Abstract
The inadequacies of the currently used small diameter, non-biodegradable synthetic grafts prompt researchers to focus on the design parameters of vascular grafts. Since the purpose is to mimic the native vessel as far as possible, the design parameters are mainly determined by the layout of cell types and proteins in the layers of the vessels and the nano and micro structure of their environments. In consequence of this, the complex structure of native vessels has become a broad source of inspiration for researchers. Electrospun fibrous scaffolds with their well accepted advantages are promising candidates and researchers are able to work with various materials with differing forms, structures, dimensions, and surface modifications according to their requirements. On the other hand, both synthetic biodegradable polymers and natural proteins are the key materials that enable researchers to take one step closer to achieving the goal of creating an autologous vessel at some time after implantation. When the priority and significance of the need for small diameter vascular grafts is considered, the research field to improve vascular grafts is worthy of note. In this regard, the objective of this review is to discuss comparatively the current studies on the design parameters of electrospun vascular grafts, defined as fiber orientation, fiber diameter, pore size and porosity, wall thickness, and material selection, based on the structure of native blood vessels, the requirements of vascular grafts and electrospinning technology, and the advantages of electrospun vascular grafts, to give an outlook for further studies.
Introduction
As the world population is getting older, cardiovascular diseases are increasing correspondingly. Vascular graft implantations are promising operations for by-pass patients. Vascular grafts of polytetrafluorethylene (ePTFE), polyethylene terephthalate (PET), and polyurethane (PU) work well in the replacement of large diameter (>6 mm), high-flow vessels. However, they fail for small diameters, mainly due to the incompatibility of their radial elasticity, insufficient porosity, and insufficient cell growth. Considering these requirements with the large number of patients in need of replacement grafts, the demand for an alternative small diameter graft is enormous and has driven scientists to search for new design ideas [1–3].
A native blood vessel is composed of three layers called the tunica intima, tunica media, and tunica adventitia. Each layer has its own cell type and protein content, which facilitate the unprecedented functions of native blood vessels. On the other hand, vascular graft design is accepted as successful to the degree that it mimics the target tissue, as in all tissue designs. Therefore, the design steps should be based on mimicking the native blood vessels' structural, mechanical, biological, and topographical properties [3].
In this review, after providing brief information about native blood vessels' histological and structural properties, the requirements for vascular grafts, the technology of electrospinning, and the advantages of electrospun vascular grafts are introduced. The design parameters of biodegradable electrospun vascular grafts, mainly such as the fiber diameter, pore size and porosity, fiber orientation, wall thickness, and material selection, are discussed in detail with recent literature works. It is intended that the blended and enriched context of this paper will help researchers to overcome the complex design problems of electrospun vascular grafts by providing them with new research areas.
Components of blood vessel
Both the pulmonary and systemic circulatory systems are made up of arteries, capillaries, and veins. Capillaries are the thinnest blood vessels with 5–10 µm diameters. These micro vessels are responsible for the connection of veins and arteries and they are also capable of exchanging water, oxygen, carbon dioxide, and other chemical substances between the blood and surrounding tissues. Without the need for extra transport mechanisms, the single layer endothelium capillary wall allows both gas and lipophilic molecules to pass through [4]. Veins are the vessels that carry the blood from the capillaries to the heart, while arteries carry the blood from the heart to the body parts. Due to the low pressure in the veins, the transport of blood depends on nearby muscular constructions. Back-flow of blood is prevented by the valves in the veins. Unlike the capillaries, the walls of veins and arteries have three layers: tunica intima, tunica media, and tunica adventitia (Figure 1) [4,5].
Layers of an artery.
The tunica intima is the layer that is in contact with blood. This layer serves as a thrombo-resistant barrier to enable laminar blood flow through the vessel. It controls vessel tone, platelet activation, adhesion and aggregation, and leukocyte adhesion [6,7]. It is composed of a single layer of endothelial cells (ECs) and sub-endothelial layers. The sub-endothelial layer is made up of collagen type IV and elastin [5]. An extra-cellular matrix (ECM) in the tunica intima also promotes diffusion, proliferation, and growth of ECs [8]. The tunica media includes many layers of smooth muscle cells (SMCs) in a matrix of collagen types I and III, elastin, and proteoglycans [5]. Collagen bundles in this layer give mechanical resistance to the vessel, while elastin enables an elastic ability. The contracting and relaxing behaviors of arteries and veins are determined by glycoproteins and proteoglycans [9]. The tunica adventitia serves as a protective sheet that consists of soft connective tissue mainly containing collagen type I mixed with elastin, nerves, and fibroblasts [5,6]. The protein structure of collagen and elastin assists the tensile performance and elasticity of the vessel while preventing it from vessel rupture and pulsatile deformation [5]. This layer consists of small vessels called vasa vasorum that nurture the arterial wall [10].
Requirements for vascular grafts
The main goal of an engineered tissue is to create living autologous neo tissues that can be used in place of damaged and diseased tissue and it should perform more like the native target tissue and less like a prosthetic material [11]. An ideal scaffold should reflect the biomechanical properties of native blood vessels, while serving as a topographically and structurally ideal backbone for cell attachment, proliferation, and diffusion [6].
Mechanical properties are essential for vascular grafts, because the grafts must withstand blood pressure and match arterial compliance to avoid flow disturbance around the anastomosis [3]. Compliance, which is one of the most important mechanical properties for basic blood vessel function, is a measure of radial elasticity. A compliance mismatch between the graft and the native vessels is one of the leading causes of graft failure in both autologous and vascular grafts [12,13]. It can cause inconsistent blood flow profiles and flow stagnation. Unstable flow through the graft can lead to stress concentrations at the suture locations, causing intimal hyperplasia (IH) [14,15], thrombosis, and even occlusion of the graft. Besides compliance performance, the graft should withstand in vivo stimuli including cyclic loading, shear stress, longitudinal stretch, circumferential stretch, and stress concentrations from sutures as well as internal pressures [2,14,16,17].
