Abstract
Peptides are one of the most important biological molecules which can be used in drug delivery applications. Peptides are composed of amino acids and are connected together by a peptide bond. These biomaterials have drawn lots of attention in recent years because of their biocompatibility, antibacterial effect, and medical and clinical applications.
In this paper, phenylalanine amino acid is used to synthesize peptide nanotubes. The method used for synthesis of peptide nanotubes was self-assembly which is efficient and spontaneous. According to the experimental evaluations of peptide nanotubes, the best concentration of amino acid solution for peptide nanotubes synthesis was 0.005 mg/ml. In this concentration, the tubes show their hollow, long structure. The average diameter of the tubes is 120 nanometer and they have an aspect ratio of 45. To investigate the drug releasing ability of nanotubes, ascorbic acid solution was loaded on them. The solution was applied on cotton fabric to examine the possibility of producing cosmetotextiles. The ability of cotton fabrics to release ascorbic acid in different temperatures and mediums were examined using different conditions. UV-Visible spectroscopy was also used to measure the amount of ascorbic acid release from peptide nanotubes. The antibacterial test was also carried out to determine the antibacterial activity of amino acid molecules on cotton fabric.
Introduction
Nanocarriers are one of the most dynamically developing branches of nanotechnology which can be used for many different applications. They are also widely used in medical science for drug delivery. One of the most important advantages of using nanocarriers is that they can react in different ways which depends on the medium they are used in. The drug delivery process of nanocarriers is under control and gradual which has been very attractive to scientists recently. Several nanocarriers can be used for medical applications such as micelles, liposomes, dendrimers, nanoparticles, and nanotubes. They can be produced in different sizes, properties, and applications [1–3].
Peptides are one of the important biological molecules that are able to form nanoscale structures by connecting amino acid groups. Several peptidic structures such as dipeptides, tripeptides, and polypeptides can be found in nature [4]. In the 1990s, it was found that amino acid groups can form tubular peptidic nanostructures which are called peptide nanotubes. One of the first investigation groups in the following years reported of design, synthesis, and evaluation of peptide nanotubes [5].
Amino acid molecules consist of two segments, which are hydroxyl and carboxyl groups. Due to their interesting structure, peptide linear molecules can form several nanoscale structures that are similar to active surfaces. They are also able to form self-assemble structures that can be hydrophilic or lipophilic depending on the types of amino acid they are made of [6–8]. As an example, Reches and Gazit suggested a new designed peptide structure that has at least one nanoscale dimension. This structure consists of several aromatic amino acids [9]. In another study, Yemini et al. could design metal electrodes that are covered with peptide nanostructures. These nanostructures are fabricated by self-assembling on the electrode surface and can enhance the conductivity of the electrodes in electrochemical reactions [10].
Peptide nanotubes are usually synthesized from peptide monomers by self-assembly method. In lots of researches, it has been reported that dipeptides and cyclic peptide can be used for the synthesis of peptide nanotubes (PNTs) [11,12]. As a cyclic peptide monomer, phenyl alanine can be used for self-assembling process of peptide nanotubes, and one of the appropriate solvents for dissolving phenyl alanine is isopropanol. This is an eco-friendly, repeatable, and rapid method which is much cheaper in comparison to other methods. There are several factors that affect the formation process of PNTs such as monomer, solvent and concentration and pH of the solution. Different properties including length, diameter, porosity, and strength of PNTs can change according to the conditions of the medium.
PNTs are one of the highly organized nanostructures; covalent and hydrogen bonds that are formed in self-assembly process can cause hydrophilicity or lipophilicity of the nanostructures [13,14]. Due to the vast range of peptide monomers and their sequence in the synthesized PNTs, these nanostructures can be used as molecular units for fabrication of nanodevices. These nanodevices have several medical applications such as using them as biosensors, drug delivery, and anti-bacterial surfaces.
