Abstract
In this paper, we investigated a functionalization process of nonwoven viscose/polyester dressings, in order to fix a wide range of drugs, using a polymer based on β-cyclodextrins. Firstly, the optimization of the treatment process was investigated. The best operating conditions were generated using Minitab 15, in order to maximize functionalization rates. Then, in order to confirm the permanent and uniform binding of cyclodextrin’s polymer, modified and virgin dressings were characterized by Fourier transformed infrared spectroscopy, thermogravimetric analysis, and the drop contact angle method used to assess the substrates hydrophilicity. The microbiological value of this chemical modification was highlighted via the application of methylene blue, used in this case as a drug model. The encapsulation properties of cyclodextrins molecules allowed its fixation into the functionalized wound dressings. The assessment of drug release kinetics confirmed a sustained release in water. Furthermore, bacteriological properties were investigated by staining and growth inhibition. Antibacterial activity of nonwoven methylene blue cyclodextrin-functionalized samples against Staphylococcus aureus and Escherichia coli before and after release are presented.
Keywords
Introduction
Wound dressings are the medical means of cleaning and protecting wounds in order to facilitate and accelerate the healing process. Developing wound dressing products have witnessed significant improvements, but their properties are still far from challenging the characteristics that chronic wounds present. Wound dressing should have some requirements including short healing time, breathability to permit gases and water vapor exchange, antibacterial properties, nontoxicity, etc. [1].
Viscose is commonly used in wound dressings manufacturing, because of its high absorbency, breathability, comfort, and softness [2,3]. Therefore, blending of cellulosic and polyester fibers, which are strong, wrinkle dirt, and microbial attacks resistant [4,5], can economically extend the products performance, to overcome the increasing consumer’s needs for innovative and multifunctional materials [6–8]. This increasing demand led to the development of textile finishing research in order to produce materials with specific characteristics that promote the health aspects while providing soft and gentle functional properties [9–14]. A wide variety of promising textiles-based drug delivery technologies have been developed, by textile finishing including microencapsulation, active softener, and the host–guest interaction of cyclodextrins (CDs). In the last method, CDs are used because they are biocompatible [15], biodegradable [16,17], and accepted in biological applications.
Cyclodextrins are torus-shaped starch derivatives that have the ability to encapsulate a wide range of organic molecules. CDs and their derivatives have been used in the textile domain since the early 1980s [18]. CD finishing offers textiles with new functionalities such as antibacterial agent, auxiliaries in washing and dyeing processes, hydrophilicity, UV protection, slow release fragrances or skin-care components, and stabilization of active ingredients [19–21].
Numerous methods were previously proposed for the permanent finishing of natural and synthetic fibers with CDs. Martel et al. [22,23] reported the CDs grafting method on natural fibers (cotton and wool) via polycarboxylic acids (PCA). In other recent studies, the possibility of CDs finishing synthetic fibers (such as polypropylene and polyester) using PCA, was also highlighted [24–26].
The aim of this work is to make possible the attachment of a wide range of drugs, and to improve the hydrophilic and release properties of a blend wound dressing, using a simple chemical modification of the biomaterial surface with CDs.
The first section concerns the development of a statistical modeling of the functionalization process in order to facilitate parameters control, and enable manufacturers to better manage chemicals and energy cost, depending on the desired result.
The impact of treatment on original properties of textiles substrates was investigated.
Furthermore, the application of a drug that was initially unable to bind to untreated fabrics intended to explain the importance of functionalization. Release mode was highlighted, and the persistence of the antibacterial effect of loaded samples with methylene blue (MB) after release was also studied.
Materials and methods
Materials
Triangular strips of Adhe-els Laboratory (Tunisia) are nonwoven (NW) wound dressings made of 67% viscose and 33% polyester (average pore size = 0.5 µm, thickness under 7.3 psi pressure = 0.8 mm, basic weight = 34 g/m2). All the NW samples were produced by random distribution for web and spun bonded thermally. Native β-CD (assay ≥ 97%), citric acid (CTR) (C6H8O7) (crosslinking agent), disodium phosphate (NaH2PO4, 2H2O) (catalyst), and Tubingal CSO (commercial softener) were all provided from Aldrich Chemicals.