Spiral blood flow is documented as a normal flow pattern for medium and large sized vessels, mostly along the inner walls of curved segments and the outer walls of arterial bifurcations. This flow enables washing of the smoother inner surface of the vessel by the blood so that atherosclerotic plaques cannot accumulate on the surface [18–20]. In small diameter vascular grafts, the blood flow rate is low and not enough to prohibit thrombus formation. Therefore, the replication of spiral flow in small diameter vascular grafts is used to solve acute occlusion due to thrombus formation [20].
Topographical features have direct effects on cell adhesion, alignment, migration, cytoskeleton protein production, and differentiation. Nanotopography, rather than micro topography, should be ideal for vascular ECs since they naturally live in a nano environment. On the other hand, forming micro porous structures is another design parameter. Micro porous structure is necessary for cell migration and tissue ingrowth since the size of SMCs is larger than 5 µm [3]. In other words, the ideal structure of a vascular graft scaffold should have an extensive network of interconnecting pores so that cells can migrate, multiply, and attach deep within the scaffolds [21].
In order to be used in surgery, the vascular grafts should be nonthrombogenic or resistant to blood clotting, nonimmunogenic, noncorrosive at body temperature and pH, and resistant to infection. They should also be biocompatible and hemocompatible [2]. Long term biostability is required for biodegradable grafts. As the biodegradable polymer degrades hydrolytically over time, cells continuously infiltrate the matrix, producing collagen, elastin, and proteoglycans to replace the degrading material. Eventually, a fully functional artery is created composed of autologous SMCs and ECs. Therefore, a long degradation time, i.e. long term biostability, is one of the most crucial properties for biodegradable vascular grafts that provides a prolonged mechanical backbone for cells to infiltrate and a low inflammatory response [7,22]. Furthermore, the vascular grafts should have low manufacturing costs, be able to withstand some type of sterilization process and be available in different specifications (diameter, length, etc.) to fit the need for the implant. Other issues to be considered might be availability in urgent cases and ease of suturing [2,16,17,21,23].
Electrospun vascular grafts
Electrospinning has shown extreme potential in the last few decades, which has promoted its usage in tissue engineering applications owing to the great advantages of electrospun fibrous surfaces. In this context, the area of vascular grafts comes to light which enables the use of biopolymers and natural proteins to design biodegradable scaffolds via electrospinnning technology [3].
Working principle of electrospinning
Electrospinning apparatus is mainly composed of three components: a high voltage supplier, a capillary tube with a tip and a collector [24]. Here, the capillary tube can have either one tip or multiple tips. On the other hand, free surfaces can also be used as a feeding unit especially for mass production [25]. The collector can be chosen as a plate shape or rotating mandrels in varying diameters based on the end use [26,27]. Production can be carried out in the vertical or horizontal directions according to the design of the electrospinning establishment [26,28].
In this process, a polymer solution or melt is held by its surface tension at the tip of a capillary tube which is subjected to an electric field. Charge is induced on the solution/melt surface by the electric field. As the intensity of the electric field is increased, the hemispherical surface of the solution/melt at the tip of the capillary tube deforms to form a conical shape, which is named a Taylor cone. When the voltage increases and the electric field reaches a critical value at which the repulsive electrical forces overcome the surface tension forces, a charged jet of the solution/melt is ejected from the tip of the Taylor cone towards the collector. Since this jet is charged, its trajectory can be controlled by an electric field [24,29]. A straight section of the jet which comes out of the Taylor cone turns sideways and forms loops in the horizontal plane under the effect of bending instabilities [28]. The loop diameter increases with time during the motion towards the counter electrode, which is called a whipping action. During this process, the jet is highly stretched and reduced. This procedure is repeated until the solvent evaporates or the melt solidifies, leaving behind a discharged interconnected polymer fiber accumulated on the collector [24]. As a result of these instabilities, fibers with diameters down to a few nanometers can be generated [28].
Since electrospinning is based on a simple working principle, producing fibers with desired properties is more complex. Thus, it is necessary to optimize the solution parameters (viscosity, concentration, conductivity, surface tension, polarity), polymer properties (molecular weight, solubility, glass transition temperature), ambient parameters (temperature, relative humidity, vapor pressure), and production parameters (voltage, feeding rate, distance between needle tip and collector) to improve the spinnability and thus the morphology of the fibers [28,30].
Advantages of electrospun vascular grafts
Electrospun scaffolds are promising candidates for tissue engineered scaffolds, i.e. vascular grafts providing a wide range of advantages [2].
Mimicking ECM is one of the advantages of electrospun vascular grafts owing to the nanotopography created by the nanofibers and interconnected pores, which results in a large surface area that promotes endothelium formation to prohibit arterial thrombosis. It is known that attachment and proliferation of ECs is better around fibers with diameters smaller than the cell diameter (<5 µm) [3]. On the other hand, the controllable pore size and porosity of electrospun scaffolds allow SMCs to grow, migrate, and regenerate through the scaffold via creating large spaces (>5 µm to allow cell diffusion).
Beside designing spiral flow guiders [31,32] for small diameter vascular grafts to replicate spiral flow and avoid thrombus formation, the surfaces of electrospun vascular grafts can be modified with protein lining, heparin incorporation, or polymer surface chemistry, which reduces thrombosis and IH while promoting the endothelium [3].
Moreover, electrospinning technology allows the design of the desired scaffolds either separately or in combinations of one scaffold with multilayers. Therefore, by designing composite scaffolds based on the variable material selection, flexibility in modification of the production technology and adjustment of the production parameters, it is possible to tune the mechanical compliance, material degradation, and surface properties according to the needs of tissue regeneration [3,12].
Design parameters for electrospun vascular grafts
When producing electrospun vascular grafts, the design parameters must be well understood in order to mimic the native blood vessels. In the last decade, there have been many studies about electrospun vascular grafts which focus on these design parameters. Although it is necessary to handle all the design parameters as a whole, it is essential to discuss them separately to understand the current situation of the literature and overcome any deficiencies.