Several researches have been carried on about the biocompatibility of peptide nanostructures. As an example, Ghanaati et al. investigated the biocompatibility of peptide nanofibers. They concluded that peptide nanofibers play an important role in healing wound and regenerating tissues that are damaged in human body [15].
In spite of the great potential of PNTs, there are also some challenges. One of these challenges is the stability of PNTs in a biological system [16].
For investigation of the stability of PNTs, Reches et al. used PNTs as a mold for fabrication of Ag nanowires. They reported that PNTs have an acceptable stability in high temperatures. These researches show that addition of thiol group to peptide structures can change them from tubular shape to sphere shape [9]. In terms of stability of PNTs in solutions, peptide nanostructures can remain in the temperature of 90℃ in the solution without being damaged [16,17].
On the other hands, one of the advantages of PNTs is their antibacterial ability. Peptides can combat the bacteria and microbes and attack their cell walls. It is suggested that the antibacterial properties of PNTs can destroy the bacteria, mainly because their distinctive amino sequences can insert into and subsequently disintegrate bacterial cell surfaces [18].
Having a net positive charge, peptides can react to the bacteria, archaea, and eukaryotes (including plants, animals, fungi, and protists) with their antimicrobial activity. Antibacterial properties of peptides are associated with two interrelated features of them: their net charge, which for the great majority of peptides is positive, and their propensity to be amphipathic, meaning that they can fold into structures with both a hydrophobic and hydrophilic surfaces. These features together facilitate the interaction of peptides with the negatively charged bacterial envelope on the surfaces of Gram-negative and Gram-positive bacteria and also with the negatively charged phospholipids of the bacterial membrane. It is assumed that after the initial electrostatic interaction of peptides with the bacterial surface, the amphipathic nature of peptides allows them to insert into the bacterial cell membrane, and by forming a pore, peptides can disrupt membrane integrity, leading to osmotic lysis of the bacterial cell [19,20].
In terms of the ability of self-assembly synthesis, peptide can be a good choice to design new structures and modify them with different functional groups. Self-assembly is a spontaneous and reversible phenomenon in which the components of a system assemble through a chemical or physical interaction to synthesize a larger unit. While the components are in the appropriate conditions, ordered structures will form spontaneously. It is proved that the dipeptide is a form of phenylalanine, which under appropriate assembling conditions can form distinct nanotubes and nanogels. Therefore, in this study phenylalanine amino acid was used to investigate the ability of PNTs on the finishing of cotton fabric.
The objective of this study was to develop a nano-sized carrier, which can be loaded on cotton fabric for the fabrication of a new cosmetotextile. For this purpose, ascorbic acid was used for being loaded in PNTs, simultaneous to the self-assembling process.
Ascorbic acid is a natural organic compound with antioxidant properties, which dissolves in water and is so effective in anti-aging creams and cosmetics.
Materials and methods
Phenylalanine and ascorbic acid were procured from Sigma-Aldrich and isopropanol was procured from Merck. Chitosan (Mw∼100,000–300,000 Da, DD = 85%) was also procured from Sigma-Aldrich. Cotton fabric (plain weave, 175 g/m2, Yazbaf, Iran) was also used as the substrate for applying peptide nanotubes on it. The pretreatment process was also done before any further treatment on cotton fabric.
Experiments were carried out in three steps:
Synthesis of peptide nanotubes and finding the optimum condition for self-assembly process Preparation of ascorbic acid-loaded nanotubes Preparation of PNTs-applied cotton samples
Assembly of phenylalanine nanotubes
For the beginning, 1 mg of phenylalanine was dissolved in 100 µl of isopropanol. The self-assembly process was started by the addition of 2 ml of distilled water to the solution. The final solution would be at the concentration of 0.5 mg/ml. An incubation time of 30 min should be considered for allowing peptide nanotube structures to form.
Naming of different solutions.

