Abstract
In this study, composite nanofibers from a solution of polyacrylonitrile (PAN), functionalized multi-walled carbon nanotubes (f-MWCNTs), and silver nitrate (AgNO3) in dimethylsulfoxide were successfully produced by the electrospinning method. Aqueous solution of hydrazinium hydroxide was used for the chemical reduction of silver ions. The effects of the simultaneous use of carbon nanotubes (either pristine or amine-functionalized) and silver nitrate in different percentages and the application of chemical reduction on the properties of the nanocomposite nanowebs were investigated. FTIR, SEM, conductivity meter, tensile tester, XRD, and DSC were used for the characterization. Antibacterial activities of the nanocomposite nanowebs were determined against
Keywords
Introduction
The incorporation of two different nanostructures with special properties in a polymer is a promising technique to produce multifunctional composites. This strategy for combining different nanostructures into polymers has been mainly applied in composite film production [1–4] but the potential to extend this strategy to fiber production offers an opportunity for the development of multifunctional fibers.
On one hand, carbon nanotubes (CNTs) and silver nanoparticles (AgNPs) have been increasingly incorporated into polymers with the aim of producing functional materials. The combination of superior mechanical, thermal, and electronic properties makes CNTs ideal candidate as a filler material in nanocomposites. The addition of CNTs is reported to improve especially mechanical and electrical properties of the composite structures [5]. A significant challenge for getting maximum benefit of CNTs in polymer composites is to achieve a good dispersion which is achieved through several approaches such as defect functionalization, covalent functionalization, and non-covalent functionalization in the literature [6,7]. On the other hand, nanocomposites with silver nanoparticles are reported to acquire catalytic, optical, and especially antibacterial properties [8,9] Through different methods of preparing a polymeric matrix containing silver nanoparticles,
There are some studies in the literature showing the advantages of the combined use of CNTs and AgNPs in films [1–4] and meltspun fibers [11]. In the studies about the composite films produced with the addition of CNTs and AgNO3, increase in conductivity [1–4], biocompatibility [1], thermal stability [3], and decrease in mechanical properties [2] are reported. Rangari et al. [11] meltspun nylon-6 fibers with Ag-coated CNTs hybrid nanoparticles (Ag/CNTs) and reported improved failure stress, modulus, thermal stability, and crystallization for the composite fibers. Besides; the composite nanofibers with Ag-coated CNTs showed significant antimicrobial activity.
Although there are some promising studies about the combined use of CNTs and silver in films and meltspun fibers in the literature, there are only very limited number of studies on the investigation of the effects of simultaneous use of CNTs and AgNPs on the properties of nanofibers produced by the electrospinning method. Ouyang et al. used electrospinning as a coating method and electrospun polyurethane nanofibers filled with carbon nanotubes and metallic silver nanoparticles on a chemical electrode. They mainly focused on the use of the structure as biosensors [12]. Other than sensor applications, nanofibers offer great advantages in many application areas such as filtration, biomedical textiles, tissue engineering, and protective clothing due to their increased surface area, very small diameters, and high porosity with small pores [13–18]. Thus, it is important to investigate the combined use of CNTs and AgNPs on nanofiber web properties which have different structural properties than melt or wet/dry spun fiber and film with regard to pore structure, fiber diameter, surface area, fiber placement, orientation, etc. In this study, composite nanofibers from a solution of polyacrylonitrile (PAN), multi-walled carbon nanotubes (CNTs), and silver nitrate (AgNO3) in dimethylsulfoxide were successfully produced by electrospinning method. The aim of the study was to produce multifunctional (conductive and antibacterial) PAN nanofibers which may find applications in filtration, protective clothing, and medical textiles applications, to investigate the properties of PAN/f-MWCNTs/AgNP composite nanofibers and synergistic effect of the simultaneous use of f-MWCNTs and AgNPs in PAN nanofiber structure and to determine of the contribution of the selected factors (f-MWCNT content, AgNO3 content, and chemical reduction application) to the variations in ultimate tensile strength, elongation, and conductivity properties of composite nanofibers. Being the first study performed on PAN/f-MWCNT/AgNP nanofibers, this study is original and contributes to the literature by comprehensively analyzing the structural, morphological, mechanical, thermal, and functional (electrical conductivity and antibacterial efficiency) properties of PAN/f-MWCNT/AgNP composite nanofibers.
Experimental details
Materials
Polyacrylonitrile (PAN) (Sigma Aldrich, 181315, average
Methods
CNT functionalization
The MWCNTs were first carboxyl-functionalized and then amine-functionalized according to the methods described in the literature [19–21].
Carboxyl-functionalized multi-walled carbon nanotubes (MWCNT–COOH) were prepared by the oxidation of pristine MWCNTs using H2SO4/HNO3 (3:1 by volume) as the oxidant. Into a 500 mL flask equipped with a condenser, pristine MWCNTs (3 g), HNO3 (65%, 25 mL), and H2SO4 (98%, 75 mL) were added with vigorous stirring. The mixture was then ultrasonicated in an ultrasonic bath (40 kHz) for 10 min and refluxed for 100 min. The temperature was gradually increased from 90℃ to 133℃ during refluxing. After cooling, the reaction mixture was diluted with 500 mL of deionized water and then vacuum-filtered using Whatman PTFE filter paper. The dilution, filtering, and washing steps were repeated until the pH of the filtrate was 7. The filtered solid was dried under vacuum for 24 h at 60℃. For amino-functionalization, 200 mg of MWCNTs-COOH was mixed with NaNO2 (580 mg) and isophoronediamine (0.5 mL). About 0.36 mL and 10 mL DMF were added. Then the mixture was stirred for 1 h at 60℃. After cooling the mixture to room temperature, DMF was added and the mixture was filtered with a PTFE membrane. The process was repeated until DMF obtained after filtration was colorless. The solid obtained was then dried at 60℃ overnight under vacuum.
