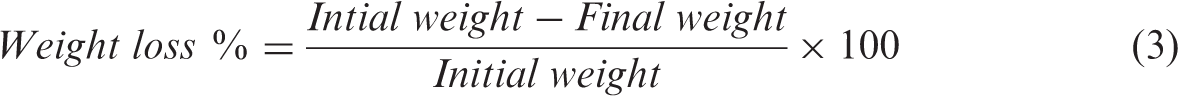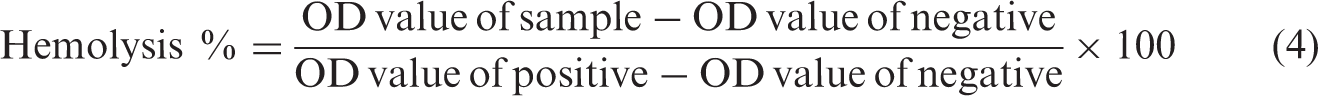Abstract
Electrospun scaffolds are being widely studied for its potential application in tissue engineering because of its nanostructure that mimics the extracellular matrices. Although it has several advantages, it lacks mechanical strength and causes structural deformation during handling of the scaffold. It is well known that the textile-based structures like woven and nonwoven fabrics have excellent structural stability. In this study, a woven and a hydroentangled nonwoven fabric were fabricated from eri silk fibroin and their characteristics were compared with electrospun scaffold. The functional groups, contact angle, thermal degradation, hemocompatibility studies showed that all the three scaffolds can be used as biomaterials. The pore and pore size distribution were better with electrospun scaffold due to smaller fiber diameter and more number of layers of fibers. The tensile behaviour was found to be better for woven and nonwoven scaffolds. The enzymatic degradation showed that the stability of woven and nonwoven scaffolds were better and electrospun scaffold degrades and disintegrates quickly. Mouse 3T3 L1 fibroblast and Human Wharton’s jelly Mesenchymal Stem Cells adhered on all the three scaffolds and a higher attachment and growth coverage were obtained on the electrospun and nonwoven scaffolds. It was found that the hydroentangled nonwoven silk scaffold exhibited similar biological characteristics as that of electrospun scaffold and also had higher mechanical strength and structural stability. Hence, it is inferred that the hydroentangled nonwoven scaffold can be considered as a suitable structure for tissue engineering applications.
Keywords
Introduction
Silk, popularly known in the textile industry for its luster and mechanical properties, is produced by silkworms. Silk filament consists of two types of self-assembled proteins: fibroin and sericin. Both these proteins contain the same 18 amino acids such as glycine, alanine and serine, but in different amounts [1]. The crystalline portion contains repetitive amino acids (-Gly-Ala-Gly-Ala-Gly-Ser-) along its sequence [2,3], resulting in β-sheet secondary structure formation and have excellent mechanical properties such as strength and toughness [4]. Eri (Samiacynthia ricini) is a type of wild silk found only in south-east Asia [5]. The eri cocoons are small, very light in color and are composed of less sericin content than that of Bombyx mori silk [6]. It has an hydrophilic to hydrophobic amino acid ratio of 0.35 [7]. In the recent years, eri silk has been studied extensively as a biomaterial for applications such as wound dressing, drug delivery, and scaffold for tissue engineering [8,9]. A number of methods, such as freeze-drying, salt leaching, gas forming and electrospinning, have been used to prepare silk scaffolds with various conformations and microstructures [10]. All these porous silk biomaterials are biocompatible and provide favorable micro-environments for both cellular infiltration and growth in in vitro and tissue regeneration in vivo [11–13].
The scaffolds are expected to provide suitable stiffness and strength for handling and should have possible fiber reinforcement for cell attachment [14]; the mechanical strength is required to withstand stress after the implantation inside the body. Most electrospun nanofibrous scaffolds lack mechanical strength and result in handling difficulty during the time of implantation. A strong correlation between scaffold stiffness and cell behavior was revealed, which showed that scaffold stiffness influences the differentiation of mesenchymal stem cells [15,16]. The unique nature of electrospun nanofibrous mat is that it has fibers of high surface to volume ratio and also the micropores which provide a better surface for the cells to adhere and grow. However, these electrospun scaffolds exhibit inferior mechanical properties, particularly for the applications requiring strength in tension. Moreover, the mat becomes brittle while sterilization and breaks during application. Hence, there is the need of experimenting other structures which would provide better strength and also meet the requirements of scaffolds used for tissue engineering applications.