Loading of ascorbic acid in PNTs
The process of preparing ascorbic acid-loaded PNTs was done by forming PNTs in ascorbic acid solution. By this method, PNTs self-assembly was carried out in the presence of ascorbic acid in the solution. Phenylalanine of 1 mg was dissolved in 100 µl of isopropanol and a solution with the concentration of 0.25 mg/ml was also added to the stock solution. The PNTs and ascorbic acid solution was stirred and incubated for 2 h at the temperature of 37℃. For better loading of ascorbic acid in PNTs, the solutions were exposed by ultrasound radiations (400 W, pulsed operation 0.5 s) for 10 min via Ultrasonic Homogenizer (FAPAN 400R, Iran).
After 24 h, the residue of the ascorbic acid which is suspended in the solution was separated by centrifuging at 4000 r/min for 10 min.
Preparation of PNTs applied cotton samples
The pretreated cotton samples were cut in given weights. The applying process of PNTs was done in two ways that includes padding and batch. In the padding process, the pick-up was set at 70% and the maximum temperature was set at 60℃. In batch process, the samples were incubated in the solution for 1 h and dried in room temperature.
Samples treated with chitosan were used as control samples to investigate the antibacterial effect of peptide structures and compare it with chitosan as an antibacterial agent. In this regard, firstly, the solvent for chitosan solution was prepared by making 1% v/v of acetic acid solution. Chitosan of 1 g was dissolved in the prepared solvent and the final solution would be at the concentration of 1% w/v. The solution was heated in the stirrer and incubated for an hour for better dissolving of chitosan.
Ascorbic acid and chitosan release from samples were examined using UV-Visible (UV-Vis) spectroscopy.
Characterization
FTIR measurements
Fourier transform infrared (FTIR, Thermo Nicolet, model: NEXUS 670, USA) was utilized to characterize the chemical structure of cotton fabric after the application of PNTs and ascorbic acid on it.
Scanning electron microscopy (SEM) microscopy
The morphology of the PNTs was determined by SEM (XL30, Philips). The samples which were 1 × 1 cm were also coated with gold vapor for SEM microscopy.
Transmission electron microscopy (TEM) microscopy
The morphology of peptide nanotubes was also evaluated by TEM microscopy. TEM of the dipeptide nanotubes was carried out using uranyl acetate negative staining method. Ascorbic acid-loaded PNTs were adsorbed on 400 mesh copper grids with carbon-coated formvar and stained with 2% uranyl acetate. Excess fluid was removed from the grid surface with a filter paper and the grid was dried at room temperature for 5–10 min. TEM pictures of nanotubes were taken using a transmission electron microscope (EM10C, Germany) operating at 80 kV.
Liquid chromatography-mass spectrometry (LC-Mass) analysis
In this study, investigation of dipeptide structures was carried out by LC-Mass analysis. It can analyze the molecular weight of PNTs structure precisely. The device used in this study was 6410 QQQ, Agilent, and the USA
Anti-bacterial activity test
The anti-bacterial test was obtained according to the AATCC test method 100-1999 as a modified colony counting method. In this test, microorganisms are grown in a liquid culture and the concentration of the test microorganism is standardized. Control fabric and the other swatches are inoculated with microorganisms. Inoculated control and test fabrics are allowed to incubate, undisturbed in sealed jars for 24 h, and after incubation, microbial concentrations are determined.

Air permeability measurements
Air permeability of the samples is the amount of air (cm3) that passes the fabric within a second with the pressure of 1 cm of water. The experiment was done using a BS5636 device. The air transition of the fabric is mentioned as volume per unit area of the fabric. The experiments were carried out by the ASTM D737 standard.
UV-Vis spectroscopy
For investigation of ascorbic acid release of PNTs, UV-Vis spectroscopy was carried on using Lambda950, Perkin-Elmer, USA.