Preparation of the solutions
The required amount of f-MWCNT and AgNO3 was added to the required amount of DMSO and homogenized with ultrasonic tip for 10 min and with ultrasonic bath for 45 min. Then PAN was added to the dispersion and dissolved by magnetically stirring at 40℃ for 3 h. The concentration of PAN was 7 w% (with respect to the weight of the solution). The content of f-MWCNT and AgNO3 was varied as 0.5, 1, and 3 w% (with respect to the weight of PAN). For comparison, nanowebs with 0.5 w% pristine MWCNTs, 0.5 w% AgNO3 and 1 w% pristine MWCNTs, and 1 w% AgNO3 were also produced.
Electrospinning
Compositions of the electrospun composite nanofiber webs.
Chemical reduction of silver ions
Pieces of the as-spun nanowebs were immersed into the aqueous solution of hydrazinium hydroxide (1:20 hydrazinium hydroxide: distilled water) for 30 min at room temperature, then washed with 100 mL distilled water two times and dried in room temperature to obtain composite nanofibers containing Ag nanoparticles.
Characterization
Fourier transform infrared (FTIR) absorption spectra for pristine carbon nanotubes and amine-functionalized carbon nanotubes were taken with Thermo Scientific Nicolet IS10 spectrometer. The scanning ranged from 4000 cm−1 to 400 cm−1. Scanning electron microscopy (SEM; EVO MA 10) was used to obtain SEM images of some selected nanofiber webs. The webs were sputter coated with gold layer before SEM analyses. The diameter of at least 50 randomly selected nanofibers was measured on SEM photomicrographs and analyzed using Image Analysis Software to obtain the average fiber diameter. Ultimate tensile strength and breaking elongation of the nanofiber webs were measured using a tensile tester with a 100 N load cell at a crosshead speed of 20 mm/min. The length and the width of the specimens were 35 mm and 5 mm, respectively. The gage length was 15 mm [22]. The thicknesses of the specimens were measured with a Mitutoyo digital micrometer. Resistance measurements were performed using a two-circular-probe connected to Microtest 6370 LCR meter with four-wire system. Conductivity in S/cm was calculated using the volume resistance value measured and the geometric dimensions of the samples [23,24]. Wide-angle X-ray diffraction traces were obtained using a Bruker® AXS D8 Advance X-ray diffractometer system using nickel-filtered CuKα radiation (
Statistical analysis
Parameters and their levels used in the full factorial experimental design.

Results and discussion
FTIR
FTIR analysis using KBr pellet method was performed to monitor the surface functional groups after functionalization of MWCNTs. The FTIR spectra of amine-functionalized MWCNTs in comparison with the pristine MWCNTs are presented in Figure 1.
FTIR spectra of (a) pristine MWCNTs and (b) f-MWCNTs.
In comparison with pristine MWCNTs, amine-functionalized MWCNTs showed some new peaks at 1348 and 1547 cm−1 which can be assigned to C–N bond stretching and N–H in-plane vibration, respectively. Peaks in the regions 3100–3500 cm−1 can be attributed to N–H stretching [20]. The peaks at 1547 cm−1 is due to the C–NH vibration while the peak at 1139 cm−1 can be attributed to the C = N and C–C vibrations. The peak at around 621 cm−1 can be assigned to the amide (N–C = O) vibrations [26]. The new peaks at the spectrum of amine-functionalized MWCNTs confirmed the successful functionalization of MWCNTs.
Morphology
SEM images of some selected composite nanofiber webs can be seen in Figure 2 while the SEM images of the selected samples after the application of the chemical reduction process can be seen in Figure 3. The average nanofiber diameters are presented in Table 3.
SEM images of (a) PAN-0.5%f-MWCNT-0.5%AgNO3, (b) PAN-0.5%CNT-pristine-0.5%AgNO3, (c) PAN-1%f-MWCNT-1%AgNO3, and (d) PAN-1%CNT-pristine-1%AgNO3. SEM images of (a) PAN-0.5%f-MWCNT-0.5%AgNO3, (b) PAN-0.5%CNT-pristine-0.5%AgNO3, (c) PAN-1%f-MWCNT-1%AgNO3, and (d) PAN-1%CNT-pristine-1%AgNO3 nanowebs after the application of chemical reduction process. Mean fiber diameters before and after the chemical reduction process.


Pure PAN nanoweb and the composite nanowebs had uniform structures. Chemical reduction process did not result in any observable surface damage to the fibers.
Additives generally cause an increase in the diameter due to the increased substance concentration [27]. Exceptionally, charge carriers such as carbon nanotubes and silver nanoparticles affect the conductivity of the electrospinning solution and can result in some opposite effects on fiber formation and diameter. They not only increase the concentration and thus may result in larger fiber formation but also increase the net charge density and may result in smaller fiber formation. The average nanofiber diameter is determined by the dominant effect [28–30].
