Abstract
Chitosan-blended cellulose monoacetate nanofibers were prepared through electrospinning process. Neat nanofibers and their sodium hydroxide-treated analogs were used as support surfaces for protease immobilization via physical adsorption method. Morphologies of the nanofibers were observed with a scanning electron microscopy. Chemical analyses were conducted with Fourier transform infrared spectroscopy, and thermal analyses were carried out with differential scanning calorimeter and thermogravimetric analyzer. Immobilized enzyme activities were measured by using casein substrate. In order to test the stability of immobilized enzymes, the tests were repeated until the immobilized enzyme activity was leveled off. The results reveal that well uniform cellulose monoacetate/chitosan nanofibers were obtained, and nanofiber structures are transformed from rounded to more flattened morphology after enzyme activation test.
Introduction
Enzymes are biological molecules that accelerate conversion of the substrates to products under appropriate conditions by lowering the activation energy of reaction [1]. These remarkable biomaterials have been used in very broad applications, including pharmaceuticals, cosmetics, biodiesel production, agrochemicals, detergent, biomedicine, textile, and food industries [2–4].
Amylases, proteases, lipases, cellulases, and catalases are major enzymes for these applications and commercially available in the market. Protease is one of the most common enzymes used in industry for broad applications including meat tenderization, detergents, cheese making, dehairing, and waste management [5].
In order to reduce the cost and provide advantages, enzyme immobilization plays an important role in both research and industries. Several advantages can be listed as increasing the reusability of the enzymes, preventing enzyme contamination in final product, improving the enzyme activity, increasing the number of enzyme molecules per unit area, preventing the loss of activity, and elimination of separation and purification for reuse [4,6–8].
There are various enzyme immobilization techniques such as physical adsorption, ionic binding, covalent binding, cross-linking, and entrapment [9]. Among all these techniques, physical adsorption technique has advantages such as reducing or avoiding enzyme leaching [10]. On the other hand, since enzymes are physically bond to the surface via van der Waals, hydrophobic interactions, or hydrogen bonds, enzyme stability is lower compared to other techniques [11,12].
There are variety of support materials for enzyme immobilization including synthetic and natural polymers, ceramic materials, and metal nanoparticles. Most commonly used supports are natural polymers including chitosan (CHI), alginate, agarose, and cellulose, and natural polymers are of great interest because of their biocompatibility, biodegradability, nontoxicity, and chemical stabilization [13–16]. Cellulose is one of the most naturally abundant biopolymers, which can be produced and extracted from various living organisms including cotton, tree, and bacteria [15]. As a natural nanostructured support, cellulose-based nanofibers (NFs) have been produced and used for enzyme immobilization. Because of their high strength, stiffness, biocompatibility, renewability, and low cost, cellulose NFs gained increased attention for the use as enzyme immobilization support [15,16]. Laccase immobilization on cellulose NF was studied by Sathishkumar et al. [17]. They reported that the relative activity of the immobilized laccase was 88% and the half-life of the immobilized enzyme was retained for up to eight cycles. Chitin is the second most abundant natural polymer after cellulose in the nature. CHI is a material that is obtained from chitin through the use of chemical or enzymatic treatments [18, 19]. CHI is considered safe and has been used for both healthcare and biomedical application [20]. CHI is used as a support material for enzyme immobilization in the literature. In order to produce CHI NFs, polyvinyl alcohol (PVA)/CHI composite NFs were produced by electrospinning and then PVA removed from the fibers by sodium hydroxide (NaOH) treatment [21]. Lipase enzyme was immobilized to as-prepared CHI NFs support. The study indicates that CHI helps robustness of the enzyme stabilization and provides well enzyme immobilization.
Using degradable materials causes enzyme loss after each use, so immobilization of enzymes can be no longer critically important. Because some enzymes break the main chain of the polymer or hydrolyze them, polymers can be enzymatically degraded. For instance, poly(ethylene terephthalate) can degrade with esterase and papain, polyamide 6, 6 papain, trypsin, and chymotrypsin [22, 23]. Less degradable polymers are required for more stable enzyme immobilization application.