Results and discussion
This study was carried on to obtain a useful method for fabrication of PNTs and using them in drug delivery systems. As shown in Figure 1(a), it is assumed that the most effective bonds that can help in the loading of ascorbic acid to the PNTs are hydrogen bonds. Using cross-linking agents such as carboxylic acids can make effective attachments of PNTs and cotton fabric. It is suggested that a cross-linking agent like citric acid can attach the hydroxyl groups of PNTs to the hydroxyl groups of cotton fabric during self-assembly process and also detach when it is needed.
Hydrogen bonding between phenylalanine and ascorbic acid molecules (a) and schematic of self-assembly process (b).
The problem of stability of PNTs were solved by not raising the temperature during the self-assembly process and there was not any sharp fluctuations in pH of the solvents.
FTIR measurements
The results of FTIR spectral of samples are shown in Figure 2. Typical absorption bands of phenylalanine and ascorbic acid can be seen in the results. The absorption band at 3200–3400 cm−1 in sample D attributes to four groups of hydroxyl in ascorbic acid and hydroxyl groups in phenylalanine and cotton fabric. It can also indicate the presence of hydroxyl groups of the cross-linking agent on cotton fabric. The higher peaks in sample D can show the increase of these groups on the cotton fabric. Having different absorption bands at 2400–3200 cm−1 in sample D proves the presence of phenylalanine on cotton fabric due to its amine groups.
FTIR of (a) untreated cotton, (b) synthetized peptide nanotube (PNT), (c) ascorbic acid, and (d) cotton fabric treated with PNT100 and ascorbic acid via padding method.
Electron microscopic analysis of nanotubes
For characterization of the morphology of the PNTs, microscopic studies have been carried on and the results of them are shown in Figure 3. Figure 3(a) and (b) shows scanning electron microscopy image of PNT and PNT 50 samples, respectively. It is obvious that the structures that are formed in self-assembly process in these concentrations are not in tubular forms. But in Figure 3(c) and (d), the SEM image and TEM micrograph of PNT100, it is obvious that distinct and ordered tubular assemblies are formed having a mean diameter of 90–120 nm, a length of 5000–5400 nm, and an average aspect ratio of 45.
