Abstract
A novel polymeric flame retardant with phosphorous–nitrogen synergism (PVP (PR)-P-DCDA) was synthesized by polyvinyl alcohol, hydrophilic polyester resin, phosphoric acid, and dicyandiamide. 100% polyester, 100% cotton, and 50/50% cotton–polyester blended fabrics were treated with PVP (PR)-P-DCDA by impregnation method. Flammability characteristics, thermal decomposition, surface morphology, and chemical structure of treated and untreated fabrics were investigated by vertical flammability test, limiting oxygen index, differential scanning calorimetry, thermogravimetric analysis, scanning electron microscopy, and Fourier-transform infrared, respectively. Results showed that PVP (PR)-P-DCDA is an eco-friendly flame retardant system, a good char-forming flame retardant agent with superior ease of application for cotton, polyester, and cotton/polyester blends. At the industrial scale, the flame retardant agent PVP (PR)-PDCDA has been commercialized under the name Fire-off EBR.
Introduction
The natural-synthetic fiber-blend fabrics are more suitable than pure natural or synthetic textile products for many purposes. Especially, cotton–polyester (CO/PET) blends are quite popular with a high usage in home textiles (furnishing, bedding), working clothing, knitwear, and outwear camping textiles where certain flame retardancy is needed [1]. However, significant differences in physical, chemical, and thermal properties of cotton and polyester induces flammability problem in these blends [2]. When burning, cotton tends to char on heating but generally maintains some structural integrity. Polyester releases a large amount of heat, and normally melts at 260℃. The molten polyester drips, and frequently tends to wick on the cotton char, resulting in the phenomenon of scaffolding, which creates favorable conditions for burning [3–11]. Because of this flammability-enhancing interaction, conferring flame retardancy to CO/PET blends is a complex and crucial problem [10,11].
The flame retardant (FR) chemicals commercially used for CO/PET blends are not sufficient to comply with the European standards and they have application difficulties such as extra equipment requirement for binding. Usage of halogenated FR chemicals is restricted due to the environmental problems. For instance, the EU community and the United States have banned brominated diphenyl derivate as they have clearly shown a high toxicity for both animals and humans [12–14]. Alternatively, FRs based on phosphorous compounds have shown that they have lower toxicity profiles as compared to halogen-based counterparts [15,16]. However, the phosphorus–nitrogen (P–N) FR systems (N/P-CH2OR(H)-type FRs or crosslinkers) such as Fyrol 76, Fyroltex HP, Pyrovatex CP New, and Proban have a formaldehyde-release problem despite showing a good N–P synergism [4,17–26].
FR treatment of fabrics is an economical and practical choice among the other methods to impart flame retardancy. In recent years, researches on FR systems for textiles (particularly referring to cotton, polyester, and their blends) have been mainly focused on topics such as the improvement of its effectiveness and replacement of toxic chemicals with counterparts that have low environmental impact and are more sustainable [27–29]. With regard to cotton, polyester, and CO/PET blends, there are ongoing attempts to develop efficient formaldehyde-free, char-former FR systems. Li et al. [10] synthesized a novel polymeric FR with P–N pendent groups (PVP-P-DCA) by polyvinyl alcohol (PVA), phosphoric acid, and dicyandiamide (DCA). This FR was applied to CO/PET fabrics with 30/70 blend ratio by pad-dry thermosol finishing process. As a result, they found that PVP-P-DCA is a reactive FR agent on CO/PET blends with high efficiency. LOI value was determined up to 26.4 after 10 times washing; however, the washing process used in the study is extremely mild and cannot meet the current standards of washing conditions [10]. As a further research, Li et al. [11] developed hydroxyl-functional, organophosphorus FR with chemical structure of 2,2-dihydroxymethylpropane-1,3-diolylbis (hydrogen phenylphosphonate) (DHDBP) and implemented to 30/70% CO/PET blends. The results indicated that DHDBP was reactive and a good char-forming agent for the CO/PET blended fabrics. However, it was not durable against 20 laundering cycles due to insufficient fixation of DHDBP on the fiber. Developing phosphorus-based UV curable FR coating systems have been attempted in the literature since 2011. In the study of Opwis et al. [30], cotton, polyester, and polyamide