Fiber diameter, pore size, and porosity
The pore size is one the most important parameters for vascular grafts. The pores must allow the infiltration of SMCs while hindering blood leakage [33,34]. Since nanofibrous surfaces serve as a useful means for cell attachment with high porosity and small pore sizes, they pose an obstacle for SMC diffusion. To overcome this challenge, several techniques were used to achieve dual porosity in either multilayer [33–36] or single-layer [37] vascular grafts.
One of the best known ways of controlling the pore size of the electrospun scaffold is to change the fiber diameter. In a study by Yalcin Enis et al. [38] the effect of additional acids (acetic acid and formic acid) on the most used polycaprolactone (PCL) (45,000 Mn) /chloroform:ethanol solution was investigated based on fiber diameter and pore size analysis. According to the results, 2.22 µm diameter micro fibers were produced from 18% PCL /chloroform:ethanol solution. With the addition of pre-determined amounts of acetic acid and formic acid to the same solution, fiber diameters were reduced to 158 nm and 256 nm for each acid type. Moreover, the average pore area was also reduced almost 50 times (from 10 µm2 to 0.2 µm2) based on fiber diameter decrement, i.e. additions of acidic solvent [38].
In three complementary studies by Bagherzadeh et al. [39,41], PCL (80,000 Mn) was dissolved in chloroform:methanol (3:1), solvent system at 10–20% polymer concentrations [39,40], and in chloroform:DMF (4:1) solvent system at 10–14% polymer concentrations [41] to examine the pore size and porosity of the scaffolds with several experimental and developed theoretical techniques. Beside a good agreement that was obtained from two experimental techniques, namely the capillary flow porometry and confocal laser scanning microscopy (CLSM) methods [39,40], a theoretical method developed by the authors was validated by these experimental techniques and compared with accepted models to determine the average pore size and pore size distribution of multilayer nanofibrous scaffolds for tissue engineering applications [41]. It was also confirmed by Bagherzadeh et al. that with an increasing concentration of polymer solution, the fiber diameter as well as the pore size, porosity, and pore interconnectivity increase significantly. Similar to the outcomes of Yalcin Enis et al. [38], these results also showed that the fiber diameter of fibrous scaffolds is a crucial structural parameter that can determine the pore structure of fibrous scaffolds [39–41].
In parallel with these outcomes, Ju et al. [33] produced an electrospun bilayer vascular graft from PCL/collagen mixture composed of different pore sizes. In order to vary the pore size, the fiber diameter was altered by changing the solution and production parameters. Both SMCs and ECs were seeded to the scaffolds in order to evaluate the cellular interactions based on fiber diameters, i.e. pore sizes. The produced bilayer vascular graft was composed of a nanofibrous inner layer (0.27 nm fiber diameter) and microfibrous outer layer (4.45 µm fiber diameter). As the outcome of the study, a nanofibrous layer with nano pore sizes served as an improved surface for EC attachment. On the other hand, the 1 µm fiber diameter was determined as a sub limit in the outer layer for SMCs infiltration [33]. Similarly, de Valence et al. [34] produced electrospun bilayer grafts with a fibrous structure of low porosity in either the inner or outer layer. It was stated that the inner layer with lower porosity reduced blood leakage while promoting cell penetration from the outer layer (through higher porosity). On the other hand, cell invasion and neovascularization were significantly reduced in grafts composed of lower porosity structure in the outer layer (Figure 2) [34].
Vascular graft structures. Two scaffolds with different porosities were electrospun sequentially to form bilayered vascular grafts. The inside-barrier graft (a) has a high-porosity layer on the adventitial side (∗, c) and a low-porosity layer on the luminal side (#, e). The outside-barrier graft (b) was constructed inversely. The transition between the two layers was continuous, with no delamination (d). Reprinted from De Valence et al. [34] with permission from Elsevier.
In a study by Wang et al. [35], 25% and 10% PCL (80,000 Mn) were dissolved in chloroform: methanol (5:1 v:v) to produce nano and micro fibers with dimensions 0.69 µm and 5.59 µm. The average pore size and average porosity of these fibrous layers were measured as 4.66 µm/65% and 40.88 µm/85% respectively. It is well understood that the thicker fibers lead to a larger pore size and higher porosities. And it is stated that larger pores allow enhanced cell invasion and vascularization in both in vivo and in vitro conditions [35]. It is also supported by Brauker et al. [36] that neovascularization is promoted by allowing cell penetration even with the pore size of the graft ranges between 0.8 µm and 8 µm. Implantation of grafts with 5 µm pore size under the skin of rats led to almost 100 times more vascular structures compared to 0.02 µm pore sized structures [36].
For the same purpose but from a different point of view, Lee et al. [37] produced three-dimensional montmorillonite-reinforced poly-L-lactic acid (PLLA) scaffolds. Nanofibrous scaffolds were produced by the electrospinning technique and then micro sized pores were achieved in nanofibrous webs by a salt leaching/gas forming technique. For the transportation of metabolic nutrient and wastes nanosized pores (100–200 nm) and for facile cell growth and blood vessel invasion micro sized (50–300 µm) pores were developed in a single layer graft. As a conclusion, dual porosity was found to be essential to meet the demands of vascular grafts [37].
Therefore, all the reported studies [33–37] placed excessive emphasis on micro sized pores especially for SMCs' invasion. Since it is not stated that a nano scaled pore size is a must for endothelialization, its advantages in hindering blood leakage [34] and allowing a fluent monolayer of ECs on lumina [42] make their use worthy of note when designing a multilayer graft.
Fiber orientation
Fiber orientation is another important parameter when designing a vascular graft. Especially the radial fiber orientation achieved in tubular scaffolds is required for enhancing the diffusion of SMCs and improving the mechanical strength against radial pulsatile pressure. There are several studies about methods such as increasing the rotational speed of the collector [43–47] or using metal plates [48], which focus on how to improve the radial orientation for tubular scaffolds.
