Abstract
Carbon nanotubes have been explored to increase the mechanical properties and electrical conductivity of polymeric fibers through compounding with polymer to be extruded into fibers. However, this route creates major challenges because carbon nanotubes have strong cohesion and tend to aggregate and precipitate due to their poor interfacial interaction with polymers. In this study, classical molecular dynamics simulations are used to predict and characterize carbon nanotubes-polymer interface mechanism in two different polymer matrices: polyvinyl butyral and polystyrene-co-glycidyl methacrylate. The dominated interface mechanisms are discovered to shed light on carbon nanotubes dispersion in solvent based systems and to explore the prerequisites for stabilized nanofluids. Our results showed that π-stacking interactions between aromatic groups and graphene surfaces of carbon nanotubes as in polystyrene-co-glycidyl methacrylate systems, play an important role in dispersion of carbon nanotubes, whereas slight repulsions between carbon nanotubes and polyvinyl butyral chains lead to large morphological differences and carbon nanotubes bundles in many chain systems. Altogether, the results indicated that polymers with structures having strong interactions with the surfaces of carbon nanotubes through π–π interactions are more effective in dispersing carbon nanotubes and caused stabilized solutions in wet fiber processing.
Introduction
Carbon nanotubes (CNTs) first reported by Oberlin et al. [1] and Iijima [2] have attracted attention of many scientists due to their unique mechanical, electrical, and thermal properties. This form of carbon containing hexagons with concentric arrangement of cylinders has three distinctive types such as single-walled nanotubes (SWNTs), double-walled nanotubes (DWNTs), and multi-walled nanotubes (MWNTs). SWNTs have one tubular rolled layer of hexagonal carbon atoms with diameters ranging from 0.4 to 3 nm (Figure 1(a)), whereas MWNTs have several coaxial cylindrical layers with diameters up to 100 nm [3]. CNTs are excellent nano-reinforcement materials due to their low mass density, large aspect ratio, and high elastic modulus (approximately 1 TPa [4]) and thus used in numerous applications such as composites [5–8], electrochemical devices [9–12], hydrogen storage [13–16], field emission devices [17–19], nanometer-sized electronic devices [20–22], sensors and probes [23–26], fibers [27–29], etc. However, the superior performance of CNTs which depends on the distribution of length and diameter, the chirality, and its defect and impurities cannot as yet be fully transferred into macro-scale composites. Many studies investigated an appropriate scheme for transferring individual nanotube properties into polymer composites [8,9,30–34]. In wet fiber processing, the major drawback is to obtain a good dispersion of CNTs by maintaining attractive interaction with the surrounding polymer matrix [8]. Moreover, it is also essential to stabilize this dispersion to prevent reaggregation of the CNTs throughout the processes.
(a) Ball and stick representation of (6,6) single-walled CNTs with diameter of 8.14 Å and length of 14.76 Å sketched by Materials Studio® 8.0 [68] and chemical structures of (b) P(St-co-GMA), (c) PVB, and (d) DMF.