fabrics were treated and all of them passed the DIN EN ISO 15025 fire test exhibiting self-extinguishing behavior. In 2012, Xing et al. [31] achieved a maximum LOI value of 24.5% and Yuan et al. [32] obtained LOI values of 27% for treated cotton fabrics and strong reduction in peak heat release rate and total heat of combustion were obtained for treated cotton. In 2015, Mayer-Gall et al. [33] developed a UV curable FR agent (allyl-functionalized polyphosphazenesally) and applied this product onto cotton and different cotton/polyester blended fabrics, using an impregnation method followed by UV-grafting to achieve permanent effect. Results showed that the treated fabrics exhibited good flame retardancy (LOI: 24-27 for CO/PET blends) with char formation. The UV-cured FR coatings showed also a good washing fastness. However, it needs improvement in terms of solubility in aqueous systems to be a commercial textile-finishing product. Apart from grafting flame retardant films on textile surfaces, an efficient approach for the covalent bonding of phosphorus-based [34] and phosphorus–nitrogen-based flame retardants [35] were developed for cotton. On subjecting to treatments, compound fabrics with LOI values of more than 30% were obtained. With regard to fabrics made of PET, a mixture of a bis-phosphonic acid derivative and ammonium sulfamate that shows not only efficient flame retardant but also anti-dripping properties when applied to PET was reported by Feng et al. [36] Padding–curing process was used to apply different ratios of these compounds onto PET fabrics. More than 28% LOI values could be achieved. Furthermore, the after flame time was 0 s and the fabrics showed an intumescent char structure after burning. Using eco-friendly and sustainable FRs instead of the current phosphorus-based synthetic products has started to be investigated since 2014. As an example of this approach, Carosio et al. [37] studied the FR effect of caseins on polyester and CO/PET (35/65%) fabrics. Fabrics were treated with an aqueous suspension of caseins through pad-dry-cure method. The results showed that caseins had enhanced the resistance of cotton, polyester, and CO/PET blends against a methane flame, strongly reducing the burning rate in the case of polyester and CO/PET blends. The number of studies related to developing P–N-based FR systems have also been increased because of synergetic effect of P and N. As an example, Jiang et al.’s [38] newly developed P–N-based FR was applied to cotton, nylon, and polyester fabrics using padding method. As a result of the vertical flammability test, treated nylon exhibited high flame retardancy with B1 grade FR, while treated cotton and polyester exhibited moderate flame retardancy (B2 grade). However, the study did not involve LOI test. Li et al. [39] developed a formaldehyde-free P–N FR chemical with multiple reactive groups, dioxo(3-triethylphosphite-5-chlorine-1-triazine) neopentyl glycol (DTCTNG) for cotton fabrics. FRs were applied to cotton fabrics via padding. FR tests and characterization test indicated that chemical cross-linking occurs between FR and the cotton matrix. LOI value of 27.3% and B1 grade of vertical flammability test were obtained at 25% add-on sample. Recent interest has also been shown with the potential for combining phosphorus, nitrogen, and silicon with cellulose substrates in order to create carbonaceous and silicaceous char formation [27]. In the study of Zhao et al. [40], a novel halogen-free phosphorus–nitrogen–silicon FR monomer with reactive siloxy groups, N-(diphenylphosphino)-1,1-diphenyl-N-(3-(triethoxysilyl)propyl) phosphinamine (DPTA) has been synthesized and was applied to the fire-resistant finishing of cotton fabrics via impregnation method. TGA, MCC, and LOI test showed that treated cotton fabrics with 29 add-on % DPTA showed 40% char residue, which apparently increased the heat release rate and LOI value of 30.7%.
In the light of the studies in literature, the initial motivation for this research was to develop an eco-friendly, easily applicable FR finishing agent at industrial scale with P–N synergism effect for CO/PET blends, which may be also compatible with both cotton and polyester. In order to enhance the durability of FR properties, polyester resin (PR) was used in addition to PVA and dicyandiamide (DCDA) in the formulation. 100% polyester, 100% cotton, and 50/50% CO/PET blends were treated via pad-dry-thermosol process with this novel polymeric flame retardant PVP (PR)-P-DCDA. FR performance and thermal properties of treated fabrics were investigated.