Matsuda et al. [43] produced a tubular electrospun scaffold for small diameter vascular grafts from PU polymer. In order to obtain the fiber orientation, 150 r/min and 3400 r/min rotational speeds (1.41 m/min and 32.02 m/min peripheral speeds) were used for a 3 mm rotating mandrel. At 3400 r/min, partial fiber orientation was achieved. The increased fiber orientation in the radial manner improved the circumferential tensile strength while decreasing the longitudinal tensile strength. However, lower compliance was achieved with the improved radial fiber orientation [43]. On the other hand, a small diameter PCL vascular graft was fabricated by Hu et al. [44] To investigate the fiber orientation and drum speed relation in detail, five peripheral speeds (240 m/min, 320.4 m/min, 400.2 m/min, 480 m/min, 640.8 m/min) were used. For their use in small diameter vascular grafts, membranes produced at 80 m/min, 320.4 m/min, and 480 m/min peripheral speeds were wrapped on a small diameter rod with varied angles. The final grafts were prepared from randomly oriented, moderately and highly aligned fibrous membranes. The results demonstrated that the alignment index (AI), i.e. fiber orientation, increased with the increased peripheral speeds (from 240 m/min to 640 m/min). The mechanical behavior of the scaffolds was found to be under the influence of fiber orientation. It was also stated that fibroblasts were aligned with the oriented fibers, indicating the potential to control the microstructure of electrospun scaffolds (Figure 3) [44].
Representative optical microscopy images of electrospun PCL fibers collected at different drum rotation speeds (a), the corresponding distributions of fiber orientations and the alignment indices (peripheral speeds from left to right: 240 m/min, 320.4 m/min, 400.2 m/min, 480 m/min, 640.8 m/min) (b), and the corresponding SEM images (c). Reproduced from Hu et al. [44] with permission from Elsevier.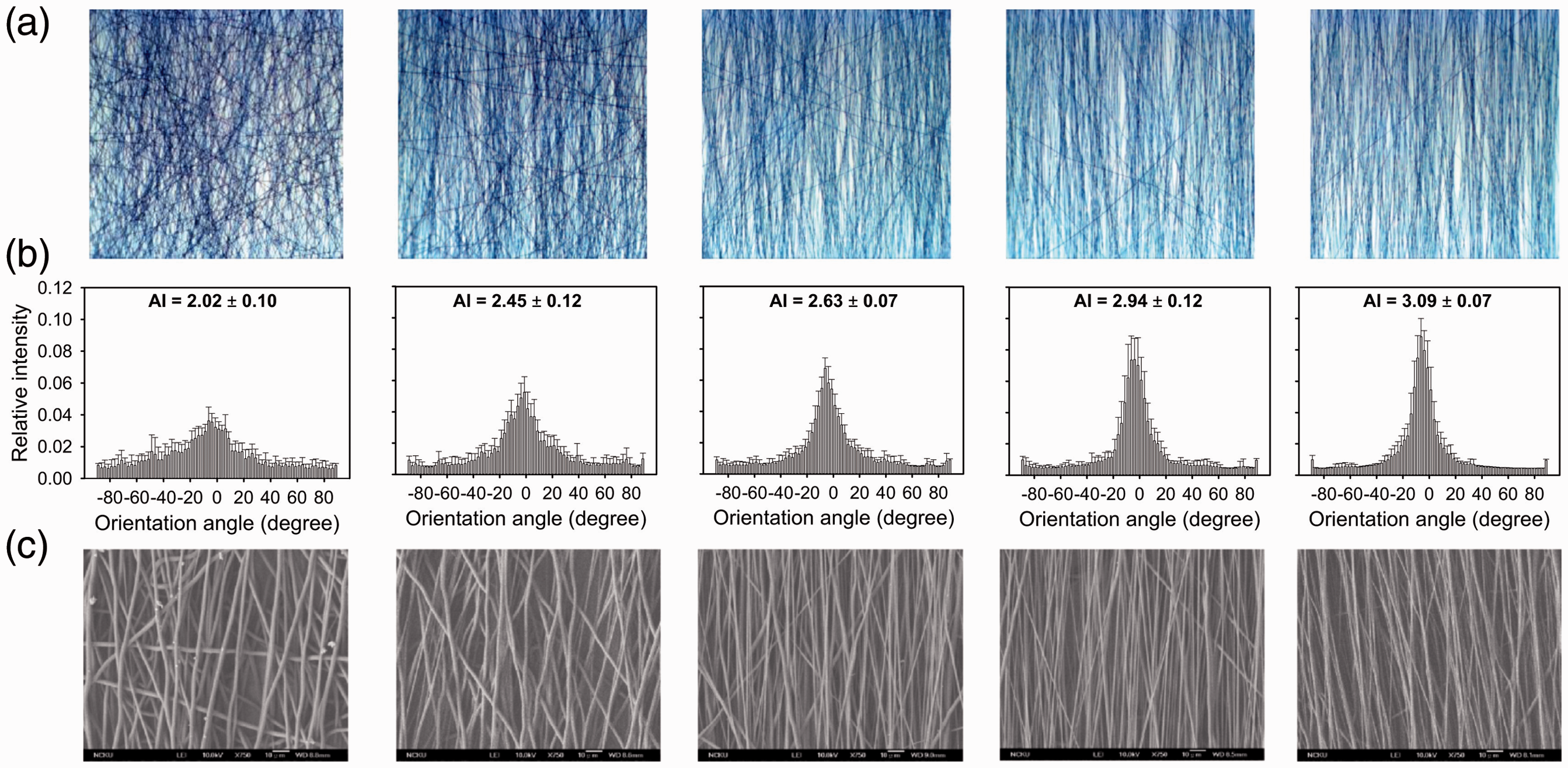
By using a rotating mandrel (32 mm in diameter) type collector, a similar study was realized by Edwards et al. [45] . In this study, the peripheral velocity of the collector varied from 0 m/min to 480 m/min to investigate PCL fiber orientations. It was reported that there is no fiber orientation up to 120 m/min peripheral velocity since the fibers are not exposed to mechanical stress. Above this limit, there is a reduction in fiber diameters that is explained by the existence of mechanical stress. The optimum fiber orientation was achieved at 150 m/min, while it rose to a maximum level at 300 m/min. Above 300 m/min, mechanical deformation was observed on the fibers because of the high mechanical stress and affected the fiber orientation in a negative way [45]. In a study by Chaparro et al. [46], randomly distributed PCL fibers were spun onto a 53 mm diameter steel rod rotated at 9000 r/min (net peripheral speed of 84.6 m/min), while aligned PCL fibers were spun onto a rotating mandrel having a 2.01 m circumference (diameter: 640 mm), rotated at 500 r/min (providing a peripheral speed of 2000 m/min) [46].