While an increase in the diameter was reported by the addition of silver nitrate, a decrease in diameter was reported with the application of the chemical reduction process [31]. On one hand, CNT addition is also reported to result in an increase in nanofiber diameter [28,32]. In this study, with the simultaneous addition of the f-MWCNTs and AgNO3 additives and application of the chemical reduction process, fiber diameters increased with respect to pure PAN nanofibers. With the increases in the additive contents, the diameters of the nanofibers further increased. Both the fillers were conductive fillers and were expected to increase the solution conductivity thereby leading to higher stretching and thinner nanofiber formation during electrospinning. On the other hand, the electrospinning solutions become more viscous on the addition of the additives. It appears that the solution viscosity had a greater impact on fiber diameter than the increase in the electrical conductivity. Besides, the increase in average nanofiber diameter on using amino-functionalized carbon nanotubes was likely due to the increased size of the carbon nanotubes after functionalization keeping the polymer macromolecules apart from each other. Chemical reduction process applied by using hydrazinium hydroxide had a thinning effect on fiber diameters. This may be due to the structural changes which might have taken place as a result of the reduction of silver ions.
When the fiber diameter differences were evaluated statistically, the average fiber diameters of PAN-1%f-MWCNT-1%AgNO3 and PAN-1%CNT-pristine-1%AgNO3 nanowebs differed significantly from PAN nanoweb after the chemical reduction process. Comparing the average nanofiber diameters before and after chemical reduction process, it was observed that the chemical reduction process affected the diameters of the nanowebs significantly. The diameters of PAN-1%f-MWCNT-1%AgNO3, PAN-0.5%CNT-pristine-0.5%AgNO3, and PAN-1%CNT-pristine-1%AgNO3 nanofibers were significantly affected with the chemical reduction process. Besides, the increased additive content resulted in significant variation on the average nanofiber diameter. The diameters of PAN-1%f-MWCNT-1%AgNO3 and PAN-1%CNT-pristine-1AgNO3 significantly differed from PAN nanofibers after chemical reduction process.
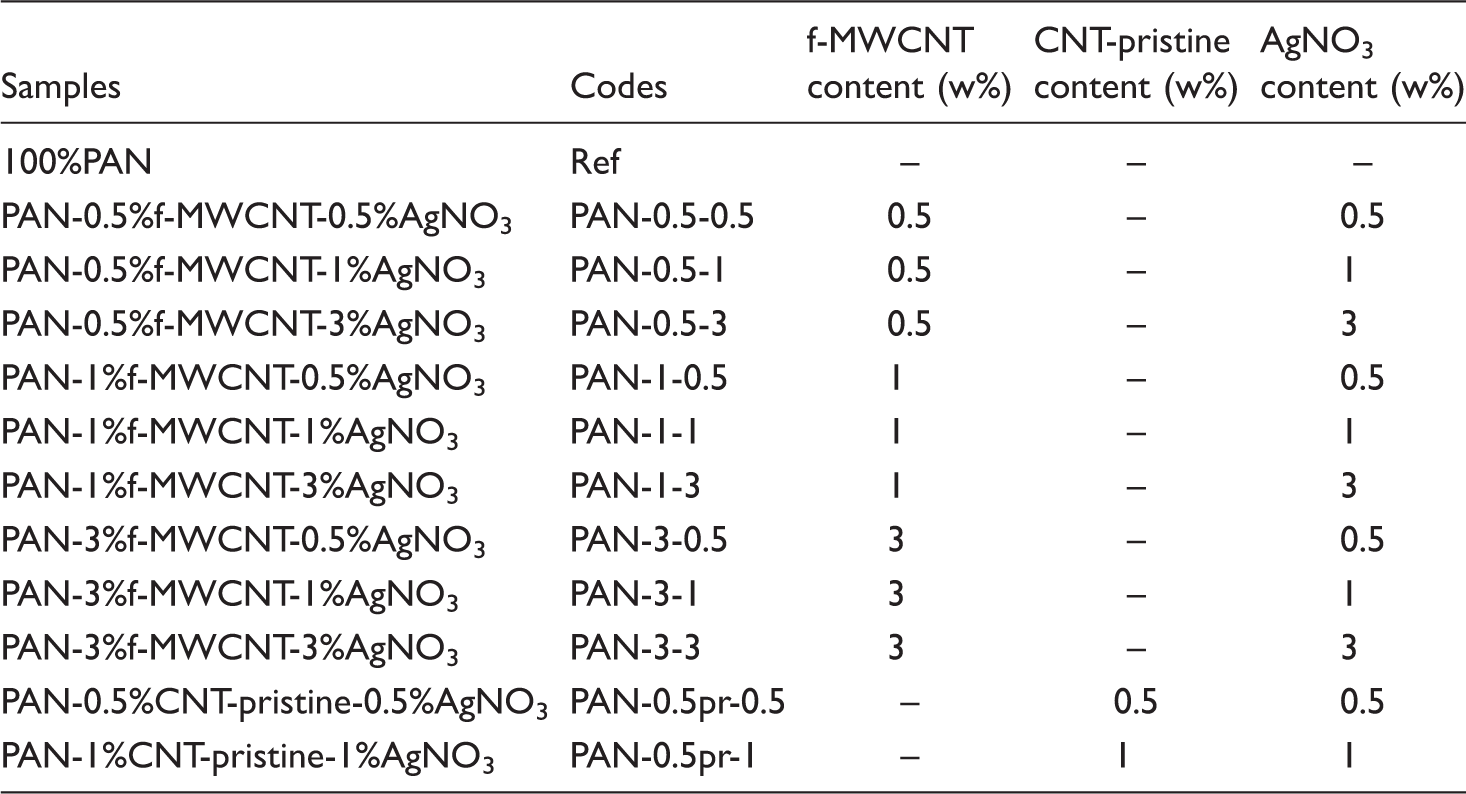
Tensile properties
Ultimate tensile strength and breaking elongation properties of the nanofiber webs before and after chemical reduction process are graphically presented in Figures 4 and 5, respectively.