The woven, knitted and nonwoven fabrics have greater strength compared to electrospun mats and can be taken into consideration to perform as scaffold for tissue engineering applications [17–19]. However, the average pore size and distribution of pore size are different for these three structures. Knitted fabric has the disadvantage that its structure is deformable and pore size is variable during handling. Close structured woven fabrics and nonwoven fabrics have definite structure and strength. Woven fabrics are produced by interlacement of threads using weaving machine. Nonwoven fabrics are produced by different methods, viz. needle punching, hydroentangling (spunlacing), melt blowing and spun bonding. Among these four nonwovens, the spunlaced nonwoven is selected for this study as it can be produced with smaller thickness with higher packing density and does not require additional polymers for bonding. Hydroentanglement is a versatile process for bonding nonwoven fabrics by using fine, closely spaced, high-velocity jets of water to rearrange and entangle loose arrays of fibers. The interaction of the water jets with fibers in the web increases the entanglement of fibers and induces displacement and rearrangement of fibers segments in the web [20]. Hydroentangled fabrics rely primarily on fiber-to-fiber friction to achieve physical integrity and are characterized by relatively high strength, softness, drape, and conformability. In this study, woven fabric, spunlaced nonwoven fabric, electrospun mat were produced from eri silk fibroin and were compared to properties essential for tissue engineering application.
Materials and methods
Material
Erisilk cocoons and yarns (Guwahati, India) were soaked in the solution of sodium carbonate maintained at 80℃ for 40–60 min to remove sericin. The degummed silk cocoons and yarns (fibroin) were kept at 40℃ in the oven for 24 h and then preserved in airtight packs.
Preparation of scaffolds
The scaffolds were prepared in three different forms, viz. woven, spunlaced nonwoven, and electrospun. The construction parameters of the scaffolds have been selected to obtain a thickness of 250 µm for all the three structures, viz. woven, nonwoven, and electrospun mat. The method of preparation is given as follows.
Nonwoven scaffold
The nonwoven scaffold was prepared using spun-lacing technique also called as hydroentanglement technique. To produce the sample, the degummed silk cocoons were cut into pieces and then opened using Shirley Trash Analyzer, the machine converts the pieces of cocoons into the fibers. A degummed eri silk fibrous web was produced using miniature carding machine. The machine has wired rollers which opens and individualizes the fibers and converts them into a sheet. The web obtained from miniature carding machine was converted into nonwoven fabric using spunlacing machine (Jetlace 1000). For obtaining nonwoven of 250 µm thickness, the card web of different linear densities was produced from miniature carding machine by keeping all the other machine parameters constant. They were converted into the nonwoven fabric. The web (90 GSM) was optimized to get nonwoven of thickness 250 ± 50 µm. The nonwoven scaffolds were then dried and kept in desiccator.
Woven fabric scaffold
Woven fabric scaffolds were developed from degummed eri silk yarn as warp and weft using a weaving machine. In order to get the fabric of 250 µm thickness, the sum of diameter of warp and weft should be kept as 125 µm. Since the same yarn was used as warp and weft, the diameter of each yarn should be of 125 µm. It was assumed that the yarn was circular in cross-section in the fabric and its packing density is 0.91 g/cm3. The yarn count predicted based on equation (1) was 53 Ne.
Since 53 Ne was an odd count and was not available commercially, 60 Ne yarn was used to make fabric. The ends per cm and picks per cm were kept at 50, and plain weave structure was used. Since the plain structure has more interlacements and hence better structural stability compared to other fabric structures, it was selected to make woven fabric. The thickness of fabric was found to be 230 ± 25 µm (80 GSM). The fabric was washed with the soap solution for 2 h to remove the starch and other substances used during weaving preparatory process and then the fabric was rinsed with distilled water. It was sterilized in 70% ethanol and pre-treated with ultraviolet rays before cell culture.
Electrospun mat scaffold
Eri silk fibroin was dissolved in trifluoroacetic acid (13% w/v). The solution was electrospun at a voltage of 20 kV. The flow rate was kept at 1 mL/h and a distance of 20 cm was maintained between syringe tip and the rotating collector in the electrospinning machine. Different volumes of solution, 1–9 mL, were taken and electrospun. A 6 mL of silk solution spun over the rotating drum at 1000 r/min gave a mat thickness of 250 ± 30 µm (20 GSM).