Experimental study
Materials
The physical characteristics of the fabrics.

50/50% CO/PET blend ratio is a crucial blend, which shows the effect of each blending component on the combustion behavior of the fabric. Because polyester-rich fabrics show generally polyester burning behavior and it is the same for cotton-rich ones [1,2]. So 50/50% CO/PET blend was chosen for the experimental study.
Chemicals namely PVA 1799 (CAS NO.: 9002-89-5, supplied by Sinochem Nanjing Corporation), orthophosphoric acid (H3PO4), hydroxyl terminated polyester resin (PR), and dicyandiamide (DCDA) supplied by private suppliers of Eksoy Company were used in the synthesis of FR agent.
Synthesis and characterization of PVP (PR)-P-DCDA
In the synthesis, PVA was chosen in terms of high reactivity, high carbonization ability, and low cost. PVA is also a good charring agent and has compatibility with cellulose. DCDA was used owing to being a good blowing agent in intumescent FR system [41–43]. Hydroxyl-terminated polyesters are commonly used as modifiers in epoxy resins [44–46] and polyurethane-based coatings [47–49] due to effective molecular weight build-up by a chain extension reaction. They are the most common polyols, which are cross-linked through isocyanate groups [50,51]. Because of its crosslinking ability, PR (hydroxyl terminated polyester resin), which is the commercial product of Eksoy Company, was used to increase the durability via crosslinking between FR chemical and cotton. The chemical formula of PR is represented in Figure 1.
Hydroxyl terminated polyester resin (PR).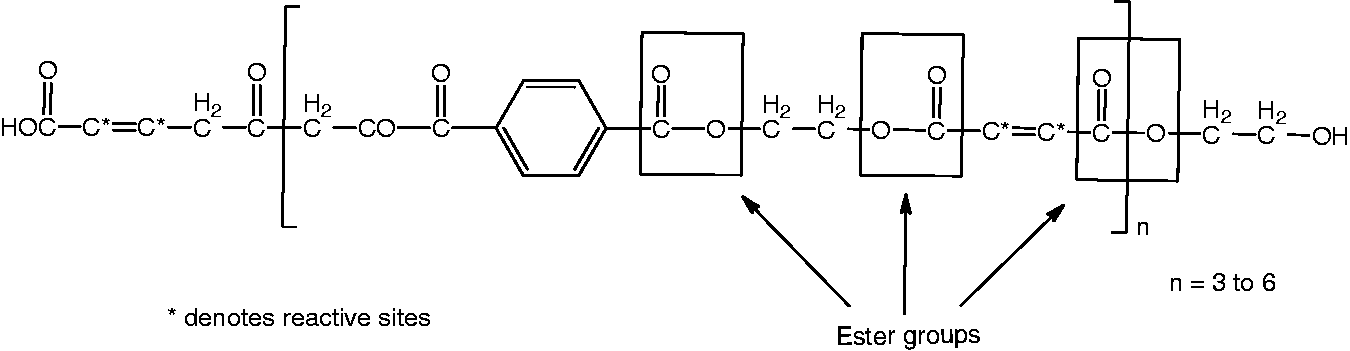
In order to produce a high yield of P–N polymer, two-step process consisting of esterification and polymerization was performed. FTIR spectra of PVP (PR)-P-DCDA were conducted with a FTIR spectrophotometry.
Synthesis of PVA-POH+ PR-POH (PVA (PR)-POH)
In the process, PVA 1799 (25 g) and hydroxyl-functional polyester resin (5 g) were added into a 500 mL glass flask equipped with mechanical stirrer, thermometer, and circumference condenser. Distilled water of 120 mL was added and the mixture was stirred for 60 min in order to obtain a homogenous mixture. Orthophosphoric acid of 55 mL (85%) was added drop by drop into the mixture for 30 min. Then, 1.2 g urea was added as a catalyst. Furthermore, the mixture was gradually heated up to 100℃. After being stirred for 3 h at 100℃, the suspended solution was slowly filtered and precipitated in 250 mL ethanol. This process was repeated for three times and the resulting white product was dried at 60℃ for 8 h (yield = 83%).
