Abstract
Composites comprising a braided poly(lactic) acid (PLA) filament and calcium phosphate bone cement (CPC) were inferred to maintain space and to pack porous fillers into restorative sites. Composites of alkalized multilayer-PLA braids and CPC (PLA/CPC) were divided into various groups according to a series of heat-treatment periods that lasted for 60, 90, 120, 150, and 180 min at 160℃; subsequently, these composites were characterized. Strength decays of samples were also compared after 24 h immersion in Hanks’s physiological solution. Results showed that the PLA/CPC specimens were toughened after treatment at 160℃ for 120 min. Furthermore, the moduli of PLA/CPC groups increased significantly when the heating time was more than 150 min; this effect was generated by the cold crystallization within the PLA filaments. The reduced stress in the composites after immersion was attributed to the fibers that protruded from the scaffold surface and to hydrolysis. The mechanical test results for the PLA/CPC composites indicated that the toughening effect was strengthened significantly under prolonged heat treatment, especially when the heating time was longer than 150 min. The cold crystallization degree of PLA increased, thereby enhancing the strength and toughness of a specimen before immersion. Thus, PLA/CPC composites can be used to simulate potential bone functions as well as to maintain three-dimensional spaces and pack porous fillers into restorative sites conveniently.
Introduction
Several techniques are employed to prepare medical composites that should meet requirements for biological application. The composites developed after implantation must not damage individual material and invoke aseptic tissue inflammation. The available importable orthopedic devices demonstrate two major trends: one is the improvement of medical device properties, such as by enhancing mechanical strength, prolonging the life of device designs, promoting bone cell growth and hastening recovery, and developing biocompatible and biodegradable devices. The other trend is the use of composite materials with bioactive factors. Grafts are obtained from implant surfaces containing peptide, plasma-rich platelets, and simplified protein for biological regeneration; this process enables osteoinduction, osteogenesis, and osteoconduction [1]. These developments and new applications mainly facilitate bone, teeth, and postoperative recovery [2].
Medical devices consist of composites that comprise a variety of materials, such as polymer and fibers, metals and their alloys, and ceramics (commonly used in biomedical applications). These materials exhibit diversified structural physiochemical and mechanical properties for prosthetic application. These composites utilize the properties of the constituent materials, thereby improving the mechanical properties and biological compatibilities of such composites. Bioabsorbable materials are the most well known among these composites; such materials are gradually dissolved into the surrounding tissues after implantation and are finally substituted by the regenerated tissues. The competing degradation and absorption rates in the materials are mainly ascribed to the combined factors of compositions, crystalline phases, and microstructures, among others. Typical examples of these degradable materials are tricalcium phosphate, amorphous phosphate, calcium deficient apatite, poly(lactic) acid (PLA), and poly(glycolic) acid.
The composition of calcium phosphate (CaP) is similar to the inorganic component of natural bones; therefore, CaPs have been investigated as bone-repair materials for more than 80 years [3]. Numerous synthetic CaPs have been evaluated and applied since 1970 [4–7]. Moreover, CaP bone cements (CPCs), which are mobile in slurry form and possess excellent biocompatibility, can be delivered through tiny pipes and can self-harden in situ [8]. CPCs can fit easily into bone defects automatically and can be used in bone restoration; therefore, these cements are excellent materials for repairing bone defects.
PLA is an aliphatic polyester that is produced via lactic acid polymerization. This polyester is a renewable fermentation product, and its melting point may vary from 171℃ to 60℃ [9]. The glass transition temperature (Tg) of PLA fibers is low at approximately 57℃, and the melting temperature (Tm) of these fibers is approximately 170℃. PLA has been presented as biodegradable and nontoxic in vivo since 1966 [10–12]; this polyester has since been well known as a bio-based polymer that is commercialized and mass produced. PLA has a degradation half-life that ranges from 6 months to 2 years depending on the size and shape of the molecules, isomer ratio, and environmental temperature [13]. PLA is primarily degraded by enzyme-catalyzed hydrolysis, which results in the release of degraded monomers with high local acid concentrations after clinical implantation. Thus, the localized inflammation induced remains a problem after PLA implantation in clinical trials [14].