Since nanostructured materials have very high specific surface area comparing to their micro and macroscale counterparts, various nanostructured materials have been used as a support material for enzyme immobilization. Zinc oxide as ceramic nanoparticle support was used for lipase immobilization, and enhanced catalytic activity was observed comparing to crude enzymes [24]. Glucose oxidase was immobilized on as-prepared three-dimensional (3D) nitrogen-doped carbon nanotubes on freestanding electrospun carbon NFs film [25]. The results demonstrate that 3D network structure could effectively avoid enzyme leakage and maintain the bioactivity of the enzymes. Glucose oxidase enzymes were also immobilized to the amino-modified iron oxide nanoparticle surfaces [26]. Amino modification greatly enhanced the enzyme activity and stability comparing to the physical adsorption method. Gustafsson et al. [27] reported that glucose oxidase and horseradish peroxidase coimmobilized to mesoporous silica nanoparticles via spatially controlled way by using mesoporous silica nanoparticles and a polycationic dendronized polymer. They showed that the technique allows the spatially controlled localization of the different types of enzymes in a simple way.
Because nanoparticles must be carefully removed from the reacting media, immobilization of enzymes on nanoparticle can be a challenge for reusing. To overcome this problem, magnetic nanoparticles have been used for enzyme immobilization [28]. Enzyme immobilized magnetic nanoparticles can be controllable removed from the reacting media just by using a magnet.
NFs are promising materials with their high aspect ratio that provide high surface-to-volume ratio to the material. NFs have great number of applications including filtration, wound healing, energy generation and storage, drug delivery, and enzyme immobilization [29]. High surface area provides lots of active sites that enzymes can immobilize to and meet more substrates. Electrospinning is a process to fabricate continuous NFs from a variety of materials including polymers, ceramics, carbon, and metals [30–32]. Electrospun nanofibrous structures have been widely used in enzyme immobilization research and numerous studies have been reported in the literature [16, 21, 33, 34]. Silk fibroin NFs were prepared by electrospinning technique and used as enzyme immobilization support [34]. α-chymotrypsin immobilized silk fibroin NFs demonstrate high stability and activity achievement. Aqueous lipase and PVA solution directly was electrospun into nanofibrous structure by Wang and Hsieh [35]. Catalytic activity of lipase-loaded NFs and crude enzymes were the same and indicate that electric charge during electrospinning had no adverse effect on enzymes. Electrospun poly(acrylonitrile-
In this study, CHI-blended cellulose acetate NFs were produced with different CHI to cellulose acetate ratio and industrial protease was immobilized on as-spun NFs. In this regard, three different concepts were attempted. First, enzymes were directly immobilized on as-spun NFs by dripping liquid enzymes. Second, as-spun CMA/CHI NFs were treated with NaOH and then enzymes were dripped on the NFs. Finally, surface of as-spun and NaOH-treated NFs were activated with GA and then enzymes were dripped on the NFs. Furthermore, enzyme activity and reusability analysis were carried out and the results were interpreted.
Materials and methods
Chemicals
List of chemicals used in experimental section and their suppliers.


Schematic illustration of electrospinning process, chemical structures of cellulose monoacetate and chitosan, and general concept of protease immobilization on as-spun nanofibers and enzyme activity test.
Preparation of CMA/CHI NFs via electrospinning method
First, 15 wt% of CMA/acetone solution was prepared by magnetic stirring for 24 h under ambient conditions. Then, by considering the weight ratio a proper amount of CHI (low: 5/1 CMA/CHI, medium: 5/2 CMA/CHI, high: 1/1 CMA/CHI, extra high: 5/7 CMA/CHI) was added into the as-prepared solution and stirred. Final stock solution (1 ml) was loaded in a plastic syringe fitted with a stainless steel needle (with 0.508 mm inner diameter). A metal collector was grounded and stayed in front of the needle where polymer solution is fed. Flow rate, applied voltage, and the distance were approximately at 0.5 ml/h, 15 kV, and 15 cm, respectively. In order to obtain uniform NFs, these parameters were varied during the electrospinning process. Because of the applied voltage to the needle, polymer jet was ejected from the needle to grounded collector, solvent was evaporated during the process, and dried NFs were collected on the grounded metal collector. Schematic illustration of electrospinning process is demonstrated in Figure 1.