Synthesis of PVP-P-DCA PR-P-DCDA (PVP (PR)-P-DCDA)
PVA(PR)-POH of 35 g and 45 mL distilled water were added to a three-necked flask equipped with magnetic stirrer, thermometer, and condenser. The mixture was stirred for 30 min to obtain a homogenous mixture. The temperature was gradually increased to 70℃. When it turned to a transparent solution, 13 g DCDA was added into the solution. After the reaction for 2 h at 100℃, the obtained white solution was cooled and precipitated in 100 mL ethanol and then filtered. This process was repeated for three times. The filtered product was dried for 8 h at 60℃ (yield = 80%). The flow chart represented in Figure 2 summarizes the synthesis process.
The flow chart of PVP (PR)-P-DCDA synthesis.
The reaction was repeated several times with optimum pH, temperature, and stirring conditions determined by specifications. Repeatability of the product was achieved and provided by characterization tests. Reaction route of PVP (PR)-P-DCDA is shown in Figure 3.
Reaction route of PVP (PR)-P-DCDA.
Finishing of fabrics with PVP (PR)-P-DCDA
Different FR baths with PVP(PR)-P-DCDA (75%) were prepared in distilled water with different concentrations such as 300 g/L, 350 g/L, 400 g/L, and 450 g/L. 100% cotton, 100% polyester, and 50/50% CO/PET fabrics were impregnated via 3 dip-3 nip padding at room temperature with 85% wet pick-up. Only for low FR concentrations, pick-up ratios dropped to around 70% for polyester samples due to their hydrophobic features. Samples were then dried at 100℃ for 3 min. They were subsequently cured at 180℃ for 3 min. The add-on (%) of FR was calculated from the following equation

Evaluation of the flame retardancy of treated fabrics
Vertical burning test was conducted according to the ISO 6940:2006 Procedure A (surface ignition). For each fabric type, 12 fabric specimens (length 200 mm ×width 50 mm) were prepared and conditioned at 20 ± 2℃ and 65 ± 2% relative humidity for 24 h before testing. The maximum flame application time (20 s) was used in the test. According to ISO 6940, materials that do not ignite with this flame application time will not normally ignite with higher flame application times. As the fabric structures are plain weave, all tests were performed in lengthwise direction of the fabric samples. Damaged lengths (Figure 4) and weight loss (%) (equation 2) of samples were measured after 20 s of flame application for in-depth investigation.
Damaged length measurement in ISO 6940 vertical flammability test.

Limiting oxygen index (LOI) test was carried out according to BS 4589-2 standard. LOI is one of the most popular scientific methods for FR evaluation. Textile materials that exhibit LOI values up to 21 vol.% burn rapidly, while they burn slowly when LOI is between 21 and 25. When LOI values are beyond 26, it indicates some FR features [52].
Characterization techniques
The chemical structure of the untreated and treated fabrics was characterized by Fourier-transform infrared spectroscopy (FTIR) using Perkin Elmer spectrum One FTIR-ATR System.
The thermal behaviors of untreated and treated polyester and CO/PET fabric samples were investigated by differential scanning calorimeter (DSC). DSC thermograms for polyester and CO/PET samples were obtained using a Perkin-Elmer model differential scanning calorimeter, from −30℃ to 275℃ with a heating and cooling rate of 10℃/min. Samples of about 4–5 mg were sealed in aluminum pans. Nitrogen was used as the purge gas to provide an inert atmosphere and to prevent sample degradation. All DSC thermograms were analyzed using Pyris software for quantitative evaluation of melting, crystallization temperatures, and their associated heat capacities and enthalpies.