MB (Aldrich Chemical) is a cationic dye well-known for its antimicrobial properties [27–29], with the chemical formula C16H18ClN3S and a molecular weight of 319.85 g/mol. It is capable of forming stable inclusion complexes with CDs [30–32]. It was selected as drug model molecule for the investigation of the sorption and drug release properties of our systems.
Bacteriological tests were carried out using two different bacteria: Staphylococcus aureus (Gram-positive) (CIP224, collection strain) and Escherichia coli (Gram-negative) (strain L70A4 recently isolated from a clinical sample). Dehydrated agar is marketed by Oxoid (PC66, France). Its pH is 7.
Functionalization of nonwoven fabrics
The nonwoven fabrics (NWF) functionalization was carried using a pad dry-cure process [23,24,33]. The impregnating solution contained CTR (100 g/L), catalyst (30 g/L), and β-CD (100 g/L) in water. Fabrics were padded, roll-squeezed, dried, thermofixed (at different temperatures and times) and finally washed several times. In order to improve the touch and remove unreacted chemicals, fabrics were impregnated in a bath containing Tubingal CSO (1 g/L), during 5 min. Samples were washed and dried for 30 min at 100℃, then cooled down to room temperature in a desiccator, before being weighed. The functionalization degree (%-FD) of NWF was reported as the weight gain of samples upon treatment with β-CDs, calculated by the following equation
Optimization of functionalization
Minitab (Version 15, State College, PA, USA) was used for the statistical analysis of data. Comparison of means was conducted using analysis of regression and variance ANOVA with post hoc Tukey’s test at p < 0.05 [34].
Study of functionalization effect on tensile strength
Tensile tests were performed according to French standard NFG 07-119. %-FD of NWF was varied. A 50 N sensor was installed. The clamps distance was 250 mm and the testing speed was set at 75 mm/min. All samples were preconditioned at 25℃ and 80% relative humidity for 24 h prior to mechanical tests. Each experimental point throughout this study is the mean of 20 experimental values.
Study of functionalization effect on whiteness index
The treatment effect on NWF was evaluated using a spectro-colorimeter. Whiteness formula WIC was recommended in 1986 [35]
Drug application on nonwoven fabrics
NW dressings were impregnated in an aqueous solution containing 2% of MB; bath ratio is 1/40. Dyeing is carried out in a weakly acid medium, using an autoclave under pressure, AHIBA nuance (Data Color International, USA) (Figure 1). When finished, dyed samples were washed with hot water, then cold water, and finally dried at room temperature.
The exhaust dyeing process.
The dyeing bath exhaustion and the NWF drug release in pure water were assessed using an UV–visible spectrophotometer (U-2000 Hitachi, National School of Engineers of Monastir, Tunisia). Wavelengths range was 200–800 nm and a quartz cell was employed for the absorbance measurements.
Characterization of nonwoven fabrics functionalization
Fourier transformed infrared (FT-IR) analysis was performed using FT-IR spectrometer (Perkin-Elmer FT-IR System 2000 Model spectrometer, CRBJ, Borj Cidria, Tunisia) in transmittance mode. Samples were ground with KBr and pressed into pellets. The spectra were collected over a range of 4000 to 400 cm−1, with a resolution of 2 cm−1. Thermal studies were monitored using thermogravimetric analysis (TGA/DTG). The measurements were performed, with a heating rate of 10℃/min. The scanning temperature ranged from 25℃ to 600℃.