To maximize the use of CPC as a matrix, this cement is combined with PLA braids, which causes CPC to release numerous ions that produce an environmental solution. This solution acts as a buffer and can be attributed to acid–base neutralization [9, 15–17]. Several studies have explored the nature of CaP and biodegradable PLA mixtures [18–20]. However, composites formed with PLA fibers or braids and CPC have not been investigated. The results from our previous study and the supplementary data show that the PLA/CPC composites heat-treated at different temperatures (100–190℃) clearly exhibit destructive and discontinuous PLA patterns, thereby inducing brittleness in PLA/CPC composites (Supplementary Figure 1). In the current study, we hypothesize that braided PLA tubes with highly brittle CPC positively affect the toughening response. To verify this hypothesis, PLA ply yarns are formed into braids with three, four, or five layers of braided PLA composites with CPC. The composites are heat-treated at 160℃, which is below the melting point of the PLA braids, under different treatment durations to measure the strengthening effects on the reinforced composites.
Materials and manufacturing
PLA filaments
PLA filaments [75 denier (D)/36f] were provided by Unitika, Ltd., Japan. Reagent-grade NaOH, stearic acid, and calcium carbonate were obtained from Osaka Drug (Shimakyu’s Pure Chemicals, Osaka, Japan). Dicalcium pyrophosphate (Alfa Aesar, Johnson Matthey Company, MA, USA) and dicalcium phosphate (DCPA, Janssen Chemical Co., Japan) were used to prepare CPC. The chemicals for simulated body fluid (SBF) preparation were obtained from Sigma–Aldrich Corporation, USA. A flowchart detailing the PLA/CPC composite fabrication procedures, heat treatments, and their duration as well as the testing regime that followed is presented in Figure 1.
Flow chart of the preparation and characterization of PLA/CPC composites under 160℃ heat treatment and their durations.
PLA braid manufacturing
The PLA braid processing and treatment procedures are described in literature [21]. A long capillary tube with an external diameter of 3.5 mm was used as a mandrel around which a 16-spindle braid machine spun 150 D PLA ply yarn into braids with three, four, and five layers; these braids were labeled as PLA3, PLA4, and PLA5, respectively. The bobbin corn with braid was placed in a thermal oven at 110℃ for 60 min to secure the exterior net of fibers [22]. The capillary was then removed to produce a hollow and porous PLA braid. The braid was then alkaline treated, rinsed with deionized water, and finally dried at 40℃ for 24 h.
CPC paste preparation
Tetracalcium phosphate (TTCP, Ca4P2O9) powder was prepared through Ca2P2O7 and CaCO3 reaction [23]. Equal molar weights of TTCP and commercial DCPA with corresponding mean particle sizes of 12.6 and 2.0 µm were mixed well and then added to 1 M phosphate-hardening solution (NaH2PO4) with a liquid/powder ratio of 0.55 ml/g to obtain a cement paste for hand layup.
PLA/CPC composite tube manufacturing
The CPC pastes were mixed well and then added into the porous PLA braids in the tubes through a hand layup process to impregnate the fibers with these pastes. The processes were carefully controlled to ensure uniformity in the capping amounts of PLA3/CPC, PLA4/CPC, and PLA5/CPC. All green PLA/CPC composites were stored under 100% humid environment at 37℃ for 24 h prior to the subsequent heat treatment. Finally, standardized sample tubes with a length of 6 mm, an inner diameter of 4 mm, and an outer diameter of approximately 5 mm (the thickness is controlled within a 10% deviation) were obtained and stored in a dehumidified environment. Once the PLA/CPC composites were fabricated, the specimens were divided into two groups. One group was not immersed in Hanks’s physiological SBF solution for the dry-strength test, and the other group followed the procedure of the wet-strength test [24]. Mechanical testing was conducted in 15 duplicates (n = 15).