NaOH treatment of CMA/CHI NFs
As-spun NFs were carefully removed from the aluminum collector and treated with 2 wt% of NaOH/water solution which was prepared under ambient condition. NFs were soaked in the solution and stayed for 30 s and then rigorously washed with distilled water several times and dried before enzyme immobilization.
Characterization of CMA/CHI NFs
The morphologies of CMA/CHI NFs were analyzed using a ZEISS EVO 40 scanning electron microscope (SEM). In order to reduce charging during SEM analysis, samples were first placed on a SEM sample holder and coated with gold–palladium (about 100 Å of thickness) using a BAL-TEC SCD005 sputter coater. Then the samples were set in the SEM chamber and SEM analyses were conducted. Acceleration voltage (20 kV) was used for SEM analysis. Attenuated total reflection Fourier transform infrared (ATR-FTIR) spectra of CMA/CHI NFs were recorded for chemical analysis in the wavenumber range of 4000–500 cm−1 at room temperature. ATR-FTIR spectra were obtained on a Bruker TENSOR37 instrument. At least 124 scans were collected to minimize noise. Thermal properties of the as-spun NFs were evaluated using a differential scanning calorimeter (DSC) with a temperature range from 25 to 250℃ (heating rate of 10℃ min−1 in nitrogen environment). DSC characterizations were conducted with a TA-Instruments Q2000 model instrument. Weight loss of the as-spun NFs in air environments was determined using a thermogravimetric analyzer (TGA) by heating from 25 to 750℃ (heating rate of 10℃ min−1). TGA analyses were carried out with a Seiko Exstar TG/DTA instrument.
Immobilization of enzymes on NFs mat
One milliliter commercial protease enzyme solution (1495 IU/ml) was dripped onto as-spun NF samples and waited for 12 h to immobilize enzymes on the NFs at the room temperature. Then, enzyme immobilized NFs were vigorously washed with distilled water to remove excess free protease. General concept of protease immobilization on as-spun NFs is shown in Figure 1. Because the scope of this study is to observe protease immobilization on the NFs via physical adsorption, proteases were also immobilized on GA-activated NFs for the comparison. For this purpose, GA was diluted with distilled water, dropped on NFs, and waited for 5 min before enzyme immobilization. The same procedure was applied to NaOH-treated NF samples.
Enzyme activity measurement
Casein substrate was used for enzyme activity test and total protease activity was measured by modification of the Anson Method [40]. Tyrosine solution (0–60 µg/ml) was used to prepare a standard curve, and the amount of enzyme required to release 1 µg/ml tyrosine expresses the protease activity. The activity yield was defined as the yield of enzyme that was immobilized in NFs and was demonstrated as

Reusability of the immobilized enzymes
In order to test the stability of immobilized protease on NFs, enzyme immobilized NFs were exposed to enzyme activity test several times for the hydrolysis reaction. Each run lasted for 20 min and then NFs with immobilized enzymes washed with distilled water and stored at 4℃ until the next use. The reaction medium was then replaced with fresh medium. Enzyme activity in the first run was defined as 100%.
Results and discussions
Concept study to illustrate protease immobilization on NFs
As a concept, first protease immobilized on as-spun NFs via physical adsorption method, stayed 12 h, then enzyme activity measurements were performed. In Figure 2, SEM images of as-spun CMA/CHI (Figure 2(a)), protease immobilized CMA/CHI NFs (Figure 2(b)), and CMA/CHI NFs after enzyme activation test (Figure 2(c)) were demonstrated. As seen from the images, as-spun NFs are uniform whereas interpenetrated fiber morphologies of protease immobilized NFs are observed after activation test. The same concept was applied to all samples prepared in this study.