Study of wettability
The surface wettability of treated and untreated NWF was measured via the drop contact angle method, using digidrop image analysis software (GBX® Digidrop, Model DS, National School of Engineers of Monastir, Tunisia). Liquid droplets were deposited on the sample surface with a precision syringe. All samples were stored in a climatic chamber at 30% of relative humidity and 25℃ prior to measurements. Contact angles were measured 5 s after placing the droplet (5 µL) on the fabric substrate at 37℃. The wetting kinetic is studied over 200 s. At least 10 measurements per sample were carried out. The used liquid is glycerol/glycerine (γL (ambient T°) = 63.4 mN/m). It was chosen because its surface energy is high enough to get a contact angle higher than 10°, and it has no chemical interactions with the tested material. Tested materials are:
Sample 0 (untreated), Sample 1 (pH = 3, temperature = 160℃, time = 30 min, β-CD concentration = 33 g/L), Sample 2 (pH = 3, temperature = 160℃, time = 30 min, β-CD concentration = 100 g/L), Sample 3 (pH = 3, temperature = 160℃, time = 30 min, β-CD concentration = 200 g/L).
Bacteriological activity
Bacteriological study is used to assess the antibacterial activity of NWF at different steps of treatment. The standardized Kirby–Bauer disk diffusion method was used to investigate the antibacterial activity of NWF against E. coli and S. aureus bacteria, by tripling measures [36]. Sample disks (diameter 11 mm) were used for each material. Four types of NWF were tested:
Untreated sample MB-loaded sample (2% of MB) CD-MB-functionalized sample (%-FD = 11.2, 2% of MB) CD-MB-functionalized sample after release (2% of MB, 10 h of release)
The bacterial culture was performed on Muller Hinton agar slant (Oxoid-PC65). Then they were incubated for 24 h at 37℃. The next day, 10 mL of ringer cysteine (RC) (PC19 - Oxoid) were added to slurry the bacteria. A drop of this solution was mixed in 10 mL of RC. The suspension’s concentration is of 105 bacteria/mL.
About 38 g of agar powder were dissolved in 1 L of distilled water and then brought to boil to be completely dissolved. The whole was sterilized for 15 min in an autoclave at 121℃. Then, the agar was sampled in small pots. Each one contained an amount of agar to form a layer of 2–3 mm in seven Petri dishes of 90 mm diameter. Spread agar was refrigerated at 2–8℃. The handling day, the agar was pouring into boxes, and 0.1 mL of the bacterial suspension at 105 bacteria/mL were spread. Then samples were placed on the agar.
After 24 h of incubation at 37℃, the bacteria grew in the bottom of the box, which was observed through the opacity of the agar. However, no growth will occur in the area surrounding the sample disk in which the MB has migrated, and a circular bacteria-free zone (inhibition zone) could be seen.
Results and discussion
Reactional mechanism
The grafting reaction of CDs onto cellulose begins with a dehydration of the PCA at the fixation temperature (Figure 2). A cyclic anhydride is obtained. Two remaining carboxylic acid groups will form a second anhydride, which can react with a hydroxyl group on the primary face of CD.
Grafting of CDs on cellulose fibers using a PCA [23].
In other recent studies, the synthetic fibers finishing using CDs, was investigated [24–26]. The grafting reaction begins with dehydration of carboxylic acid at the fixation temperature which leads to a cyclic anhydride (Figure 2). Due to the absence of hydroxyl functions (or amine functions) on synthetic fibers, it is impossible for the anhydride to react with them. It will therefore react with hydroxyl functions of CD to form a crosslinked polymer network.
Optimization of functionalization
The functionalization parameters study has been previously reported [37].
The optimal conditions of the functionalization process were predicted by response optimizer tool of Minitab 15 software (Figure 3). It indicated that for a %-FD of 11.44, the optimum values for each independent variable were bath pH = 2.8, thermo fixation temperature = 160℃, thermofixation duration = 29.5 min, and β-CD concentration = 100 g/L.
Response optimization of functionalization process.
The results obtained when applying the optimal combination were in agreement with the theoretical result. Indeed, a %-FD of 11.51 was obtained versus a theoretical value of 11.44.