Testing methods and analysis
Phase vaporization tests
The differential scanning calorimetry (DSC) method was used in this study, and temperatures had to reach a certain level to facilitate the analysis of the differences in the melting temperatures (Tm) and crystallization temperatures (Tc) of PLA braids. DSC curves were also measured in situ (MDSC 2920 and TG 2950 analyzers, TA Instrument, Inc.). The temperatures in the DSC tests were varied at a heating rate of 10℃/min, and the nitrogen air flow rate was set to 100 ml/min. The general weight of the specimen for thermal analysis was approximately 2 mg, and Al2O3 was utilized as the reference powder.
Fiber volume fraction and mechanical testing
The fiber volumes in the final PLA/CPC composite tubes are critical for strength. The fiber volume distribution in vol/vol% of the multilayered PLA braids was investigated (n = 10). The compressive strengths of PLA/CPC composites were measured at a speed of 1 min/mm via Instron 5566 (Instron Asia Ltd., USA). Preliminary studies showed that the PLA/CPC group heated for 120 min was considerably toughened and that the moduli of these groups increased when the heating time was more than 150 min. Therefore, five different heating durations were considered in this study: 60, 90, 120, 150, and 180 min. Furthermore, the composite strengths of the PLA/CPC composites that were not subject to heating treatment were particularly low because of the loose structural fabrics, the exterior fiber nets of which should be secured by heat. Moreover, no interactions were observed with CPC. Therefore, this study recorded the strengths, moduli, and the typical strength versus extension curves (i.e., the sample deformation under applied strength before fracture) of the experimental PLA/CPC composites heat-treated at 160℃ under different treatment durations (60, 90, 120, 150, and 180 min) before and after 24 h SBF immersion. Finally, a stereomicroscope (Olympus B061, Japan) was used to observe the surface of the resultant composites at a magnification of 0.67. A scanning electron microscope (SEM, S3000N, Hitachi, Japan) was utilized to examine the fiber distribution in the fractured composites at magnifications of 500× and 2000×.
Statistical analysis
Data were statistically analyzed with JMP 9.0 software (SAS Institute Inc., Cary, NC, US); differences in strength and moduli with p < 0.05 were considered statistically significant. One-way ANOVA and Tukey’s honest significant difference test were also conducted to analyze significant variations.
Results and discussion
DSC curves of the PLA sample
No significant difference was observed in the infrared spectra of PLA3, PLA4, and PLA5/CPC composites after heat treatment at 160℃ (Supplementary Figure 2); therefore, the PLA braids displayed a crystallization peak (Tc) near the small exothermal peaks at approximately 93℃ (Tc1) and 168℃ (Tc2) during the DSC test. Two melting peaks (Tm) were also detected at approximately 163℃ (Tm1) and 169℃ (Tm2) (Figure 2). PLA exhibited a Tg in the range of 50–60℃, and this polyester was rigid and brittle at lower temperatures [16]. When the heating temperature was above Tg and Tc1, the long-chain polymer began to move slowly and the ordered structure of cold crystallization was apparently [25–27]. At temperatures higher than Tm1, however, the degree of crystallinity in PLA was significantly higher than that of unheated composites. This phenomenon is attributed to the high amount of free energy that facilitates chain mobility, thereby reducing the strength and toughness of the specimen. The melting and crystallization behaviors occurred when the treatment temperature was higher than Tm1, thereby significantly enhancing strength. However, this strength suddenly declined subsequently. This result was observed with composites containing PLA with different and discontinuous fiber crystallinity; the entire fiber was molten into short fibers.