In literature, several polymer matrices are reported for CNTs reinforced nanofiber/fibers that benefit from CNTs-polymer attractive interactions [35–37]. For instance, recent experimental observations indicated that polystyrene-co-glycidyl methacrylate (P(St-co-GMA)) containing aromatic rings have attractive π–π interaction with the graphitic side walls of CNTs [38], which is called “π-stacking.” The same interaction leads to CNTs bundles, which are major obstacles to their processing [39]. There are several techniques reported to overcome this challenge, particularly in wet fiber processing [40–43], where homogenously dispersed polymer/nanotube solutions lead to well-oriented CNTs in the resultant nanofibers/fibers. For instance, adding surfactants (e.g. sodium dodecyl sulfate), large amphiphilic polymers (e.g. polyvinyl pyrrolidone), and natural macromolecules (e.g. polysaccharide, Gum arabic), which can be adsorbed onto the hydrophobic nanotubes help to stabilize CNTs dispersions [44,45]. In addition, ultrasonication is another useful technique to overcome the entanglement of nanotubes and to break up the agglomerates. However, it might cause some defects and irregularities into the CNTs [46]. Chemical functionalization of CNTs also helps to disperse CNTs, but it dominantly affects their electronic and photonic properties [47]. Different types of polymers, including polystyrene, polyurethane, P(St-co-GMA), and poly(vinyl butyral) (PVB) have been used as polymer matrices to produce CNTs composite nanofibers [47–50]. Polymers containing aromatic rings are able to wrap CNTs via π–π stacking and van der Waals interactions between the polymer chain and nanotube surface [51]. Ozden-Yenigun et al. used surface reactive copolymer (P(St-co-GMA)) and successfully produced CNTs reinforced electrospun nanofibers without any modification, they reported the enhancement in flexural strength (15%) and flexural modulus (20%) within the incorporation of CNTs into epoxy resin [38]. The researchers also used PVB as matrix in CNTs integrated nanocomposites to enhance mechanical, electrical and thermal properties [52,53]. Later, Imaizumi et al. produced MWNTs/PVB composite electrospun nanofibers and twisted these nanofibers to have composite nanofiber yarns [54]. Enhanced mechanical, electrical, and thermal properties were reported and explained by referring CNTs-PVB interactions. As seen in literature, P(St-co-GMA) (Figure 1(b)) and PVB (Figure 1(c)) are two different polymers that successfully promise CNTs oriented nanofibers. However, dominated mechanism in liquid state prior to fiber forming was not explained in detail. Herein, we aimed to explore these two different polymer matrices’ dispersing ability of CNTs, which are in different chemical nature. Classical molecular dynamic (MD) simulation can describe in detail the interface and morphology of polymer chains adsorbed on CNTs surfaces and the interaction mechanisms that take place in different supramolecular systems [55,56]. On the other hand, computational intensiveness of such simulations might prevent exorbitant experimental sampling [56,57]. MD simulation is frequently used to explore CNTs and their interactions in polymer matrices [58–62]. For instance, Uddin et al. investigated the effect of surfactant chemistry in CNTs/surfactant in water-based solutions via MD [63]. They also reported the dominated dispersive interactions in CNTs/polyethylene oxide/water systems and pointed out the hydrophobic interactions of CNTs in aqueous solutions [64]. Pang et al. explored the dispersion state of CNTs and the wrapping motion of silicon surfactants experimentally and computationally and noted that van der Waals attractions leaded to steric stabilization [65]. Sohrabi et al. used MD simulations to probe both pure and mixed surfactants adsorption onto nanotubes [66]. They stated at low surfactant concentration all surfactant molecules, lie flat on the surface or wrap around CNTs and only a small fraction of surfactant heads in pure cationic or cationic-rich mixtures are extended toward the water. Xiao et al. functionalized CNTs by covalent linking of alkyl chains and investigated the wrapping ability of functional molecules around CNTs, the miscibility of functional molecules with polyethylene matrix, and the aggregation behavior of functionalized CNTs using MD [67]. Their investigations demonstrated that CNTs without alkyl chains experienced a smooth pullout, whereas those with alkyl chains underwent an uneven pullout with five failure stages. They also stated that crystallization of polyethylene molecules occurs in the vicinity of CNT and it increases the interfacial shear strength by 15% [67].
In this study, MD simulations are used to predict and characterize polymer-CNTs interface mechanisms in PVB and P(St-co-GMA) polymer solutions, which are proven good polymer matrices for composite fibers [38,54]. Thus, P(St-co-GMA) and PVB polymers are introduced into atomistic models to investigate and compare their interactions with CNTs by monitoring the effect of aromatic groups on CNTs dispersion. We also incorporate solvent molecules dimethylformamide (DMF) (Figure 1(d)) to simulate solvent-based fiber solutions, as in wet-fiber processing. Local structure is evaluated by analyzing radial distribution function (RDFs) of CNTs concentration profile and radius of gyration (RG) of polymer to understand dominated particle-polymer interaction mechanism [69,70]. Clustering behavior and single polymer dynamics of neat and CNTs containing polymer solutions are determined to provide insight into the nanofluid behavior at the macro-scale.