Study of functionalization effect on tensile strength
The functionalization via a polymer of β-CD was performed in conditions which could affect the mechanical behavior of fabrics. Figure 4 shows the evolution of tensile strength of NWF while varying the thermofixation temperature. We observe that NWF tensile properties were maintained constant until 170℃. Beyond this value, there was a slight decrease in breaking strength of 10%. High temperatures up to 160℃ affect in particular viscose fibers. This could be explained by the fact that during the treatment process at higher temperatures, pronounced structural changes occurred especially in amorphous region, and long cellulose macromolecules split into shorter fragments. The results are generally consistent with early study from Yu and Wu, that the glycosidic bonds in the chain segments within the amorphous portion of cellulose started to be ruptured at temperatures higher than 150℃ [38], which have a direct impact on the resistance to mechanical attacks of the dressing.
Evolution of the breaking strength of NWF.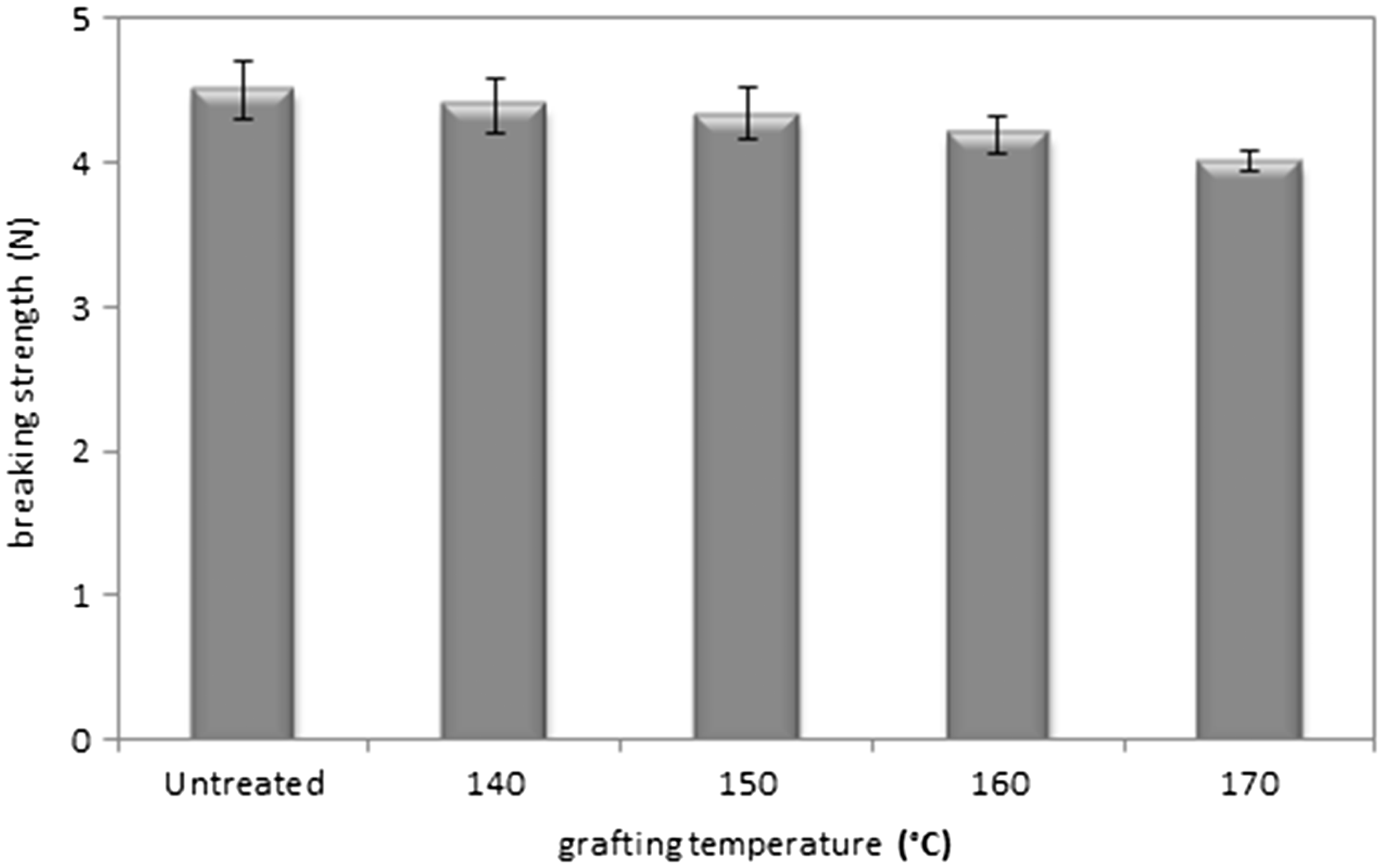
Study of functionalization effect on whiteness index
According to Figure 5, increasing temperature from 0℃ to 160℃ generates a hardly detectable yellowing. At 170℃ a decrease of 18.5% of NWF whiteness Index is detected. Therefore, the observed color change of the fibers could be related to a partial degradation of cellulose via successive degradation of carbohydrate structures to furans or aromatic rings) that would ultimately lead to the production of char at high severity. Coloration of the fibers was also observed by Ibrahim et al. [39] who explained this phenomenon via sugar degradation after steam pre-treatment at higher severity factor.