SEM image of (a) PNT, (b) PNT50, and (c) PNT100, and (d) TEM image of PNT100.
LC-Mass analysis
For investigation of components that form PNTs, LC-Mass test has been carried on. Mass spectroscopy analysis has been widely used recently for sensitive and quantitative detection of peptides and proteins [21]. It is expected that dipeptide structures participate in the self-assembly process and form tubular PNTs. In this reaction, the carboxyl group of one amino acid molecule reacts with the amino group of the other molecule, releasing a molecule of water. The results of this test are shown in Figure 4, and the peak in 320.9 m/z proves the presence of dipeptide structures in the solution.
LC-Mass results of PNT100 solution.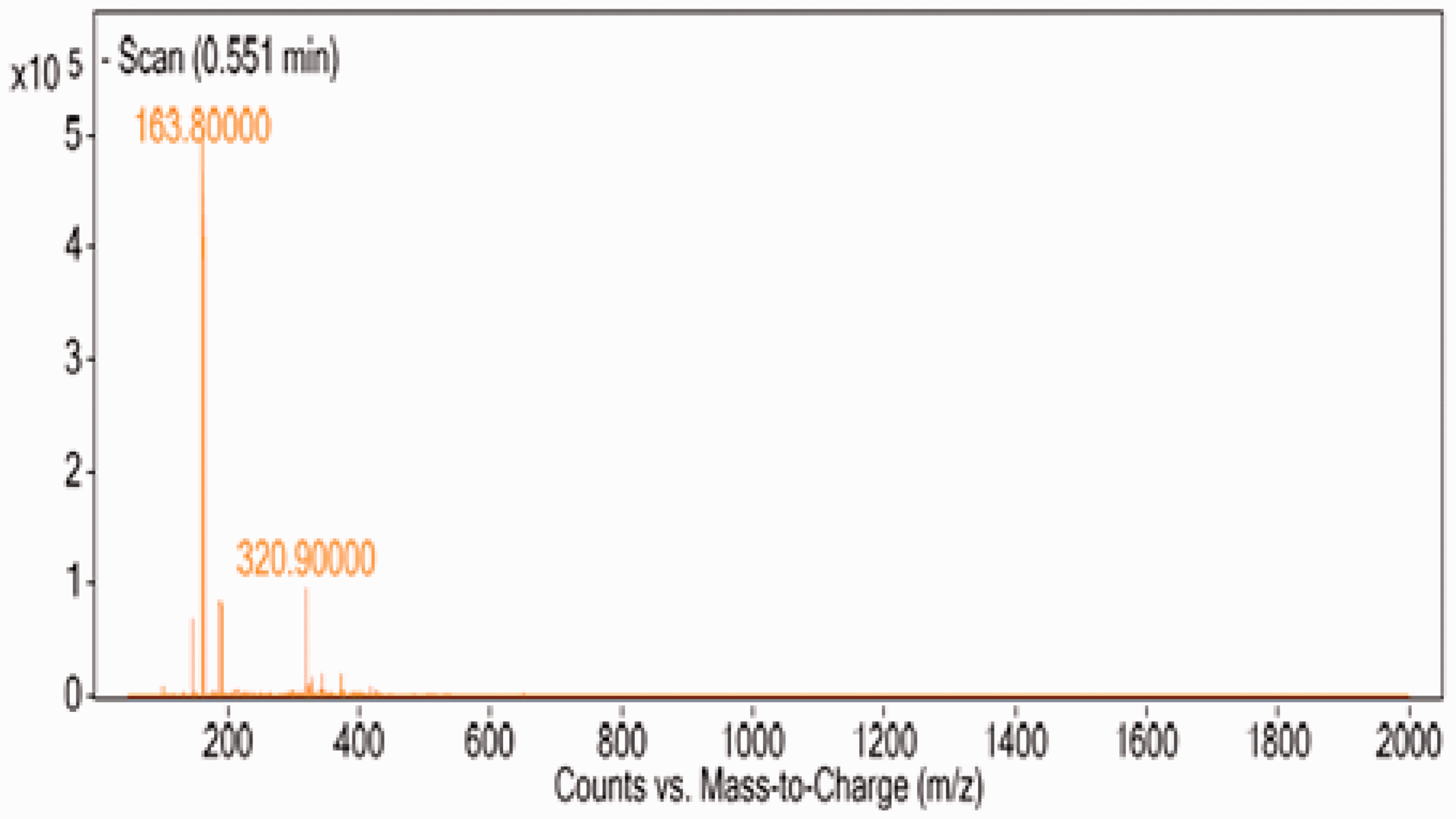
Antibacterial activity test
As mentioned earlier, one of the advantages of peptide structures is their anti-bacterial property. Peptides can be used as a natural and biocompatible anti-bacterial agent on fabrics to improve their properties for special applications.
Cyclic peptides in PNTs possess unique structural features which cannot be found in the natural class of peptide such as wool or silk. Under conditions that favor hydrogen bonding, such as adsorption on to lipid membranes, the cyclic peptides can stack to form hollow, β-sheet-like tubular structures that are open-ended, presenting the amino acid side chains on the outside surface of the ensemble. Therefore, appropriately designed cyclic peptides may be able to selectively target, self-assemble in bacterial membranes, and exert antibacterial activity by increasing the membrane permeability [22].
Each peptide was designed to bear at least one basic residue to enhance its target septicity towards negatively charged bacterial membrane. Studies on model membranes have indicated that peptide nanotubes provoke an increase in plasma membrane permeability. A first step in the mechanism of action is the electrostatic interaction between the cationic peptide and the negatively charged components of the membrane of the pathogen; hence, an increase in positive charge of the peptide will increase bacteriostatic activity. On the other hand, many positive charges can lead to a loss of activity of bacteria [23].