Materials and methods
Simulated models of PVB and P(St-co-GMA), construction parameters and their cell parameters for MD simulations.

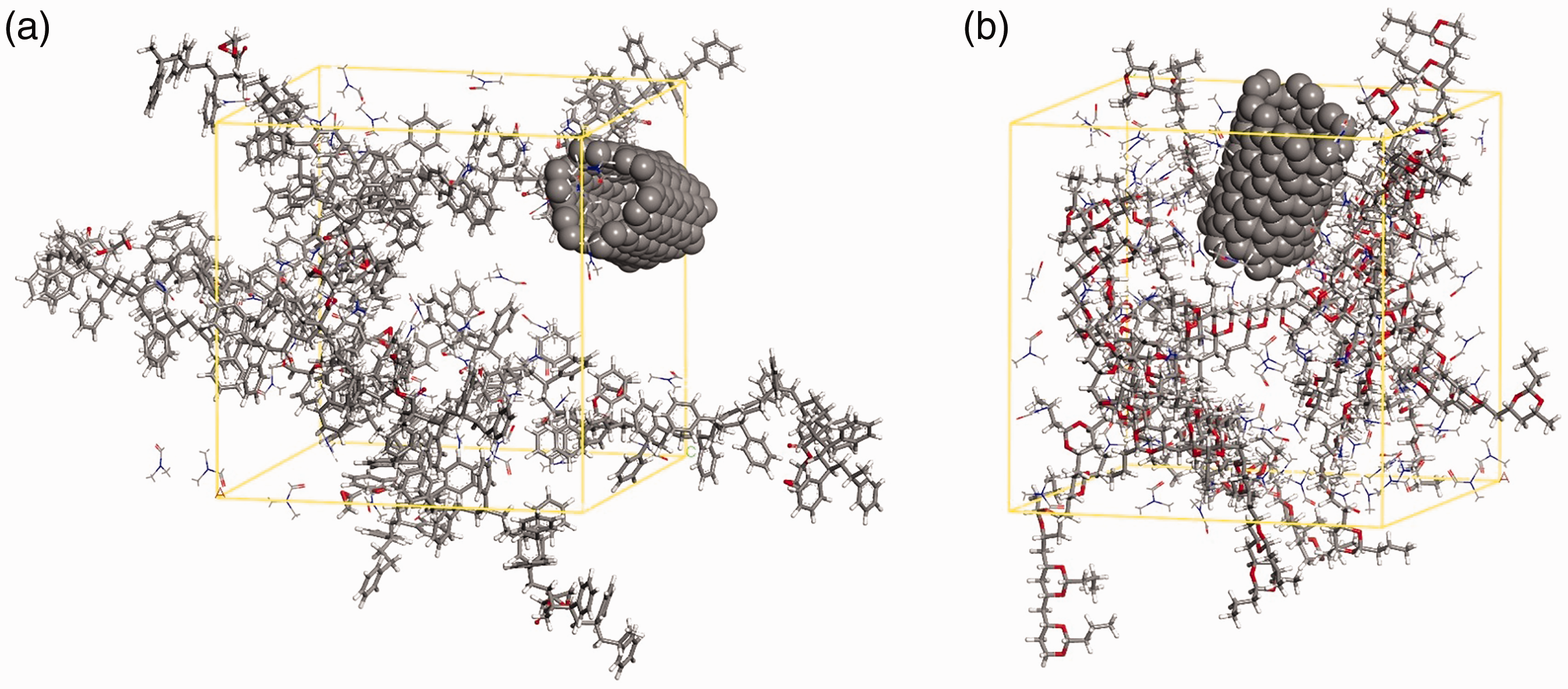
Constructed simulation boxes of (a) PSTcoGMA-1-CNTs and (b) PVB-1-CNTs systems, which are described in Table 1. For better description, CNTs were displayed in CPK representation, while stick and line representations are preferred for polymer and solvent molecules, respectively.