Characterization of scaffolds
Physical and chemical characterization
The woven, nonwoven, and electrospun scaffolds were subjected to the following physical and chemical characterization. The morphology of the three scaffolds was observed using a scanning electron microscope (SEM). The samples were vacuum dried and sputter-coated with gold before testing. Images obtained were processed using ImageJ software (NIH, USA). For analyzing the functional groups, the Fourier Transform Infrared spectra (Agilent, Cary 630) were obtained for the scaffolds with an attenuated total reflectance accessory. Each spectrum was acquired in reflectance mode on a Ge crystal by the accumulation of 16 scans with a resolution of 4 cm−1 and a spectral range of 4000 to 600 cm−1.
The thermal property of the scaffolds was analyzed using Thermo-gravimetric analyzer (TA, Q50). The measurements were carried out under nitrogen atmosphere at 40 to 60 mL/min; the samples were heated up to 700℃ at a heating rate of 10℃ min−1. The wettability of scaffolds was studied by sessile droplet method using dynamic contact angle tester. The water contact angle was measured using Goniometer (DSA-10, Kruss188, Germany) at room temperature. Millipore water was dropped on the scaffold surface, and the contact angle was measured from the images taken using digital microscope. Three samples were analyzed in triplicate.
The tensile strength of scaffolds were measured using Universal tensile tester (Instron, 3369). The nanofiber samples were cut into 40 mm length and 10 mm width. Adhesive tapes were pasted on the corners of each sample to avoid the jaw slippage. The test was conducted at 20 mm gauge length and at 1 mm/min test speed [21]. The tensile property of the woven and non-woven scaffolds was tested as per the standard ASTM D5034.
The porosity of the scaffolds was measured using Capillary Porometer (Porous Materials Inc., USA). The samples were measured for their thicknesses and were cut into a square of 30 mm width. The scaffolds were initially wetted with Galwick solution (15.9 dynes/cm) and were placed in a vacuum chamber. The samples were subjected to nitrogen gas flow to obtain wet and dry curves. The porosity characteristics were determined from the wet and dry curves by automated CFP software.
The stimulated body fluid (SBF) uptake capacity of scaffolds was measured by immersing 2 × 2 cm pieces of scaffolds in 50 mL of stimulated body fluid [22] kept at 37℃. The dry weight of scaffold was measured (Wdry). They were immersed in SBF and the wet weight of scaffolds was measured at different time intervals (Wwet). Before weighing, the wet samples were kept on the filter paper to remove excess amount of solution present on the surface of the scaffolds. The SBF uptake capacity of the scaffolds was calculated using the following formula.
To analyze enzymatic degradation characteristic, the silk scaffolds were cut into a size of 4 × 1 cm and each of the scaffold samples was individually weighed. The enzyme solution was prepared by dissolving protease XIV (P8811, 5.9 U/mg, Sigma Aldrich) in 0.05 M Phosphate buffer solution (pH 7.4). Eri silk scaffolds were immersed in the enzyme solution and kept at 37℃ for 15 days. For every 24 h, the enzyme solution was replaced fresh. Samples were collected at specified intervals of time, rinsed with distilled water, dried, and weighed. The percentage weight loss of silk scaffolds after different periods of degradation was calculated using equation (2).
Biological characterization
For conducting hemolysis study, the scaffolds were cut into small pieces of 1 × 1 cm and aged for 24 h in physiological saline. Anti-coagulated human adult blood (0.2 mL) was diluted with 4 mL saline and then added to each scaffold samples taken in tubes and they were maintained at 37℃ for 60 min. The positive and negative controls were produced by adding 0.2 mL of anti-coagulated human adult blood to 4 mL of distilled water and saline water, respectively. Then, all the samples were centrifuged at 3000 r/min for 10 min. The supernatant liquid was taken for the optical density (OD) examination at 545 nm using UV spectrophotometer. The percent hemolysis was calculated using the below equation
The procedure used for cell viability study is given as follows: The scaffolds were cut into 5 × 5 mm pieces and were sterilized with ethanol and UV irradiation. After sterilization, the samples (n = 3) were placed in a 24-well plate. Mouse 3T3 L1 fibroblast cells and Human Wharton’s Jelly MSCs were used for testing their viability on the scaffolds. Wharton’s Jelly mesenchymal stem cells (hMSCs) were procured from Himedia Laboratories, India. A 200 µL of fresh stem cell expansion medium (Himedia) was added into the well plates and the scaffolds were conditioned for 1 h. The medium was then removed and the hMSCs (3 × 103/ cm2) were seeded and incubated in CO2 chamber. Mouse 3T3 L1 fibroblast was obtained from NCCS, Pune, India. The same protocol that was used for hMSCs was followed for 3T3 L1 (2 × 104/cm2) fibroblasts, but DMEM was used as the medium. After 48 h, the medium was carefully removed and then the scaffolds were washed with phosphate-buffered solution (PBS).