The results of anti-bacterial tests are shown in Figure 5 and Table 2. Samples shown in Figure 5(a) and (b) are the control samples which are not treated with PNTs solution. According to the results, it is deducted that although PNT50 sample has better results than PNT100 sample due to its higher concentration, chitosan-loaded PNTs can have a satisfying result even in a less concentration of amino acids. Chitosan itself has the ability to kill the bacteria and this effect is noticeable in the sample that is treated with chitosan-loaded PNTs.
Bacteria culture in various samples, (a) control sample against Number of colonies on various samples.

Air permeability analysis
Air permeability is one of the important parameters in fabrication of textiles with special features. It can also be used to provide an indication of the breathability of fabrics. Construction factors and finishing techniques can have an effect upon air permeability [24].
Air permeability results of the samples.

UV-Vis spectroscopy
The release of ascorbic acid and chitosan from PNTs was examined using UV-Vis spectroscopy. One of the effective factors on the loading of ascorbic acid in PNTs is incubation time. For the comparison of different incubation times, two cotton samples were treated with solutions that were incubated for 2 h and 4 h. As it is shown in Figure 6, the assembly appears to have better results when the incubating time is more than 2 h. Therefore, it can be deducted that longer incubation times can help PNTs have more uptake of ascorbic acid in comparison to shorter incubation times. Since self-assembly is a reversible reaction, release of some dipeptides from the nanotube surface in longer times may have facilitated further entrapment of ascorbic acid within the nanotubes.
Comparison of incubation times of PNTs. (a) 2 h incubation time. (b) 4 h incubation time.
For comparison of two methods of application of PNTs and ascorbic acid solution on cotton fabric, UV-Vis spectroscopy was carried out with two different samples. The results show that the padding method can probably cause some faults in the UV-Vis spectroscopy results. It can be due to the uneven pressure of padding machine during the process. Additionally, the pressure of this method can damage the hollow structure of peptide nanotubes. As a result, batch method can be a suitable method for application of PNTs and ascorbic acid on the cotton fabric. Figure 7 shows the results of UV-Vis spectroscopy of two methods.
Comparison of batch and padding methods for application of PNTs on cotton fabric. (a) Batch. (b) Padding.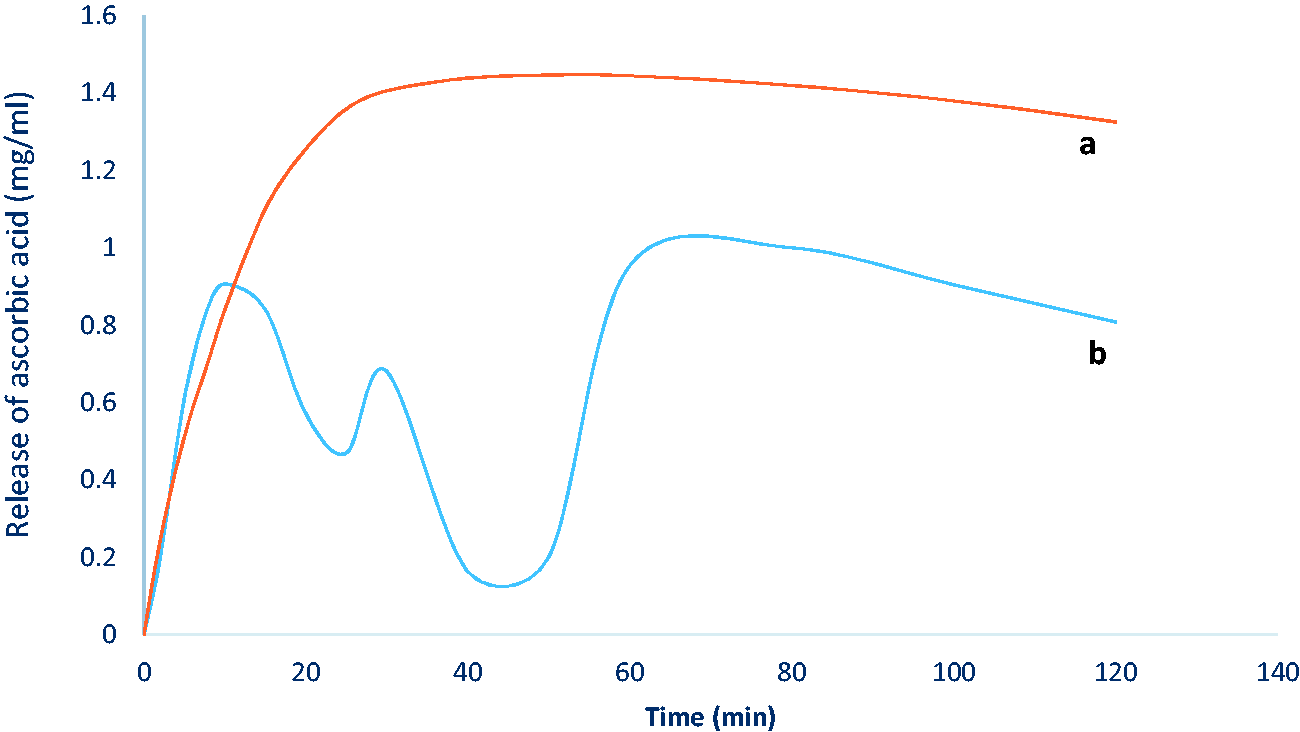