SEM images of (a) as-spun NFs, (b) enzyme immobilized on NFs, and (c) NFs after enzyme activity test. NF: nanofiber; SEM: scanning electron microscope.
SEM analysis of as-spun CMA and CMA/CHI NFs
Morphologies of as-spun neat CMA and CMA/CHI-blended NFs and their NaOH-treated analogs were evaluated via low and high magnification SEM imaging and shown in Figures 3 and 4, respectively. In order to obtain uniform NFs, electrospinning conditions (applied voltage, needle-to-collector distance, and solution flow rates) were varied during the electrospinning process. As observed from Figure 3(A1) and (A2), neat CMA NFs are bead free and uniform. The NF diameter varied from the nanometer to micrometer scale (Figure 3(B1), (C1), (D1)). Since CHI was not dissolved very well during electrospinning solution preparation, it is speculated that CHI particles were dispersed in CMA/acetone solution and some CHI particles were encapsulated by CMA NFs to form CMA/CHI composite nanofibrous structure.
As-spun CMA/CHI nanofibers: (A1, A2) CMA, (B1, B2) CMA/CHI (low), (C1, C2) CMA/CHI (medium), and (D1, D2) CMA/CHI (high). Two percent NaOH/water-treated CMA/CHI nanofibers: (A1, A2) CMA, (B1, B2) CMA/CHI (low), (C1, C2) CMA/CHI (medium), and (D1, D2) CMA/CHI (high). CHI: chitosan; CMA: cellulose monoacetate.

SEM analysis of NaOH-treated CMA and CMA/CHI NFs
SEM images of all the samples in Figure 3 after NaOH treatment are shown in Figure 4. As seen from the images, morphologies of NFs are dramatically changed after NaOH treatment. Nanofibers are flattened and more closely packed because aqueous NaOH diffused in the fibers and dissolved them a little bit (Figure 4). Size of the pores among the fibers decreases dramatically comparing with nontreated NFs.
Chemical analysis of NFs via FTIR
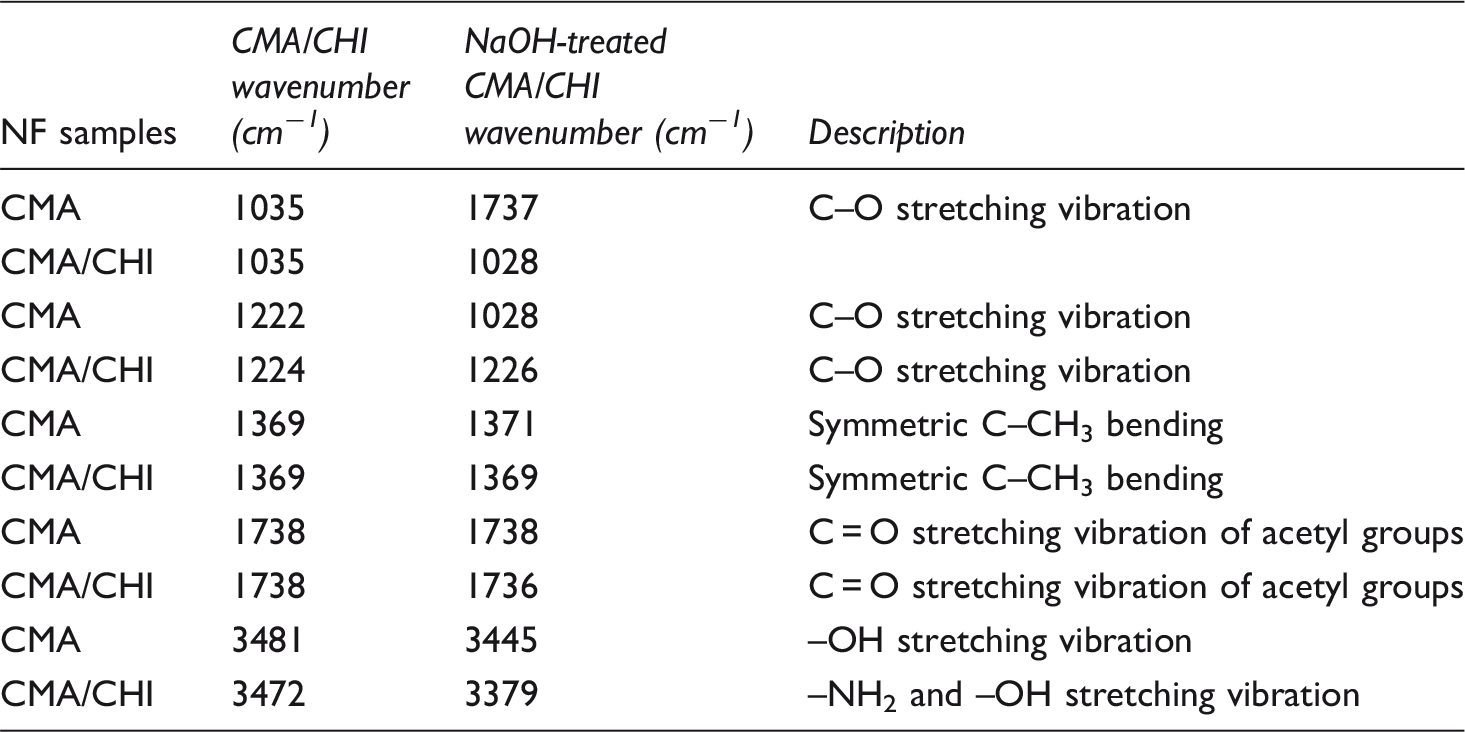