The viability of the mouse 3T3 L1 fibroblast and hMSCs on the scaffolds were quantified through assay by labeling the cytoplasm of the living cells with green fluorescent calcein AM. The fluorescence intensity is directly proportional to number of viable cells present. A 200 µL of calcein AM was added and absorbance was measured at 480 nm using microplate reader. Cells without scaffold were taken as control, and survival rate after 48 h was set as 100%. The morphology of adhered cells on the scaffolds was analyzed using SEM. The scaffolds were washed with PBS and fixed with 5% of glutaraldehyde. 0.1% osmium tetroxide was added in dark and incubated for 30 min. Then, the scaffolds were washed with PBS and treated with series of ethanol solution 60%, 70%, 80%, 90%, and 100%. The samples were allowed to dry overnight and sputter coated with palladium prior to imaging by SEM. The morphology of adhered cells on the scaffolds was analyzed using fluorescent microscope. Since the scaffolds were made of peptides and amino acids, it was difficult to dye the cell and scaffolds separately. Hence, lipophilic carbocyanines were used as stain to avoid the background false indicators from the scaffolds. Stock DIL (1, 1′-Dioctadecyl-3, 3, 3′, 3′-tetramethylindocarbocyanine perchlorate) stain was produced according to the manufacturer’s protocol. The dye was added to the scaffolds seeded with hMSCs (3 × 103 cells/well) and incubated for 30 min at 37℃ and then washed and analyzed under a fluorescence microscope at 549 nm.
Statistical analysis
The data in the all characterizations are represented as mean ± standard deviation (SD) with sample size as 3. ANOVA was used for statistical analysis. Significant difference was *p<0.05.
Results and discussion
Morphology
The SEM images and fiber diameter histogram of woven, nonwoven, and electrospun scaffolds are shown in Figure 1. The SEM images show that the majority of the fibers in the woven scaffold have the diameter in the range of 14 to 22 µm, and in the nonwoven scaffold, it varies from 12 to 20 µm, whereas in the electrospun mat, the fibers have the diameter ranges from 0.6 to 1.2 µm. The woven scaffold shows twisted, flat, and relatively smooth-surfaced microfibers, and the nonwoven and nanofiber scaffolds show a highly randomized fibrous network. The random microfibrous alignment is most important for the increase of surface to volume ratio of the scaffolds which promotes cell adhesion and cell growth [23]. The average fiber diameter of the three scaffolds is listed in Table 1.
SEM images and fiber diameter histogram of eri silk fibroin (a) woven, (b) nonwoven and (c) electrospun scaffolds. Mean fiber diameter and standard deviation of woven, nonwoven, and nanofiber scaffolds.

The results indicate that electrospun scaffolds have more number of fibers and layers per unit volume followed by nonwoven and then woven scaffolds.
Functional group analysis
Figure 2 shows the FTIR spectra of raw silk cocoon, woven, nonwoven, and electrospun scaffolds. The spectra of cocoon and three forms of scaffolds show the absorption bands at 1620 cm−1, 1510 cm−1, 1226 cm−1, which respectively, indicate the presence of amide I, amide II, and amide III of protein. Amide I absorption primarily represents C=O stretching vibration of the amide group, amide II absorption contains both N–H bending and C–N stretching vibrations, and amide III peak is arised from C–N stretching vibration coupled with N–H in plane bending vibration. The spectral range 1698–1623 cm−1 show the presence of β sheets in the scaffolds [24]. Thus, the FTIR pattern shows that there is no structural difference between three forms of scaffolds. There is no structural change due to dissolving of fibroin in trifluoroacetic acid and electrospinning of scaffold.