Ultimate tensile strength values of composite nanofiber webs before and after chemical reduction process (B: before reduction; A: after reduction). Breaking elongation values of composite nanofiber webs before and after chemical reduction process (B: before reduction; A: after reduction).

In the literature, there are studies which reported increase in the strength with the addition of carbon nanotubes [5,33] and with the addition of silver nanoparticles [34]. In this study, both carbon nanotubes and silver nitrate were added to the electrospinning solution, and it was observed that the ultimate tensile strength of the nanofibers decreased as the additive content increased. As additive content increased, the nanoparticles might have formed agglomerates which might have acted as stress points during the tensile testing. The decrease in ultimate tensile strength is higher for the nanowebs produced with the addition of pristine nanotubes.
On application of the chemical reduction process, there was an increase in ultimate tensile strength values which might have occurred due to the formation of coordination bonds between the nitrile groups and Ag nanoparticles. Furthermore, the shrinkage of 3% in nanofiber web dimensions, which we have observed during the chemical reduction process, might have resulted in a more compact structure with less porosity and this may be another reason for the higher strength values obtained after the reduction process. Besides, it is reported in literature that treatment with hydrazinium hydroxide may form crosslinking between PAN chains which can lead to an enhanced tensile strength [35,36].
It is observed that the breaking elongation values increased with the addition of the additives. This might have been due to the increased voids which might have formed around the nanoparticles. The increase in breaking elongation is higher for the samples produced with the addition of pristine carbon nanotubes. After the reduction process, breaking elongation decreased as the nanowebs became more compact and the mobility of the macromolecules decreased with the formation of coordination bonds between the nitrile groups and the silver nanoparticles.
Analysis of variance for ultimate tensile strength.

Analysis of variance for elongation.
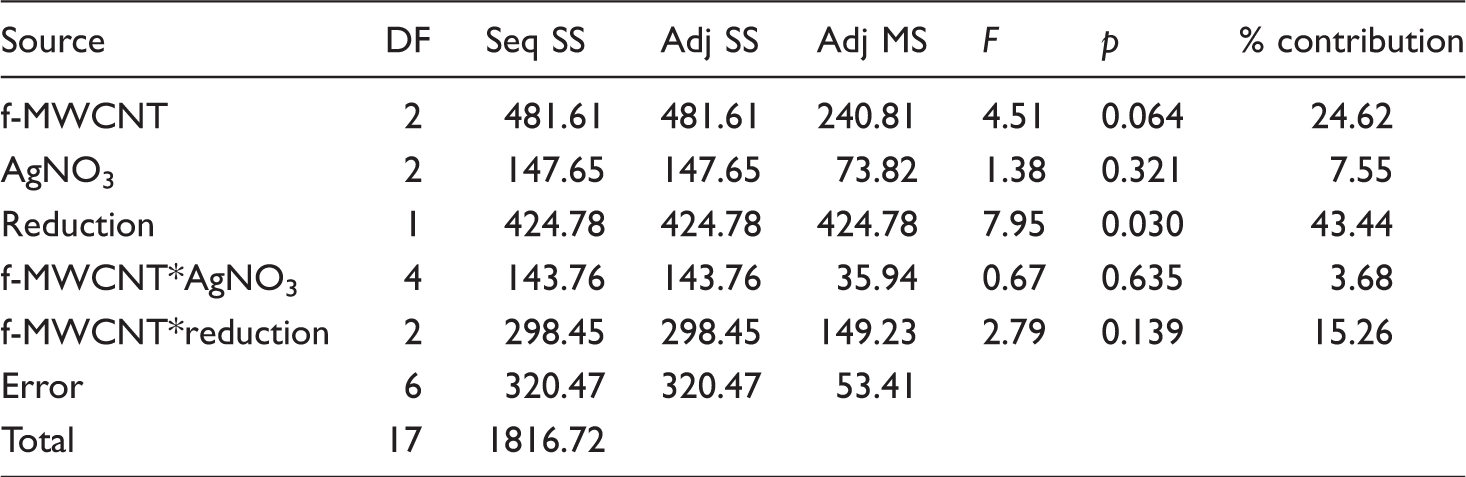
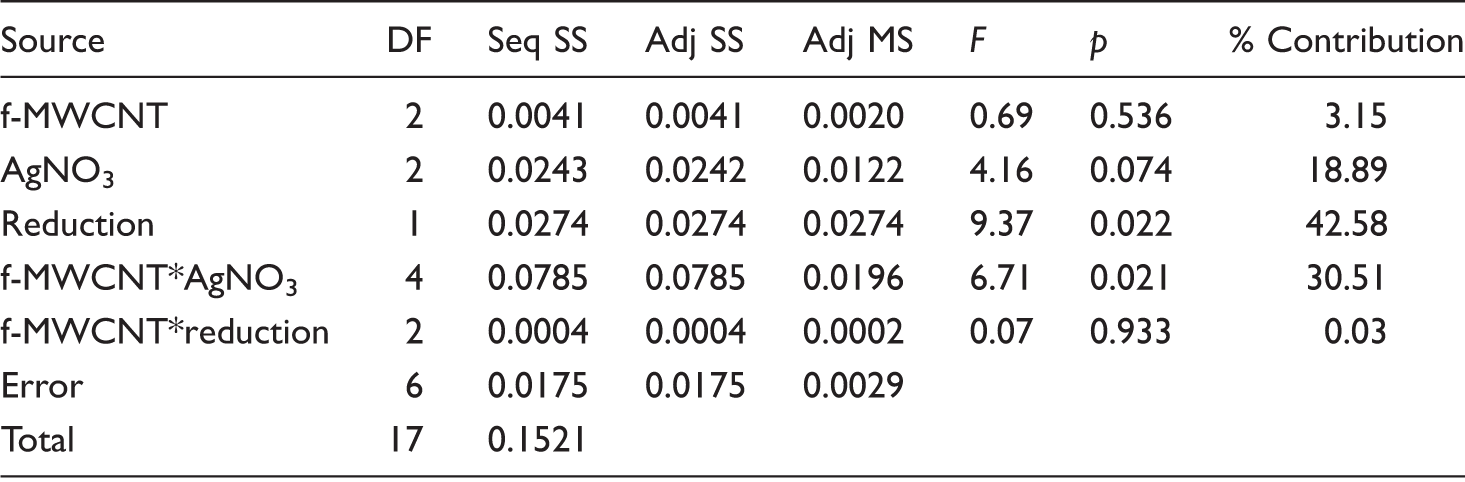
Conductivity
Conductivity values of the composite nanofiber webs.

The addition of MWCNTs, either pristine or functionalized and AgNO3 provided conductive properties into PAN nanofiber which is normally an insulator with a conductivity value of 10−12 S/cm [37]. The conductivity of the nanofibers increased with respect to neat PAN nanofibers with the addition of conductive additives, but was not much affected with the increase in additive content, especially for the samples which are produced using f-MWCNTs. Compared with the addition of pristine MWCNTs, the addition of f-MWCNTs resulted in higher conductivity values which was attributed to the better dispersion of f-MWCNTs. For the samples produced with pristine MWCNTs, conductivity decreased as the additive content increased which might have been due to the possible agglomeration of the additives. Silver reduction process generally resulted in a 10 times increase in the conductivity which is mainly due to the formation of silver nanoparticles in the nanofiber structure. Furthermore, as the structure became more compact during the reduction process, the conductive particles might have become closer which resulted in an increase in conductivity.