The above studies show that, although high rotational speeds improve the fiber orientation, it is not easy to reach such high speeds (>10,000 r/min) by using small diameter (<6 mm) rotating mandrels. Therefore, most of the studies use large diameter collectors (630 mm [42], 102 mm [44], 32 mm [45], and 640 mm [46]) for accessible rotational speeds. Matsuda et al. [43] reported that they achieved partial fiber orientation at 32.02 m/min for a 3 mm rotating mandrel diameter [43] which had been explained by Edwards et al. as having an insufficient peripheral velocity [45]. Moreover, the type of polymer should be taken into consideration when optimizing the speed for fiber orientation.
To overcome the speed limitations, Yalcin et al. [47] produced a PCL (45,000 Mn) vascular graft by a custom-designed electrospinning unit modified with a rotating mandrel powered by a high-speed motor. Radial fiber orientation was achieved by varying the rotational speed (250–5000–10,000–15,000 r/min) of a 6 mm diameter rotating mandrel. Fiber orientation was achieved at 5000 r/min (94.2 m/min) and 10,000 r/min (188 m/min). The prevailing fiber orientation in the radial direction was improved with the increasing rotational speed up to 15,000 r/min (282.6 m/min) [47]. In order to obtain radial fiber orientation, peripheral velocities were found in the range of previously confirmed values (150 m/min to 300 m/min by Edwards et al. [45]). No fiber orientation is observed at 250 r/min (4.71 m/min) [47] since the fibers are not exposed to a notable extension force at less than 120 m/min speed [45]. On the other hand, elongated cell shapes were observed, which suggests that cells tend to orient themselves with the fibers in the radial direction. The increase in the nuclei elongation ratios, which were calculated as 1.22, 1.75, 1.87, 1.94 for 250, 5000, 10,000, and 15,000 r/min, confirmed the cell orientation along the fibers (Figure 4) [47].
Fluorescence microscopy pictures of 3T3 mouse fibroblasts nuclei stained with propidium iodide after 1 day of culturing anisotropic structure (4.71 m/min) (a), and isotropic structure (188 m/min) (b). Scale bars 25 µm. Reprinted from Yalcin et al. [47].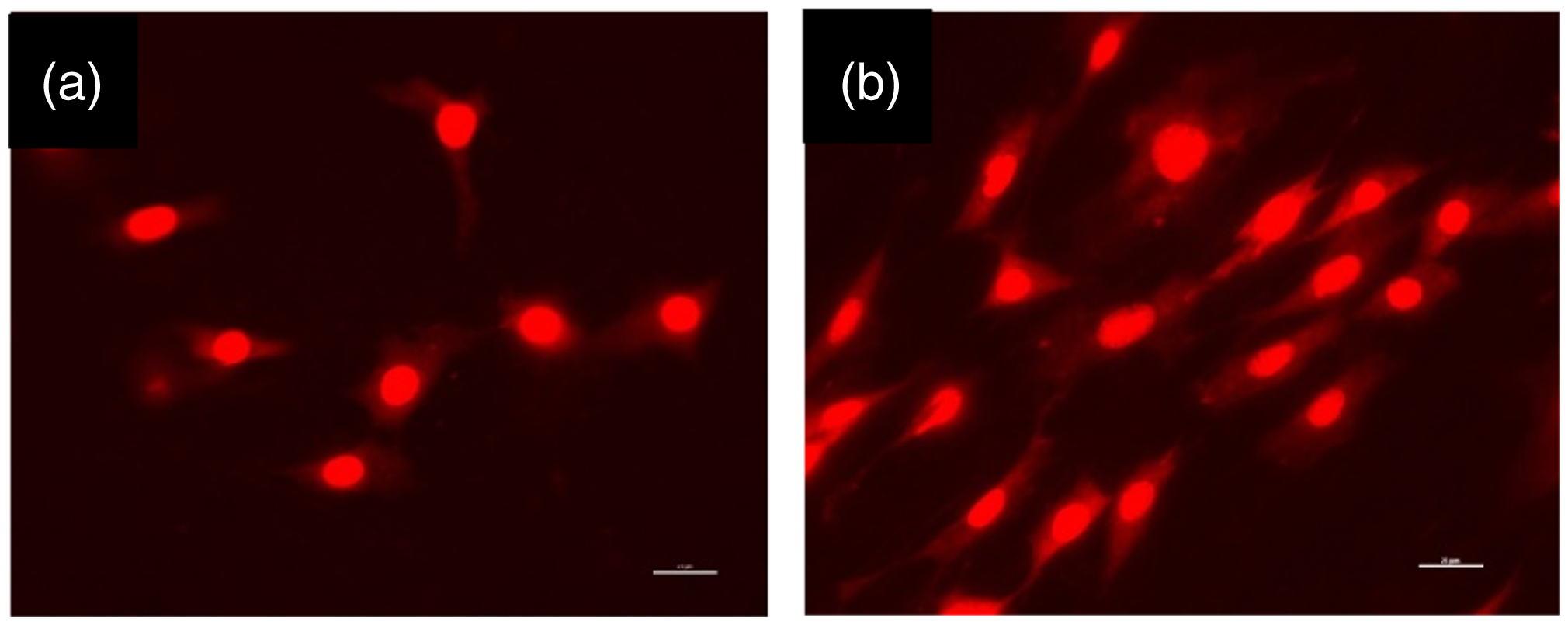
Modification of the electrospinning unit with metal plates to improve the fiber orientations of electrospun small diameter vascular grafts was studied by Wu et al. [48]. A 4.5 mm rotating mandrel was chosen for small diameter vascular graft production while 50 r/min and 300 r/min rotational speeds (0.7 m/min and 4.2 m/min peripheral speeds) were used for the longitudinal and radial fiber orientations, respectively. For both orientations the electrospinning unit was supported by directionally positioned metal plates. The results demonstrated that by using metal plates it is possible to achieve fiber orientations in both directions. Although cell attachment was observed in both oriented and non-oriented PCL scaffolds, it was noted that the oriented fibers facilitated and regulated the cell attachment. Directional mechanical properties were achieved by using oriented fibers. This study concluded that designing a multilayer vascular graft consisting of both longitudinal and radial oriented fibers was found to be ideal for improved mechanical performance and cell response [48]. As discussed in Hu et al. [44] and Edwards et al. [45], 300 r/min is insufficient for a 4.5 mm collector diameter (4.2 m/min peripheral speeds) to develop the fiber orientation radially. However, Wu et al. stated that with the modification of the electrospinning with directionally positioned metal plates, excellent radial fiber orientations can be obtained [48].
