melt blends compatibilized by PLA-g-(St-co-GMA) for MB were prepared, and the morphology, crystallization, thermal, and rheological behaviors were fully investigated. PLA-g-(St-co-GMA) was proved to efficiently improve the interface between PLA and PA11. Spherically dispersed PA11 phases promoted the cold crystallization and evidently enhance the thermal stability of PLA matrix. In comparison with PLA, PLA/PA11/PLA-g-(St-co-GMA) blends also displayed non-Newtonian shear-thinning behaviors while the viscosity was slightly increased.
PLA/PA11/PLA-g-(St-co-GMA) MBs were also successfully manufactured, and the structure, mechanical properties, and filtration performance were evaluated. In comparison with PLA MBs, the average fiber diameter and pore size of PLA/PA11/PLA-g-(St-co-GMA) MBs were slightly increased, but the strength and elongation were both efficiently improved. The stress and the strain at the maximum stress of PLA/PA11/PLA-g-(St-co-GMA) (70/15/15) MBs were increased by 68.2% and 223.23% compared with those of PLA MBs. All the PLA/PA11/PLA-g-(St-co-GMA) MBs presented high air permeability and low filtration resistance. The filtration efficiency of PLA/PA11/PLA-g-(St-co-GMA) decreased compared with that of PLA when the diameter of target particle was less than 1 µm, whereas high filtration efficiency (>99.8%) was still obtained for the larger particles (>2.5 µm). It was also believed that PLA/PA11/PLA-g-(St-co-GMA) toughened MBs with better comprehensive performance could be obtained by optimizing MB machine and processing parameters.
Footnotes
Acknowledgements
We appreciate the help from Prof. Gangjin Chen of Hangzhou Dianzi University, China, and Prof. Darko Ujevic of University of Zagreb, Croatia.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Key Lab. of Industrial Textile Materials and Manufacturing Tech. of Zhejiang Province, and Zhejiang Provincial Project of Science and Technology (2017C33077).