Thermogravimetric analyses (TGA) of the samples were performed from 30℃ to 800℃ at a heating rate of 10℃/min under a dry nitrogen atmosphere (purging rate 20 mL/min) using a SEIKO Exstar 6200 TG/DTA instrument (Olomouc, Czech Republic). The sample masses were 4–5 mg, placed in open platinum pans.
The surface morphology of the untreated and treated samples was investigated using an EVO MA10 scanning electron microscope at an acceleration voltage of 15 kV and about 20 mm working distance. Samples were mounted on aluminum specimen stubs with double-sided adhesive tape. They were sputter-coated with a gold layer in rarefied argon using a Cressington Sputter Coater with a current of 20 mA for 180 s.
Results and discussion
Synthesis and characterization of PVP(PR)-P-DCDA
The esterification reaction occurred between PVA and H3PO4. The production of polymer with an appropriate phosphorus: nitrogen (P : N) synergy was completed by the polymerization reaction of PVA-POH PR-POH and dicyandiamide (DCDA). The white product PVA-based, P–N FR (PVP (PR)-P-DCDA) was obtained with a yield of 80%.
FTIR spectrum of PVP (PR)-P-DCDA is illustrated in Figure 5. –OH stretching vibrations appeared around 3500 cm−1. NH2 and N–H stretching vibrations were around 3330 and 3200 cm−1, respectively. The wave number of 2800 cm−1 exhibited –CH2− vibration. The C≡N stretching vibrations were found at around 2390 cm−1. The wavelengths of 1745 and 1700 cm−1 displayed C=O and C=NH stretching vibrations. Wave numbers of 1595 and 1578 cm−1 were bend vibrations of C–C and C=NH2+, respectively. P=O stretching vibration was found at around 1222 cm−1. The stretching vibrations of –O–O–P and C–O–P appeared around 1112 and 1090 cm−1, respectively. P(V)–O stretching vibration is seen in the wavelength of 951 cm−1. The characteristic absorption peaks of ammonium appeared is an evidence of the salt forming.
FTIR spectrum of PVP (PR)-P-DCDA.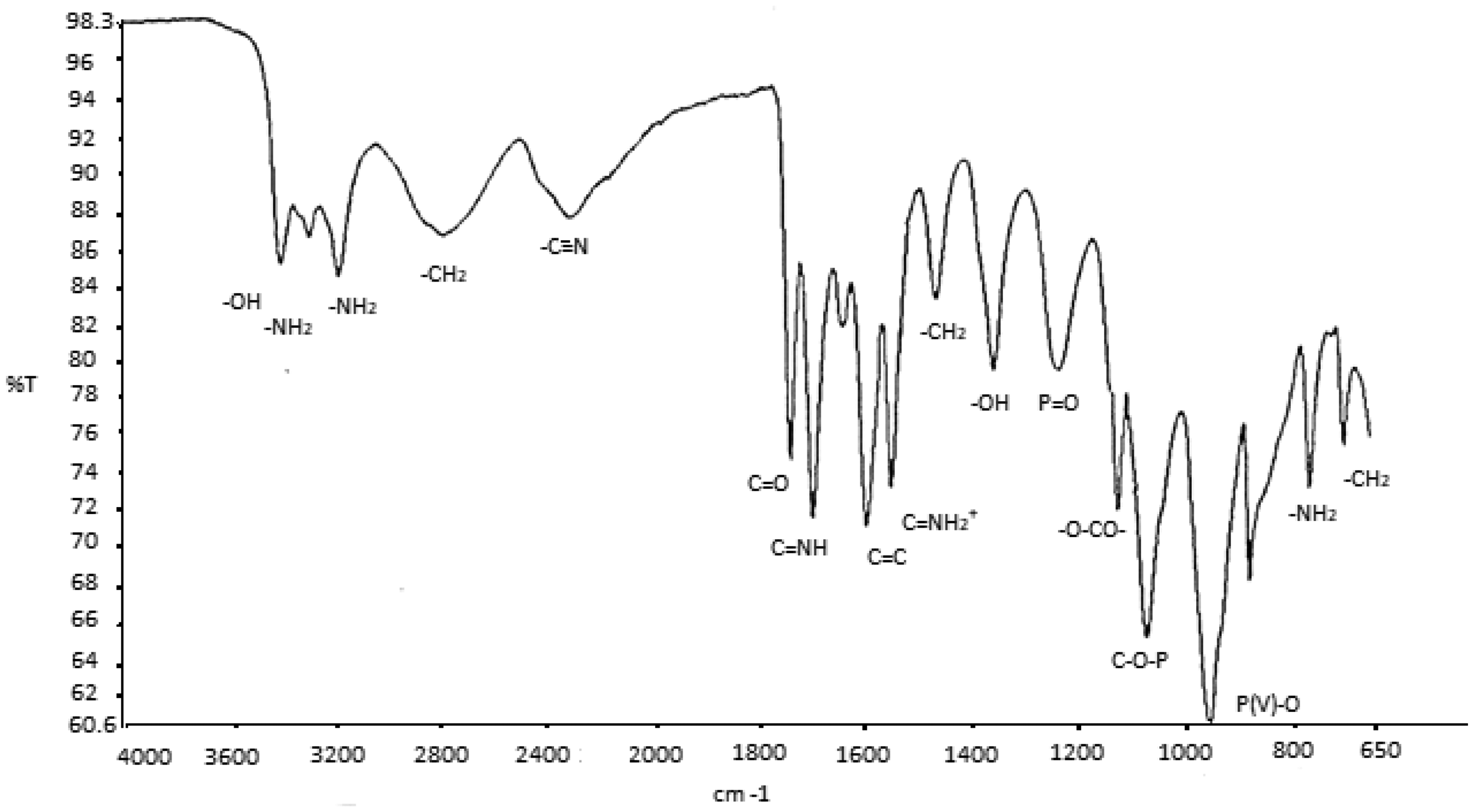
Evaluation of the flame retardancy of treated fabrics