Wall thickness
Beside the mentioned morphological parameters such as fiber diameter, pore size, and fiber orientations, the wall thickness of the scaffold as a dimensional property is also deterministic on the mechanical and biological properties of the vascular grafts. If the other production and solution parameters are kept constant, the production time is directly affected by the wall thickness [47]. It is possible to obtain a homogeneous wall thickness during electrospinning by the reciprocal movement of either the feeding unit [47] or the collector mechanism [49]. On the other hand, the ease of removing the polymeric scaffold in tubular form from the mandrel after production enables the graft's inner diameter to be fixed as the same as the collector diameter.
Yalcin et al. [47] carried out a histological analysis of native artery when designing an electrospun vascular graft. The wall thickness of the analyzed native artery was measured in a range of 400–1000 µm along its length. Moreover, detailed information about the structural properties of the native blood vessel was given in the study. Microscopic images reveal that ECs are arranged through a single layer covering the luminal surface, whereas SMCs are organized into numerous layers inside the tunica media. Elastin, which gives the vessel elasticity, was found densely in the lamina elastica interna, as well as the tunica media and the tunica adventitia. The experimental results were also proven by the literature [22,50–54]. It was also observed that fibrous collagen bundles oriented in a radial manner appear in micrometer scales (Figure 5) [47].
Hematoxylin eosin staining (a), collagen (b), and elastin (c) in the tunica intima (TI), tunica media (TM), and tunica adventitia (TA). Scale bars: 100 µm. Collagen nanofibers arranged in microfibrous bundles are depicted in SEM picture intercalated in (b), scale bar: 10 µm. Black arrows in picture (c) indicate vasa vasorum in the tunica adventitia. Reprinted from Yalcin et al. [47].
In a study by Inoguchi et al. electrospun poly (L-lactid-co-ε-caprolactone) PLCL vascular grafts with a thinner wall thickness demonstrated improved compliance properties [55]. On the other hand, the outcomes of Johnson et al.'s [56] study indicate that the compliance of PCL tubes remains steady within a range of 2–4% / mmHg when the wall thicknesses range between 400 µm and 1000 µm. However, the thinner PCL grafts exhibit a large variance in compliance measurements. Moreover, when the wall thickness increases from 200 to 1000 µm, the burst pressure values increase in a linear fashion from 0.6 MPa to 2.9 MPa [56]. In the study by Thijssen et al. [57], the impact of the wall thickness on vascular responsiveness was investigated. The conduit diameter, wall thickness and wall to lumen ratio (W: L) of the arteries taken from the lower and upper limbs of healthy men were examined. The results indicated the regional heterogeneity is present between arteries. Especially small diameter arteries had higher W:L ratios than large diameter arteries. It was concluded that this regional heterogeneity can influence the vasoactive stimuli [57]. Variation of the W:L ratio within a single artery was also demonstrated by Bjarnegard and Länne [58].
Material selection
Since electrospinning enables a wide range of polymer selection for production, electrospun vascular grafts can be produced from non-biodegradable synthetic polymers (PU [35], PET [59]), biodegradable synthetic polymers (PCL [47,53,60], polylactic acid (PLA) [53], poly(glycerol sebacate) (PGS) [61,62], poly glycolic acid (PGA) [63]), structural proteins (collagen [64,65], elastin [66], fibrin [67], silk fibroin [68,69]), and their mixtures [60,70,71] or copolymers [22,72,73].
Compared to natural polymers, synthetic biodegradable polymers generally demonstrate better mechanical properties with high reproducibility. PCL, PLA, PGS, and PGA are the polymers approved by the Food and Drug Administration (FDA) for human clinical use [17,21,74].
PGA is a semicrystalline, thermoplastic aliphatic polyester synthesized by the ring-opening polymerization of glycolide, which degrades completely in vivo within six months [17]. PLA is a thermoplastic aliphatic polyester synthesized by ring-opening polymerization of lactic acid which exists in two isoforms: PLLA and poly-D-lactic acid (PDLA) [74,75]. The racemic mixture is termed poly-L,D-lactic acid (PDLLA) or PLA. PLLA is semicrystalline (∼37%) while PDLLA is amorphous [74]. The degradation rate for PLLA fibrous scaffold is 42 days, whereas the PLA scaffold is shrunk significantly after only three days [75]. PLA shows good biocompatibility and mechanical properties [74]. PLA in scaffolds is mostly in copolymers because of its low degradation rate [75]. PCL is a semicrystalline, aliphatic polyester synthesized by the ring-opening polymerization of ε-caprolactone. PCL has improved elasticity that closely matches native tissue. PCL has good biocompatibility and its degradation rate is very slow in vivo (within almost 1 year) [17]. PGS is an elastomer synthesized by polycondensation of glycerol and sebacic acid. It has good biocompatibility, with elastomeric and tough behavior. PGS degrades in vivo by hydrolysis in two months [17,74]. To match the native tissue properties, many researchers focus on improving the inadequate mechanical properties and enhancing the biocompatibility of the scaffolds by either blending these polymers or using their copolymers.