FTIR spectra of eri silk cocoon, woven scaffold, nonwoven scaffold, and electrospun scaffold.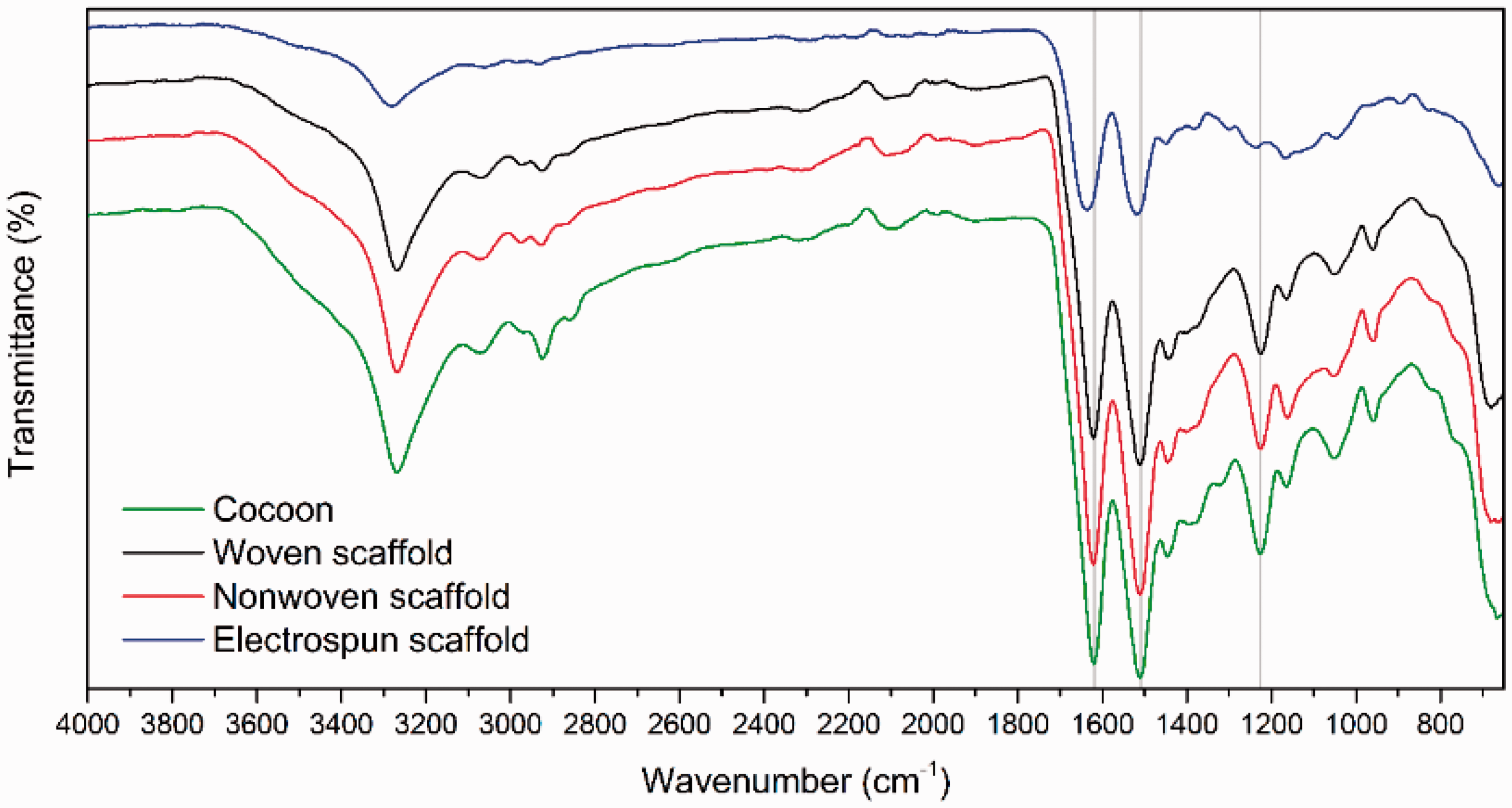
Thermal behavior
Figure 3 shows the thermograms of three forms of eri silk scaffolds. All the three curves show the initial weight loss during 60–100℃ due to removal of moisture.
Thermograms of woven, nonwoven, and electrospun nanofibrous scaffold.
The eri silk woven and nonwoven scaffold start to degrade at 275℃ and decomposes at 390℃. In the case of electrospun eri silk scaffold, the degradation starts early at 200℃ and decomposes at 390℃. The decomposition is due to degradation of peptide bonds in the proteins. The differential curves of all the three forms of scaffolds show a same decomposition temperature of 390℃. It is found that the all the three scaffolds have the same decomposition temperature and it is higher than the temperature at which sterilization is carried out [25]. Hence, all the three forms of scaffolds can be sterilized safely and used for tissue engineering applications.