Differential scanning calorimetry heating curves (green lines) of untwisted PLA ply yarn and its secured exterior fiber net when pretreated at 70℃ and 110℃ for 30 and 60 min (the arrows indicate the corresponding quantities of heat flows at the specified temperatures).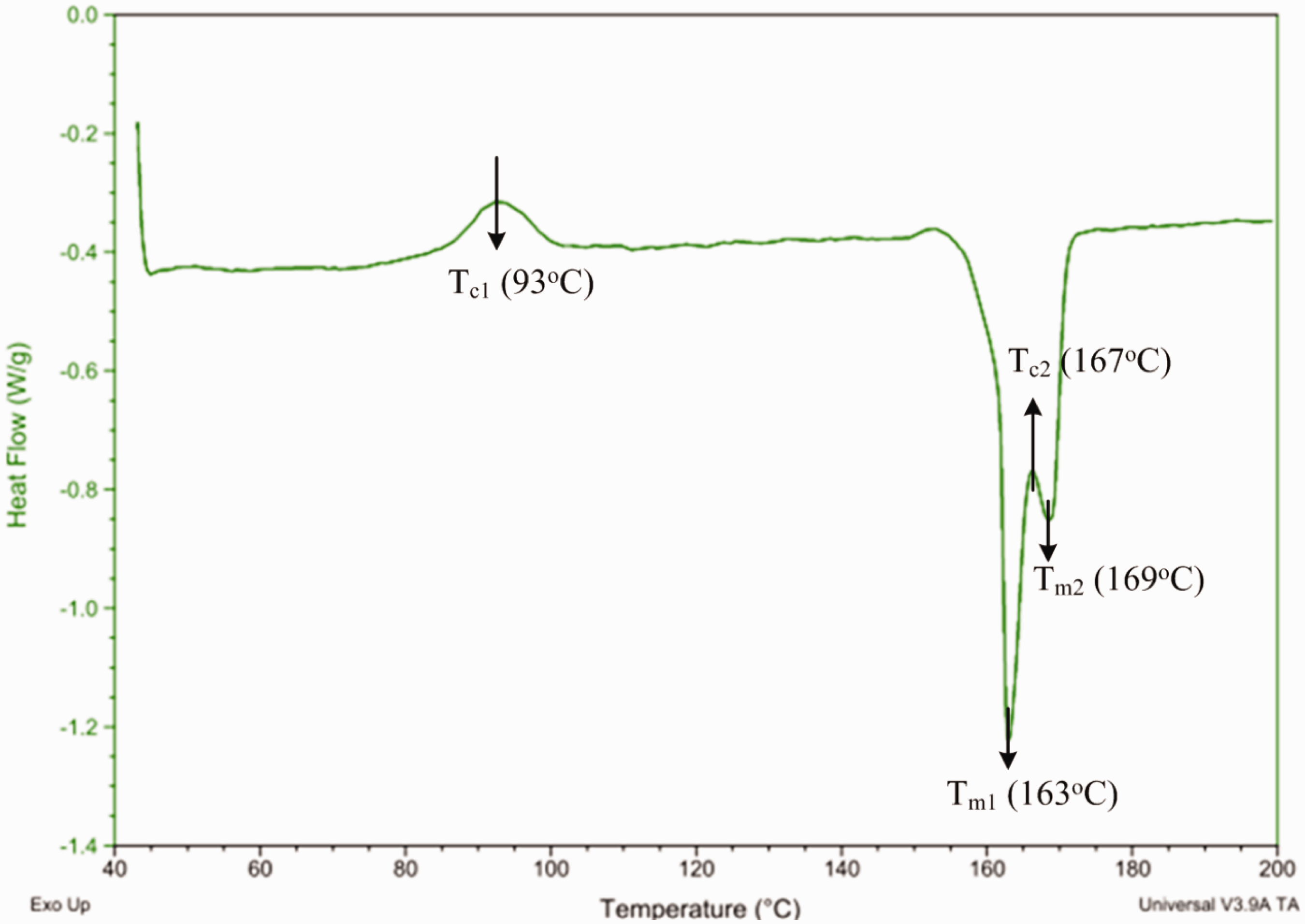
Fiber fractions and morphologies of PLA/CPC composite tubes
The PLA/CPC composite is shown in Figure 3 and the individual volumes percentage (vol%) of PLA, CPC and the PLA and CPC volume in composites are observation. The volume (vol). The prepared PLA/CPC composites had a straight hollow 3D structure with slight dimensional variations. CPC contents and PLA volumes were all shown apparently in linear proportion to braided PLA tubes with more PLA layers (Figure 3a). The increasing rate of PLA in multilayered PLA/CPC composites was higher than the rate of CPC (Figure 3b). The PLA vol% in composites of PLA3/CPC, PLA4/CPC, and PLA5/CPC were in proportion to about 20, 25, and 30 in vol/vol%. Figure 4 shows the fractures of the PLA3/CPC, PLA4/CPC, and PLA5/CPC groups treated for 60, 90, 120, 150, and 180 min at 160℃ after the strength tests. The results indicated that the fracture patterns of the PLA/CPC composites gradually change with prolonged heating; at heating durations of more than 120 min, crack growth and fractures are mainly detected along the PLA yarn braid because the PLA/CPC composites absorb energy by developing cracks in the fiber braid direction. Consequently, these composites are toughened (Figure 4). No significant difference was observed at the fracture interfaces of the PLA3/CPC, PLA4/CPC, and PLA5/CPC composites after heat treatment at 160℃ under various treatment durations after compressive testing. Typical SEM images were obtained for PLA5/CPC (Figure 5) and PLA3/CPC (Supplementary Figure 3).