Flammability of treated fabrics was tested according to ISO 6940 with 20 s of flame subjection. All fabric types showed strong resistance to burn, whereas cotton and CO/PET samples formed charring; while polyester samples showed melting with with hole forming into fabrics during the flame application. The pictures of fabrics after the test are depicted in Figure 6. After removal of the flame source, neither afterflame nor afterglow was observed on any kind of fabrics used in the study.
Pictures of (a) treated cotton, (b) CO/PET samples, and (c) polyester fabrics (300 to 450 g/L) at the end of vertical flammability test.
Flammability results of untreated and treated cotton, polyester, and CO/PET fabrics.

LOI: limiting oxygen index; FR: flame retardant.
As clearly reported in Table 2, the damaged lengths and weight losses of the all three treated types of fabric are inversely associated with the concentrations of the FR agent. Higher FR agent concentrations lead to shorter char lengths and lesser loss of mass in the treated cotton and CO/PET fabrics. However, this trend is more obvious for cotton than CO/PET as also can be seen in Figure 6. It is negligible and less deterministic for polyester samples owing to their lightweight and sensitivity of fabric.
As it is also obvious in Table 2 (third column), PVP (PR)-P-DCDA treatment are able to remarkably increase the LOI values of cotton (17.6 to 27.1), polyester (22.5 to +38), and CO/PET (17.5 to 26.3) as compared to the untreated fabrics. Cotton samples demonstrate flame retardancy at ∼15% add-ons, whereas CO/PET samples require minimum add-on of 17% to achieve a LOI of 26. On the other hand, the LOI value of polyester rises up to 33 even at 8–9% add-ons.
To sum up, FR concentration in finishing treatments is strongly related to the LOI values, damaged length, and weight loss of fabrics. Polyester fabrics give more desirable results than CO/PET and cotton fabrics in terms of damaged areas, weight loss after vertical burning test, and superior LOI values.
FTIR analysis of untreated and treated fabrics
FTIR spectra of untreated and PVP (PR)-P-DCDA (400 g/L) treated cotton, polyester, and CO/PET fabrics are given in Figure 7.
FTIR spectra of untreated and PVP (PR)-P-DCDA: (a) treated cotton, (b) polyester, and (c) CO/PET fabrics (cm−1: data is wavenumber, A: data is absorbance %).