Each system was subject to detailed MD simulations up to 20 ns. The trajectories of equilibrated systems were saved and analyzed by Materials Studio® Forcite Analysis Module to monitor structural changes. Local structure evolution was completed by analyzing structural parameters such as RDFs, CNTs concentration profile, and RG of polymers of the equilibrated trajectories. Pair correlation functions of each system including intra and intermolecular components were calculated at cutoff 12.5 Å. Last 5 ns trajectories were averaged out to determine concentration profiles of CNTs-CNTs pairs and RDFs of CNTs-CNTs, CNTs-DMF, and CNTs-PVB/P(St-co-GMA) pairs at different CNT concentrations, and to understand dominated clustering and dispersion properties, while RG of PVB and P(St-co-GMA) molecules were also explored to reveal single chain dynamics in presence of CNTs.
Results and discussion
Dispersion of CNTs in PVB/DMF systems
Molecular interactions between CNTs, polymer, and DMF solvent molecules were studied at different CNTs number densities as given in Table 1 to provide detailed information at the molecular level as well as to understand the dominated interaction mechanism. First, RDFs of PVB systems were calculated. Figure 3(a) and (b) describes RDFs of CNTs-CNTs and PVB-CNTs at different CNTs number densities, and reveal short and long range of order in presence of CNTs in PVB/DMF solutions. Figure 3(a) demonstrates that the first coordination shell of CNTs-CNTs interactions ends at ca. 5 Å, displaying sharper distributions in PVB-2-CNTs and PVB-3-CNTs. RDFs of CNTs-PVB at this distance showed that increasing CNTs concentration also altered polymer-CNTs interaction in short distances due to increase in probability of finding each other. While, at longer distances, as beyond 10 Å, CNTs-CNTs interactions and CNTs-PVB interaction were balanced. But still, because of the tendency of CNTs to prefer CNTs over polymer molecules, there will be an uneven distribution of CNTs and polymer molecules elsewhere. This could lead to CNTs clustering particularly in CNTs rich suspensions, which also pointed one of the major drawbacks to use CNTs at high loading levels (above 10 wt%). PVB-2-CNTs and PVB-3-CNTs systems revealed that although CNTs tend to agglomerate in low concentrations due to the “π-stacking” phenomena [74], in higher concentration models as in PVB-4-CNTs and PVB-5-CNTs, CNTs have to occupy the space in every coordinates (Figure 3(a)). It could be related to the abundance of the CNTs in the same cell box dimensions. We should also note that Imaizumi et al. [54] observed highly oriented and individual MWNTs along fiber axis, at 10 wt% MWNTs concentration, which was due to the electrified liquid flow formed during electrospinning. On the other hand, additive effect of the MWNTs on the electrical conductivity of the as-spun composite fiber was quite limited. They concluded that the PVB matrix polymer blocked the electrical contacts between the nanotubes. We rationalize this outcome by considering the distribution of CNTs. In PVB systems, CNTs tend to cluster more with its own kind than the other species, which leads to prevent to build required network for electrical conductivity.
(a) Radial distribution function (RDF) of CNTs-CNTs in PVB-2-CNTs, PVB-3-CNTs, PVB-4-CNTs, PVB-5-CNTs models; (b) RDF of PVB-CNTs in PVB-1-CNTs, PVB-2-CNTs, PVB-3-CNTs, PVB-4-CNTs, PVB-5-CNTs models; and (c) RDF of DMF-CNTs in PVB-1-CNTs, PVB-2-CNTs, PVB-3-CNTs, PVB-4-CNTs, and PVB-5-CNTs models.
Even though, DMF solvent molecules are mobile enough, the tendency of DMF molecules toward CNTs are reduced by increased CNTs concentrations, as seen in Figure 3(c). Therefore, we observed that solvent choice does not provide additive effect to disperse CNTs in PVB systems. Single chain dynamics and dimensional stability could provide insight into attractive and repulsive interactions around polymer chains. The analysis on RG expressed polymer chain conformations and exhibited how the gyration radius changed due to these repulsive/attractive interactions. As seen in Figure 4, RG of PVB chains were calculated at different CNTs concentrations. For better description, Gaussian fits of curves given in Figure 4 while raw data are also provided in Figure S1.