Comparisons of multilayered PLA braids and hand lay-up CPC (blue-line) versus the PLA volumes (red-line) (a) individual volumes of PLA and CPC and (b) the PLA and CPC volume percentage (%) in PLA/CPC composite tubes after hardening (p < 0.001; n = 10). Original morphologies and fractures of PLA/CPC composites containing 3, 4, and 5 layers of PLA after compressive testing and after heat treatment at 160℃ under different treatment durations. Typical SEM images of a fractured specimen based on compression tests after heat treatment at 160℃ under various treatment durations. The images are captured at 500× and 2000× magnifications. The fractured surfaces of the PLA5/CPC tube specimen groups are shown before (dry-compressive forces) and after 24 h of immersion (wet-compressive forces). The white arrowheads indicate various groups that do not differ significantly from one another, and the gray arrowheads reveal the fibers protruding from the PLA fiber surface.


Strength and modulus of PLA/CPC composite tubes
Due to the treatment temperatures of 160℃ were approached to Tm1 of PLA at 163℃, thus, the crystallinity induced in PLA was dependent on the treatment periods. PLA crystallization was highlighted when the treatment was extended to 150 min because the high chain mobility interfered with the regular arrangement of the polymer chain (especially in the specimen group dried for 180 min, as indicated by the white arrow in Figure 5). PLA can be cold crystallized, as reflected by the exothermal peak at 90℃. However, the heat treatment at 120 min was too brief to induce PLA crystallization at 160℃ because of insufficient time for chain mobility. Cold crystallization became apparent with the further extension of the treatment to 150 min; this process enhanced compressive strength and moduli (Figure 6). A high ratio of PLA fiber to CPC matrix generally generated a higher strength and modulus due to the larger vol% amount of fiber facilitates stress transfer from the matrix to the fiber (Figures 3 and 5).