Lee et al. [60] produced small diameter vascular grafts by electrospinning from collagen/elastin alone and a mixture of 45% collagen, 15% elastin, and 40% biodegradable synthetic polymers (PLLA, PLGA, PCL and PLCL), which was stated as the real ratio of native blood vessels. According to the results, although the collagen/elastin scaffolds degraded in a few days after production, PCL was found to be the best for 28 day patency levels. On the other hand, the lowest mechanical strength was obtained for scaffolds of PCL mixtures. SMCs were seeded on the scaffolds and in vitro test results indicated that all the scaffolds are biocompatible and promote cell response (Figure 6) [60].
Dimensional stability of the electrospun scaffolds. (a) Gross observation and (b) percentage of remaining diameters of the electrospun nanofiber scaffolds in culture condition. The scaffolds were immersed in culture medium at 37 ℃ for 56 days. *The electrospun collagen/elastin scaffold without synthetic polymer was collapsed after 4 days incubation. Reprinted from Lee et al. [60] with permission from Wiley Periodicals, Inc.
Wise et al. [70] produced PCL/elastin vascular grafts by an electrospinning unit. The increase in elastin content improved the cell growth. On the other hand, the increase in the PCL ratio resulted in better mechanical performance [70]. Lee et al. [71] fabricated a vascular graft composed of PCL and collagen by electrospinning. A 520 ± 14 nm fiber diameter and 22.7 ± 9.6 µm2 pore area were achieved from PCL/collagen (1:1) composite scaffold. PCL vascular grafts were produced as control scaffolds. The burst pressure, compliance, and yield strength of the PCL/collagen (4915 ± 155 mmHg, 5.6 ± 1.6% and 4.0 ± 0.4 MPa respectively) were improved as compared to PCL scaffolds (914 ± 130 mmHg, 3.5 ± 1.4% and 1.5 ± 0.7 MPa respectively). Although there was a decrease in the suture retention strength of the PCL/collagen scaffold, the results were found to be above the sub-limit, which is generally accepted as 2 N. Both ECs and SMCs had good adhesion to the PCL/collagen scaffolds [71].
In a study by He et al. [72], a vascular graft composed of P(LLA-CL70:30) copolymer was fabricated. Collagen coating was done to the surface of the scaffold. The tensile strengths of the scaffolds were found to be 7.0 MPa and 3.9 MPa in the longitudinal (LD) and circumferential directions (CD). The tensile strength of commercial Teflon grafts is stated as 85.2 MPa in LD and 66.7 MPa in CD, while the tensile strength of the abdominal aorta is stated as 1.47 MPa in LD and 5.29 MPa in CD. The in vitro biodegradation results indicate that the electrospun P(LLA-CL) scaffolds did not lose their structural integrity until eight months later. After eight months, most of the nanofibers broke into small segments of fibers. When the scaffold was implanted in a rabbit, seven weeks patency was observed (Figure 7) [72]. Similarly, Dong et al. [53] produced a vascular graft from P(LLA-CL 70:30). Mainly, the long term cell viability and degradation of the scaffold were studied. According to the results, the molecular weight of the P(LLA-CL) nanofibers (average fiber diameter 580 nm) decreased sharply after 60 days. The estimation realized in the study showed that after 230 days, P(LLA-CL) nanofibers will completely degrade [53].
SEM images showing morphology of the P(LLA-CL) nanofiber meshes biodegraded in 1 × PBS at 37 ℃ for (a and b) 0 month, (c and d) 3 months, and (e and f) 8 months. (b, d, and f) are magnified images of (a, c, and e), respectively. Reprinted from He et al. [72] with permission from Wiley Periodicals, Inc.
Kim et al. [73] produced a small diameter vascular graft from PLGA copolymer and modified it with stem cells. Sufficient mechanical properties together with better cell growth were achieved. However, shortly after implanting the graft into a dog, arterial occlusion was observed. The results revealed the difficulty of using PLGA biodegradable polymer in vascular grafts [73]. Del Gaudio et al. [76] fabricated electrospun PCL vascular grafts using both DMF:THF (1:1) and chloroform solvents. The resultant fiber diameters were 0.8 ± 0.2 µm and 3.6 ± 0.8 µm, respectively. Human umbilical vein ECs were seeded to the scaffolds. Although sufficient cell adhesion was achieved in both nano and micro scaled fibers, the viability of the cells was better in the micro scaled fibers, with improved cell–fiber interactions [76]. Pfeiffer et al. [42] produced three-layer grafts from different polymer blends. In their first design, the inner and outer layers were produced from a PCL/PLA blend with 1.61–1.43 µm fiber diameter and 5.8–5.3 µm pore size. In the second design, the inner and outer layers were produced from PCL/PLA/polyethylene glycol (PEG) blend with 1.76–1.63 µm fiber diameter and 5.8–6.7 µm pore size. Between the two layers, PCL was used as a barrier layer. Endothelialization was observed in a comparison of ePTFE grafts. The morphological results indicate that only a few cells with a round shape were seen on the native ePTFE grafts, whereas some clusters with partly spreading cells could be observed on the PCL/PLA scaffolds. The best results were obtained on PCL/PLA/PEG grafts where the ECs showed good adherence and many cell-to-cell contacts (Figure 8) [42].