Gaussian fits of radius of gyration (RG) of PVB molecules in all equilibrated PVB models.
The measures on chain size, RG, carry information on the local chain geometry, which depends on valence and dihedral angle distributions as well as local interactions [75]. Figure 4 describes that with an increase in CNTs number density, RG of PVB molecules slightly reduces. Due to repulsive interactions between CNTs-PVB, polymer molecules suffer from chain contractions. Moreover, three distinctive peaks were observed at higher polymer concentrations (e.g. PVB-4-CNTs and PVB-5-CNTs), and this could lead to CNTs rich and polymer rich regions in the polymer solutions, caused by uneven distribution of molecules. The small variations observed in chain dimensions and slight repulsions between CNTs and PVB chains might lead to large morphological differences in many chain systems and eventually phase separations. Thus, the tendency of CNTs to agglomerate in PVB/DMF systems was explored by CNTs concentration profile analysis. Last 5 ns chunks of total 20 ns dynamics of each equilibrated systems were analyzed to obtain CNTs concentration profiles in (100), (010), and (001) hkl planes. Figure 5 illustrates the CNTs concentration profiles of PVB models in these three different planes, respectively. In high concentration models, due to high number fraction of CNTs in the same cell-box dimensions, CNTs are standing more close to each other. In PVB models, even in high CNTs concentration models, phase separation of CNTs aggregates occurs (e.g. PVB-5-CNTs model at ca. 13 Å in (001) plane). Thus, it is clear that PVB polymers are not effective enough to disperse CNTs individually at atomistic level, they may require additional dispersing agents such as surfactants and/or dispersing method in wet fiber processing or electrohydrodynamic spinning process such as electrospinning.
CNTs concentration profile of PVB models at different CNT concentrations in given planes: (a) (100), (b) (010), and (c) (001).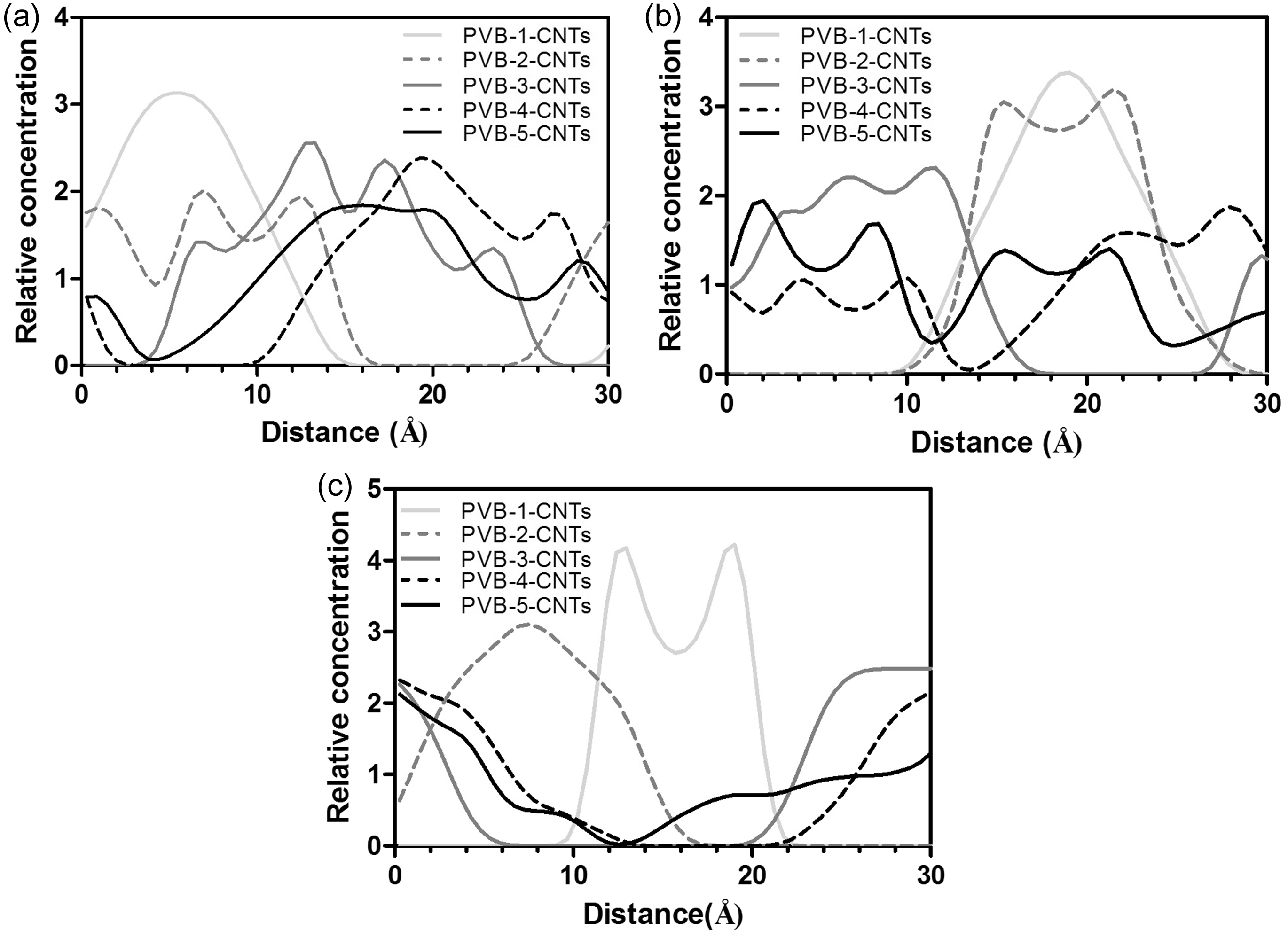
Dispersion of CNTs in P(St-co-GMA)/DMF systems