Pore size distribution
Figure 4 shows the pore size distribution of woven, nonwoven, and electrospun scaffolds. The size of the pore and its distribution in the scaffolds depends on the fiber diameter and scaffold’s areal density [26]. It can be seen from Figure 4 that majority of pores in both woven and nonwoven scaffolds have pore size diameter in range of 5 to 50 µm. In the case of woven scaffold, the individual pores are bigger between the interlacement points of warp and weft threads, and pores are straight through from top to bottom and the number of pores are less than that of nonwoven. Since the present method of measurement involves penetration of pressurized fluid through the scaffold, the nonwoven scaffold also shows a similar pore size distribution as that of woven scaffold. However, it can be noticed that the pore size as seen through microscope is very less as the fibers in one layer overlay on the fibers in the next layer, and hence no through pore is available in nonwoven scaffold, but have the tortuosity effect. The nonwoven scaffolds have uniform pore size distribution due to the random alignment of fibers with superimposition of fibers in one layer over the fibers another layer. The pore size distribution is found to be 0.7 to 1 µm in electrospun scaffold, which is the least compared to other two scaffolds. Majority of the pores have the size of 0.7 µm. The diameter of the fiber is less in electrospinning and number of layers per unit thickness is higher than that of nonwoven scaffold. The randomized arrangement and superimposing of layers of fibers are same in nonwoven, and electrospun scaffold; however, the diameter of fibers is different.
Pore size distribution of scaffolds.
Tensile behavior
The stress–strain graphs of woven, nonwoven, and electrospun scaffolds are given in Figure 5. The fiber alignment and overall geometrical structure have an influence on the mechanical properties of the scaffolds [27].
Stress–strain curves of woven, nonwoven, and electrospun scaffolds.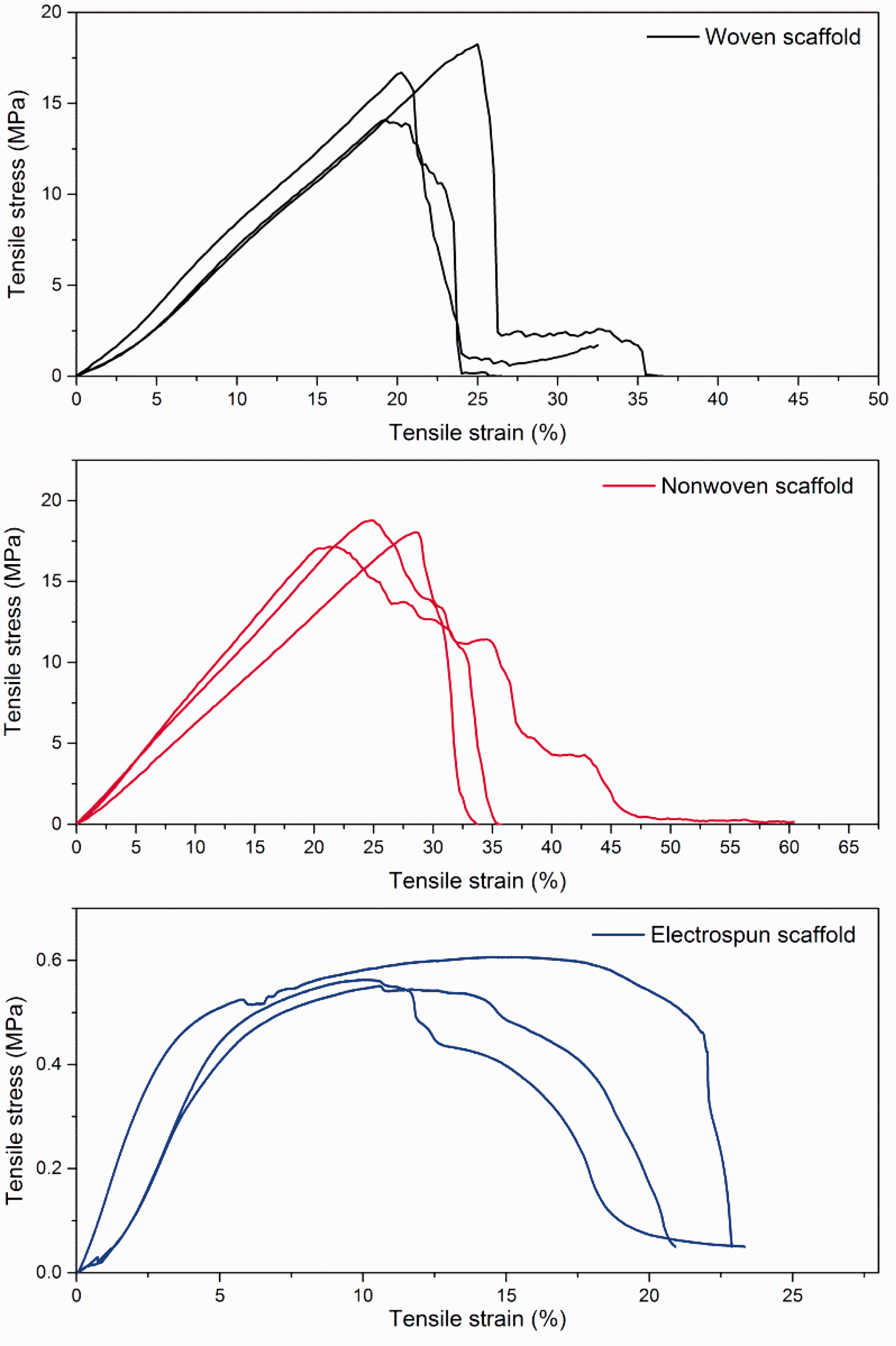
The tensile stress, strain, and modulus of woven scaffold are found to be 16.36 MPa, 0.215 mm/mm, and 92.4 MPa, respectively. In the case of nonwoven scaffold, they are, respectively, 17.99 MPa, 0.249 mm/mm, and 81.88 MPa. For electrospun scaffold, they are 0.578 MPa, 0.132 mm/mm, and 14.314 MPa. No significant difference in tensile stress, strain, and Young’s modulus observed between woven and nonwoven scaffolds. Both the scaffolds have superior tensile properties than that of electrospun. It indicates that the woven and nonwoven scaffolds can withstand the stress applied to them during tissue engineering applications. The higher tensile strength of scaffold would make it withstand multiple sutures during in vivo application. The electrospun scaffolds would not provide adequate strength and may tear during suturing [28].