Figure 5(a) and (b) shows FTIR spectra of the produced CMA/CHI and NaOH-treated CMA/CHI NFs with different CHI ratio, respectively. FTIR results were demonstrated in Table 2 for bond assignments. In pure CMA NFs, strong peaks at 1739 cm−1 (C = O stretching vibration of acetyl groups) come from carbonyl absorption band corresponding to the acetyl group of cellulose acetate [39]. Also, peaks at 1037 and 1222 cm−1 (C–O stretching vibration), and 1369 cm−1 (symmetric C–CH3 bending) come from CMA NFs [42]. There is no distinctive difference between the neat CMA and CMA/CHI blends NF samples. It is speculated that the reason for not observing any CHI vibrational bands for CMA/CHI-blended NFs is due to the encapsulation of CHI by CMA during electrospinning since acetone was used as solvent in ES solution. Since only surface functional groups are detected by FTIR with ATR attachment, vibrational bands of amide groups in CHI could not be observed for these samples. The intensity of the band at 3000–3750 cm−1 due to the OH groups increases and broaden with increased CHI content in NaOH-treated CMA/CHI NFs samples. At CMA/CHI samples, a characteristic band around 3450 cm−1 is seen and this is attributed to –NH2 and –OH stretching vibration [43, 44]. This band increases after NaOH treatment and the intensity increases with increasing CHI content. The peak at 3497 cm−1 shifts to lower value with increasing CHI content in CMA indicates the increase of amine groups that comes from CHI [45]. These results prove our speculations that CHI encapsulated by CMA during electrospinning and encapsulated CHI in CMA/CHA NFs were shown up with NaOH treatment.
ATR-FTIR spectra of (A) CMA/CHI: (a) Pure CMA, (b) CMA/CHI (low), (c) CMA/CHI (medium), and (d) CMA/CHI (high); and (B) 2% NaOH-treated-CMA/CHI: (a) Pure CMA, (b) CMA/CHI (low), (c) CMA/CHI (medium), and (d) CMA/CHI (high). ATR-FTIR: attenuated total reflection Fourier transform infrared; CHI: chitosan; CMA: cellulose monoacetate. FTIR spectral results of neat and NaOH-treated CMA/CHI nanofibers. CMA/CHI was the CMA/CHI (high) sample. Note: CHI: chitosan; CMA: cellulose monoacetate; FTIR: Fourier transform infrared; NF: nanofiber.