Analysis of variance for conductivity.
X-ray diffraction
The polymer chains are expected to experience an extremely strong shear force during the electrospinning process due to the rapid stretching of the electrified jet and the rapid evaporation of the solvent. The strain rate on the order of around 104 s−1 in the electrospinning jets forces the polymer molecules to orient in the direction of elongation. On one hand, the rapid stretching and solidification during electrospinning can prevent the polymer chains from relaxing back to their equilibrium conformations resulting in high degree of molecular orientation, on the other hand, they lead to the retardation of crystallization process as the stretched chains do not have enough time for crystal formation. Although the polymer chains are non-crystalline in the nanofibers, they are highly oriented [38].
Figure 6 shows the curve fitting of equatorial X-ray diffraction traces of electrospun pure PAN and PAN-1%f-MWCNT-1%AgNO3 (after chemical reduction) composite nanofibers obtained in this study.
Curve fittings of equatorial X-ray diffraction traces of electrospun (a) pure PAN and (b) PAN-1%f-MWCNT-1%AgNO3 (after chemical reduction) nanofibers.
Qualitative examination of equatorial X-ray diffraction traces of PAN nanoweb shows two prominent reflections with d-spacings of 0.52 and 0.3 nm which can be indexed as (100) and (110) reflections of hexagonal unit cell [39] with basal plane dimensions of
Crystallinity values of pure PAN and composite nanofiber (PAN-1%f-MWCNT-1%AgNO3 (after reduction) webs.

In the literature, an increase in crystallinity is observed with the addition of certain amount of AgNPs due to the formation of some cyclic structure in the PAN chains [10]. Besides; addition of 1 w% MWCNTs is also reported to improve crystallinity [21]. In this study, simultaneous addition of f-MWCNTs and AgNO3 into nanofiber structure resulted in improved crystallinity which may be attributed to the dominant effect of the increased conductivity of the electrospinning solutions with the addition of the conductive additives. Higher conductivity of electrospinning solution is expected to result in higher stretching during electrospinning [41–43], leading to higher orientation of the macromolecules [41]. Besides, it is likely that additives help the PAN chains to become highly linear and pack more efficiently thereby causing an increase in the degree of order (orientation) [44]. The highly oriented domains in nanofibers are metastable and may slowly revert to crystallites [41]. Higher orientation together with the late evaporation of the solvent providing some more time for crystallization to take place might have resulted in higher crystallinity values for PAN-1% f-MWCNTs-1%AgNO3 nanofiber webs.
Thermal properties
The thermal performance of a fiber material is an important property since it is closely related to fiber’s post-processing and utilization performance [14]. It is well established in the literature that PAN goes through the cyclization reactions which is exothermic in nature when heated. The cyclization of nitrile groups of pure PAN initiated through a free radical is considered to take place as the result of intra or intermolecular jumping of radicals [44–47]. During this process, PAN undergoes a number of physical and chemical changes that converts the linear PAN molecular chains to an aromatic ladder structure [48].