Evolution of whiteness of NWF.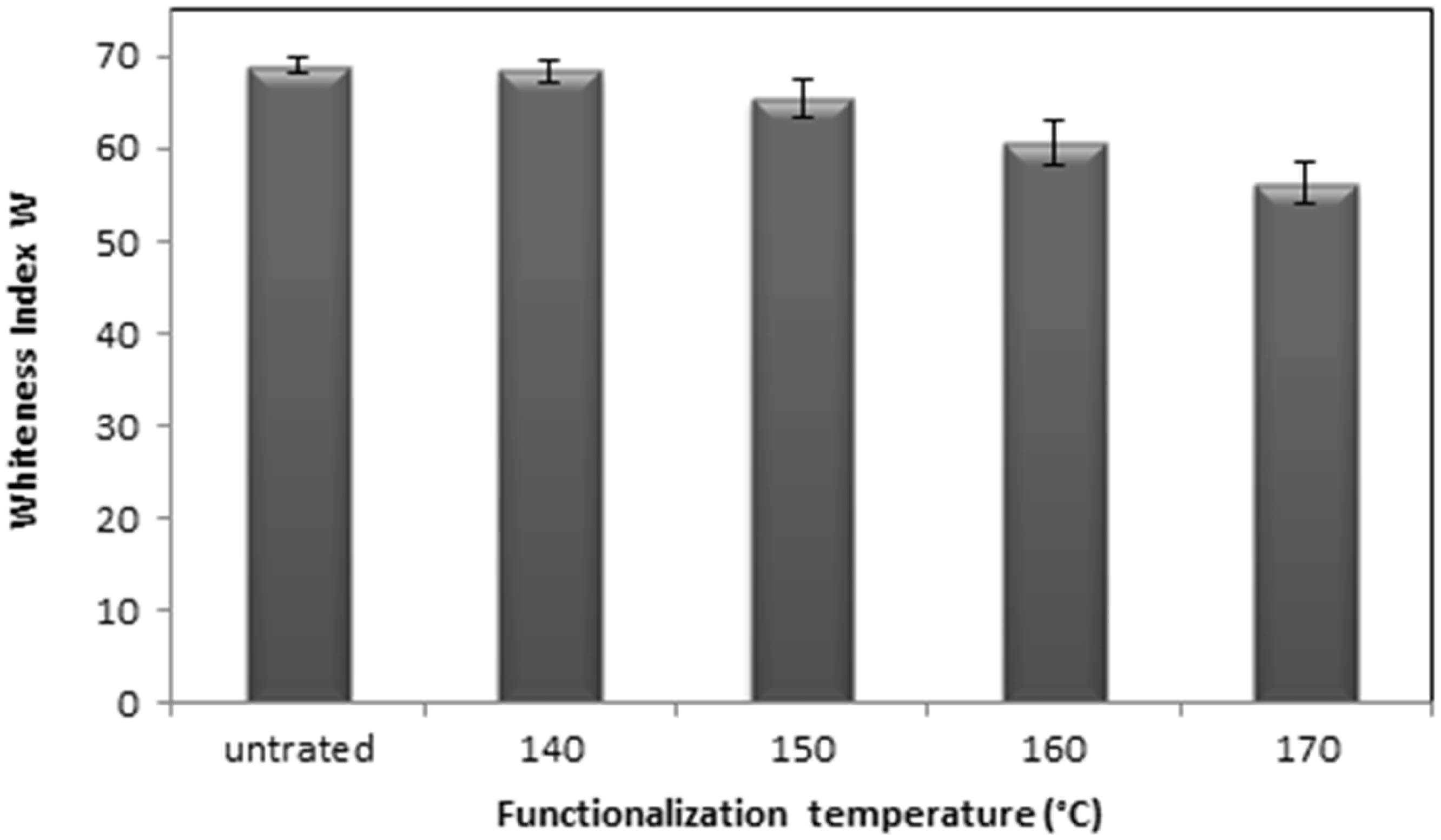
Indeed, high temperatures affect mainly cellulose fibers, but it has almost no effect on the polyester fibers. For this reason, the processing temperature must not exceed 160℃.
Characterization of functionalized nonwoven fabrics
FT-IR spectroscopy analysis
Structures of the modified and virgin NWF were assessed by comparative inspection of their FT-IR spectra (Figure 6).
FT-IR spectra of untreated NWF and functionalized NWF via a polymer of β-CD.
The FT-IR spectra of NWF show the broad and intense peak at 3300 cm−1, indicating the presence of O–H stretching vibration in cellulose. The peak at 2890 cm−1corresponds to the C–H stretching. The absorption peak at 1707 cm−1 corresponds to the C=O ester bond of polyester. Furthermore, in functionalized NWF spectrum, the significant increase of the intensity of the characteristic band of esters in 1707 cm−1 is the result of the mechanism of the functionalization of viscose/polyester dressings via poly-esterification.
In summary, the FT-IR studies confirmed the appearance of esters after the chemical modification. They are induced by the poly-esterification reaction that occurred between citric acid, cellulose and β-CD, or also between citric acid and β-CD.
Thermal analysis
The thermal stability and decomposition behavior of the functionalized NWF were studied (Figure 7). Samples are placed in a nacelle then subjected to the temperature program. TGA/DTG thermograms of untreated and functionalized NW dressings were detailed. The first stage exhibited a mass loss of 4%, which is due to the loss of the adsorbed water, and could be observed for the two supports. The second stage reached mass losses of about 96% for untreated materials, and 90% for modified materials.
TGA (a) and DTG (b) curves of untreated and functionalized NWF via a polymer based on β-CD.
The DTG curves revealed that virgin NWF decomposed at 350℃, whereas the functionalized NWF decomposed at 295℃. Chemical modification has resulted in a thermally less stable material. The residual mass is more important in the case of functionalized NWF, which confirms the binding or the appearance of new chemical entities. The TGA curve shows that the %-FD of treated sample is around 9.5%, which is in agreement with weighing results.
Study of fuctionalization effect on wettability
The wetting contact angle of virgin and treated NWF surfaces is measured. Then it is possible to conclude whether a material surface is hydrophilic (θ < 90°), hydrophobic (90° < θ < 150°) or super-hydrophobic (θ > 150°). By increasing β-CD concentration in the functionalization bath (Figure 8), the contact angle decreases. We can conclude that increasing the concentration of β-CD in the finishing bath improves the NWF hydrophilicity. The amphiphilic CDs have a macrocyclic structure with a hydrophilic outside and a hydrophobic inside. In addition, the presence of the carboxylic entities of the PCA crosslinking agent is also responsible for enhancing the hydrophilicity. Thus the NWF functionalization via polymers based on β-CD makes the material more hydrophilic.