The experimental observations pointed out that long-term stability of CNTs could be achieved by the manipulation of π–π interactions in P(St-co-GMA)/DMF systems [18]. Thus, as seen in Figure 6, P(St-co-GMA) models demonstrate different interaction mechanisms than PVB models. Figure 6(a) and (b) exhibits RDF of CNTs-CNTs and CNTs-P(St-co-GMA) at different CNTs concentrations, respectively. As seen in Figure 6(a), there was no long range order for CNTs. A peak at around 12.5 Å caused by the neighboring walls of CNTs considering the diameter of individual CNTs, which is approximately 8.14 Å. The tendency of clustering is not as strong as it is in PVB systems. Figure 6(b) addressed that polymer has affinity toward CNTs both in short- and long-range distances. This difference might be based on the presence of aromatic groups in P(St-co-GMA) that provide strong short-range interactions with nanotube sidewalls. Figure 6(b) also shows that (St-co-GMA) molecules were positioned closer to sidewalls than PVB molecules due to the existence of aromatic groups. This further supports our prediction that there could be strong alignment of aromatic rings of P(St-co-GMA) parallel to the CNTs surface [43,76]. As has been recently demonstrated with aromatic amino acids [76], the stacking of aromatic groups onto the SWNTs surface allows for the dispersion of individual SWNTs. Moreover, the incorporation of aromatic groups decreases the ability of the peptides to self-associate, which was also revealed by atomic force microscope images. Intensive π–π interaction between these polycyclic aromatic hydrocarbons and the external SWNTs surface were supported by experimental findings [77,78]. As in P(St-co-GMA) molecules, rigid-backbone polymers can be able to form ordered molecular structures surrounding the nanotubes with n-fold symmetry, as expressed by Nish et al. [79]. Beyond 12.5 Å, P(St-co-GMA) molecules still maintain their tendency toward CNTs as seen in Figure 6(b). This can be interpreted as the dispersing ability of styrene-based polymers, which could be an effective agent in wet-fiber processing. Thus, it is predictable that isolation of individual CNTs could be achieved with a polymer design containing aromatic groups. RDFs of DMF-CNTs (Figure 6(c)), in presence of P(St-co-GMA), display almost same profile as in PVB models and, at longer distances, DMF-CNTs interactions are diminished while increasing CNTs concentrations, whereas styrene-based polymers are more soluble in DMF than PVB. The selective dispersion have been found to be strongly influenced by the polymer structures and solvent used [80].