The thermal properties of pure PAN and composite nanowebs were examined by DSC analysis in the temperature range from 30 to 400 ℃ with a heating rate of 20 ℃/min. DSC thermograms of the pure PAN and a selected composite nanoweb (PAN-1%f-MWCNTs-1%AgNO3-after reduction) are presented in Figure 7. Table 9 shows the cyclization (i.e., exothermic peak) temperature ( DSC thermograms of (a) pure PAN and (b) composite nanofiber (PAN-1%f-MWCNT-1%AgNO3-after reduction) webs. DSC results showing the 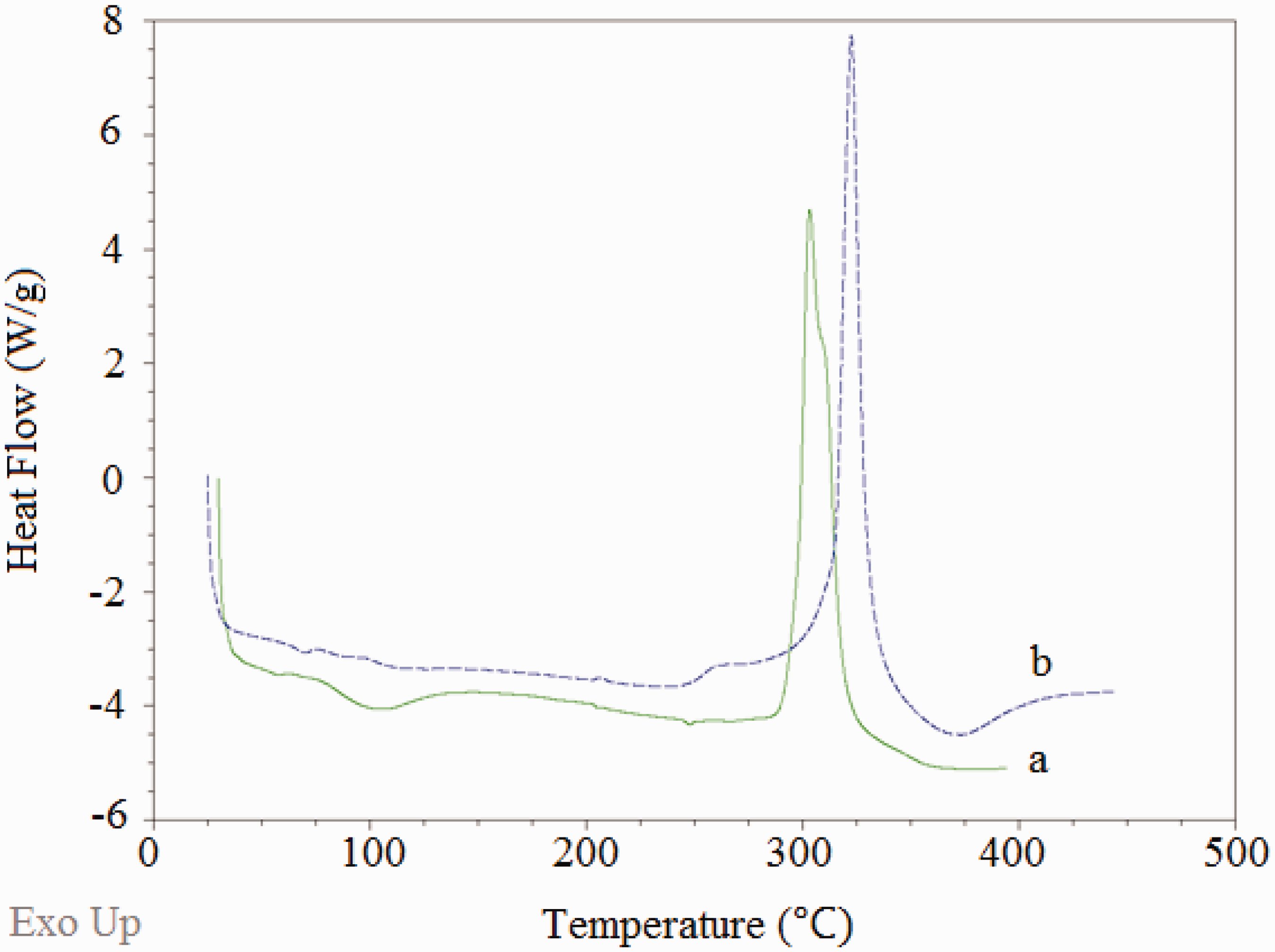
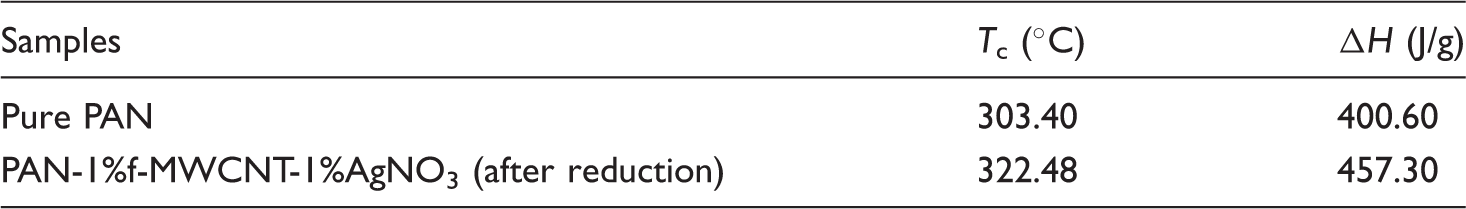
The main exothermic peak of pure PAN located around 315 ℃ results from a combination of three main reactions, mainly dehydrogenation, instantaneous cyclization, and crosslinking reactions, which are known to be mainly exothermic in nature [49]. Out of these three thermal reactions, the predominant one is known to be the instantaneous cyclization reaction [46,50,51]. Since the DSC thermograms are obtained under an inert atmosphere, no oxidation reactions are expected to take place. The large and sharp peak of pure PAN indicates the presence of instantaneous cyclization of nitrile groups into an extended conjugated ring system in the nitrogen atmosphere [51]. The fast reaction of electrospun pure PAN nanofibers in nitrogen atmosphere may be attributed to the facile formation of free radicals on the nitrile groups and subsequent recombinations between the radical groups intermolecularly or intramolecularly [52].