Protease degradation
The degradation behavior of silk fibroin has a close relationship with the molecular weight and structural characteristics like fiber diameter, their interlacement, and packing. The protease XIV enzyme quickly degrades the surface of the silk scaffold particularly the amorphous regions of silk I fibroin, whereas the silk II fibroin which provides the tensile strength have a controlled degradation over the period [29]. The degradation of scaffold decreases its structural stability and hence its strength. Figure 6 shows the degradation of three forms of scaffolds over a period of 15 days by protease XIV. It was observed that the enzymes have etched the surface of the scaffold and degradation was due to surface erosion, which is similar to the findings of Muthumanickkam et al. [30]. Due to the degradation of surface, the structure of the scaffold disintegrates. The woven scaffold shows 39% degradation in 15 days, whereas the nonwoven silk fibroin scaffold shows a degradation of 30%. It was also observed that there was no structural disintegration during this period. The electrospun scaffold showed a higher degradation rate of about 85% in 15 days. The structure also totally disintegrated during this period. The woven and nonwoven scaffolds have slower degradation rate and possess stronger structural stability compared to that of electrospun scaffolds and can be ideal material for implantations where longer service life is required.
Comparison of degradation (%) effects of protease XIV degradation on scaffolds at various time intervals at 37℃, n = 3.
Contact angle
Since all scaffolds were made from silk fibroin, which is hydrophilic in nature, the water was absorbed immediately by the scaffolds. The contact angle was not formed during this process. Hence, all the three scaffolds exhibit hydrophilicity and they can be considered as suitable material for cell adhesion [31].
SBF absorption
The SBF uptake of the three scaffolds is shown in Figure 7. The uptake is maximum for nonwoven scaffold (844%) followed by electrospun scaffold (496%) and then woven scaffold (235%). In all the three cases, uptake reaches the saturation level at the first day itself, since the structure of the scaffolds is found to be stable in the wet condition and also have good absorption, the scaffolds can be considered for tissue engineering.
SBF uptake percentage of woven, nonwoven, and electrospun scaffolds at different time intervals.