(a) RDF of CNTs-CNTs in PSTcoGMA-2-CNTs, PSTcoGMA-3-CNTs, PSTcoGMA-4-CNTs, and PSTcoGMA-5-CNTs models; (b) RDF of CNTs-P(St-co-GMA) in PSTcoGMA-1-CNTs, PSTcoGMA-2-CNTs, PSTcoGMA-3-CNTs, PSTcoGMA-4-CNTs, and PSTcoGMA-5-CNTs models; (c) RDF of DMF-CNTs in PSTcoGMA-1-CNTs, PSTcoGMA-2-CNTs, PSTcoGMA-3-CNTs, PSTcoGMA-4-CNTs, and PSTcoGMA-5-CNTs models.
Figure 7 displays RG of P(St-co-GMA) molecules at different CNTs number densities. For better description, Gaussian fits of curves are given in Figure 7 while raw data are also provided in Figure S2. The mean peak value of RG for the P(St-co-GMA) molecule in absence of CNTs (e.g. PSTcoGMA-0-CNTs) was about 20 Å, whereas in PSTcoGMA-5-CNTs model, two distinctive peaks were observed and the mean value was shifted to 15.6 Å. We observed that with an increase in CNTs concentration, RG of P(St-co-GMA) molecules slightly decreases, as in PVB models. Furthermore, the shift of RG in presence of CNTs was below 5 Å (from 20.0 Å to 15.6 Å), whereas the end to end distance of P(St-co-GMA) was approximately 70 Å. In case of PVB, this dimensional change was about 3 Å and the end to end distance of PVB molecule was ca. 50 Å. Therefore, we conclude that PVB molecules have almost the same contraction value (%) with P(St-co-GMA) molecules. However, in case of P(St-co-GMA) systems, polymer molecules suffer more from intra-molecular π–π interactions than inter-molecular P(St-co-GMA)-CNTs interactions. Concentration profile of CNTs given in Figure 8 would also assist to understand these same-kind interactions. Unlike PVB models, P(St-co-GMA) models exhibited better dispersion state in high CNTs loadings. Thus, no phase separation was observed in PSTcoGMA-4-CNTs and PSTcoGMA-5-CNTs models (Figure 8). This is further evidence of how that better dispersion of CNTs in the vicinity of P(St-co-GMA) molecules was achieved due to the increased π–π stacking with a strong orientation preference.
Gaussian fits of RG of P(St-co-GMA) molecules in all equilibrated PSTcoGMA models. CNTs concentration profile of P(St-co-GMA) models at different concentrations in given planes: (a) (100), (b) (010), and (c) (001).

Conclusion
In conclusion, local structures and conformational analysis in presence of CNTs are performed to provide insight into dispersing ability of PVB and P(St-co-GMA) molecules. It is seen that due to π–π interactions between the aromatic rings of P(St-co-GMA) and CNTs, P(St-co-GMA) molecules were more effective in dispersing CNTs and/or preventing CNTs clusters than PVB molecules. PVB-CNTs interactions influence the degree of nanotube dispersion and polymer reaggregation. Additional dispersing agents such as surfactants in wet fiber processing or electrohydrodynamic spinning process were required to form composite PVB fibers. We can conclude that polymers with aromatic groups are better candidates to disperse CNTs in wet-fiber processing. Manipulating the inter-molecular π–π interactions would eliminate the need of dispersing agents in fiber forming.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work presented here was supported by TUBITAK 3001 (Project No.: 213M618).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