In the spectrum of untreated cotton fabric, the major vibration absorption peaks of cotton (cellulose) can be found. The stretching vibration of the hydroxyl groups of cellulose was 3333 cm−1 and that of CH2 was 2896 cm−1. The characteristic peaks of CH wagging were 1424 and 1315 cm−1. The stretching vibrations of C–O were around 1027 and 1052 cm−1. The characteristic absorption peaks of P and N in the PVP (PR)-P-DCDA can easily be identified on treated cotton fabric. For instance, P was observed at around 831, 905, and 1232 cm−1 and –NH2 was observed at around 1659 and 3333 cm−1 on the treated fabrics. These bands indicated the presence of the P–N containing chemical on cotton fabrics.
With regard to Figure 7(b), the major vibration absorption peaks of polyester could be seen on the spectrum of untreated polyester fabrics. The intense band located at 1713 cm−1 corresponds to the stretching vibration of the carbonyl group of the ester bonds. At 1408, 1338, 1242, and 1094 cm−1 were identified absorption bands corresponding to the bending vibration of C–O and asymmetric stretching vibration of C–O–C. The characteristic peak of CH2 in polyester was 722 cm−1. On the spectrum of polyester fabrics treated with PVP(PR)-P-DCDA, PVP(PR)-P-DCDA characteristics IR absorptions were found at 3426, 2967, and 988,793, which correspond to the stretching vibrations of –NH2, –CH2, P–O, and –NH2, respectively. P was also observed at 902 like treated cotton fabric. These bands revealed that P and N was successfully transferred to the polyester fabric.
The spectrum of untreated CO/PET fabric includes major vibration absorption peaks of polyester and cotton (cellulose). The stretching vibration of hydroxyl groups in polyester and cotton was 3333 cm−1. The stretching vibration of CH2 was seen in 2900 cm−1. The 1713 cm−1, which is a very strong band, is the stretching vibration of C=O in polyester. The stretching vibrations of C–O were 1017 and 1052 cm−1. Another strong band at 1104 cm−1 was the stretching vibration of C–O–C. The bending vibration of CH2 in polyester was at 722 cm−1. For CO/PET blends treated with PVP(PR)-P-DCDA, PVP(PR)-P-DCDA characteristics IR absorptions were easily identified at around 3333, 1242, 905, and 831 cm−1, which correspond to the stretching vibrations of –NH2, and P–O, respectively. These bands indicated that PVP (PR)-P-DCDA was successfully applied to the CO/PET fabrics.
Differential scanning calorimeter results of treated and untreated fabrics
Figure 8 presents the first-heating and cooling DSC scans. Table 3 shows the collected data for untreated and treated polyester and CO/ PET fabrics.
First heating and cooling DSC scans for polyester and CO/PET fabrics: (a) untreated polyester fabrics; (b) treated polyester fabrics; (c) untreated CO/PET fabrics; (d) treated CO/PET fabrics. Collected data from DSC thermograms.

From the DSC scans of the polyester fabrics (Figure 8(a) and (b)), it is seen that the untreated polyester fabric melts and shows a sharp peak at 252.56℃ with 68.77 J/g heat absorption. Unlike treated polyester fabrics, melting process is slower and occurs at lower temperature (247.58℃) with lower heat absorption (56.50 J/g). Treated polyester fabric developed crystallinity upon cooling from the melt at 208.63℃ with 48.54 J/g heat desorption and became semi-crystalline largely during cooling above its glass transition. On the other hand, crystallinity process of untreated polyester is slower with crystallinity temperature of 194.7℃ and 51.51 J/g heat desorption.
With regard to CO/ PET samples, it should be noted that their DSC scans are closely similar to those of polyester samples. The only difference is the observation of a slight endothermic peak at heating cycle due to evaporation of water at ∼30–110℃. The findings indicate that the FR treatment with PVP(PR)-P-DCDA has resulted in the change in the thermal characters of polyester and CO/PET fabrics and lead to enhanced char formation.