A shift to higher temperature is observed in exothermic peak position when 1 w% f-MWCNTs and 1 w% AgNO3 are added to PAN nanofibers. The addition of 1 w% AgNO3 and 1 w% f-MWCNTs also increased the value of the heat of the exothermic peak (i.e., enthalpy, Δ
Antimicrobial efficiency
According to FTTS-FA-002 standard, samples with 99% or over 99% antimicrobial efficiency can be called antimicrobial [53]. While pure PAN nanofibers do not display antimicrobial effect [54], most of the nanoparticles show antimicrobial effect due to their large surface area [55].
Nanoparticles of Ag have been widely investigated for their antimicrobial property [56–59]. The AgNPs may get attached to the cell membrane and damage it, and also penetrate inside the bacteria and damage the DNA, or attack the respiratory chain, finally leading to cell death. The antimicrobial mechanism of AgNPs could also be related to the formation of free radicals and subsequent free radical-induced membrane damage [60–62]. Carbon nanotubes were also found to exhibit high antimicrobial activity. It was evidenced that the size of carbon nanotubes (diameter, length, and aggregation), functionalization, concentration, impurities, incubation time, and culture solutions’ chemistry were able to affect the antimicrobial efficiency of carbon nanotubes. The single-walled CNTs were found to be much more toxic to bacteria than that of multi-walled CNTs. NH2 functionalized CNTs were reported to show antibacterial effect at higher concentrations when compared to –OH and –COOH functionalized CNTs [63]. Possible mechanism of antimicrobial activity of carbon nanotubes was proposed as physical interaction with cell membrane, formation of cell-CNTs aggregates, and induction of the cell membrane disruption [64].
Pure PAN nanoweb and composite nanowebs (PAN/f-MWCNTs, PAN/AgNO3 and PAN/f-MWCNT/AgNO3) were evaluated for their antibacterial activity by measuring the cell concentration after an incubation step and a 24-h culture step on agar plates. Figure 8(a)–(d) displays the photographs of agar plates after the 24-h culture step. Antimicrobial efficiency percentages of composite nanofibers are presented in Table 10. With the simultaneous addition of f-MWCNTs and AgNPs, it was expected to see a synergistic increase in antibacterial activity against Photographs of petri dishes containing bacterial solutions: (a) untreated; (b) treated with PAN/f-MWCNTs; (c) treated with PAN/AgNO3; (d) treated with PAN/f-MWCNT/AgNO3. Antimicrobial efficiency of nanofibers. (−) represent not enough antimicrobial activity; (+) represent antimicrobial activity.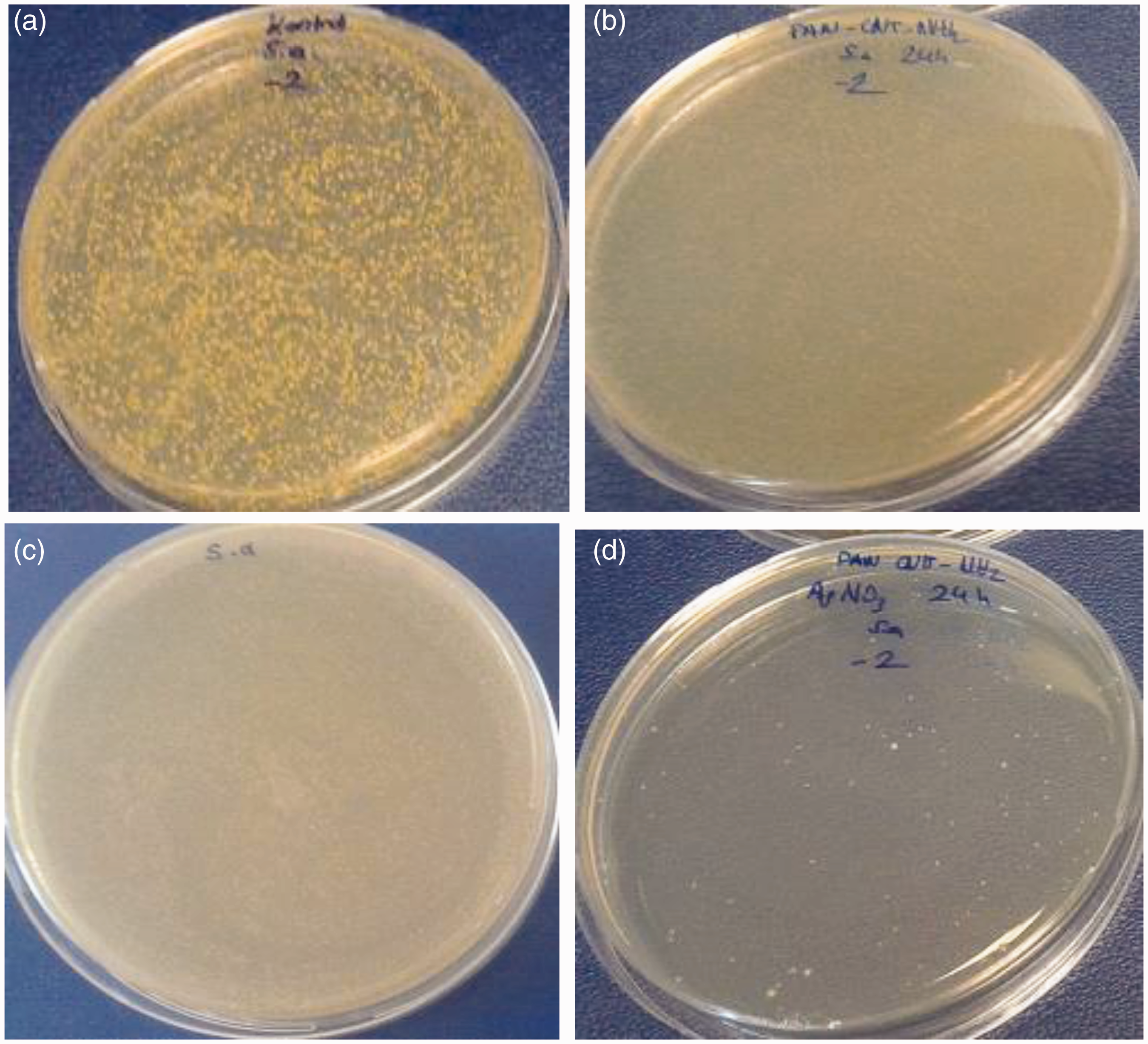

While nanofibers with 1% AgNO3 and 1% f-MWCNTs were unable to provide sufficient antimicrobial effect due to low additive concentration and weak antibacterial properties of CNTs, respectively, the antibacterial efficiency of PAN-1%f-MWCNT-1%AgNO3 composite nanofibers reached up to 99.83% after the chemical reduction process, due to the synergistic effect of the additives. The simultaneous addition of f-MWCNTs and AgNO3 might have affected the morphology of the nanofibers. The nanoparticles and the possible agglomerates might have resulted in the formation of some voids in the nanofiber structure. The fiber structure becoming more porous with the addition of different types of the additives might have led to the higher release of the silver ions resulting in higher antibacterial efficiency [65]. Besides, the interplay between the large surface area and strong adsorption property of the MWCNTs and high bactericidal activities of the AgNPs might have resulted in improvement of antibacterial efficiency [66].