For the investigation of the release of chitosan from PNT100 solution, the cotton samples were treated by the solution of chitosan-loaded PNT100. This solution was prepared by adding 1% w/v of chitosan solution and PNT100 solution. The cotton fabric was treated with chitosan-loaded PNT100 solution for 4 h with batch method. The results are shown in Figure 8.
Comparison of the release of chitosan from PNT100 in two solvents. (a) Phosphate-buffered saline. (b) Acetic acid.
As shown in Figure 8, release of chitosan in acetic acid is more than phosphate-buffered saline which represents the liquid in human's body. This is due to the solubility of chitosan in acetic acid. Therefore, it can be deducted that PNTs are sensitive to pH of the environment and can react to the fluctuations in some parameters. Therefore, PNTs are one of the smart nanocarriers, which release the drug in specific conditions depending on different parameters.
Conclusion
In this study, self-assembled peptide nanotubes were developed for functional finishing of cosmetotextiles. A great advantage of using self-assembling peptides to build nanostructures in a “bottom-up” approach is that specific features such as effective antibacterial effect and releasing behavior can be incorporated.
The surface morphology of PNTs has been evaluated using electron microscopy. Ordered tubular assemblies are formed having a mean diameter of 90–120 nm, a length of 5000–5400 nm, and an average aspect ratio of 45. Additionally, antibacterial tests of cotton samples treated with different PNT solutions with/without chitosan as a famous bio antibacterial agent indicated that however samples that are treated with chitosan-loaded PNTs have better results than the other ones, the antibacterial effect of fabric treated with PNTs without chitosan is remarkable. This property is due to the appropriately designed cyclic structure of peptides that may be able to selectively target, self-assemble in bacterial membranes, and exert antibacterial activity by increasing the membrane permeability. The electrostatic interaction between the cationic group of peptide and the negatively charged components of the membrane of the pathogen increase the bacteriostatic activity of PNTs.
The release of ascorbic acid as model molecule in different incubation times was also examined by UV-Vis spectroscopy. The results show that the release of ascorbic acid from PNTs depends on the fluctuations of the environmental parameters, which turn them into one of the smart nanocarriers. As a conclusion, since PNTs are compatible structures to human's bodies, dipeptide nanotubes may be developed as different applications such as antibacterial agent and drug delivery systems for the finishing of bio-functional textiles.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