Thermogravimetric analysis of treated and untreated fabrics
TGA is the most common method for thermal stability assessments of various polymers [53]. Phosphorous containing FRs are known to form a cross-linked network within the cellulose and PET that can inhibit the release of volatile combustible fragments and enhance char formation. They can reduce cellulose and polyester inflammability, primarily by dehydration, phosphorylation, and phosphate-ester decomposition mechanisms. In general, FR is expected to decompose before or near the decomposition temperature of the substrate to interfere with the burning process [18]. Figure 9 illustrates TG curves of untreated and treated 100% cotton fabric, 50/50% CO/PET fabrics, and 100% polyester fabrics with PVP (PR)-P-DCDA (400 g/L). The collected data are summarized in Table 4.
TG curves of untreated and treated fabrics. TGA data of untreated and treated fabrics.

With regard to cotton, it usually pyrolyses in nitrogen through two alternative pathways, which involve the decomposition of the glycosyl units to an aliphatic char at lower temperatures and the depolymerization of such units to volatile products containing levoglucosan at higher temperature [54]. In this case, Figure 8 shows that cotton is thermally degraded by only one step because of the charring of the glycosyl units. The presence of FR treatments is responsible for the strong reduction of the cellulose decomposition temperature, as revealed by the initial decomposition (
Surface morphology
The morphology of CO/PET fabrics before and after treatment is shown by SEM images in Figure 10. SEM images of untreated CO/PET fabrics demonstrate the typical micrographs of a smooth fiber surface. On the other hand, fabric surface after treatment became relatively rough, firm, and intact including many packed species on the fiber, which showed the presence of the FR materials bound to the surface of fabric.
SEM micrographs of 50/50% CO/PET fabrics: (a) untreated CO/PET fabric; (b) treated CO/PET fabric.
Conclusion
A novel reactive FR, PVP (PR)-P-DCDA, was synthesized for cotton, polyester, and CO/PET blend fabrics and the fabrics were applied with different concentrations via impregnation method. FR performance as well as thermal properties and chemical structures of treated fabrics were investigated. FTIR results verified that FR was successfully transferred onto the fabrics. The thermal stability of the treated fabrics in nitrogen, as well as their resistance to a flame application has proven to be strongly affected by PVP (PR)-P-DCDA treatment. DSC showed that treated polyester fabrics tend to melt in lower temperatures and crystalize quickly in higher temperatures than the untreated ones. TGA results indicated that decomposition temperature of the treated fabrics were reduced by treatment and PVP (PR)-P-DCDA are able to protect all three types of fabrics against their thermal degradation and favoring the formation of a stable char, which differs from the untreated ones. These results suggested that the addition of FRs onto the cotton, polyester, and CO/PET might have reduced the flammability via PVP (PR)-P-DCDA dehydration into char. With regard to the FR performance of fabrics, the treated fabrics were not ignited and did not show afterglow in the vertical flammability test. Indeed, in the case of polyester, a significant increase in its LOI (from 22.5% to 33%) has been reached even at 8–9% add-ons. In the case of cotton and CO/PET blends, PVP (PR)-P-DCDA turned out to slow down in char length and weight loss and FR properties (LOI values of +26) were obtained in higher concentrations. At larger scale, Eksoy Chemical Company commercialized PVP (PR)-P-DCDA under the name Fire-off EBR.
In conclusion, Fire-off finish applied to lightweight fabrics is suitable for daily use and finds applications in many areas including curtains, upholstery, and garment fabrics. While commercial FR products such as Pyrovatex CP (N-methylol phosphonopropionamide) can be used for cotton or its polyester blends up to 30% synthetics fiber content, Fire-off can be applied to cotton, polyester, and CO/PET fabrics in any blending ratios [54–56]. Washing durability and mechanical assessments of CO/PET fabrics treated with Fire-off will be discussed in future papers.
Footnotes
Acknowledgements
This project was carried out with EKSOY Kimya Sanayi ve Ticaret A.Ş. and Istanbul Technical University. The authors wish to thank Zorlu Mensucat for fabrics.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Ministry of Science, Industry and Technology as SAN-TEZ (No: 0080.STZ.2013-1) and BAP (No: 39775) projects.