DSC analysis of as-spun NFs
DSC thermograms of the as-spun CMA/CHI NFs were recorded at temperatures ranging from 25 to 275℃ and demonstrated in Figure 6. The freshly electrospun CMA NFs exhibit a relatively strong exothermic peak centered at ca. 230℃, which is associated with melting temperature of CMA NFs [46]. Glass transition temperature of CMA was observed at around 160℃ [47]. With addition of CHI in CMA NF melting temperature and glass transition temperature went to lower temperature and this trend continued with increasing CHI content in CMA/CHI NFs. Jung et al. [43] reported that cellulose acetate has transition temperature near 220–230℃, CHI has lower transition temperature near 180℃, and blending these two polymers shifts to transition to a lower temperature as a result of interactions among polymers.
DSC thermal analysis of (a) CMA, (b) CMA/CHI (low), (c) CMA/CHI (medium) and (d) CMA/CHI (high). CHI: chitosan; CMA: cellulose monoacetate.
TGA analysis of as-spun and NaOH-treated NFs
TGA plots observed for as-spun and NaOH-treated CMA/CHI NFs in air atmosphere as shown in Figure 7(a) and (b). As observed by inspecting these plots, decomposition and degradation of the CMA/CHI NFs mostly take place in the temperature range of 280−350℃ (Figure 7(a)). An obvious weight loss in this temperature range is related to decomposition of the polymers and the removal of volatile components. The residuals of the samples after TGA measurement increase with increasing CHI content in CMA/CHI NFs and the results are fluctuated. The initial weight loss between 25 and 120℃ is proportional to CMA ratio in the NaOH-treated CMA/CHI NFs that can be attributed to the absorbed moisture from the environment. The onset decomposition temperature of NaOH-treated CMA/CHI NFs is lower comparing to untreated samples and increases with increasing CHI content (Figure 7(b)). As a result, thermal stability increases at NaOH treated samples with increasing CHI content even though addition of CHI and increase its content in CMA NFs does not dramatically affect the thermal stability of as-spun NFs.
TGA thermograms of (a) CMA/CHI and (b) 2% NaOH/water-treated CMA/CHI nanofibers. CHI: chitosan; CMA: cellulose monoacetate; TGA: thermogravimetric analyzer.
Enzyme immobilization yield and stability measurement
Effect of number of reused cycles of immobilized protease enzymes on CMA/CHI nanofibers when free enzyme activity is 1495 IU/ml.

Note: CHI: chitosan; CMA: cellulose monoacetate; GA:
A: pure CMA/Chitosan NFs, B: GA-activated CMA/chitosan NFs, C: NaOH-treated CMA/chitosan NFs, and D: NaOH-treated and GA-activated CMA/chitosan NFs.
Immobilization yield was determined from the comparison of free enzyme activity and immobilized enzyme activity at first cycle. Among all the samples, the highest immobilization yield was obtained as about 83% with GA-activated CMA/CHI samples. Other samples have lower immobilization yield, are 63, 70, 82, respectively, for CMA, CMA/CHI, and GA-activated CMA NFs. Immobilization yield decreased more after NaOH treatment. Immobilization yield was measured as 67, 69, 55, and 68% for NaOH-treated CMA, CMA/CHI, GA-activated CMA, and GA-activated CMA/CHI NF samples, respectively. The reuse efficiency of immobilized enzyme decreased after being used several times. The remaining activities of the immobilized enzymes on CMA and CMA/CHI were found as 55 and 48%, respectively, after four times of reuse. Among all samples the immobilization efficiency decreased gradually after five uses, retained activity at NaOH-treated and GA-activated samples were around 34% for CMA and 19% for CMA/CHI NF samples after seven reuses. Hence, even though the onset immobilization efficiency at NaOH-treated samples is the lowest, NaOH-treated and GA-activated samples have the highest operational stability among all samples.