Hemolysis assay
Figure 8 shows the percentage hemolysis occurred in three scaffolds. Hemolysis rate (%) is found to be 1.6, 1.2, and 2.6, respectively, for woven, nonwoven, and electrospun scaffolds. Hence, all the three scaffolds can be considered as biomaterial as they have hemolysis (%) less than 5.
Exposure of scaffolds on whole blood cells for 60 min. Image represents the data of mean and standard deviation of lysed blood cells (n = 3).
Cell viability
Calcein AM assay
Comparison of viability of living cells after 48 h of treatment is shown in Figure 9. The fluorescence intensity confirms the proliferation of 3T3 L1 and hMSCs on all the scaffolds. Cells on electrospun scaffolds show a prominent steady slow population over a period of time. The scaffolds support the proliferation of both the cell lines. Further, observation through the microscope indicated that there was no effect or change in morphology of cells.
Fluorescence intensity of (a) 3T3 L1 fibroblast cells and (b) hMSCs on scaffolds after 48 h, in both cell lines n = 10.
SEM imaging
The SEM images in Figure 10 show the distribution of 3T3 L1 and MSCs over the surface of the scaffolds. It can be seen from the image that the cells spread over the surface of the fiber bundle of the twisted yarn and the cells could not hook up between two individual yarns in the structure, leading to the low adhesion of cells with woven scaffolds. In the case of nonwoven scaffold, the cells adhere to the fibers on the surface of scaffold due to the reason that the inter fiber gap is very less and is occupied by fibers in another layer. It can be seen that the microenvironment of nanofiber scaffolds with negligible inter-fiber space keeps higher adhesion of cells than other two scaffolds. While considering the structural stability and cell adhesion distribution, the nonwoven scaffolds may be the better substitute for electrospun scaffolds.
SEM images of mouse 3T3 L1 fibroblasts on different scaffolds (a) woven, (b) nonwoven, (c) electrospun. SEM micrographs of hMSCs on (d) woven, (e) nonwoven, and (f) electrospun scaffolds.
Fluorescence imaging
The cells spread over the surface of the scaffolds start to proliferate and formed the spindle morphology after 24 h of seeding. It can be observed in Figure 11 that the electrospun exhibits a higher confluence than the woven and nonwoven scaffolds. However, the proliferation and cell distribution on nonwoven scaffold are found to be higher than that of woven scaffold and are close to the nanofiber scaffold. All the scaffolds support the morphological formation of the hMSCs in in vitro condition.
Morphology of hMSCs on different scaffolds stained with DIL stain after 48 hours observed through fluorescence microscopy at 549 nm (magnification 20× corresponds to all images) (a) woven, (b) nonwoven and (c) electrospun silk scaffold.
Conclusion
The scaffolds for tissue engineering applications were prepared from woven, nonwoven fabrics, and electrospun mats. They were studied for physical and biological characters relevant for tissue engineering application. The results showed that the mean diameter of fibers in woven and non woven was 16 µm and in electrospun it was 907 nm. The Thermogram values proved that all the three scaffolds were thermally stable up to 200℃ which is higher than the temperature at which sterilization is carried out. Woven and nonwoven scaffolds showed pore size diameter distribution of 5 to 55 µm which was much higher than that of electrospun scaffold having pore of 0.7 to 1 µm. Nonwoven scaffolds showed an increased resistance to degradation by protease XIV enzyme, while the electrospun scaffold in structurally degrades in 15 days. The super hydrophilicity of the scaffolds was confirmed by water contact angle study. All the three scaffolds were found to be hemocompatible. Cell adherence was found to be better with nonwoven and electrospun scaffolds. It can be inferred from the results that eri silk woven, nonwoven, and nanofiber scaffolds can be used as scaffold for tissue engineering purposes. Woven and nonwoven scaffolds have good mechanical strength and structural stability compared to the electrospun scaffold and also scaling up is easier with these scaffolds. However, the cell adherence was better with nonwoven and electrospun scaffolds. On comparison, nonwoven scaffold exhibited the better mechanical ability, structural integrity, and effectively supported cell the growth and proliferation. It is suggested that nonwoven scaffold may be a better candidate for application in 3D tissue regeneration compared to the electrospun scaffolds.
Footnotes
Acknowledgement
The authors would like to thank Vasanthan Ravichandran, Indian Institute of Technology Madras, India for his help in cell culture assays and fluorescence techniques.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are thankful to CEMA, MHRD, India and University Grant Commission’s Basic Research Fellowship (UGC-BSR), India.