Dried strengths (a), moduli (b), and typical strength versus extension curves (c) of PLA/CPC composites with 3, 4, and 5 layers of PLA after heat treatment at 160℃ under various treatment durations. The compressive model of PLA5/CPC tube specimen groups is subjected to dry-compressive forces before immersion. Groups with similar letters denote no significant difference, whereas different letters reflect a statistical significance.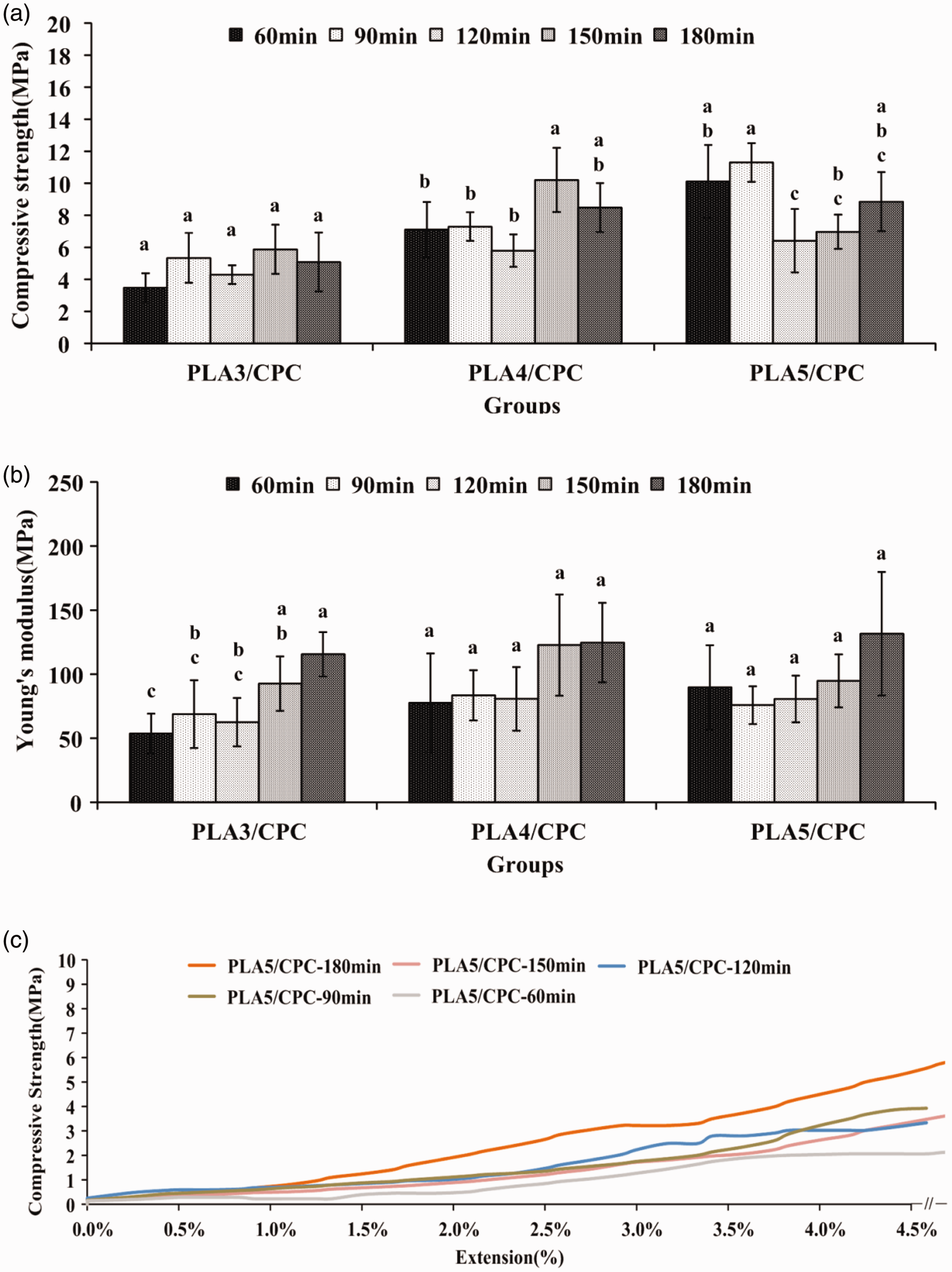
The wet and dry compressive strengths of the PLA/CPC groups in artificial body fluids were determined through day soaking and exhibited reduction tendencies that differed from those of the group treated for more than 150 min (Figures 6 and 7). Strength was significantly reduced in the PLA5/CPC group treated at 160℃ for 150 min. Furthermore, defects and bubbles were observed in the PLA after immersion when the heating time was prolonged to 150 min (the gray arrowheads in the bottom right corner of Figure 5). This phenomenon suggests that the isotropical melting of PLA yarns results in the loss of the braid parallel to the fibers. Thus, apparent cold crystallization eliminated the toughening effect of the braid after immersion (Figure 7).