Live/dead analysis of different endothelialized vascular grafts after fibronectin coating: Cells were stained with calcein AM (green fluorescence) and Annexin V (red fluorescence) to distinguish live (green) from dead cells (red). Numerous dead cells were observed on ePTFE (a) grafts. In contrast, PCL/PLA (b) and PCL/PLA/PEG (c) grafts only sporadically showed apoptotic cells. Scale bar 200 µm. Viability counting of endothelial cells revealed lower attachment and viability of endothelial cells on fibronectin-coated ePTFE compared to PCL/PLA and PCL/PLA/PEG grafts. Reprinted from Pfeiffer et al. [42] with permission from Wiley Periodicals, Inc.
Wu et al. [62] designed cell-free biodegradable elastomeric grafts that degrade rapidly to yield neo-arteries that are nearly free of foreign materials three months after interposition grafting in rat abdominal aorta. The bilayer vascular graft is composed of two polymers; a PGS inner and PCL outer layer. After three months of implantation, the neo-arteries composed of PGS and PCL allowed ECs and SMCs substitution in vivo respectively, as the polymeric scaffold degraded. It is also stated that elastin, collagen, and glycosaminoglycan are synthesized and that tough and compliant mechanical properties were achieved [62].
In a study by Tillman et al. [77], the biomechanical strength of PCL (inherent viscosity: 1.77 dL/g):collagen type I derived from calf skin (1:1) tubular scaffolds fabricated by electrospinning was investigated. The in vivo mechanical characterization of the scaffolds indicated that the tensile strength of the grafts measured before implantation was measured at around 1.7 MPa and decreased to 0.9 MPa at one month post-implantation. The resultant tensile strength after implantation was found to be still comparable to native rabbit aorta, which demonstrates that these scaffolds maintain an adequate degree of biomechanical strength in in vivo conditions [77]. Similarly, the pre-implant and post-implant mechanical performances of bilayer grafts composed of synthetic elastin inner layer and PCL (Mn = 70,000–90,000) outer layer were compared with each other by Wise et al. [78]. The results indicated that the outer diameters of the pre-implant and post-implant grafts were unchanged. Improvements of compliance were found in the post-implant grafts, while the burst pressure decreased slightly based on the expected degradation of PCL. All the results were comparable with the control samples, which demonstrates the mechanical integrity of bilayer grafts in in vivo conditions [78].
Based on these studies, it is claimed that the long-term patency levels of PCL were found to be better than PLGA, PLLA, and PLCL biodegradable scaffolds, while PCL has the lowest mechanical strength [60], and the compliance of PCL vascular grafts is higher than commercial artificial blood vessels [62]. When PCL was used together with structural proteins it improved the mechanical strength [70], and the use of different solvents resulted in both nano and micro scaled fibers [38,76], whereas better cell growth was achieved in micro scaled fibers [76]. PCL:collagen [77] and SE:PCL [78] vascular grafts demonstrated comparable biomechanical performance in in vivo conditions. On the other hand, PLA, PLLA, and PLGA polymers with improved mechanical strength are mostly preferred in vascular graft designs [60]. However, these polymers show fast biodegradation after implantation, thus they show poor long-term patency levels [73], which can therefore cause crucial handicaps when these polymers are used alone in the structure.
Conclusion
Electrospinning, providing a wide range of polymer selection and allowing various scaffolds in differing shapes and structures, is a promising technique for the production of fibrous vascular grafts. Biodegradable electrospun fibrous scaffolds have many advantages when mimicking the native vessel based on the resemblance to ECM, while behaving like a backbone for the cells during their proliferation and synthesis of their own proteins.
It can be considered from the above discussed studies that researchers tend to focus on electrospun multilayer scaffolds for vascular grafts by either varying the materials or using different pore sizes and fiber orientations in the layers. Thus, the histology of the native blood vessel has a crucial significance when determining these design parameters. To draw general boundaries for future attempts at vascular graft design, it can be said that the tunica intima should promote endothelialization to achieve nonthrombogenicity. The pore size of this layer should be limited to blood cell dimensions in order to hinder blood leakage. Nanofibrous layers with nano pores can be preferred for this layer, which can also be modified with heparin bonding to prohibit platelet activation. Against the tunica intima, the tunica media should have larger pore sizes than SMCs in order to provide a porous layer for their diffusion through the wall thickness. Therefore, the layer with micro fibers can be electrospun, including larger pores that enable the diffusion of SMC through the tunica media. Besides adjusting the fiber diameter to control the pore size, there are other techniques for electrospun fibrous scaffolds, such as the gas forming/salt leaching technique that creates larger pores to allow cell diffusion. Since the tunica media is the layer responsible for the mechanical performance of the vessel, improved fiber orientation is required in this layer for sufficient tensile properties in order to withstand the blood pressure after implantation. By adjusting the rotational speeds of the rotating mandrel in the light of the sub-limits determined in the literature, extreme radial fiber orientation can be achieved. Moreover, positioned metal plates can be used to modify the collector system of the electrospinning technology, which improves the radial fiber orientation. However, the reduced compliance properties with improved radial fiber orientation should also be taken into consideration. In this respect, the wall thickness and the selection of material also become important parameters to obtain adequate tensile strength with improved radial elasticity to overcome compliance mismatch of the scaffold with the sewn vessel. Moreover, the degradation rate of the polymer should be studied for multilayer biodegradable fibrous scaffolds in order to determine the minimum time that is required for biodegradable scaffolds to allow cells to synthesize their own proteins before the biological degradation of the scaffold. Therefore, the final objectives of designing a vascular graft should be the degradation of the scaffold after a sufficient time of implantation by leaving its place to an autologous vessel which is similar to native vessel.
This review compiles knowledge of the electrospinning technology with its flexibility in modifications to create ideal scaffolds for vascular grafts by exploiting the advantages of electrospun biodegradable scaffolds as “autologous vessels”. The aim is that the blended and enriched context of this review will enable researchers to overcome the complex design problems of electrospun vascular grafts by encouraging them to explore new research areas.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