When enzyme activity plots were inspected in Figure 8(a) to (d), GA-activated (Figure 8(b)) and nonactivated (Figure 8(a)) pure CMA and CMA/CHI NFs demonstrate similar cycle numbers and most of the enzyme activity was lost up to seven cycles. But after NaOH treatment, GA activation effectively increases the cycle number and about 20% of enzyme activity was still retained after seven cycles (Figure 8(d)). This percentage is higher at CMA NFs than CMA/CHI NFs and measured around 33.5%. In a similar study, Huang et al. [48] prepared cellulose acetate NFs and then exposed these NFs to alkaline hydrolysis to convert NF surfaces into regenerated form. Surface of NF activated with aldehyde groups for lipase immobilization. Comparing free enzymes, the immobilized lipase exhibited significantly higher thermal stability and durability. Immobilized enzyme activity decayed with repeated uses and the remaining enzyme activity on cellulose NFs was about 30% after eight cycles. In this study, NaOH-treated and GA-activated CMA NF samples have 33.5% remaining enzyme activity after seven cycles. Huang et al. immobilized lipase on CHI NFs via GA activation. They reported that residual activities of the immobilized enzymes were 46% after 10 cycles [21]. On the other hand, at high GA concentration enzyme activity decreases, so the GA activation must be limited. Croisier and Jerome [20] reported the GA rapidly reacts with the amino groups of the CHI and GA can covalently bond to enzyme. Because CHI has amino groups, it was expected that existence of CHI in CMA NFs can increase the cycle number but it was not happened as it was expected. Because amino groups of CHI were not functionalized properly for chemical bonding with GA, enzymes were not bond properly to NFs.
Reusability of immobilized protease enzyme. (a) CMA/CHI NFs, (b) CMA/CHI NFs coated with GA, (c) CMA/CHI NFs treated with 2% NaOH, and (d) GA coated CMA/CHI NFs coated with GA and treated with 2% NaOH. All values are averages of results from triplicate trials; error bars indicate the SD values. CHI: chitosan; CMA: cellulose monoacetate; GA: 
SEM images of NFs after enzyme activity test
SEM images of protease immobilized NF after activation test are demonstrated in Figures 9 and 10. More flattened and interpenetrated NF fibers morphology of protease immobilized NFs activation test (Figures 9 and 10) are seen. More clogged void morphologies are seen in NaOH-treated samples comparing nontreated samples (Figure 10). NF uniformity was still intact, but physical integrity of the NFs was lost after enzyme activity test as a result of conducting the test in tyrosine solution. This was mostly observed at NaOH-treated samples. Because aqueous NaOH solution diffused in the fibers and dissolved them a little bit, NFs morphologies transformed from rounded to more flattened and closely packed structure, and hence the morphology can be similar after enzyme activity test. On the other hand, because immobilized and used enzymes stayed among the fibers, clogged structure and interpenetrated morphology were inevitable.
NFs after activations test: (A1, A2) CMA, (B1, B2) CMA/CHI (low), (C1, C2) CMA/CHI (medium), and (D1, D2) CMA/CHI (high). CHI: chitosan; CMA: cellulose monoacetate; NF: nanofiber. Two percent NaOH-treated NFs after activations test: (A1, A2) CMA, (B1, B2) CMA/CHI (low), (C1, C2) CMA/CHI (medium), and (D1, D2) CMA/CHI (high). CHI: chitosan; CMA: cellulose monoacetate; NF: nanofiber.

Conclusion
In the present study, protease was immobilized on as-spun and NaOH-treated CMA/CHI NFs via physical adsorption method. Morphological investigation reveals that uniform NF structure is converted to interpenetrated and more flattened NF structure after enzyme activity test. Immobilized enzyme activity was measured by using casein substrate and compared with free enzymes. GA activation has positive effect on NaOH-treated samples, and the highest immobilization yield observed for GA-treated CMA/CHI samples is about 83%. NaOH treatment before GA activation shows the best results for protease immobilization on CMA and CMA/CHI NFs. Operational stability increases after NaOH treatment and GA activation. After NaOH treatment, GA activation effectively increases the cycle number and about 20% of enzyme activity was still retained after seven cycles at CMA/CHI samples. This percentage is higher at pure CMA NFs than CMA/CHI NFs and measured around 33.5%. Comparing free protease, immobilized protease can be used repeatability in targeted applications such as detergent and textile industries.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by Scientific Research