Conclusions
PAN/f-MWCNT/Ag composite nanofibers were successfully prepared via electrospinning method using different contents of amine-functionalized MWCNTs and AgNO3. Chemical reduction of silver ions using an aqueous solution of hydrazinium hydroxide was conducted for the synthesis of silver nanoparticles. While an increase was observed in average nanofiber diameter, breaking elongation, conductivity, crystallinity, cyclization temperature, and enthalpy values, decrease was observed in ultimate tensile strength with the simultaneous addition of f-MWCNTs and AgNO3. Chemical reduction process had a thinning effect on fiber diameters. There was an increase in ultimate tensile strength and a decrease in breaking elongation of nanofiber webs with the application of the chemical reduction process. The conductivity of the nanofiber webs increased with respect to neat PAN nanofibers with the addition of conductive additives, but there was little or no effect with the increase in additive content, especially for the samples which are produced using f-MWCNTs. Compared with the addition of pristine CNTs, the addition of f-MWCNTs resulted in higher conductivity values. For the samples produced with pristine CNTs, conductivity decreased with the increased additive content. Silver reduction process generally resulted in a 10 times increase in conductivity. While nanofibers with 1 w% AgNO3 and 1 w% f-MWCNTs were unable to provide sufficient antibacterial effect due to low additive concentration and weak antibacterial properties of CNTs, respectively, the nanofiber webs produced with simultaneous addition of 1 w% f-MWCNTs and 1 w% AgNO3 displayed up to 99.83% antibacterial efficiency due to the synergistic effect of the additives. Among the selected factors of f-MWCNT content, AgNO3 content and application of reduction, the reduction process made the highest contribution to the ultimate tensile strength, elongation and conductivity of the nanowebs. f-MWCNT content had negligible effect on conductivity of the nanowebs.
Considering all the results obtained in this study, composite nanofiber webs of PAN with 1 w% f-MWCNTs and 1 w% AgNO3 can be suggested for use as antistatic and antibacterial material. Combining the advantages of nanofibers such as increased surface area, very small diameters, and high porosity with small pores with functional properties such as antistatic and antibacterial properties, these nanofiber webs can be used in biomedical textiles, tissue engineering, protective clothing, etc. with higher effectiveness.
Footnotes
Acknowledgements
The authors would like to thank Prof. Dr. Aysen Onen, Prof. Dr. Ismail Karacan, Msc. Eng. Olcay Eren, and Msc. Eng. Nesrin Demirsoy for their supports in some of the experimental studies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to thank TUBITAK for supporting this study with Project (112M877).