Evolution of contact angle of the viscose/polyester NW dressings with glycerin drop depending on β-CD concentration (pH = 3, temperature = 160° C, time = 30 min).
Bacteriological study
A NWF is functionalized under optimal conditions (%-FD = 11.51). We propose to encapsulate MB molecules.
Measure of the bath exhaustion
Material sorption capacity of MB was measured for virgin and CD-functionalized fabrics using spectrophotometry at λ = 655 nm. Very low bath exhaustion rate is obtained with untreated NWF (Figure 9), compared to the one given by the modified dressing. So during treatment there has been creation or attachment of chemical entities able to fix the cationic dye molecules; these are β-CD host molecules and probably free carboxylic groups of CTR, having not reacted with viscose and β-CD.
Dyebath exhaustion of treated and untreated NW dressings.
Methylene blue release
Upon exposure of viscose/polyester-MB-CD dressing (%-GD = 11.51, %-MB = 2, bath ratio = 1/40) to pure water, at room temperature, the total amount of released MB from the device was determined by UV–Visible spectrophotometry.
Indeed, in antibacterial drug release system, it is necessary to involve a considerable initial release. According to Figure 10, an initial burst release of MB is detected which is followed by a sustained zero-order release. The first amount of drug is supposed to kill bacteria in wounds. The second step is useful to prevent re-growth of bacteria. The amount of active ingredient, which has been fixed in NWF, is distributed in two modes of fixation:
A first moving part, the molecules included in the β-CD molecule, which is released over time, for about 7 h. It presents 31.5% of the quantity of MB fixed on the viscose/polyester dressing. A static second part, which remains fixed on the same support after impregnation in water for several days. This part presents the amount of the active ingredient fixed directly on the fibers or by free carboxylic ends, which will be in continuing contact with the wound. Release profile of MB of functionalized viscose/polyester NWF.

Bacteriological assays
The inhibitory effect was presented in Figure 11, through measuring the difference between the diameter of untreated sample and inhibition zone of modified samples. The presence of an inhibition halo reveals the bactericidal effect of the pellet. Virgin NWF does not have any antibacterial effect. The β-CD functionalized NWF present a larger inhibition zone compared to untreated materials with CDs. This confirms that functionalization enabled the dressing to fix the drug molecules.
Antibacterial activity of untreated and functionalized NWF disks with MB on S. aureus and E. coli bacteria.
MB–CD–NWF, which undergoes a MB release for 24 h, still has an antibacterial activity. This activity is due to the MB present in NWF, but which was not encapsulated in the molecules of CDs.
The largest activity is revealed in the last case of β-CD-MB functionalized samples. It is explained by the release of the large amount of MB, which was encapsulated in CDs molecules.
Conclusion
In this work, blend NWF (viscose/polyester) were finished via a polymer of β-CD in order to provide the feature of different drugs binding. This chemical modification also helped to improve hydrophilicity to finallyobtain dressings with good healing properties.
On one hand, statistical analysis software was used to optimize functionalization efficiency. On the other hand, mechanical properties and whiteness index were studied to confirm that functionalization treatment conditions do not alter the original NWF properties. The assessment of the wettability before and after functionalization has shown that the presence of the polymer based β-CD molecules produced super absorbent textiles, which is a desirable feature for wound dressings. After that, we confirmed through characterization techniques (FT-IR spectroscopy and thermogravimetric analysis) the efficiency of functionalization treatment. In addition to that, a drug application was carried out to show the usefulness of our treatment in the binding of drugs molecules to dressing thanks to the encapsulation capacity of CDs molecules. Dressings were successfully loaded with MB and the obtained drug delivery system showed a sustained release in pure water.
The originality and applications of our materials are numerous due to their functionalities. Our study provides a clearly defined antibacterial application. In fact, we worked on a dressing structure already marketed and very widespread. In addition to that, the used preparation is simple (one step), ecologic (water as reaction medium), and very low cost and, consequently, these results highlight the potential of the presented techniques to be industrially applied.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