Wet strengths (a), moduli (b), and typical strength versus extension curves (c) of PLA/CPC composites with 3, 4, and 5 layers of PLA after heat treatment at 160℃ under various treatment durations. The wet compressive modes of the PLA5/CPC tube specimen groups are processed after 24 h immersion. Groups with similar letters do not differ significantly from one another.
The brittleness and bearing load of CaP scaffolds with highly interconnected pores limit the application of this compound in bone restoration [28, 29]. The compressive strength of porous scaffolds with CaP as the matrix is only 3 MPa [30], and scaffolds with bioactive borosilicate glass can lead to dense struts and a maximum compressive strength of approximately 10 MPa given an overall porosity of approximately 70% [31, 32]. This study employed an approach that combines a multilayer PLA composite with CPC matrix; the composite displayed high-compressive strength of over 10 MPa before immersion. These results are comparable to those for a trabecular bone and are considerably higher than those for pure, highly porous ceramic scaffolds. The PLA/CPC group heated for 120 min was toughened, as indicated by the strength versus extension (specimen deformation) curve. This effect is caused by the cold crystallization within the PLA filaments. The moduli of the PLA/CPC groups tended to increase when the heating time was more than 150 min; this characteristic resulted in a material with an improved capability to absorb external applied energy. Thus, the PLA/CPC tubes can resist the initial fracture after implantation. Therefore, the optimal heat treatment time should be selected. Furthermore, the designed PLA/CPC composites that comprise a cortical layer of CPC with biodegradable PLA braids can be responsible for the anticrack behavior observed during implantation in vivo, especially in load-bearing restoration. As per this study, increasing the treatment time at 160℃ can increase the breaking strength of PLA braids; thus, an optimal treatment should be developed that would toughen and reinforce PLA/CPC composites. Although PLA exhibits high tensile strength and biocompatibility at initial implantation, the decay and fixation of PLA fabrics after long-term restoration in situ has raised several issues because the acidic degradation of PLA may cause toxicity. The CPC composed of TTCP and DCPA [8], and their reactors mainly neutralize these acidic properties after reaction; as a result, the biocompatibility of this material is high in PLA/CPC composites. Furthermore, oversaturated Ca2+ and phosphate ions are involved in apatite precipitation after the generated CPC reacts with native bones; the bio-mechanisms and subsequent regeneration of osteoclasts may support osteoregeneration before PLA degradation [33]. The fabricated CPC prevents acidic degradation of PLA and serves as a PLA component to contribute to the ductility of PLA/CPC composites.
Conclusion
This study successfully creates the three different composite bone scaffolds by infusing CPC pastes into braided PLA tubes. The braided PLA tubes in highly brittle CPC could positively affect toughening response, and the cold crystallization in PLA could enhance the strength and toughness of the composites that were attributed to the energy absorbed via crack propagation in the fiber braid direction. According to the experimental results, the optimal outcome was apparent in the PLA5/CPC group with its concentration in vol/vol% of about 30% PLA/70%CPC treated at a heating time that should be limited to not more than 150 min. Such composite bone scaffolds have optimal compressive strength when they contain highly PLA volume fractions of as well as in groups with thick PLA layers of PLA5/CPC group. As shown in SEM observation, multilayers of PLA groups that were heat-treated for more than 150 min were create crystallized agglomeration and led to the strength decay effects of PLA/CPC composites. PLA/CPC composites can be future applied clinically in simulate potential bone functions as well as to maintain three-dimensional spaces and pack porous fillers into restorative sites conveniently.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the financial support provided by the Ministry of Science and Technology, Taiwan, under contract numbers MOST 102, 103, and 104-2622-E-035-006-CC2 and MOST 101-2815-C-035-026-E.
